Abstract
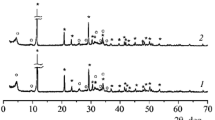
Brushite (CaHPO4 · 2H2O) powder has been synthesized in aqueous 1.0 M solutions of calcium nitrate dipotassium hydrogen phosphate, and disodium hydrogen phosphate at a Ca/P ratio of unity, without adjusting the pH of the reaction. After synthesis and drying, the fraction of a reaction by-product (NaNO3, KNO3, or their mixture) in the powder was about 20 wt %. After firing at temperatures from 800 to 1000°C, the ceramics prepared using the powder synthesized from Ca(NO3)2 and Na2HPO4 consisted of β-Ca2P2O7 and β-NaCaPO4. After firing at temperatures from 900 to 1100°C, the ceramics prepared using the powder synthesized from Ca(NO3)2 and K2HPO4 consisted of Са10К(РО4)7 and СаК2Р2О7. The ceramic composites produced in this study can be recommended as materials for resorbable bone implants.
Similar content being viewed by others
References
Danil’chenko, S.N., Structure and properties of calcium apatites from the viewpoint of biomineralogy and biomaterials research (a review), Visn. Sumsk. Derzh. Univ., Ser. Fiz., Mat., Mekh., 2007, no. 2, pp. 33–59.
Gerk, S.A. and Golovanova, O.A., Normal and pathological elemental compositions of human bone tissue, Vestn. Omsk. Univ., 2015, no. 4 (78), pp. 39–44.
Barinov, S.M., Calcium phosphate-based ceramic and composite materials for medicine, Russ. Chem. Rev., 2010, vol. 79, no. 1, pp. 13–29.
Putlayev, V.I., Safronova, T.V., Veresov, A.G., Klimashina, E.S., and Filippov, Ya.Yu., Resorbable inorganic materials for bone implants: searching strategy, Advanced Metals, Ceramics and Composites: The 12th China–Russia Symposium on Advanced Materials and Technologies (CRSAMT 2013), Kunming: Yunnan Science and Technology, 2013, vol. 1, pp. 225–230.
Putlyaev, V.I. and Safronova, T.V., A new generation of calcium phosphate biomaterials: the role of phase and chemical compositions, Glass Ceram., 2006, vol. 63, nos. 3–4, pp. 99–102.
Bohner, M., Resorbable biomaterials as bone graft substitutes (review), Mater. Today, 2010, vol. 13, nos. 1–2, pp. 24–30.
Putlayev, V.I., Evdokimov, P.V., Garshev, A.V., Prosvirin, D.V., Klimashina, E.S., Safronova, T.V., and Ivanov, V.K., Strength characteristics of resorbable osteoconductive ceramics based on diphosphates of calcium and alkali metals, Russ. Phys. J., 2014, vol. 56, no. 10, pp. 1183–1189.
Evdokimov, P.V., Putlyaev, V.I., Ivanov, V.K., Garshev, A.V., Shatalova, T.B., Orlov, N.K., Klimashina, E.S., and Safronova, T.V., Phase equilibria in the tricalcium phosphate–mixed calcium sodium (potassium) phosphate systems, Russ. J. Inorg. Chem., 2014, vol. 59, no. 11, pp. 1219–1227.
Safronova, T.V. and Putlyaev, V.I., Powder systems for calcium phosphate ceramics, Inorg. Mater., 2017, vol. 53, no. 1, pp. 17–26.
Putlyaev, V.I., Kukueva, E.V., Safronova, T.V., Ivanov, V.K., and Churagulov, B.R., Features of octacalcium phosphate thermolysis, Refract. Ind. Ceram., 2014, vol. 54, no. 5, pp. 420–424.
Safronova, T.V., Mukhin, E.A., Putlyaev, V.I., Knotko, A.V., Evdokimov, P.V., Shatalova, T.B., Filippov, Ya.Yu., Sidorov, A.V., and Karpushkin, E.A., Amorphous calcium phosphate powder synthesized from calcium acetate and polyphosphoric acid for bioceramics application, Ceram. Int., 2017, vol. 43, pp. 1310–1317.
Khimicheskaya tekhnologiya keramiki (Chemical Technology of Ceramics), Guzman, I.Ya., Ed., Moscow: Stroimaterialy, 2003.
Safronova, T.V., Phase composition of ceramic based on calcium hydroxyapatite powders containing byproducts of the synthesis reaction, Glass Ceram., 2009, vol. 66, nos. 3–4, pp. 136–139.
Gustafsson, J.P., Visual MINTEQ Version 3.1, Stockholm: Department of Sustainable Development, Environmental Science and Engineering, KTH, 2013. https://vminteq.lwr.kth.se/.
PDF4+ Database, Newtown Square: International Centre for Diffraction Data, 2010. http://www.icdd. com/products/pdf2.htm.
Kramer, C.M. and Wilson, C.J., The phase diagram of NaNO3–KNO3, Thermochim. Acta, 1980, vol. 42, no. 3, pp. 253–264.
Voskresenskaya, N.K., Evseeva, N.N., Berul’, S.I., and Vereshchetina, I.P., Spravochnik po plavkosti sistem iz bezvodnykh neorganicheskikh solei (Melting Behavior of Anhydrous Inorganic Salt Systems: A Handbook), vol. 1: Dvoinye sistemy (Binary Systems), Moscow: Akad. Nauk SSSR, 1961.
Lur’e, Yu.Yu., Spravochnik po analiticheskoi khimii (Handbook of Analytical Chemistry), Moscow: Khimiya, 1971, 4th rev. ed.
Berak, J. and Znamierowska, T., Phase equilibria in the system CaO–Na2O–P2O5. Part II. The partial system Ca(PO3)2–Na2O–P2O5, Rocz. Chem., 1972, vol. 46, no. 10, pp. 1697–1708.
Znamierowska, T., Phase equilibriums in the system calcium oxide–potassium oxide–phosphorus(V) oxide. Part II. Partial system calcium phosphate–calcium potassium pyrophosphate–potassium metaphosphate–calcium pyrophosphate, Pol. J. Chem., 1978, vol. 52, no. 6, pp. 1127–1134.
Safronova, T.V., Putlyaev, V.I., Shekhirev, M.A., and Kuznetsov, A.V., Composite ceramic containing a bioresorbable phase, Glass Ceram., 2007, vols. 64, nos. 3–4, pp. 102–106.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.V. Safronova, V.I. Putlyaev, Ya.Yu. Filippov, T.B. Shatalova, D.S. Fatin, 2018, published in Neorganicheskie Materialy, 2018, Vol. 54, No. 2, pp. 210–220.
Rights and permissions
About this article
Cite this article
Safronova, T.V., Putlyaev, V.I., Filippov, Y.Y. et al. Ceramics Based on Brushite Powder Synthesized from Calcium Nitrate and Disodium and Dipotassium Hydrogen Phosphates. Inorg Mater 54, 195–207 (2018). https://doi.org/10.1134/S0020168518020127
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168518020127