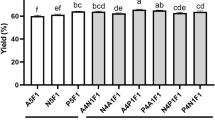
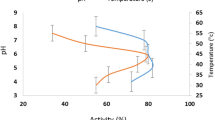
Abstract
Wheat gluten is a group of water-insoluble proteins of wheat endosperm. As a co-product of the starch and bioethanol production, gluten is available in fairly large quantities. Therefore, the search for optimal ways of its industrial utilization is an important economic task. The food industry is a traditional area for gluten application. However, the study of its structural and functional properties allowed expanding the area of the gluten use due to its various modifications, in particular, enzymatic hydrolysis. As compared to native gluten, the gluten hydrolysates have better foaming and emulsifying abilities, as well as higher water solubility. Biologically active properties of hydrolysates (antioxidant, hepatoprotective and angiotensin-converting enzyme inhibiting) were also shown. The search for and selection of proteolytic enzymes or their combinations, and optimization of the technological process will permit to obtain the hydrolysis products with the required characteristics, expand their use in industry and reduce the cost of their production.



Similar content being viewed by others
Notes
Alkalase is a Bacillus licheniformis serine protease.
Pronase is a mixture of proteinases isolated from culture liquid of Streptomyces griseus that consists mainly of serine proteases.
REFERENCES
Balashov, A.I., Production Management (Production Organization) at the Enterprise, St. Petersburg: Peter, 2009, pp. 33–35.
Deep Grain Processing and Biofuel Production. Investment Memorandum of the Project. Developed on the basis of marketing research of the market of raw materials and products of deep processing of grain and biofuel production, LLC Navigate Consulting, 2015, pp. 4–16.
Day, L., Batey, I.L., Wrigley, C.W., and Augustin, M.A., Gluten Uses and Food Industry Needs. Value Added Wheat CRC Project Report, 2004, pp. 1–38.
Lu, B., Evaluation of Vital wheat gluten as a source of protein in extruded diets for juvenile Giant croaker (Nibea japonica): feed technological properties and biological responses, Master Thesis, Norway: Norwegian University of Life Sciences, 2015.
Wieser, H., Chemistry of gluten proteins, Food Microbiol., 2007, pp. 24, 115.
Shewry, P.R., Halford, N.G., Belton, P.S., and Tatham, A.S., The structure and properties of gluten: an elastic protein from wheat grain, Philos. Trans. R. Soc. Lond. B. Biol. Sci., 2002, vol. 357, no. 1418, pp. 133–142. doi 10.1098/rstb.2001.1024
Wang, P., Jin, Z., and Xu, X., Physicochemical alterations of wheat gluten proteins upon dough formation and frozen storage—a review from gluten, glutenin and gliadin perspectives, Trends Food Sci. Technol., 2015, pp. 1–10. doi 10.1016/j.tifs.2015.10.005
Balakireva, A.V. and Zamyatnin, A.A., Jr., Properties of gluten intolerance: gluten structure, evolution, pathogenicity and detoxification capabilities, Rev. Nutr., 2016, vol. 8, no. 644, pp. 1–27. doi 10.3390/nu8100644
Cousineau, J., Production and characterization of wheat gluten films, Master’s Thesis, Ontario: University of Waterloo, 2012.
Wu, Q., Yu, Sh., Kollert, M., et al., Highly absorbing antimicrobial biofoams based on wheat gluten and its biohybrids, ACS Sustainable Chem. Eng., 2016, vol. 4, no. 4, pp. 2395–2404. doi 103390/nu8100644
Wu, Q., Biofoams and biocomposites based on wheaten gluten proteins, Doctoral Thesis, Sweden, Stockholm: KTH Royal Institute of Technology, 2017.
Wouters, A., Rombouts, I., Fierens, E., et al., Relevance of the functional properties of enzymatic plant protein hydrolysates in food systems, Compr. Rev. Food Sci. Food Saf., 2016, vol. 15, pp. 786–800. doi 101111/1541-433712209
Woerdeman, D., Veraverbeke, W., Verpoest, I., et al., Gluten biopolymers, US Patent no. 7520929, 2009.
Blomfeldt, T.O.J., Kuktaite, R., Johansson, E., Hedenqvist, M.S., et al., Mechanical properties and network structure of wheat gluten foams, Biomacromolecules, 2011, vol. 12, pp. 1707–1715. doi 101021/bm200067f
Takeda, K., Matsumura, Y., and Shimizu, M., Emulsifying and surface properties of wheat gluten under acidic conditions, J. Food Sci., 2001, vol. 66, no. 3, pp. 393–399. doi 101111/j1365-26212001tb16116x
Day, L., Augustin, M.A., Batey, I.L., and Wrigley, C.W., Wheat-gluten uses and industry needs, Trends Food Sci. Technol., 2006, vol. 17, no. 2, pp. 82–90. doi 101016/ jtifs200510003
Gontard, N., Guilbert, S., and Cuq, J-L., Edible wheat gluten films: influence of the main process variables on film properties using response surface methodology, J. Food Sci., 1992, vol. 57, no. 1, pp. 190–199. doi 101111/j1365-26211992tb05453x
Zubeldia, F., Ansorena, M.R., Marcovich, N.E., and Zubeldia, F., Wheat gluten films obtained by compression molding, Polym. Test., 2015, vol. 43, pp. 68–77. doi 101016/jpolymertesting201502001
Herald, T.J., Gnanasambandam, R., Mcguire, B.H., and Hachmeister, K.A., Degradable wheat gluten films: preparation, properties and applications, J. Food Sci., 1995, vol. 60, no. 5, pp. 1147–1150. doi 101111/j1365-26211995tb06311x
Reddy, N., Jiang, Q., and Yang, Y., Novel wheat protein films as substrates for tissue engineering, J. Biomater. Sci., Polym. Ed., 2011, pp. 2063–2077. doi 101163/092050610X532638
Popineau, Y., Huchet, B., Larre, C., and Berot, S., Foaming and emulsifying properties of fractions of gluten peptides obtained by limited enzymatic hydrolysis and ultrafiltration, J. Cereal Sci., 2002, vol. 35, pp. 327–335. doi101006/jcrs20010437
Kong, X., Zhou, H., and Qian, H., Enzymatic hydrolysis of wheat gluten by proteases and properties of the resulting hydrolysates, Food Chem., 2007, vol. 101, pp. 615–620. doi 101016/jfoodchem200606062
Wang, J., Zhao, M., and Jiang, Y., Effects of wheat gluten hydrolysate and its ultrafiltration fractions on dough properties and bread quality, Food Technol. Biotechnol., 2007, vol. 45, no. 4, pp. 410–414.
Adler-Nissen, J., Determination of the degree of hydrolysis of food protein hydrolysates by trinitrobenzenesulfonic acid, J. Agric. Food Chem., 1979, vol. 27, no. 6, pp. 1256–1262. doi 101021/jf60226a042
Wang, J-S., Zhao, M-M., Zhao, Q-Z., et al., Characterization of hydrolysates derived from enzymatic hydrolysis of wheat gluten, JFS C: Food Chem. Toxicol., 2007, vol. 72, no. 2, pp. 103–107. doi 101111/j1750-3841200600247x
L’vovich, Ch.D., Method for manufacture of food product with foaming properties, RF Patent no. 2536967, 2014.
Joye, J. and McClements, D.J., Emulsifying and emulsion-stabilizing properties of gluten hydrolysates, J. Food Chem. Biochem., 2014, vol. 62, pp. 2623–2630. doi 101021/jf5001343
Wouters, A., Rombouts, I., Legein, M., Fierens, E., et al., Air-water interfacial properties of enzymatic wheat gluten hydrolyzates determine their foaming behavior, Food Hydrocolloids, 2016, vol. 55, pp. 155–162. doi 101016/jfoodhyd201511017
Wouters, A., Fierens, E., Rombouts, I., Brijs, K., et al., Exploring the relationship between structural and air-water interfacial properties of wheat (Triticum aestivum L.) gluten hydrolysates in a food system relevant pH range, J. Agric. Food Chem., 2017, vol. 65, no. 6, pp. 1263–1271. doi 101021/acsjafc6b05062
Wouters, A., Fierens, E., Rombouts, I., Brijs, K., et al., Air-water interfacial properties of enzymatically hydrolyzed wheat gluten in the presence of sucrose, Food Hydrocolloids, 2017, vol. 73, pp. 284–294. doi 101016/jfoodhyd201707014
Wouters, A., Fierens, E., Rombouts, I., Brijs, K., et al., Impact of ethanol on the air-water interfacial properties of enzymatically hydrolyzed wheat gluten, Colloids Surf., A: Physicochem. Eng. Asp., 2017, vol. 529, pp. 659–667. doi 101016/jcolsurfa201706013
Wouters, A., Air-water interfacial properties of enzymatically hydrolysed wheat (T. aestivum L.) gluten, Doctoral Thesis, Leuven, Belgium: KU Leuven, 2017.
Dadzie, R.G., Ma, H., Abano, E.E., Qu, W., et al., Optimization of process conditions for production of angiotensin i converting enzyme (ace) inhibitory peptides from vital wheat gluten using response surface methodology, Food Sci. Biotechnol., 2013, vol. 22, no. 6, pp. 1531–1537. doi 101007/s10068-013-0248-9
Zhang, Y., Li, J., Li, S., Ma, H., and Zhang, H., Mechanism study of multimode ultrasound pretreatment on the enzymolysis of wheat gluten, J. Sci. Food Agric., 2018, vol. 98, no. 4, pp. 1530–1538. doi 101002/jsfa8624
Sato, K., Egashira, Y., Ono, S., Mochizuki, S., et al., Identification of a hepatoprotective peptide in wheat gluten hydrolysate against D-galactosamine-induced acute hepatitis in rats, J. Agric. Food Chem., 2013, vol. 61, no. 26, pp. 6304–6310. doi 10102 1/jf400914e
Widyarani, W., Sari, Y.W., Ratnaningsih, E., Sanders, J.P., and Bruins, M.E., Production of hydrophobic amino acids from biobased resources: wheat gluten and rubber seed proteins, Appl. Microbiol. Biotechnol., 2016, vol. 100, no. 18, pp. 7909–7920. doi 101007/s00253-016-7441-8
Koo, S.H., Bae, I.Y., Lee, S., Lee, D-H., et al., Evaluation of wheat gluten hydrolysates as taste-active compounds with antioxidant activity, J. Food Sci. Technol., 2011, vol. 51, no. 3, pp. 535–542. doi 101007/s13197-011-0515-9
Park, E.Y., Imazu, H., Matsumura, Y., et al., Effects of peptide fractions with different isoelectric points from wheat gluten hydrolysates on lipid oxidation in pork meat patties, J. Agric. Food Chem., 2012, vol. 60, no. 30, pp. 7483–7488. doi 101021/jf301532
Maloletnikova, I.M., Zaryankina, A.I., and Lozovik, S.K., Gluten enteropathy in children, Probl. Health Ecol., 2013, vol. 4, no. 38, pp. 143–147.
Liflyandsky, V.G., Smolyansky, B.L., Lavrenova, G.V., and Solovyova, V.A., Encyclopedia of Diagnosis and Treatment from A to Z, Koreshkina, I.A., Ed., Moscow: Publishing house OLMA Media Group, 2013, pp. 186–187.
Serena, G., Camhi, S., Sturgeon, C., Yan, S., et al., The role of gluten in celiac disease and type 1 diabetes, Nutrients, 2015, vol. 7, no. 9, pp. 7143–7162. doi 103390/nu7095329
Waga, J., Structure and allergenicity of wheat gluten proteins—a review, Pol. J. Food Nutrit. Sci., 2004, vol. 13/54, no. 4, pp. 327–338. doi 101371/journalpone0172819
Stenman, S., Coeliac disease-inducing gluten, Academic Dissertation, Tampere, Finland: University of Tampere, 2011.
Li, Y.Yu.J., Goktepe, I., and Ahmedna, M., The potential of papain and alcalase enzymes and process optimizations to reduce allergenic gliadins in wheat flour, Food Chem., 2016, vol. 196, pp. 1338–1345. doi 101016/jfoodchem201510089
Tereshchenkova, V.F., Goptar, I.A., Kulemzina, I.A., et al., Dipeptidyl peptidase 4—an important digestive peptidase in Tenebrio molitor larvae, Insect Biochem. Mol. Biol., 2016, vol. 76, pp. 38–48. doi 101016/jibmb201607003
Stepniak, D., Spaenij-Dekking, L., Mitea, C., et al., Highly efficient gluten degradation with a newly identified prolyl endoprotease: implications for celiac disease, AJP Gastrointest. Liver Physiol., 2006, vol. 291, no. 4, pp. 621–629. doi 101152/ajpgi000342006
Gutierrez, S., Perez-Andres, J., Martinez-Bianco, H., et al., The human digestive tract has proteases capable of gluten hydrolysis, Mol. Metab., 2017, vol. 6, no. 7, pp. 693–702. doi 101016/jmolmet201705008
Rizzello, C.G., Montemurro, M., and Gobbetti, M., Characterization of the bread made with durum wheat semolina rendered gluten free by sourdough biotechnology in comparison with commercial gluten-free products, J. Food Sci., 2016, vol. 81, no. 9, pp. 2263–2272. doi 101111/1750-384113410
Gass, J.D., Chaitan, K., Bethune, M., and Siegel, M.J., Combination enzyme therapy for digestion of dietary gluten, Patent no. EP2136833A1, 2007.
Rey, M., Yang, M., Lee, L., et al., Addressing proteolytic efficiency in enzymatic degradation therapy for celiac disease, Sci. Rep., 2016. doi 101038/srep30980
Eugster, P.J., Salamin, K., Grouzmann, E., and Monod, M., Production and characterization of two major Aspergillus oryzae secreted prolyl endopeptidases able to efficiently digest proline-rich peptides of gliadin, Microbiology, 2015, vol. 161, no. 12, pp. 2277–2288. doi 101099/mic0000198
Wolf, C., Siegel, J.B., Tinberg, C., et al., Engineering of Kuma030: a gliadin peptidase that rapidly degrades immunogenic gliadin peptides in gastric conditions, J. Am. Chem. Soc., 2015, vol. 137, no. 40, pp. 13106–13113. doi 101021/jacs5b08325
Zamyatnin, A.A., Savvateeva, L.V., Gorokhovets, N.V., Makarov, V.A., et al., Method of wheat cysteine protease proteins family (Triticum aestivum) producing and preparation of tritikale-alpha protein, obtained using said method, RF Patent no. 2603054C2, 2015.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Abbreviations: ACE, angiotensin-converting enzyme; HMS, high molecular weight subunits; LMS, low molecular weight subunits.
Rights and permissions
About this article
Cite this article
Asrarkulova, A.S., Bulushova, N.V. Wheat Gluten and Its Hydrolysates. Possible Fields of Practical Use. Appl Biochem Microbiol 54, 825–833 (2018). https://doi.org/10.1134/S0003683818090107
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683818090107