Abstract
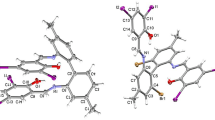
The complexes of 3d-metal 5-sulfosalicylates with phenylacethydrazide are synthesized: [Co(L)3]HSSal · 3H2O (I), [Ni(L)3]HSSal · 3H2O (II), and [Zn(L)3]HSSal · 6H2O (III) (L is phenylacethydrazide, and HSSal2– is 5-sulfosalicylic acid anion). Compounds I–III are characterized by the methods of chemical analysis, IR spectroscopy, diffuse reflectance spectroscopy, luminescence, and thermogravimetry. Compound II is studied by X-ray diffraction analysis (СIF file CCDC no. 1819696). In the complex cation [Ni(L)3]2+, the nickel atom has a face octahedral coordination mode by three O atoms and three N atoms of three bidentate chelate ligands L. The complex cations, HSSal2– anions, and crystallization water molecules are joined by a branched system of hydrogen bonds. Complexes I–III are luminescent, and complex III exhibits the brightest luminescence.



Similar content being viewed by others
REFERENCES
Arjunan, V., Rani, T., Mythili, C.V., and Mohan, S., Spectrochim. Acta, Part A, 2011, vol. 79, no. 3, p. 486.
Ul Ain, Q., Ashiq, U., Ara, J.R., and Mahrooof-Tahir, M., Spectrochim. Acta, Part A, 2013, vol. 115, p. 683.
Fan, S.-R. and Zhu, L.-G., J. Mol. Struct., 2007, vol. 827, nos. 1–3, p. 188.
Mistri, S., Zangrando, E., Farnetti, E., et al., Polyhedron, 2015, vol. 89, p. 250.
Yan, Ch.-F., Jiang, F.-L., Chen, L., et al, J. Solid State Chem., 2009, vol. 182, no. 11, p. 3162.
Lamshöft, M., Storp, J., Ivanova, B., et al., Polyhedron, 2011, vol. 30, no. 15, p. 2564.
Koksharova, T.V., Sergienko, V.S., Surazhskaya, M.D., et al., Russ. J. Inorg. Chem., 2017, vol. 62, no. 12, p. 1568. doi 10.1134/S0036023617120105
Xia, M. and Wei, C.-L., J. Struct. Chem., 2013, vol. 54, no. 1, p. 110.
Gao, Sh., Huo, L.-H., Zhao, H., et al., Acta Crystallogr., Sect E: Struct. Rep. Online, 2005, vol. 61, no. 2, p. m290.
Han, Q., Wang, X.-Ch., Li, X.-Y., et al., Acta Crystallogr., Sect E: Struct. Rep. Online, 2009, vol. 65, no. 11, p. m1282.
Yenikaya, C., Sari, M., Ilkimen, H., et al., Polyhedron, 2011, vol. 30, no. 3, p. 535.
Chen, Zh.-F., Shi, Sh.-M., Hu, R.-X., et al., Chin. J. Chem., 2003, vol. 21, no. 8, p. 1059.
Sun, Y.-Q., Fan, L.-L., Gao, D.-Zh., et al., Dalton Trans., 2010, vol. 39, no. 40, p. 9654.
Antsyshkina, A.S., Koksharova, T.V., Sergienko, V.S., et al., Russ. J. Inorg. Chem., 2014, vol. 59, no. 12, p. 1417. doi 10.1134/S0036023614120031
Lagorio, M.G., Dicelio, L.E., Litter, M.I., et al., J. Chem. Soc., Faraday Trans., 1998, vol. 94, no. 3, p. 419.
Bril, A. and de Jager-Veenis, A.W., J. Electrochem. Soc., 1976, vol. 123, no. 3, p. 396.
Nazarov, M., Noh, D.Y., Sohn, J., et al., J. Solid State Chem., 2007, vol. 180, no. 9, p. 2493.
APEX2, SAINT, SADABS, Madison: Bruker AXS Inc., 2008–2009.
Altomare, A., Gascsrano, G., Giacovazzo, C., and Guagliard, A., J. Appl. Cryst. A, 1993, vol. 26, p. 343.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Odunola, O.A., Adeoye, I.O., and Woods, J.A.O., Synth. React. Inorg. Metal-Org. Chem., 2002, vol. 32, no. 4, p. 801.
Jensen, J.B., Acta Chem. Scand., 1956, vol. 10, no. 4, p. 667.
Gordon, A. and Ford, R., The Chemist’s Companion: A Handbook of Practical Data, Techniques, and References, New York: Wiley, 1972.
Ul Ain, Q., Ashiq, U., Ara, J.R., et al., Arab. J. Chem., 2017, vol. 1, no. 4, p. 488.
Issa, R.M., El-Shazly, M.F., and Iskander, M.F., Z. Anorg. Allg. Chem., 1967, vol. 354, nos. 1−2, p. 90.
Zidan, A.S.A., Synth. React. Inorg. Metal-Org. Chem., 2004, vol. 34, no. 4, p. 743.
Gogorishvili, P.V., Karkarashvili, M.V., and Kalandarishvili, D.Z., Zh. Neorg. Khim., 1969, vol. 14, no. 6, p. 1516.
Dutta, A.A. and Chaudhuri, N.R., J. Inorg. Nucl. Chem., 1971, vol. 33, no. 1, p. 189.
Odunola, O.A., Adeoye, I.O., Woods, J.A.O., et al., Synth. React. Inorg Metal-Org. Chem., 2003, vol. 33, no. 2, p. 205.
Narang, K.K. and Singh, M., Synth. React. Inorg. Metal-Org. Chem., 1985, vol. 15, no. 6, p. 821.
Narang, K.K., Pandey, J.P., Singh, K.P., et al., Synth. React. Inorg. Metal-Org. Chem., 1990, vol. 20, no. 10, p. 1301.
Fan, S.-R. and Zhu, L.-G., Chin. J. Chem., 2005, vol. 23, no. 10, p. 1292.
Varghese, T.H., Panicker, Y.C., and Philip, D., J. Raman Spectrosc., 2007, vol. 38, no. 3, p. 309.
Nakanishi, K. Infrared Spectra Absorption Spectroscopy, Tokyo: Nankodo Company, 1962.
Lever, A., Inorganic Electronic Spectroscopy, New York: Elesvier, 1984, vol. 2.
Fabbrizzi, L., Licchelli, M., Pallavicini, P., et al., Analyst, 1996, vol. 121, p. 1763.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Koksharova, T.V., Sergienko, V.S., Surazhskaya, M.D. et al. Syntheses and Characterization of Coordination Compounds of 3d-Metal 5-Sulfosalicylates with Phenylacethydrazide: Crystal Structure of [Ni(L)3]HSSal · 3H2O (L is Phenylacethydrazide, and HSSal2– is 5-Sulfosalicylic Acid Anion). Russ J Coord Chem 44, 678–687 (2018). https://doi.org/10.1134/S1070328418110040
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328418110040