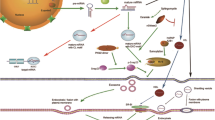
Abstract
The detection of miRNAs in plasma and other body fluids opened up a fascinating possibility that animal noncoding RNAs can act as extracellular signaling molecules. In this review, we discuss recent progress in the field including the ability of miRNAs to participate in intercellular communication in vitro and in vivo, and the application of circulating miRNAs as diagnostic markers of a wide range of diseases. Special attention is paid to the relevance of the development and unification of current techniques for isolation of circulating miRNAs.
Similar content being viewed by others
Abbreviations
- c-miRNAs:
-
circulating microRNAs
- HDL:
-
high-density lipoproteins
- RNP:
-
ribonucleoprotein
References
Pasquinelli, A. E. (2012) MicroRNAs and their targets: recognition, regulation, and an emerging reciprocal relationship, Nat. Rev. Genet., 13, 271–282.
Huntzinger, E., and Izaurralde, E. (2011) Gene silencing by microRNAs: contributions of translational repression and mRNA decay, Nat. Rev. Genet., 12, 99–110.
Friedman, R. C., Farh, K. K., Burge, C. B., and Bartel, D. P. (2009) Most mammalian mRNAs are conserved targets of microRNAs, Genome Res., 19, 92–105.
Palanichamy, J. K., and Rao, D. S. (2014) miRNA dysregulation in cancer: towards a mechanistic understanding, Front. Genet., 5, 54.
Valadi, H., Ekstrom, K., Bossios, A., Sjostrand, M., Lee, J. J., and Lotvall, J. O. (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells, Nat. Cell Biol., 9, 654–659.
Chim, S. S., Shing, T. K., Hung, E. C., Leung, T. Y., Lau, T. K., Chiu, R. W., and Lo, Y. M. (2008) Detection and characterization of placental microRNAs in maternal plasma, Clin. Chem., 54, 482–490.
Mitchell, P. S., Parkin, R. K., Kroh, E. M., Fritz, B. R., Wyman, S. K., Pogosova-Agadjanyan, E. L., Peterson, A., Noteboom, J., O’ Briant, K. C., Allen, A., Lin, D. W., Urban, N., Drescher, C. W., Knudsen, B. S., Stirewalt, D. L., Gentleman, R., Vessella, R. L., Nelson, P. S., Martin, D. B., and Tewari, M. (2008) Circulating microRNAs as stable blood-based markers for cancer detection, Proc. Natl. Acad. Sci. USA, 105, 10513–10518.
Lawrie, C. H., Gal, S., Dunlop, H. M., Pushkaran, B., Liggins, A. P., Pulford, K., Banham, A. H., Pezzella, F., Boultwood, J., Wainscoat, J. S., Hatton, C. S., and Harris, A. L. (2008) Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large Bcell lymphoma, Br. J. Haematol., 141, 672–675.
Kosaka, N., Izumi, H., Sekine, K., and Ochiya, T. (2010) microRNA as a new immune-regulatory agent in breast milk, Silence, 1, 7.
Park, N. J., Zhou, H., Elashoff, D., Henson, B. S., Kastratovic, D. A., Abemayor, E., and Wong, D. T. (2009) Salivary microRNA: discovery, characterization, and clinical utility for oral cancer detection, Clin. Cancer Res., 15, 5473–5477.
Hanke, M., Hoefig, K., Merz, H., Feller, A. C., Kausch, I., Jocham, D., Warnecke, J. M., and Sczakiel, G. (2010) A robust methodology to study urine microRNA as tumor marker: microRNA-126 and microRNA-182 are related to urinary bladder cancer, Urol. Oncol., 28, 655–661.
Wang, C., Yang, C., Chen, X., Yao, B., Zhu, C., Li, L., Wang, J., Li, X., Shao, Y., Liu, Y., Ji, J., Zhang, J., Zen, K., Zhang, C. Y., and Zhang, C. (2011) Altered profile of seminal plasma microRNAs in the molecular diagnosis of male infertility, Clin. Chem., 57, 1722–1731.
Cogswell, J. P., Ward, J., Taylor, I. A., Waters, M., Shi, Y., Cannon, B., Kelnar, K., Kemppainen, J., Brown, D., Chen, C., Prinjha, R. K., Richardson, J. C., Saunders, A. M., Roses, A. D., and Richards, C. A. (2008) Identification of miRNA changes in Alzheimer’s disease brain and CSF yields putative biomarkers and insights into disease pathways, J. Alzheimer’s Dis., 14, 27–41.
Weber, J. A., Baxter, D. H., Zhang, S., Huang, D. Y., Huang, K. H., Lee, M. J., Galas, D. J., and Wang, K. (2010) The microRNA spectrum in 12 body fluids, Clin. Chem., 56, 1733–1741.
Schwarzenbach, H., Nishida, N., Calin, G. A., and Pantel, K. (2014) Clinical relevance of circulating cell-free microRNAs in cancer, Nat. Rev. Clin. Oncol., 11, 145–156.
Sheinerman, K. S., Tsivinsky, V. G., and Umansky, S. R. (2013) Analysis of organ-enriched microRNAs in plasma as an approach to development of universal screening test: feasibility study, J. Transl. Med., 11, 304.
Finnegan, E. F., and Pasquinelli, A. E. (2013) MicroRNA biogenesis: regulating the regulators, Crit. Rev. Biochem. Mol. Biol., 48, 51–68.
Cai, X., Hagedorn, C. H., and Cullen, B. R. (2004) Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs, RNA, 10, 1957–1966.
Lee, Y., Kim, M., Han, J., Yeom, K. H., Lee, S., Baek, S. H., and Kim, V. N. (2004) MicroRNA genes are transcribed by RNA polymerase II, EMBO J., 23, 4051–4060.
Winter, J., Jung, S., Keller, S., Gregory, R. I., and Diederichs, S. (2009) Many roads to maturity: microRNA biogenesis pathways and their regulation, Nat. Cell Biol., 11, 228–234.
Borchert, G. M., Lanier, W., and Davidson, B. L. (2006) RNA polymerase III transcribes human microRNAs, Nat. Struct. Mol. Biol., 13, 1097–1101.
Lee, Y., Ahn, C., Han, J., Choi, H., Kim, J., Yim, J., Lee, J., Provost, P., Radmark, O., Kim, S., and Kim, V. N. (2003) The nuclear RNase III Drosha initiates microRNA processing, Nature, 425, 415–419.
Yi, R., Qin, Y., Macara, I. G., and Cullen, B. R. (2003) Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs, Genes Dev., 17, 3011–3016.
Braun, J. E., Huntzinger, E., and Izaurralde, E. (2013) The role of GW182 proteins in miRNA-mediated gene silencing, Adv. Exp. Med. Biol., 768, 147–163.
Vasudevan, S., Tong, Y., and Steitz, J. A. (2007) Switching from repression to activation: microRNAs can up-regulate translation, Science, 318, 1931–1934.
Kim, D. H., Saetrom, P., Snove, O., Jr., and Rossi, J. J. (2008) MicroRNA-directed transcriptional gene silencing in mammalian cells, Proc. Natl. Acad. Sci. USA, 105, 16230–16235.
Forman, J. J., Legesse-Miller, A., and Coller, H. A. (2008) A search for conserved sequences in coding regions reveals that the let-7 microRNA targets Dicer within its coding sequence, Proc. Natl. Acad. Sci. USA, 105, 14879–14884.
Lee, I., Ajay, S. S., Yook, J. I., Kim, H. S., Hong, S. H., Kim, N. H., Dhanasekaran, S. M., Chinnaiyan, A. M., and Athey, B. D. (2009) New class of microRNA targets containing simultaneous 5′-UTR and 3′-UTR interaction sites, Genome Res., 19, 1175–1183.
Lytle, J. R., Yario, T. A., and Steitz, J. A. (2007) Target mRNAs are repressed as efficiently by microRNA-binding sites in the 5′-UTR as in the 3′-UTR, Proc. Natl. Acad. Sci. USA, 104, 9667–9672.
Bartel, D. P. (2009) MicroRNAs: target recognition and regulatory functions, Cell, 136, 215–233.
Basso, K., Sumazin, P., Morozov, P., Schneider, C., Maute, R. L., Kitagawa, Y., Mandelbaum, J., Haddad, J., Jr., Chen, C. Z., Califano, A., and Dalla-Favera, R. (2009) Identification of the human mature B cell miRNome, Immunity, 30, 744–752.
Larsen, M. T., Hother, C., Hager, M., Pedersen, C. C., Theilgaard-Monch, K., Borregaard, N., and Cowland, J. B. (2013) MicroRNA profiling in human neutrophils during bone marrow granulopoiesis and in vivo exudation, PLoS One, 8, e58454.
Polikepahad, S., and Corry, D. B. (2013) Profiling of Thelper cell-derived small RNAs reveals unique antisense transcripts and differential association of miRNAs with argonaute proteins 1 and 2, Nucleic Acids Res., 41, 1164–1177.
Landgraf, P., Rusu, M., Sheridan, R., Sewer, A., Iovino, N., Aravin, A., Pfeffer, S., Rice, A., Kamphorst, A. O., Landthaler, M., Lin, C., Socci, N. D., Hermida, L., Fulci, V., Chiaretti, S., Foa, R., Schliwka, J., Fuchs, U., Novosel, A., Muller, R. U., Schermer, B., Bissels, U., Inman, J., Phan, Q., Chien, M., Weir, D. B., Choksi, R., De Vita, G., Frezzetti, D., Trompeter, H. I., Hornung, V., Teng, G., Hartmann, G., Palkovits, M., Di Lauro, R., Wernet, P., Macino, G., Rogler, C. E., Nagle, J. W., Ju, J., Papavasiliou, F. N., Benzing, T., Lichter, P., Tam, W., Brownstein, M. J., Bosio, A., Borkhardt, A., Russo, J. J., Sander, C., Zavolan, M., and Tuschl, T. (2007) A mammalian microRNA expression atlas based on small RNA library sequencing, Cell, 129, 1401–1414.
Turchinovich, A., Weiz, L., Langheinz, A., and Burwinkel, B. (2011) Characterization of extracellular circulating microRNA, Nucleic Acids Res., 39, 7223–7233.
Kubota, S., Chiba, M., Watanabe, M., Sakamoto, M., and Watanabe, N. (2015) Secretion of small/microRNAs including miR-638 into extracellular spaces by sphingomyelin phosphodiesterase 3, Oncol. Rep., 33, 67–73.
Gould, S. J., and Raposo, G. (2013) As we wait: coping with an imperfect nomenclature for extracellular vesicles, J. Extracell. Vesicles, 2; doi: 10.3402/jev.v2i0.20389.
Raposo, G., and Stoorvogel, W. (2013) Extracellular vesicles: exosomes, microvesicles, and friends, J. Cell Biol., 200, 373–383.
Colombo, M., Raposo, G., and Thery, C. (2014) Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles, Annu. Rev. Cell Dev. Biol., 30, 255–289.
Mitchell, P., Petfalski, E., Shevchenko, A., Mann, M., and Tollervey, D. (1997) The exosome: a conserved eukaryotic RNA processing complex containing multiple 3′→5′ exoribonucleases, Cell, 91, 457–466.
Akers, J. C., Gonda, D., Kim, R., Carter, B. S., and Chen, C. C. (2013) Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies, J. Neurooncol., 113, 1–11.
Gibbings, D. J., Ciaudo, C., Erhardt, M., and Voinnet, O. (2009) Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity, Nat. Cell Biol., 11, 1143–1149.
Boon, R. A., and Vickers, K. C. (2013) Intercellular transport of microRNAs, Arterioscler. Thromb. Vasc. Biol., 33, 186–192.
Vickers, K. C., Palmisano, B. T., Shoucri, B. M., Shamburek, R. D., and Remaley, A. T. (2011) MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins, Nat. Cell Biol., 13, 423–433.
Wang, K., Zhang, S., Weber, J., Baxter, D., and Galas, D. J. (2010) Export of microRNAs and microRNA-protective protein by mammalian cells, Nucleic Acids Res., 38, 7248–7259.
Arroyo, J. D., Chevillet, J. R., Kroh, E. M., Ruf, I. K., Pritchard, C. C., Gibson, D. F., Mitchell, P. S., Bennett, C. F., Pogosova-Agadjanyan, E. L., Stirewalt, D. L., Tait, J. F., and Tewari, M. (2011) Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma, Proc. Natl. Acad. Sci. USA, 108, 5003–5008.
Kowal, J., Tkach, M., and Thery, C. (2014) Biogenesis and secretion of exosomes, Curr. Opin. Cell Biol., 29, 116–125.
Lee, Y. S., Pressman, S., Andress, A. P., Kim, K., White, J. L., Cassidy, J. J., Li, X., Lubell, K., Lim do, H., Cho, I. S., Nakahara, K., Preall, J. B., Bellare, P., Sontheimer, E. J., and Carthew, R. W. (2009) Silencing by small RNAs is linked to endosomal trafficking, Nat. Cell Biol., 11, 1150–1156.
Trajkovic, K., Hsu, C., Chiantia, S., Rajendran, L., Wenzel, D., Wieland, F., Schwille, P., Brugger, B., and Simons, M. (2008) Ceramide triggers budding of exosome vesicles into multivesicular endosomes, Science, 319, 1244–1247.
Kosaka, N., Iguchi, H., Yoshioka, Y., Takeshita, F., Matsuki, Y., and Ochiya, T. (2010) Secretory mechanisms and intercellular transfer of microRNAs in living cells, J. Biol. Chem., 285, 17442–17452.
Mittelbrunn, M., Gutierrez-Vazquez, C., Villarroya-Beltri, C., Gonzalez, S., Sanchez-Cabo, F., Gonzalez, M. A., Bernad, A., and Sanchez-Madrid, F. (2011) Unidirectional transfer of microRNA-loaded exosomes from T-cells to antigen-presenting cells, Nat. Commun., 2, 282.
Turchinovich, A., and Cho, W. C. (2014) The origin, function and diagnostic potential of extracellular microRNA in human body fluids, Front. Genet., 5, 30.
Laterza, O. F., Lim, L., Garrett-Engele, P. W., Vlasakova, K., Muniappa, N., Tanaka, W. K., Johnson, J. M., Sina, J. F., Fare, T. L., Sistare, F. D., and Glaab, W. E. (2009) Plasma microRNAs as sensitive and specific biomarkers of tissue injury, Clin. Chem., 55, 1977–1983.
Corsten, M. F., Dennert, R., Jochems, S., Kuznetsova, T., Devaux, Y., Hofstra, L., Wagner, D. R., Staessen, J. A., Heymans, S., and Schroen, B. (2010) Circulating microRNA-208b and microRNA-499 reflect myocardial damage in cardiovascular disease, Circ. Cardiovasc. Genet., 3, 499–506.
Zhang, Y., Jia, Y., Zheng, R., Guo, Y., Wang, Y., Guo, H., Fei, M., and Sun, S. (2010) Plasma microRNA-122 as a biomarker for viral-, alcohol-, and chemical-related hepatic diseases, Clin. Chem., 56, 1830–1838.
Akat, K. M., Moore-McGriff, D., Morozov, P., Brown, M., Gogakos, T., Correa Da Rosa, J., Mihailovic, A., Sauer, M., Ji, R., Ramarathnam, A., Totary-Jain, H., Williams, Z., Tuschl, T., and Schulze, P. C. (2014) Comparative RNA-sequencing analysis of myocardial and circulating small RNAs in human heart failure and their utility as biomarkers, Proc. Natl. Acad. Sci. USA, 111, 11151–11156.
Turchinovich, A., Weiz, L., and Burwinkel, B. (2012) Extracellular miRNAs: the mystery of their origin and function, Trends Biochem. Sci., 37, 460–465.
Banzet, S., Chennaoui, M., Girard, O., Racinais, S., Drogou, C., Chalabi, H., and Koulmann, N. (2013) Changes in circulating microRNAs levels with exercise modality, J. Appl. Physiol. (1985), 115, 1237–1244.
Uhlemann, M., Mobius-Winkler, S., Fikenzer, S., Adam, J., Redlich, M., Mohlenkamp, S., Hilberg, T., Schuler, G. C., and Adams, V. (2012) Circulating microRNA-126 increases after different forms of endurance exercise in healthy adults, Eur. J. Prev. Cardiol., 21, 484–491.
Hannafon, B. N., and Ding, W. Q. (2013) Intercellular communication by exosome-derived microRNAs in cancer, Int. J. Mol. Sci., 14, 14240–14269.
Turchinovich, A., Samatov, T. R., Tonevitsky, A. G., and Burwinkel, B. (2013) Circulating miRNAs: cell–cell communication function? Front. Genet., 4, 119.
Salido-Guadarrama, I., Romero-Cordoba, S., Peralta Zaragoza, O., Hidalgo-Miranda, A., and Rodriguez-Dorantes, M. (2014) MicroRNAs transported by exosomes in body fluids as mediators of intercellular communication in cancer, Onco Targets Ther., 7, 1327–1338.
Saleem, S. N., and Abdel-Mageed, A. B. (2014) Tumorderived exosomes in oncogenic reprogramming and cancer progression, Cell. Mol. Life Sci., 72, 1–10.
Tabet, F., Vickers, K. C., Cuesta Torres, L. F., Wiese, C. B., Shoucri, B. M., Lambert, G., Catherinet, C., Prado-Lourenco, L., Levin, M. G., Thacker, S., Sethupathy, P., Barter, P. J., Remaley, A. T., and Rye, K. A. (2014) HDLtransferred microRNA-223 regulates ICAM-1 expression in endothelial cells, Nat. Commun., 5, 3292.
Chevillet, J. R., Kang, Q., Ruf, I. K., Briggs, H. A., Vojtech, L. N., Hughes, S. M., Cheng, H. H., Arroyo, J. D., Meredith, E. K., Gallichotte, E. N., Pogosova Agadjanyan, E. L., Morrissey, C., Stirewalt, D. L., Hladik, F., Yu, E. Y., Higano, C. S., and Tewari, M. (2014) Quantitative and stoichiometric analysis of the microRNA content of exosomes, Proc. Natl. Acad. Sci. USA, 111, 14888–14893.
Kogure, T., Lin, W. L., Yan, I. K., Braconi, C., and Patel, T. (2011) Intercellular nanovesicle-mediated microRNA transfer: a mechanism of environmental modulation of hepatocellular cancer cell growth, Hepatology, 54, 1237–1248.
Nolte-’t Hoen, E. N., Buermans, H. P., Waasdorp, M., Stoorvogel, W., Wauben, M. H., and’ t Hoen, P. A. (2012) Deep sequencing of RNA from immune cell-derived vesicles uncovers the selective incorporation of small non-coding RNA biotypes with potential regulatory functions, Nucleic Acids Res., 40, 9272–9285.
Li, C. C., Eaton, S. A., Young, P. E., Lee, M., Shuttleworth, R., Humphreys, D. T., Grau, G. E., Combes, V., Bebawy, M., Gong, J., Brammah, S., Buckland, M. E., and Suter, C. M. (2013) Glioma microvesicles carry selectively packaged coding and noncoding RNAs which alter gene expression in recipient cells, RNA Biol., 10, 1333–1344.
Guduric-Fuchs, J., O’Connor, A., Camp, B., O’ Neill, C. L., Medina, R. J., and Simpson, D. A. (2012) Selective extracellular vesicle-mediated export of an overlapping set of microRNAs from multiple cell types, BMC Genomics, 13, 357.
Koppers-Lalic, D., Hackenberg, M., Bijnsdorp, I. V., van Eijndhoven, M. A., Sadek, P., Sie, D., Zini, N., Middeldorp, J. M., Ylstra, B., de Menezes, R. X., Wurdinger, T., Meijer, G. A., and Pegtel, D. M. (2014) Nontemplated nucleotide additions distinguish the small RNA composition in cells from exosomes, Cell Rep., 8, 1649–1658.
Ji, H., Chen, M., Greening, D. W., He, W., Rai, A., Zhang, W., and Simpson, R. J. (2014) Deep sequencing of RNA from three different extracellular vesicle (EV) subtypes released from the human LIM1863 colon cancer cell line uncovers distinct miRNA-enrichment signatures, PLoS One, 9, e110314.
Villarroya-Beltri, C., Gutierrez-Vazquez, C., Sanchez-Cabo, F., Perez-Hernandez, D., Vazquez, J., Martin Cofreces, N., Martinez-Herrera, D. J., Pascual-Montano, A., Mittelbrunn, M., and Sanchez-Madrid, F. (2013) Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs, Nat. Commun., 4, 2980.
Hergenreider, E., Heydt, S., Treguer, K., Boettger, T., Horrevoets, A. J., Zeiher, A. M., Scheffer, M. P., Frangakis, A. S., Yin, X., Mayr, M., Braun, T., Urbich, C., Boon, R. A., and Dimmeler, S. (2012) Atheroprotective communication between endothelial cells and smooth muscle cells through miRNAs, Nat. Cell Biol., 14, 249–256.
Le, M. T., Hamar, P., Guo, C., Basar, E., Perdigao Henriques, R., Balaj, L., and Lieberman, J. (2014) miR200-containing extracellular vesicles promote breast cancer cell metastasis, J. Clin. Invest., 124, 5109–5128.
Fabbri, M., Paone, A., Calore, F., Galli, R., Gaudio, E., Santhanam, R., Lovat, F., Fadda, P., Mao, C., Nuovo, G. J., Zanesi, N., Crawford, M., Ozer, G. H., Wernicke, D., Alder, H., Caligiuri, M. A., Nana-Sinkam, P., Perrotti, D., and Croce, C. M. (2012) MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response, Proc. Natl. Acad. Sci. USA, 109, 2110–2116.
Lehmann, S. M., Kruger, C., Park, B., Derkow, K., Rosenberger, K., Baumgart, J., Trimbuch, T., Eom, G., Hinz, M., Kaul, D., Habbel, P., Kalin, R., Franzoni, E., Rybak, A., Nguyen, D., Veh, R., Ninnemann, O., Peters, O., Nitsch, R., Heppner, F. L., Golenbock, D., Schott, E., Ploegh, H. L., Wulczyn, F. G., and Lehnardt, S. (2012) An unconventional role for miRNA: let-7 activates Toll-like receptor 7 and causes neurodegeneration, Nat. Neurosci., 15, 827–835.
Saikumar, J., Ramachandran, K., and Vaidya, V. S. (2014) Noninvasive micromarkers, Clin. Chem., 60, 1158–1173.
Lagana, A., Russo, F., Veneziano, D., Bella, S. D., Giugno, R., Pulvirenti, A., Croce, C. M., and Ferro, A. (2013) Extracellular circulating viral microRNAs: current knowledge and perspectives, Front. Genet., 4, 120.
Makarova, J. A., Maltseva, D. V., Galatenko, V. V., Abbasi, A., Maximenko, D. G., Grigoriev, A. I., Tonevitsky, A. G., and Northoff, H. (2014) Exercise immunology meets miRNAs, Exerc. Immunol. Rev., 20, 135–164.
Hoy, A. M., Lundie, R. J., Ivens, A., Quintana, J. F., Nausch, N., Forster, T., Jones, F., Kabatereine, N. B., Dunne, D. W., Mutapi, F., Macdonald, A. S., and Buck, A. H. (2014) Parasite-derived microRNAs in host serum as novel biomarkers of helminth infection, PLoS Negl. Trop. Dis., 8, e2701.
Sato-Kuwabara, Y., Melo, S. A., Soares, F. A., and Calin, G. A. (2015) The fusion of two worlds: non-coding RNAs and extracellular vesicles — diagnostic and therapeutic implications (review), Int. J. Oncol., 46, 17–27.
Eichelser, C., Stuckrath, I., Muller, V., Milde-Langosch, K., Wikman, H., Pantel, K., and Schwarzenbach, H. (2014) Increased serum levels of circulating exosomal microRNA-373 in receptor-negative breast cancer patients, Oncotarget, 5, 9650–9663.
Bala, S., Petrasek, J., Mundkur, S., Catalano, D., Levin, I., Ward, J., Alao, H., Kodys, K., and Szabo, G. (2012) Circulating microRNAs in exosomes indicate hepatocyte injury and inflammation in alcoholic, drug-induced, and inflammatory liver diseases, Hepatology, 56, 1946–1957.
Taguchi, Y. H., and Murakami, Y. (2014) Universal disease biomarker: can a fixed set of blood microRNAs diagnose multiple diseases? BMC Res. Notes, 7, 581.
Taguchi, Y. H., and Murakami, Y. (2013) Principal component analysis based feature extraction approach to identify circulating microRNA biomarkers, PLoS One, 8, e66714.
Keller, A., Leidinger, P., Vogel, B., Backes, C., El Sharawy, A., Galata, V., Muller, S. C., Marquart, S., Schrauder, M. G., Strick, R., Bauer, A., Wischhusen, J., Beier, M., Kohlhaas, J., Katus, H. A., Hoheisel, J., Franke, A., Meder, B., and Meese, E. (2014) miRNAs can be generally associated with human pathologies as exemplified for miR-144, BMC Med., 12, 224.
Ple, H., Landry, P., Benham, A., Coarfa, C., Gunaratne, P. H., and Provost, P. (2012) The repertoire and features of human platelet microRNAs, PLoS One, 7, e50746.
Stakos, D. A., Gatsiou, A., Stamatelopoulos, K., Tselepis, A. D., and Stellos, K. (2013) Platelet microRNAs: from platelet biology to possible disease biomarkers and therapeutic targets, Platelets, 24, 579–589.
Cheng, H. H., Yi, H. S., Kim, Y., Kroh, E. M., Chien, J. W., Eaton, K. D., Goodman, M. T., Tait, J. F., Tewari, M., and Pritchard, C. C. (2013) Plasma processing conditions substantially influence circulating microRNA biomarker levels, PLoS One, 8, e64795.
Van Deun, J., Mestdagh, P., Sormunen, R., Cocquyt, V., Vermaelen, K., Vandesompele, J., Bracke, M., De Wever, O., and Hendrix, A. (2014) The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling, J. Extracell. Vesicles, 18, 3.
Monleau, M., Bonnel, S., Gostan, T., Blanchard, D., Courgnaud, V., and Lecellier, C. H. (2014) Comparison of different extraction techniques to profile microRNAs from human sera and peripheral blood mononuclear cells, BMC Genomics, 15, 395.
Kim, D. J., Linnstaedt, S., Palma, J., Park, J. C., Ntrivalas, E., Kwak-Kim, J. Y., Gilman-Sachs, A., Beaman, K., Hastings, M. L., Martin, J. N., and Duelli, D. M. (2012) Plasma components affect accuracy of circulating cancerrelated microRNA quantitation, J. Mol. Diagn., 14, 71–80.
Page, K., Guttery, D. S., Zahra, N., Primrose, L., Elshaw, S. R., Pringle, J. H., Blighe, K., Marchese, S. D., Hills, A., Woodley, L., Stebbing, J., Coombes, R. C., and Shaw, J. A. (2013) Influence of plasma processing on recovery and analysis of circulating nucleic acids, PLoS One, 8, e77963.
Koberle, V., Pleli, T., Schmithals, C., Augusto Alonso, E., Haupenthal, J., Bonig, H., Peveling-Oberhag, J., Biondi, R. M., Zeuzem, S., Kronenberger, B., Waidmann, O., and Piiper, A. (2013) Differential stability of cell-free circulating microRNAs: implications for their utilization as biomarkers, PLoS One, 8, e75184.
Becker, N., and Lockwood, C. M. (2013) Pre-analytical variables in miRNA analysis, Clin. Biochem., 46, 861–868.
Pritchard, C. C., Kroh, E., Wood, B., Arroyo, J. D., Dougherty, K. J., Miyaji, M. M., Tait, J. F., and Tewari, M. (2012) Blood cell origin of circulating microRNAs: a cautionary note for cancer biomarker studies, Cancer Prev. Res. (Philadelphia), 5, 492–497.
Adams, S. V., Newcomb, P. A., Burnett-Hartman, A. N., Wurscher, M. A., Mandelson, M., Upton, M. P., Zhu, L. C., Potter, J. D., and Makar, K. W. (2014) Rare circulating microRNAs as biomarkers of colorectal neoplasia, PLoS One, 9, e108668.
Wang, K., Yuan, Y., Cho, J. H., Mc Clarty, S., Baxter, D., and Galas, D. J. (2012) Comparing the microRNA spectrum between serum and plasma, PLoS One, 7, e41561.
Huang, Z., Huang, D., Ni, S., Peng, Z., Sheng, W., and Du, X. (2010) Plasma microRNAs are promising novel biomarkers for early detection of colorectal cancer, Int. J. Cancer, 127, 118–126.
Giraldez, M. D., Lozano, J. J., Ramirez, G., Hijona, E., Bujanda, L., Castells, A., and Gironella, M. (2013) Circulating microRNAs as biomarkers of colorectal cancer: results from a genome-wide profiling and validation study, Clin. Gastroenterol. Hepatol., 11, 681–688.
Kanaan, Z., Roberts, H., Eichenberger, M. R., Billeter, A., Ocheretner, G., Pan, J., Rai, S. N., Jorden, J., Williford, A., and Galandiuk, S. (2013) A plasma microRNA panel for detection of colorectal adenomas: a step toward more precise screening for colorectal cancer, Ann. Surg., 258, 400–408.
Wang, Q., Huang, Z., Ni, S., Xiao, X., Xu, Q., Wang, L., Huang, D., Tan, C., Sheng, W., and Du, X. (2012) Plasma miR-601 and miR-760 are novel biomarkers for the early detection of colorectal cancer, PLoS One, 7, e44398.
Toiyama, Y., Takahashi, M., Hur, K., Nagasaka, T., Tanaka, K., Inoue, Y., Kusunoki, M., Boland, C. R., and Goel, A. (2013) Serum miR-21 as a diagnostic and prognostic biomarker in colorectal cancer, J. Natl. Cancer Inst., 105, 849–859.
Farina, N. H., Wood, M. E., Perrapato, S. D., Francklyn, C. S., Stein, G. S., Stein, J. L., and Lian, J. B. (2014) Standardizing analysis of circulating microRNA: clinical and biological relevance, J. Cell. Biochem., 115, 805–811.
Moret, I., Sanchez-Izquierdo, D., Iborra, M., Tortosa, L., Navarro-Puche, A., Nos, P., Cervera, J., and Beltran, B. (2013) Assessing an improved protocol for plasma microRNA extraction, PLoS One, 8, e82753.
Witwer, K. W., Buzas, E. I., Bemis, L. T., Bora, A., Lasser, C., Lotvall, J., Nolte-’t Hoen, E. N., Piper, M. G., Sivaraman, S., Skog, J., Thery, C., Wauben, M. H., and Hochberg, F. (2013) Standardization of sample collection, isolation, and analysis methods in extracellular vesicle research, J. Extracell. Vesicles, 2.
Kanwar, S. S., Dunlay, C. J., Simeone, D. M., and Nagrath, S. (2014) Microfluidic device (ExoChip) for onchip isolation, quantification, and characterization of circulating exosomes, Lab. Chip, 14, 1891–1900.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2015, Vol. 80, No. 9, pp. 1344–1355.
Rights and permissions
About this article
Cite this article
Makarova, J.A., Shkurnikov, M.U., Turchinovich, A.A. et al. Circulating microRNAs. Biochemistry Moscow 80, 1117–1126 (2015). https://doi.org/10.1134/S0006297915090035
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297915090035