Abstract
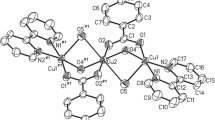
The molecular and crystal structures of complexes [Cu2(HL)2(CH3OH)2](BF4)2 ∙ C6H3Cl3 (I), [Cu2(HL)2(H2O)2](1,4-O2CС6Н4CO2) (II), and [Cu2(HL)2(H2O)2](4,4’-O2CС6Н4С6Н4CO2) (III) (H2L is 4,4’-[2-(3-hydroxyiminobutyl)imino]biphenyl, C6H3Cl3 is 1,2,4-trichlorobenzene) are determined by X-ray diffraction analyses. The crystalline lattices of complexes I and II contain discrete binuclear cations Cu2(HL) 2+2 , whereas the crystalline lattice of compound III contains both discrete cations Cu2(HL) 2+2 and polymer chains [Cu2(HL) 2+2 ]n. In compounds I–III, the crystalline lattice units are joined by hydrogen bonds. The analysis of the temperature dependence of the magnetic susceptibility shows that the magnetic properties of compound III are determined only by antiferromagnetic interactions of the Cu2+ ions within the cations Cu2(HL) 2+2
Similar content being viewed by others
References
Li, J.-R., Sculley, J., and Zhou, H.-C., Chem. Rev., 2012, vol. 112, p. 869.
Corma, A., García, H., Llabrés, I., and Xamena, F.X., Chem. Rev., 2010, vol. 110, p. 4606.
Li, H.-Y., Wei, Y.-L., Dong, X.-Y., et al., Chem. Mater., 2015, vol. 27, p. 1327.
Stavila, V., Talin, A.A., and Allendorf, M.D., Chem. Soc. Rev., 2014, vol. 43, p. 5994.
Wriedt, M., Yakovenko, A.A., Halder, G.J., et al., J. Am. Chem. Soc., 2013, vol. 135, p. 4040.
Sotnik, S.A., Polunin, R.A., Kiskin, M.A., et al., Inorg. Chem., 2015, vol. 54, p. 5169.
Schoedel, A., Wojtas, L., Kelley, S.P., et al., Angew. Chem., Int. Ed. Engl., 2011, vol. 50, p. 11421.
Beauvais, L.G., Shores, M.P., and Long, J.R., J. Am. Chem. Soc., 2000, vol. 122, p. 2763.
Kolotilov, S.V., Cador, O., Pointillart, F., et al., J. Mater. Chem., 2010, vol. 20, p. 9505.
Zhang, Q.-F., Niu, Y., Leung, W.-H., et al., Chem. Commun. (Cambridge, UK), 2001, p. 1126.
Dorofeeva, V.N., Kolotilov, S.V., Kiskin, M.A., et al., Chem.-Eur. J., 2012, vol. 18, p. 5006.
Polunin, R.A., Kolotilov, S.V., Kiskin, M.A., et al., Izv. Akad. Nauk, Ser. Khim., 2010, vol. 59, p. 1192.
Nakata, K., Miyasaka, H., Sugimoto, K., et al., Chem. Lett., 2002, p. 658.
Haldar, R., Sikdar, N., and Maji, T.K., Mater. Today, 2015, vol. 18, p. 97.
Kolotilov, S.V., Cador, O., Gavrilenko, K.S., et al., Eur. J. Inorg. Chem., 2010, p. 1255.
Mikhaleva, E.A., Kolotilov, S.V., Gavrilenko, K.S., et al., Teor. Eksp. Khim., 2008, vol. 44, p. 233.
Huang, F.-P., Yang, C., Li, H.-Y., et al., Dalton Trans., 2015, vol. 44, p. 6593.
Lan, Y.-Q., Jiang, H.-L., Li, S.-L., et al., Inorg. Chem., 2012, vol. 51, p. 7484.
Sheldrick G.M., SHELXS-97 and SHELXL-97, Göttingen Univ. of Göttingen, 1997.
Drabent, K., Ciunik, Z., and Chmielewski, P.J., Eur. J. Inorg. Chem., 2003, p. 1548.
Akitsu, T. and Einaga, Y., Acta Crystallogr., Sect. E: Struct. Rep. Online, 2004, vol. 60, p. m436.
Toganoh, M., Kawabe, Y., Uno, H., et al., Angew. Chem., Int. Ed. Engl., 2012, vol. 51, p. 8753.
Rancan, M., Tessarolo, J., Casarin, M., et al., Inorg. Chem., 2014, vol. 53, p. 7276.
Sinn, E., Coord. Chem. Rev., 1970, vol. 5, p. 313.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © R.A. Polunin, K.S. Gavrilenko, M.A. Kiskin, I.L. Eremenko, V.M. Novotortsev, S.V. Kolotilov, 2016, published in Koordinatsionnaya Khimiya, 2016, Vol. 42, No. 8, pp. 451–458.
Rights and permissions
About this article
Cite this article
Polunin, R.A., Gavrilenko, K.S., Kiskin, M.A. et al. Effect of the counterion and guest molecules on the crystal structures of the coordination compounds with the Cu2(HL) 2+2 cation (H2L = 4,4’-[2-(3-hydroxyiminobutyl)imino]biphenyl): Syntheses, structures, and magnetic properties. Russ J Coord Chem 42, 487–493 (2016). https://doi.org/10.1134/S1070328416080054
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328416080054



