Abstract
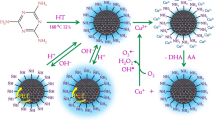
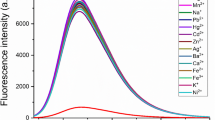
A quinoline moiety was used as a building block for designing a probe for the selective detection of copper ions in a partially aqueous medium. We have developed a molecular sensing system which gives insight into the complex physiological and redox aspects of labile copper. The probe provides a colorimetric approach for distinguishing cuprous and cupric ions along with their simultaneous discrimination from other metal ions in the visible range of the spectrum. The chemosensor showed a remarkable fluorescence enhancement along with a significant bathochromic shift of about 35 nm. The detection limit of the probe was found to be 1.03 μM which is optimally favorable to be applied in real-time monitoring. Fabrication of paper strips with the probe was done to detect the presence of cuprous ions in the real sample. The value of the binding constant (1.37 × 104 M−1) suggests stable complex formation between the metal ion and the sensing probe. The photoluminescence and structural aspects of the chemosensor were characterized by using fluorescence, absorption, ESI-MS, and 1H NMR spectroscopy. Furthermore, the cytotoxic nature and bioimaging properties of the probe were interpreted in vitro on RAW 264.7 macrophage cell lines and peripheral blood mononuclear cells (PBMCs) respectively.
Similar content being viewed by others
References
T. A. Dickinson, J. White, J. S. Kauer and D. R. Walt, A chemical-detecting system based on a cross-reactive optical sensor array, Nature, 1996, 382, 697–700.
C. Bargossi, M. C. Fiorini, M. Montalti, L. Prodi, N. Zaccheroni, V. Selmi and I. Bologna, Recent developments in transition metal ion detection by luminescent chemosensors, Coord. Chem. Rev., 2000, 208, 17–32.
D. Staneva, M. S. I. Makki, T. R. Sobahi, P. Bosch, R. M. Abdel-Rahman, A. Asiri and I. Grabchev, Synthesis and spectral characterization of a new blue fluorescent tripod for detecting metal cations and protons, J. Lumin., 2015, 162, 149–154.
V. Kumar, S. Kumar, R. Singh, L. P. Singh, S. K. Shoora and B. Sethi, Cadmium(II) ion sensing through p-tert-butyl calix [6]arene based potentiometric sensor, J. Mol. Liq., 2014, 195, 65–68.
F. Firdaus, A. Farhi, M. Faraz and M. Shakir, Benzidine based fluorescent probe for the sensitive detection of heavy metal ions via chelation enhanced fluorescence mechanism- A multiplexed sensing platform, J. Lumin., 2018, 199, 475–482.
M. Lütfi, V. Kumar, T. Eren and A. E. S, A novel electro analytical nanosensor based on graphene oxide/silver nanoparticles for simultaneous determination of quercetin and morin, Electrochim. Acta, 2014, 120, 204–211.
V. Kumar, B. Sethi, R. A. Sharma, S. Agarwal and A. Bharti, Mercury selective potentiometric sensor based on low rim functionalized thiacalix [4]-arene as a cationic receptor, J. Mol. Liq., 2013, 177, 114–118.
V. K. Gupta, H. Karimi-maleh and R. Sadegh, Simultaneous Determination of Hydroxylamine, Phenol and Sulfite in Water and Waste Water Samples Using A Voltammetric Nanosensor, Int. J. Electrochem. Sci., 2015, 10, 303–316.
S. K. Srivastava, V. K. Gupta and S. Jain, PVC-Based 2,2,2-Cryptand Sensor for Zinc Ions, Anal. Chem., 1996, 68, 1272–1275.
S. K. Srivastava, V. K. Gupta and S. Jain, Determination of Lead Using a Poly(vinyl chloride)-based Crown Ether Membrane, Analyst, 1995, 120, 495–498.
L. Prodi, F. Bolletta, M. Montalti, V. Selmi and I. Bologna, Luminescent chemosensors for transition metal ions, Coord. Chem. Rev., 2000, 205, 59–83.
M. Yang, Ju B. Chae, C. Kim and R. G. Harrison, A visible chemosensor based on carbohydrazide for Fe(II), Co(II) and Cu(II) in aqueous solution, Photochem. Photobiol. Sci., 2019, 18, 1249–1258.
J. Wang and Q. Zong, A new turn-on fluorescent probe for the detection of copper ion in neat aqueous solution, Sens. Actuators, B, 2015, 216, 572–577.
J. Chen, W. Su, E. Wang and Y. Liu, for Cu(II) and its application in bioimaging, J. Lumin., 2016, 180, 301–305.
C. Wu, J. Wang, J. Shen, C. Zhang, Z. Wu and H. Zhou, A colorimetric quinoline-based chemosensor for sequential detection of copper ion and cyanide anions, Tetrahedron, 2017, 73, 5715–5719.
R. An, D. Zhang, Y. Chen and Y. Cui, Sensors and Actuators B: Chemical A “turn-on” fluorescent and colorimetric sensor for selective detection of Cu(II) in aqueous media and living cells, Sens. Actuators, B, 2016, 222, 48–54.
Q. Lin, Y. Fan, P. Mao, L. Liu, J. Liu and Y. Zhang, Pillar[5] arene-Based Supramolecular Organic Framework with Multi-Guest Detection and Recyclable Separation Properties, Chem. – Eur. J., 2018, 24, 777–783.
M. J. Schnermann, Organic dyes for deep bioimaging, Nature, 2017, 551, 176–177.
H. Karimi-maleh, F. Tahernejad-javazmi, N. Atar, M. Lu, V. K. Gupta and A. A. Ensa, A Novel DNA Biosensor Based on a Pencil Graphite Electrode Modified with Polypyrrole/Functionalized Multiwalled Carbon Nanotubes for Determination of 6- Mercaptopurine Anticancer Drug, Ind. Eng. Chem. Res., 2015, 54, 3634–3639.
V. Kumar, N. Mergu, L. Kumar and A. Kumar, Selective naked-eye detection of Magnesium(II) ions using a coumarin-derived fluorescent probe, Sens. Actuators, B, 2015, 207, 216–223.
V. K. Gupta, N. Mergu, L. K. Kumawat and A. K. Singh, A reversible fluorescence “off- on-off” sensor for sequential detection of Aluminum and Acetate/Fluoride ions, Talanta, 2015, 144, 80–89.
V. K. Gupta, A. K. Singh and L. K. Kumawat, Thiazole Schiff base turn-on fluorescent chemosensor for Al3+ ion, Sens. Actuators, B, 2014, 195, 98–108.
H. Zhu, J. Fan, B. Wang and X. Peng, Chem Soc Rev sensors for the first-row d-block metal ions, Chem. Soc. Rev., 2015, 44, 4337–4366.
H. Yao, Y. Zhang, T. Wei and Q. Lin, Aggregation-Induced Emission Supramolecular Organic Framework (AIE SOF) Gels Constructed from Supramolecular Polymer Networks Based on Tripodal Pillar[5]arene for Fluorescence Detection and Efficient Removal of Various Analytes, ACS Sustainable Chem. Eng., 2019, 7, 11999–12007.
K. J. Waldron, J. C. Rutherford, D. Ford and N. J. Robinson, Metalloproteins and metal sensing, Nature, 2009, 460, 823–830.
L. Tang, J. Zhao, M. Cai, P. Zhou, K. Zhong, S. Hou and Y. Bian, An efficient sensor for relay recognition of Zn2+ and Cu2+ through fluorescence “off – on – off” functionality, Tetrahedron Lett., 2013, 53, 6105–6109.
Y. Liu, Y. Feng, R. Wang, T. Jiao, J. Li, Y. Rao, Q. Zhang, Z. Bai and Q. Peng, Self- Assembled Naphthylidene-Containing Schiff Base Anchored Polystyrene Nanocomposites Targeted for Selective Cu(II) Ion Removal from Wastewater, ACS Omega, 2019, 4, 12098–12106.
S. Liu, Y. Wang and J. Han, Reviews Fluorescent chemosensors for copper(II) ion: Structure, mechanism, and application, J. Photochem. Photobiol., C, 2017, 32, 78–103.
L. K. Kumawat, N. Mergu, A. K. Singh and V. K. Gupta, A novel optical sensor for copper ions based on phthalocyanine tetrasulfonic acid, Sens. Actuators, B, 2015, 212, 389–394.
L. Tang, M. Cai, Z. Huang, K. Zhong and S. Hou, Rapid and highly selective relay recognition of Cu(II) and sulfide ions by a simple benzimidazole-based fluorescent sensor in water, Sens. Actuators, B, 2013, 185, 188–194.
L. Tang, P. Zhou, K. Zhong and S. Hou, Fluorescence relay enhancement sequential recognition of Cu2+ and CN− by a new quinazoline derivative, Sens. Actuators, B, 2013, 182, 439–445.
V. Kumar, L. P. Singh, R. Singh, N. Upadhyay, S. P. Kaur and B. Sethi, A novel copper(II) selective sensor based on Dimethyl 4, 4′ (o-phenylene) bis(3-thioallophanate) in PVC matrix, J. Mol. Liq., 2012, 174, 11–16.
A. Pralle, L. Zeng, E. Y. Isacoff, C. J. Chang and E. W. Miller, A Selective Turn-On Fluorescent Sensor for Imaging Copper in Living Cells, J. Am. Chem. Soc., 2006, 128, 10–11.
M. Saleem and K. Lee, Selective fluorescence detection of Cu(II) in aqueous solution and living cells, J. Lumin., 2014, 145, 843–848.
L. Liu, F. Dan, W. Liu, X. Lu, Y. Han, S. Xiao and H. Lan, A high-contrast colorimetric and fluorescent probe for Cu2+ based on benzimidazole-quinoline, Sens. Actuators, B, 2017, 247, 445–450.
J. Cody and C. J. Fahrni, Fluorescence sensing based on cation-induced conformational switching: copper-selective modulation of the photoinduced intramolecular charge transfer of a donor–acceptor biphenyl fluorophore, Tetrahedron, 2004, 60, 11099–11107.
A. Farhi, F. Firdaus and M. Shakir, Design and application of a tripodal on-off type chemosensor for discriminative and selective detection of Fe2+ ions, New J. Chem., 2018, 42, 31–36.
S. R. Bhatta, B. Mondal, G. Vijaykumar and A. Thakur, ICT-Isomerization-Induced Turn-On Fluorescence Probe with a Large Emission Shift for Mercury Ion: Application in Combinational Molecular Logic, Inorg. Chem., 2017, 56, 11577–11590.
M. Suresh, A. K. Mandal, S. Saha, E. Suresh, A. Mandoli, R. Di Liddo, P. P. Parnigotto and A. Das, Azine-based receptor for recognition of Hg2+ ion: Crystallographic evidence and imaging application in live cells, Org. Lett., 2010, 12, 5406–5409.
J. S. Wu, W. M. Liu, X. Q. Zhuang, F. Wang, P. F. Wang, S. L. Tao, X. H. Zhang, S. K. Wu and S. T. Lee, Fluorescence turn on of coumarin derivatives by metal cations: A new signaling mechanism based on C=N isomerization, Org. Lett., 2007, 9, 33–36.
J. Wu, W. Liu, J. Ge, H. Zhang and P. Wang, New sensing mechanisms for the design of fluorescent chemosensors emerging in recent years, Chem. Soc. Rev., 2011, 40, 3483–3495.
Z. Xu and D. R. Spring, Fluorescent chemosensors for Zn2+, Chem. Soc. Rev., 2010, 39, 1996–2006.
Y. Ma, F. Wang, S. Kambam and X. Chen, A quinoline-based fluorescent chemosensor for distinguishing cadmium from zinc ions using cysteine as an auxiliary reagent, Sens. Actuators, B, 2013, 188, 1116–1122.
M. Saleem and K. H. Lee, Optical sensor: A promising strategy for environmental and biomedical monitoring of ionic species, RSC Adv., 2015, 5, 72150–72287.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c9pp00247b
Rights and permissions
About this article
Cite this article
Farhi, A., Firdaus, F., Saeed, H. et al. A quinoline-based fluorescent probe for selective detection and real-time monitoring of copper ions – a differential colorimetric approach. Photochem Photobiol Sci 18, 3008–3015 (2019). https://doi.org/10.1039/c9pp00247b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00247b