Abstract
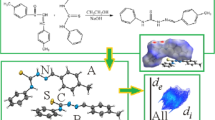
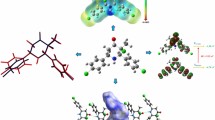
A new Schiff base compound, (Z)-1-(2-(4-nitrophenyl)hydrazineylidene)naphthalen-2(1H)-one, was prepared and structurally characterized by single-crystal X-ray diffraction. The compound crystallizes in the P2/n monoclinic space group with a = 15.9421(6) Å, b = 7.7607(3) Å, c = 21.8052(9) Å, and β = 92.946(3)°. The molecular geometry of the compound was optimized using density functional theory calculation at HSEH1PBE/cc-pvdz level. Hirshfeld surface analyses for the compound were presented and discussed. The computational findings show that theoretical geometric parameters are consistent with experimental ones. The calculated and experimental results show the title compound tends to the hydrazone (NH) form. Stabilization of the crystal structure was achieved by means of intramolecular N–H⋯O and intermolecular C–H⋯O interactions.










Similar content being viewed by others
REFERENCES
T. A. Khattab, A. A. Allam, S. I. Othman, M. Bin-Jumah, H. M. Al-Harbi, and M. M. G. Fouda. Synthesis, solvatochromic performance, pH sensing, dyeing ability, and antimicrobial activity of novel hydrazone dyestuffs. J. Chem., 2019, 2019, 7814179. https://doi.org/10.1155/2019/7814179
H. Valizadeh and A. Shomali. A new nitrite ionic liquid (IL-ONO) as a nitrosonium source for the efficient diazotization of aniline derivatives and in-situ synthesis of azo dyes. Dyes Pigm., 2012, 92, 1138-1143. https://doi.org/10.1016/j.dyepig.2010.11.010
X. Su and I. Aprahamian. Hydrazone-based switches, metallo-assemblies and sensors. Chem. Soc. Rev., 2014, 43, 1963-1981. https://doi.org/10.1039/C3CS60385G
L. A. Tatum, X. Su, and I. Aprahamian. Simple hydrazone building blocks for complicated functional materials. Acc. Chem. Res., 2014, 47, 2141-2149. https://doi.org/10.1021/ar500111f
L. Popiołek. Hydrazide–hydrazones as potential antimicrobial agents: overview of the literature since 2010. Med. Chem. Res., 2017, 26, 287-301. https://doi.org/10.1007/s00044-016-1756-y
L. Popiołek, B. Rysz, A. Biernasiuk, and M. Wujec. Synthesis of promising antimicrobial agents: Hydrazide-hydrazones of 5-nitrofuran-2-carboxylic acid. Chem. Biol. Drug Des., 2020, 95, 260-269. https://doi.org/10.1111/cbdd.13639
H. M. Marwani, A. M. Asiri, and S. A. Khan. Green-synthesis, characterization, photostability and polarity studies of novel Schiff base dyes using spectroscopic methods. Russ. J. Bioorg. Chem., 2012, 38, 533-538. https://doi.org/10.1134/S1068162012050056
K. C. Gupta and A. K. Sutar. Catalytic activities of Schiff base transition metal complexes. Coord. Chem. Rev., 2008, 252, 1420-1450. https://doi.org/10.1016/j.ccr.2007.09.005
A. Caballero, R. Martinez, V. Lloveras, I. Ratera, J. Vidal-Gancedo, K. Wurst, A. Tarraga, P. Molina, and J. Veciana. Highly selective chromogenic and redox or fluorescent sensors of Hg2+ in aqueous environment based on 1,4-disubstituted azines. J. Am. Chem. Soc., 2005, 127, 15666/15667. https://doi.org/10.1021/ja0545766
R. L. Sheng, P. F. Wang, W. M. Liu, X. H. Wu, and S. K. Wu. A new colorimetric chemosensor for Hg2+ based on coumarin azine derivative. Sens. Actuators B, 2008, 128, 507-511. https://doi.org/10.1016/j.snb.2007.07.069
S. H. Kim, S. Y. Gwon, S. M. Burkinshaw, and Y. A. Son. The synthesis and proton-induced spectral switching of a novel azine dye and its boron complex. Dyes Pigm., 2010, 87, 268-271. https://doi.org/10.1016/j.dyepig.2010.04.006
J. M. Locke, R. Griffith, T. D. Bailey, and R. L. Crumbie. Competition between cyclisation and bisimine formation in the reaction of 1,3-diaminopropanes with aromatic aldehydes. Tetrahedron, 2009, 65, 10685-10692. https://doi.org/10.1016/j.tet.2009.10.060
S. Rayati, S. Zakavi, M. Koliaei, A. Wojtczak, and A. Kozakiewicz. Electron-rich salen-type Schiff base complexes of Cu(II) as catalysts for oxidation of cyclooctene and styrene with tert-butylhydroperoxide: A comparison with electron-deficient ones. Inorg. Chem. Commun., 2010, 13, 203-207. https://doi.org/10.1016/j.inoche.2009.11.016
K. Butsch, T. Gunther, A. Klein, K. Stirnat, A. Berkessel, and J. Neudorfl. Redox chemistry of copper complexes with various salen type ligands. Inorg. Chim. Acta, 2013, 394, 237-246. https://doi.org/10.1016/j.ica.2012.08.016
H. P. Ebrahimi, J. S. Hadi, Z. A. Abdulnabi, and Z. Bolandnazar. Spectroscopic, thermal analysis and DFT computational studies of salen-type Schiff base complexes. Spectrochim. Acta, Part A, 2014, 117, 485-492. https://doi.org/10.1016/j.saa.2013.08.044
S. Menati, A. Azadbakht, R. Azadbakht, A. Taeb, and A. Kakanejadifard. Synthesis, characterization, and electrochemical study of some novel, azo-containing Schiff bases and their Ni(II) complexes. Dyes Pigm., 2013, 98, 499-506. https://doi.org/10.1016/j.dyepig.2013.04.009
W. Radecka-Paryzek, V. Patroniak, and J. Lisowski. Metal complexes of polyaza and polyoxaaza Schiff base macrocycles. Coord. Chem. Rev., 2005, 249, 2156-2175. https://doi.org/10.1016/j.ccr.2005.02.021
S.-Z. Li, L. Tong, X. Li, and W.-K. Dong. New insight into two penta-coordinated multinuclear copper(II) single-armed salamo-based complexes. Inorg. Chim. Acta, 2022, 540, 121047. https://doi.org/10.1016/j.ica.2022.121047
S.-Z. Li, Y.-X. Wei, Y. Huang, and W.-K. Dong, Counteranion-driven self-assembly of di- and tetra-nuclear Zn(II) single-armed salamo-type complexes. J. Mol. Struct., 2022, 1265, 133473. https://doi.org/10.1016/j.molstruc.2022.133473
C. T. Grainger and J. F. McConnell. The crystal structure of 1-p-nitrobenzeneazo-2-naphthol (Para Red) from overlapped twin-crystal data. Acta Crystallogr. B, 1969, 25, 1962-1970. https://doi.org/10.1107/S0567740869005036
A. Whitaker. Crystal data for a second polymorph (β) of C.I. Pigment Red 1,1-[(4-nitrophenyl)azo]-2-naphthol. J. Appl. Crystallogr., 1979, 12, 626/627. https://doi.org/10.1107/S0021889879013509
A. Whitaker. Crystal data for a third polymorph (γ) of C.I. Pigment Red 1,1-[(4-nitrophenyl)azo]-2-naphthol. J. Appl. Crystallogr., 1980, 13, 458/459. https://doi.org/10.1107/S002188988001254X
A. Whitaker. The crystal structure of a second polymorph (β) of C.I. Pigment Red 1,1-[(4-nitrophenyl)azo]-2-naphthol. Z. Kristallogr., 1980, 152, 227-238. https://doi.org/10.1524/zkri.1980.152.14.227
O. S. Bushuyev, T. A. Singleton, and C. J. Barrett. Fast, reversible, and general photomechanical motion in single crystals of various azo compounds using visible light. Adv. Mater., 2013, 25, 1796-1800. https://doi.org/10.1002/adma.201204831
X-AREA Version 1.18, X-RED32 Version 1.04. Darmstadt, Germany: Stoe and Cie, 2002.
L. Palatinus and G. Chapuis. SUPERFLIP - a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Crystallogr., 2007, 40, 786-790. https://doi.org/10.1107/S0021889807029238
G. M. Sheldrick. Crystal structure refinement with SHELXL. Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71(1), 3-8. https://doi.org/10.1107/s2053229614024218
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard, and H. Puschmann. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr., 2009, 42, 339-341. https://doi.org/10.1107/S0021889808042726
J. Heyd, G. E. Scuseria, and M. Ernzerhof. Erratum: “Hybrid functionals based on a screened Coulomb potential” (J. Chem. Phys., 118, 8207 (2003)). J. Chem. Phys., 2006, 124(21), 219906. https://doi.org/10.1063/1.2204597
A. V. Krukau, O. A. Vydrov, A. F. Izmaylov, and G. E. Scuseria. Influence of the exchange screening parameter on the performance of screened hybrid functionals. J. Chem. Phys., 2006, 125, 224106. https://doi.org/10.1063/1.2404663
T. H. Dunning Jr. Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys., 1989, 90, 1007-1023. https://doi.org/10.1063/1.456153
A. K. Wilson, T. van Mourik, and T. H. Dunning Jr. Gaussian basis sets for use in correlated molecular calculations. VI. Sextuple zeta correlation consistent basis sets for boron through neon. J. Mol. Struct.: THEOCHEM, 1996, 388, 339-349. https://doi.org/10.1016/S0166-1280(96)80048-0
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox. Gaussian09, Revision E.01. Wallingford, CT, USA: Gaussian Inc., 2009.
F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen, and R. Taylor. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lengths in organic compounds. J. Chem. Soc. Perkin Trans. 2, 1987, (12), S1. https://doi.org/10.1039/p298700000s1
J. Bernstein, R. E. Davis, L. Shimoni, and N.-L. Chang. Patterns in hydrogen bonding: functionality and graph set analysis in crystals. Angew. Chem., Int. Ed. Engl., 1995, 34(15), 1555-1573. https://doi.org/10.1002/anie.199515551
M. A. Spackman and J. J. McKinnon. Fingerprinting intermolecular interactions in molecular crystals. CrystEngComm, 2002, 4, 378-392. https://doi.org/10.1039/B203191B
M. A. Spackman and P. G. Byrom. A novel definition of a molecule in a crystal. Chem. Phys. Lett., 1997, 267, 215-220. https://doi.org/10.1016/S0009-2614(97)00100-0
J. J. McKinnon, A. S. Mitchell, and M. A. Spackman. Hirshfeld surfaces: A new tool for visualising and exploring molecular crystals. Chem. Eur. J., 1998, 4, 2136-2141. https://doi.org/10.1002/(SICI)1521-3765(19981102)4:11<2136::AID-CHEM2136>3.0.CO;2-G
J. J. McKinnon, D. Jayatilaka, and M. A. Spackman. Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem. Commun., 2007, 37, 3814-3816. https://doi.org/10.1039/B704980C
A. L. Rohl, M. Moret, W. Kaminsky, K. Claborn, J. J. Mckinnon, and B. Kahr. Hirshfeld surfaces identify inadequacies in computations of intermolecular interactions in crystals: Pentamorphic 1,8-dihydroxyanthraquinone. Cryst. Growth Des., 2008, 8, 4517-4525. https://doi.org/10.1021/cg8005212
T. Maity, H. Mandal, A. Bauza, B. C. Samanta, A. Frontera, and S. K. Seth. Quantifying conventional C–H⋯π(aryl) and unconventional C–H⋯π(chelate) interactions in dinuclear Cu(II) complexes: Experimental observations, Hirshfeld surface and theoretical DFT study. New J. Chem., 2018, 42, 10202-10213. https://doi.org/10.1039/C8NJ00747K
S. K. Seth. The importance of CH⋯X (X = O, π) interaction of a new mixed ligand Cu(II) coordination polymer: Structure, Hirshfeld surface and theoretical studies. Crystals, 2018, 8, 455. https://doi.org/10.3390/cryst8120455
P. R. Spackman, M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, D. Jayatilaka, and M. A. Spackman. CrystalExplorer: A program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals J. Appl. Crystallogr., 2021, 54(3), 1006-1011. https://doi.org/10.1107/S1600576721002910
R. G. Parr, L. Szentpaly, and S. Liu. Electrophilicity index. J. Am. Chem. Soc., 1999, 121, 1922-1924. https://doi.org/10.1021/ja983494x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author declares that he has no conflicts of interests.
Additional information
Text © The Author(s), 2023, published in Zhurnal Strukturnoi Khimii, 2023, Vol. 64, No. 8, 114403.https://doi.org/10.26902/JSC_id114403
Rights and permissions
About this article
Cite this article
Yildirim Gümüşhan, I. Synthesis, Crystal Structure, Hirshfeld Surface Analysis and DFT Studies of (Z)-1-(2-(4-Nitrophenyl)Hydrazineylidene)Naphthalen-2(1H)-One. J Struct Chem 64, 1435–1447 (2023). https://doi.org/10.1134/S0022476623080085
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0022476623080085