Abstract
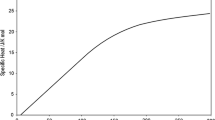
The paper analyzes all of the experimental data available in publications and main reviews on the thermodynamic properties of pure vanadium in a condensed state. Based on the analysis, we proposed a new temperature dependence of the specific heat in the range of 298.15–2650 K at standard pressure. It is represented by a single equation for the solid phase. The result was prepared for uploading to the IVTANTERMO information system.



Similar content being viewed by others
REFERENCES
Gurvich, L.V., Veits, I.V., Medvedev, V.A., et al., Termodinamicheskie svoistva individual’nykh veshchestv. Spravochnoe izdanie, Glushko, V.P., Ed., Moscow: Nauka, 1982, vol. 4 (Thermodynamic Properties of Individual Substances), Glushko, V.P., Ed., New York: Hemisphere Publishing, 1994, vol. 3.
Gurvich, L.V., Vestn. Akad. Nauk SSSR, 1983, no. 3, p. 54.
Belov, G.V., Iorish, V.S., and Yungman, V.S., CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 1999, vol. 23, no. 2, p. 173.
Thermodynamic properties of individual substances, vol. 6. http://www.chem.msu.su/rus/tsiv/.
Arblaster, J.W., J. Phase Equilib. Diffus., 2017, vol. 38, no. 1, p. 51.
Belov, G.V., Dyahkov, S.A., Levashov, P.R., et al., J. Phys.: Conf. Ser., 2018, vol. 946, no. 1, 012120.
Shomate, C.H., J. Am. Chem. Soc., 1944, vol. 66, no. 6, p. 928.
Chemistry WebBook, The National Institute of Standards and Technology. http://webbook.nist.gov/chemistry/.
Belov, G.V., Morozov, I.V., Sineva, M.A., et al., J. Phys.: Conf. Ser., 2019, vol. 1385, 012025.
Aristova, N.M., Belov, G.V., Morozov, I.V., and Sineva, M.A., High Temp., 2018, vol. 56, no. 5, p. 652.
Jaeger, F.M. and Veenstra, W.A., Recl. Trav. Chim. Pays-Bas, 1934, vol. 53, no. 8, p. 677.
Berezin, B.Ya. and Chekhovskoi, V.Ya., Teplofiz.Vys. Temp., 1977, vol. 15, no. 4, p. 772.
Gathers, G.R., Shaner, J.W., Hixson, R.S., and Young, D.A., High Temp.—High Pressure, 1979, vol. 11, no. 6, p. 653.
Chekhovskoi, V.Ya. and Kalinkina, R.G., Teplofiz.Vys. Temp., 1973, vol. 11, no. 4, p. 885.
Kohlhaas, R., Braun, M., and Vollmer, O., Z. Naturforsch.,A: Phys. Sci., 1965, vol. 20, no. 8, p. 1077.
Lin, R. and Frohberg, M.G., Z. Metallkd., 1991, vol. 82, no. 1, p. 48.
Schaefers, K., Rösner-Kuhn, M., and Frohberg, M.G., Mater. Sci. Eng., A, 1995, vol. 197, no. 1, p. 83.
Berezin, B.Ya., Chekhovskoi, V.Ya., and Sheindlin, A.E., High Temp. Sci., 1972, vol. 4, no. 6, p. 478.
Takahashi, Y., Nakamura, J.I., and Smith, J.F., J. Chem. Thermodyn., 1982, vol. 14, no. 10, p. 977.
Cezairliyan, A., Righini, F., and McClure, J.L., J. Res. Natl. Bur. Stand.,Sect. A, 1974, vol. 78, no. 4, p. 509.
Chekhovskoi, V.Ya., Enthalpy, heat capacity, heat and entropy of melting of some refractory metals, in Obzory po teplo izicheskim svoistvam veshchestv (Reviews on the Thermophysical Properties of Substances), Moscow: Akad. Nauk SSSR, 1979, no. 6.
Gurvich, L.V., Pure Appl. Chem., 1989, vol. 61, no. 6, p. 1027.
ACKNOWLEDGMENTS
The authors are grateful to E.L. Osina, L.N. Gorokhov, and J. Arblaster for their valuable discussion and assistance in preparing the work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Zhukova
Rights and permissions
About this article
Cite this article
Lineva, V.I., Sineva, M.A., Morozov, I.V. et al. Thermodynamic Properties of Vanadium in a Condensed State. High Temp 58, 44–49 (2020). https://doi.org/10.1134/S0018151X20010113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0018151X20010113