Abstract—
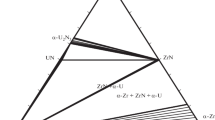
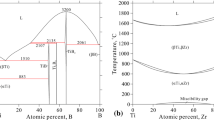
A thermodynamic model is proposed for condensed phases in the ternary system Nb–Zr–N in the range 298–3000 K. The model is based on available experimental data and previously reported models of the constituent binaries Nb–Zr, Nb–N, and Zr–N. It is consistent with a previously reported subsolidus phase diagram of the Nb–Zr–N system and allows one to assess phase equilibria involving liquid phase.





Similar content being viewed by others
REFERENCES
Sheftel, E.N. and Bannykh, O.A., Niobium-base alloys, Int. J. Refract. Met. Hard Mater., 1993–1994, vol. 12, no. 5, pp. 303–314.https://doi.org/10.1016/0263-4368(93)90038-H
Ban’kovskii, O.I., Moiseev, V.F., Pechkovskii, E.P., and Trefilov, V.I., Phase composition of cast Nb–Zr–N alloys at solidus temperatures, Metallofizika, 1974, no. 53, pp. 103–109.
Holleck, H., Binary and Ternary Carbides and Nitrides of the Transition Metals and Their Phase Relations, Karlsruhe: Kernforschungszentrum, 1981.
Barabash, O.M., Kozyrskii, G.Ya., Shul’zhenko, V.K., and Shurin, A.K., Structure and strength of Nb–ZrN alloys, Izv. Akad. Nauk SSSR, Met., 1976, no. 3, pp. 220–225.
Saunders, N. and Miodownik, A.P., CALPHAD (Calculation of Phase Diagrams): A Comprehensive Guide, Amsterdam: Elsevier, 1998.
Dinsdale, A.T., SGTE data for pure elements, CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 1991, vol. 15, no. 4, pp. 317–425.https://doi.org/10.1016/0364-5916(91)90030-N
Duwez, P. and Odell, F., Phase relationships in the binary systems of nitrides and carbides of zirconium, columbium, titanium, and vanadium, J. Electrochem. Soc., 1950, vol. 97, no. 10, pp. 299–304.
Toth, L.E., Yen, C.M., Rosner, L.G., and Anderson, D.E., Superconducting critical fields, currents and temperatures in the Nb–Zr–N ternary system, J. Phys. Chem. Solids, 1966, vol. 27, nos. 11–12, pp. 1815–1819. https://doi.org/10.1016/0022-3697(66)90112-0
Lafaye, P., Toffolon-Masclet, C., Crivello, J.-C., and Joubert, J.-M., Experimental investigations and thermodynamic modelling of the Cr–Nb–Sn–Zr system, CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2019, vol. 64, pp. 43–54.https://doi.org/10.1016/j.calphad.2018.11.002
Huang, W., Thermodynamic assessment of the Nb–N system, Metall. Mater. Trans. A, 1996, vol. 27, no. 11, pp. 3591–3600.https://doi.org/10.1007/BF02595450
Sridar, S., Kumar, R., and Hari Kumar, K.C., Thermodynamic modelling of Ti–Zr–N system, CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2017, vol. 56, pp. 102–107.https://doi.org/10.1016/j.calphad.2016.12.003
Xiaoyan Ma, Changrong Li, Kewu Bai, Ping Wu, and Weijing Zhang, Thermodynamic assessment of the Zr–N system, J. Alloys Compd., 2004, vol. 373, nos. 1–2, pp. 194–201.https://doi.org/10.1016/j.jallcom.2003.10.051
Hillert, M., The compound energy formalism, J. Alloys Compd., 2001, vol. 320, no. 2, pp. 161–176.https://doi.org/10.1016/S0925-8388(00)01481-X
Chen, S.L., Zhang, J.Y., Lu, X.G., et al., Application of Graham scan algorithm in binary phase diagram calculation, J. Phase Equilib. Diffus., 2006, vol. 27, no. 2, pp. 121–125.https://doi.org/10.1007/s11669-006-0034-y
Voskov, A.L., Dzuban, A.V., and Maksimov, A.I., TernAPI program for the calculation of ternary phase diagrams with isolated miscibility gaps by the convex hull method, Fluid Phase Equilib., 2015, vol. 388, pp. 50–58.https://doi.org/10.1016/j.fluid.2014.12.028
Funding
This work was supported by the Russian Science Foundation, project no. 20-13-00392 (federal state budget funded science institution Baikov Institute of Metallurgy and Materials Science, Russian Academy of Sciences).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Tsarev
Rights and permissions
About this article
Cite this article
Voskov, A.L., Kovalev, I.A., Kochanov, G.P. et al. Thermodynamic Modeling of Phase Equilibria in the Nb–Zr–N System. Inorg Mater 58, 509–515 (2022). https://doi.org/10.1134/S0020168522050119
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168522050119