Abstract
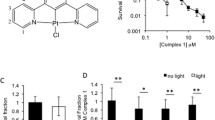
Photodynamic therapy (PDT) is a minimally invasive approach to treat various forms of cancer, based on the ability of certain non-toxic molecules (photosensitizers) to generate reactive oxygen species (ROS) after excitation by light of a certain wavelength and eventually induce strong phototoxic reactions against malignant cells and other pathogens. Curcumin is one of the most extensively investigated phytochemcals with a wide range of therapeutic properties and has been shown to induce strong photocytotoxic effects in micromolar concentrations against a variety of cancer cell lines. Curcumin (1) is comparatively evaluated with the naturally occurring bisdemethoxy Curcumin (2), which lacks the two methoxy groups, as well as two newly synthesized curcuminoids, the cinnamaldehyde derivative (3) and the dimethylamino one (4), designed to increase the absorption maximum and hence the tissue penetration. The synthetic curcuminoids were successfully synthesized in sufficient amounts and their photophysical properties such as absorption, fluorescence, photobleaching and free radical generation were investigated Compound 4 exhibited a significant increase in peak absorption (497 nm) and strong fluorescent emission signals were recorded for all curcuminoids. Photobleaching of 4 was comparable to 1 whereas 2 and 3 showed more extended photobleaching but much higher ROS production in very short irradiation times. Compounds 2 and 4 exhibited specific intracellular localization. After dark and light cytotoxicity experiments against LNCaP prostate cancer cell line for all curcuminoids, concentration of 3 µM and irradiance of 6 mW cm−2 were selected for the PDT application which resulted in remarkable results with very short LD50. Curcuminoids 2 and 4 exhibited a significant dose-dependent PDT effect. The biphasic doseresponse photodynamic effect observed for 1 and 3 may provide a strategy against prolonged and sustained photosensitivity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. Agostinis, K. Berg, K. A. Cengel, T. H. Foster, A. W. Girotti, S. O. Gollnick, S. M. Hahn, M. R. Hamblin, A. Juzeniene, D. Kessel, M. Korbelik, J. Moan, P. Mroz, D. Nowis, J. Piette, B. C. Wilson and J. Golab, Photodynamic therapy of cancer: An update, Ca-Cancer J. Clin., 2011, 61 ,250–281.
K. Plaetzer, M. Berneburg, T. Kiesslich and T. Maisch, New applications of photodynamic therapy in biomedicine and biotechnology, BioMed Res. Int., 2013, 2013 ,161362.
Á. Juarranz, P. Jaén, F. Sanz-Rodríguez, J. Cuevas and S. González, Photodynamic therapy of cancer. Basic principles and applications, Clin. Transl. Oncol., 2008, 10 ,148–154.
A. P. Castano, T. N. Demidova and M. R. Hamblin, Mechanisms in photodynamic therapy: part two-cellular signaling, cell metabolism and modes of cell death, Photodiagn. Photodyn. Ther., 2005, 1 ,1–23.
C. Hopper, Photodynamic therapy: a clinical reality in the treatment of cancer, Lancet Oncol., 2000, 1 ,212–219.
L. B. Josefsen and R. W. Boyle, Photodynamic therapy and the development of metal-based photosensitisers, Met.- Based Drugs, 2008, 2008 ,276109.
M. Salem, S. Rohani and E. R. Gillies, Curcumin, a promising anti-cancer therapeutic: a review of its chemical properties, bioactivity and approaches to cancer cell delivery, RSCAdv., 2014, 4 ,10815–10829.
A. B. Kunnumakkara, D. Bordoloi, C. Harsha, K. Banik, S. C. Gupta and B. B. Aggarwal, Curcumin mediates anticancer effects by modulating multiple cell signaling pathways, Clin. Sci., 2017, 131 ,1781–1799.
H. H. Tønnesen, H. de Vries, J. Karlsen and G. Beijersbergen van Henegouwen, Studies on curcumin and curcuminoids. IX: Investigation of the photobiological activity of curcumin using bacterial indicator systems, J. Pharm. Sci., 1987, 76 ,371–373.
T. A. Dahl, W. M. McGowan, M. A. Shand and V. S. Srinivasan, Photokilling of bacteria by the natural dye curcumin, Arch. Microbiol., 1989, 151 ,183–185.
T. Haukvik, E. Bruzell, S. Kristensen and H. H. Tønnesen, Photokilling of bacteria by curcumin in selected polyethylene glycol 400 (PEG 400) preparations. Studies on curcumin and curcuminoids, XLI, Die Pharmazie, 2010, 65, 600–606.
L. Nardo, A. Andreoni, M. Masson, T. Haukvik and H. H. Tønnesen, Studies on curcumin and curcuminoids. XXXIX. Photophysical properties of bisdemethoxycurcumin, J. Fluoresc., 2011, 21 ,627–635.
E. M. Bruzell, E. Morisbak and H. H. Tønnesen, Studies on curcumin and curcuminoids. XXIX. Photoinduced cytotoxicity of curcumin in selected aqueous preparations, Photochem. Photobiol. Sci., 2005, 4 ,523–530.
L. Nardo, A. Andreoni, M. Bondani, M. Másson, T. Haukvik and H. H. Tønnesen, Studies on Curcumin and Curcuminoids. XLVI. Photophysical Properties of Dimethoxycurcumin and Bis-dehydroxycurcumin, J. Fluoresc., 2012, 22 ,597–608.
L. Nardo, A. Andreoni, M. Bondani, M. Másson and H. Hjorth Tønnesen, Studies on curcumin and curcuminoids. XXXIV. Photophysical properties of a symmetrical, non-substituted curcumin analogue, J. Photochem. Photobiol., B, 2009, 97 ,77–86.
N. C. Araújo, C. R. Fontana, M. E. M. Gerbi and V. S. Bagnato, Overall-Mouth Disinfection by Photodynamic Therapy Using Curcumin, Photomed Laser Surg., 2012, 30 ,96–101.
P. R. Kunwar, A. Barik, B. Mishra, K. I. Priyadarsini and K. Rathinasamy, Differential up-take and fluorescence of curcumin, a yellow pigment from turmeric, in normal vs tumor cells, BARC Newsl., 2007, 285 ,202–207.
C. F. Chignell, P. Bilskj, K. J. Reszka, A. G. Motten, R. H. Sik and T. A. Dahl, Spectral and photochemical properties of curcumin, Photochem. Photobiol., 1994, 59 ,295–302.
F. G. Rego-Filho, M. T. de Araujo, K. T. de Oliveira and V. S. Bagnato, Validation of Photodynamic Action via Photobleaching of a New Curcumin-Based Composite with Enhanced Water Solubility, J. Fluoresc., 2014, 24 ,1407–1413.
A. Sreedhar, I. Sarkar, P. Rajan, J. Pai, S. Malagi, V. Kamath and R. Barmappa, Comparative evaluation of the efficacy of curcumin gel with and without photo activation as an adjunct to scaling and root planing in the treatment of chronic periodontitis: A split mouth clinical and microbiological study, J. Nat. Sci., Biol. Med., 2015, 6 ,102–109.
N. Komerik and A. J. MacRobert, Photodynamic therapy as an alternative antimicrobial modality for oral infections, J. Environ. Pathol., Toxicol. Oncol., 2006, 25 ,487–504.
K. I. Priyadarsini, Photophysics, photochemistry and photobiology of curcumin: Studies from organic solutions, bio-mimetics and living cells, J. Photochem. Photobiol., C, 2009, 10 ,81–95.
K. Panagiotaki, Z. Sideratou, S. Vlahopoulos, M. Paravatou-Petsotas, M. Zachariadis, N. Khoury, V. Zoumpourlis and D. Tsiourvas, Triphenylphosphonium-Functionalized Mitochondriotropic Nanocarrier for Efficient Co-Delivery of Doxorubicin and Chloroquine and Enhanced Antineoplastic Activity, Pharmaceuticals, 2017, 10 ,91.
W. Wichitnithad, U. Nimmannit, S. Wacharasindhu and P. Rojsitthisak, Synthesis, Characterization and Biological Evaluation of Succinate Prodrugs of Curcuminoids for Colon Cancer Treatment, Molecules, 2011, 16 ,1888–1900.
D. P. V. Leite, F. R. Paolillo, T. N. Parmesano, C. R. Fontana and V. S. Bagnato, Effects of Photodynamic Therapy with Blue Light and Curcumin as Mouth Rinse for Oral Disinfection: A Randomized Controlled Trial, Photomed Laser Surg., 2014, 32 ,627–632.
K. Khorsandi, R. Hosseinzadeh and M. Fateh, Curcumin intercalated layered double hydroxide nanohybrid as a potential drug delivery system for effective photodynamic therapy in human breast cancer cells, RSC Adv., 2015, 5 ,93987–93994.
H. Koon, A. W. N. Leung, K. K. M. Yue and N. K. Mak, Photodynamic effect of curcumin on NPC/CNE2 cells, J. Environ. Pathol., Toxicol. Oncol., 2006, 25 ,205–215.
S. Banerjee, P. Prasad, A. Hussain, I. Khan, P. Kondaiah and A. R. Chakravarty, Remarkable photocytotoxicity of curcumin in HeLa cells in visible light and arresting its degradation on oxovanadium(IV) complex formation, Chem. Commun., 2012, 48 ,7702–7704.
Y. J. Wang, M. H. Pan, A. L. Cheng, L. I. Lin, Y. S. Ho, C. Y. Hsieh and J. K. Lin, Stability of curcumin in buffer solutions and characterization of its degradation products, J. Pharm. Biomed. Anal., 1997, 15 ,1867–1876.
P. Anand, A. B. Kunnumakkara, R. A. Newman and B. B. Aggarwal, Bioavailability of Curcumin: Problems and Promises, Mol. Pharm., 2007, 4 ,807–818.
L. C. Price and R. W. Buescher, Kinetics of alkaline degradation of the food pigments curcumin and curcuminoids, J. Food Sci., 2006, 62 ,267–269.
H. H. Tønnesen and J. Karlsen, Studies on curcumin and curcuminoids - VI. Kinetics of curcumin degradation in aqueous solution, Z. Lebensm.-Unters. Forsch., 1985, 180 ,402–404.
M. Cozzolino, P. Delcanale, C. Montali, M. Tognolini, C. Giorgio, M. Corrado, L. Cavanna, P. Bianchini, A. Diaspro, S. Abbruzzetti and C. Viappiani, Enhanced photosensitizing properties of protein bound curcumin, Life Sci., 2019, 233 ,116710.
M. A. Subhan, K. Alam, M. S. Rahaman, M. A. Rahman and R. Awal, Synthesis and Characterization of Metal Complexes Containing Curcumin (C21H20O6) and Study of their Antimicrobial Activities and DNA Binding Properties, J. Sci. Res., 2013, 6 ,97–109.
D. Patra and C. Barakat, Synchronous fluorescence spectroscopic study of solvatochromic curcumin dye, Spectrochim. Acta, Part A, 2011, 79 ,1034–1041.
H. Van Nong, L. X. Hung, P. N. Thang, V. D. Chinh, L. Van Vu, P. T. Dung, T. Van Trung and P. T. Nga, Fabrication and vibration characterization of curcumin extracted from turmeric (Curcuma longa) rhizomes of the northern Vietnam, SpringerPlus, 2016, 5 ,1147.
A. Ormond and H. Freeman, Dye Sensitizers for Photodynamic Therapy, Materials, 2013, 6 ,817–840.
G. Jori, Tumour photosensitizers: approaches to enhance the selectivity and efficiency of photodynamic therapy, J. Photochem. Photobiol., B, 1996, 36 ,87–93.
R. Jagannathan, P. M. Abraham and P. Poddar, Temperature-Dependent Spectroscopic Evidences of Curcumin in Aqueous Medium: A Mechanistic Study of Its Solubility and Stability, J. Phys. Chem. B, 2012, 116 ,14533–14540.
K. Priyadarsini, The Chemistry of Curcumin: From Extraction to Therapeutic Agent, Molecules, 2014, 19 ,20091–20112.
M. Kharat, Z. Du, G. Zhang and D. J. McClements, Physical and Chemical Stability of Curcumin in Aqueous Solutions and Emulsions: Impact of pH, Temperature, and Molecular Environment, J. Agric. Food Chem., 2017, 65 ,1525–1532.
M. Ghosh, A. T. K. Singh, W. Xu, T. Sulchek, L. I. Gordon and R. O. Ryan, Curcumin nanodisks: formulation and characterization, Nanomedicine, 2011, 7 ,162–167.
R. Bonnett and G. Martínez, Photobleaching of sensitisers used in photodynamic therapy, Tetrahedron, 2001, 57 ,9513–9547.
D. Deeb, Y. X. Xu, H. Jiang, X. Gao, N. Janakiraman, R. A. Chapman and S. C. Gautam, Curcumin (diferuloyl-methane) enhances tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in LNCaP prostate cancer cells, Mol. Cancer Ther., 2003, 2 ,95–103.
A. Petri, D. Yova, E. Alexandratou, M. Kyriazi and M. Rallis, Comparative characterization of the cellular uptake and photodynamic efficiency of Foscan® and Fospeg in a human prostate cancer cell line, Photodiagn. Photodyn. Ther., 2012, 9 ,344–354.
V. Ellerkamp, N. Bortel, E. Schmid, B. Kirchner, S. Armeanu-Ebinger and J. Fuchs, hotodynamic Therapy Potentiates the Effects of Curcumin on Pediatric Epithelial Liver Tumor Cells, Anticancer Res., 2016, 36 ,3363–3372.
Z. Hussain, H. E. Thu, M. W. Amjad, F. Hussain, T. A. Ahmed and S. Khan, Exploring recent developments to improve antioxidant, anti-inflammatory and antimicrobial efficacy of curcumin: A review of new trends and future perspectives, Mater. Sci. Eng., C, 2017, 77 ,1316–1326.
A. S. Martirosyan, H. R. Vardapetyan, S. G. Tiratsuyan and A. A. Hovhannisyan, Biphasic dose-response of antioxidants in hypericin-induced photohemolysis, Photodiagn. Photodyn. Ther., 2011, 8 ,282–287.
M. Tanaka, M. Kinoshita, Y. Yoshihara, N. Shinomiya, S. Seki, K. Nemoto, M. R. Hamblin and Y. Morimoto, Photodynamic therapy using intra-articular Photofrin for murine MRSA arthritis: biphasic light dose response for neutrophil-mediated antibacterial effect, Lasers Surg. Med., 2011, 43 ,221–229.
M. Tanaka, P. Mroz, T. Dai, M. Kinoshita, Y. Morimoto and M. R. Hamblin, Photodynamic therapy can induce nonspecific protective immunity against a bacterial infection, Proc. SPIE 8224, Biophotonics Immun. Respons. VII, 2012, 8224, 822403.
A. P. Castano, T. N. Demidova and M. R. Hamblin, Mechanisms in photodynamic therapy: part one-photosensitizers, photochemistry and cellular localization, Photodiagn. Photodyn. Ther., 2004, 1 ,279–293.
A. A. Rosenkranz, D. A. Jans and A. S. Sobolev, Targeted intracellular delivery of photosensitizers to enhance photodynamic efficiency, Immunol. Cell Biol., 2000, 78 ,452–464.
H. Abrahamse and M. R. Hamblin, New photosensitizers for photodynamic therapy, Biochem. J., 2016, 473 ,347–364.
S. Jiang, R. Zhu, X. He, J. Wang, M. Wang, Y. Qian and S. Wang, Enhanced photocytotoxicity of curcumin delivered by solid lipid nanoparticles, Int. J. Nanomed., 2016, 12 ,167–178.
A. Kunwar, A. Barik, B. Mishra, K. Rathinasamy, R. Pandey and K. I. Priyadarsini, Quantitative cellular uptake, localization and cytotoxicity of curcumin in normal and tumor cells, Biochim. Biophys. Acta, 2008, 1780 ,673–679.
S. Ben-Zichri, S. Kolusheva, M. Danilenko, S. Ossikbayeva, W. J. Stabbert, J. L. Poggio, D. E. Stein, Z. Orynbayeva and R. Jelinek, Cardiolipin mediates curcumin interactions with mitochondrial membranes, Biochim. Biophys. Acta, Biomembr., 2019, 1861 ,75–82.
A. Moustapha, P. Pérétout, N. Rainey, F. Sureau, M. Geze, J.-M. Petit, E. Dewailly, C. Slomianny and P. Petit, Curcumin induces crosstalk between autophagy and apoptosis mediated by calcium release from the endoplasmic reticulum, lysosomal destabilization and mitochondrial events, Cell Death Discovery, 2015, 26 ,15017.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Kazantzis, K.T., Koutsonikoli, K., Mavroidi, B. et al. Curcumin derivatives as photosensitizers in photodynamic therapy: photophysical properties and in vitro studies with prostate cancer cells. Photochem Photobiol Sci 19, 193–206 (2020). https://doi.org/10.1039/c9pp00375d
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00375d