Abstract
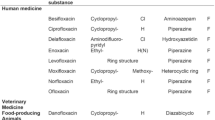
A method for preparing antibodies that specifically recognize antibiotics of the fluoroquinolone group with the same radical in the first position of the quinolone nucleus is proposed. The specificity of rabbit antisera prepared at different cycles of immunization by changing the structure of the hapten in the composition of immunogens was characterized. The selected antibodies provided a group-specific analysis of 16 representatives of fluoroquinolones, including a combination of the following compounds that are controlled in animal products: ciprofloxacin, norfloxacin, pefloxacin, ofloxacin, and enrofloxacin. Using these antibodies, an indirect competitive enzyme-linked immunosorbent assay was developed that was characterized by a detection limit of ciprofloxacin of 0.2 ng/mL and a duration of 2 h. The assay was approbated for the detection of fluoroquinolones in milk.




Similar content being viewed by others
REFERENCES
Ezelarab, H.A.A., Abbas, S.H., Hassan, H.A., and Abuo-Rahma, G.E.A., Arch. Pharm. (Weinheim), 2018, vol. 351, no. 9. e1800141.
Blondeau, J.M., Surv. Ophthalmol., 2004, vol. 49, no. 2, pp. 73–S78.
Gouvea, R., Dos, S.F.F., Aquino, M.H.C.D., and Pereira, V.L.D., Bras. J. Poultry Sci., 2015, vol. 17, no. 1, pp. 1–10.
Brown, S.A., J. Vet. Pharm. Therapeutics, 1996, vol. 19, no. 1, pp. 1–14.
Stahlmann, R. and Lode, H.M., Expert Opin. Drug Saf., 2013, vol. 12, no. 4, pp. 497–505.
Council Regulation (EEC) No 2377/90 of 26 June 1990 laying down a Community procedure for the establishment of maximum residue limits of veterinary medicinal products in foodstuffs of animal origin.
Uniform sanitary-epidemiological and hygienic requirements for goods subject to sanitary-epidemiological supervision (control). Approved by the Decision of the Commission of the Customs Union dated May 28, 2010, no. 299.
De, A.K., Bera, A.K., and Pal, B., Int. J. Pharm. Sci. Res., vol. 7, no. 2, pp. 531–542.
Sousa, J., Alves, G., Fortuna, A., and Falcao, A., Anal. Bioanal. Chem., 2012, vol. 403, no. 1, pp. 93–129.
Lehotay, S.J. and Chen, Y.B., Anal. Bioanal. Chem., 2018, vol. 410, no. 22, pp. 5331–5351.
Xu, F., Ren, K., Yang, Y.Z., Guo, J.P., Ma, G.P., Liu, Y.M., Lu, Y.Q., and Li, X.B., J. Integr. Agricult., 2015, vol. 14, no. 11, pp. 2282–2295.
Duffy, G.F. and Moore, E.J., Anal. Lett., 2017, vol. 50, no. 1, pp. 1–32.
Gaudin, V., Biosens. Bioelectron., 2017, vol. 90, pp. 363–377.
Dzantiev, B.B., Byzova, N.A., Urusov, A.E., and Zherdev, A.V., TrAC, Trends Anal. Chem., 2014, vol. 55, pp. 81–93.
Tittlemier, S., Gelinas, J.-M., Dufresne, G., Haria, M., Querry, J., Cleroux, C., and Godefroy, S.B., Food Anal. Methods, 2008, vol. 1, no. 1, pp. 28–35.
Huet, A.-C., Charelier, C., Tittlemier, S.A., Singh, G., Benrejeb, S., and Delahaut, P., J. Agric. Food Chem., 2006, vol. 54, no. 8, pp. 2822–2827.
Liu, Y.Z., Zhao, G.X., Wang, P., Liu, J., Zhang, H.C., and Wang, J.P., J. Environ. Sci. Health, 2013, vol. 48, no. 2, pp. 139–146.
Li, Y., Ji, B., Chen, W., Liu, L., Xu, C., Peng, C., and Wang, L., Food Agric. Immunol., 2008, vol. 19, no. 4, pp. 251–264.
Wang, Z., Zhu, Y., Ding, S., He, F., Beier, R.C., Li, J., and Shen, J., Anal. Chem., 2007, vol. 79, no. 12, pp. 4471–4483.
Bucknall, S., Silverlight, J., Coldham, N., Thorne, L., and Jackman, R., Food Additives Contam., 2003, vol. 3, no. 3, pp. 221–228.
Burkin, M.A., Food Agric. Immunol., 2008, vol. 19, no. 2, pp. 131–140.
Wen, K., Nolke, G., Schillberg, S., Wang, Z., Zhang, S., Wu, C., and Shen, J., Anal. Bioanal. Chem., 2012, vol. 403, no. 9, pp. 2771–2783.
Lu, S., Zhang, Y., Liu, J., Zhao, C., Liu, W., and Xi, R., J. Agric. Food Chem., 2006, vol. 54, no. 19, pp. 6995–7000.
Chu, F.S., Lau, H.P., Fan, T.S., and Zhang, G.S., J. Immunol. Methods, 1982, vol. 55, no. 3, pp. 73–78.
Liu, Z.Q., Lu, S.X., Zhao, C.H., Ding, K., Cao, Z.Z., Zhan, J.H., Ma, C., Liu, J.T., and Xi, R.M., J. Sci. Food Agric., 2009, vol. 89, no. 7, pp. 1115–1121.
Funding
The work was performed with partial financial support from the Russian Foundation for Basic Research (Grant no. 18-58-00038) and the Belarusian Republican Foundation for Fundamental Research (Grant no. H18P-060).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Shanin, I.A., Zvereva, E.A., Eremin, S.A. et al. Development of an Immunoenzyme Assay to Control the Total Content of Antibiotics of the Fluoroquinolone Group in Milk. Appl Biochem Microbiol 55, 563–569 (2019). https://doi.org/10.1134/S0003683819050132
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683819050132