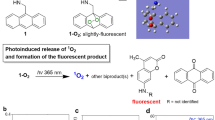
Abstract
Here we report the activatable photosensitizer BromoAcroB, a brominated BODIPY dye incorporating a reactive acrolein warhead. The acrolein moiety serves as an intramolecular switch, deactivating the BODIPY dye in its singlet and triplet excited states via internal conversion. Thiolate addition to this moiety disables the intramolecular quenching mechanism restoring the photosensitizing properties of the parent dye, characterized by a quantum yield of singlet oxygen photosensitization of 0.69 ± 0.02. In cell cultures, and upon thiol adduct formation, BromoAcroB induced light-dependent cell death in MRC-5 and HeLa cell lines. Using fluorescence microscopy and upon measuring the low yet non-negligible emission of the activated compound, we show that the phototoxicity of the dormant photosensitizer correlated with the quantity of BromoAcroB adducts generated. BromoAcroB thus serves as a dormant photosensitizer sensitive to intracellular electrophile response. Our results highlight the effective control of a triplet state process by modulation of an unsaturated moiety on the BODIPY scaffold and underscore the mechanistic opportunities arising for controlled singlet oxygen production in cells specifically sensitive to electrophile stress.
Similar content being viewed by others
Notes and references
Two lifetimes were measured: τ1 = 1.727 ± 0.005 ns (96%), τ2 = 0.61 ± 0.05 ns (4%). The shorter lifetime we attribute to an artefact.
J. Chan, S. C. Dodani and C. J. Chang, Nat. Chem., 2012, 4, 973–984.
A. Nadler and C. Schultz, Angew. Chem., Int. Ed., 2013, 52, 2408–2410.
W. Chyan and R. T. Raines, ACS Chem. Biol., 2018, 13, 1810–1823.
J. Zhang, X. Chai, X. P. He, H. J. Kim, J. Yoon and H. Tian, Chem. Soc. Rev., 2019, 48, 683–722.
L. E. Greene, R. Lincoln and G. Cosa, Photochem. Photobiol., 2019, 95, 192–201.
R. Lincoln, L. E. Greene, K. Krumova, Z. Ding and G. Cosa, J. Phys. Chem. A, 2014, 118, 10622–10630.
H. L. Kee, C. Kirmaier, L. Yu, P. Thamyongkit, W. J. Youngblood, M. E. Calder, L. Ramos, B. C. Noll, D. F. Bocian, W. R. Scheidt, R. R. Birge, J. S. Lindsey and D. Holten, J. Phys. Chem. B, 2005, 109, 20433–20443.
M. K. Kuimova, G. Yahioglu, J. A. Levitt and K. Suhling, J. Am. Chem. Soc., 2008, 130, 6672–6673.
R. Lincoln, L. E. Greene, C. Bain, J. O. Flores-Rizo, D. S. Bohle and G. Cosa, J. Phys. Chem. B, 2015, 119, 4758–4765.
G. Zheng, J. Chen, K. Stefflova, M. Jarvi, H. Li and B. C. Wilson, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 8989–8994.
J. Chen, T. W. Liu, P. C. Lo, B. C. Wilson and G. Zheng, Bioconjugate Chem., 2009, 20, 1836–1842.
J. Chen, K. Stefflova, M. J. Niedre, B. C. Wilson, B. Chance, J. D. Glickson and G. Zheng, J. Am. Chem. Soc., 2004, 126, 11450–11451.
X. J. Jiang, P. C. Lo, Y. M. Tsang, S. L. Yeung, W. P. Fong and D. K. P. Ng, Chem. – Eur. J., 2010, 16, 4777–4783.
V. Lebrun and P. Faller, Chem, 2018, 4, 663–665.
A. M. Durantini, L. E. Greene, R. Lincoln, S. R. Martinez and G. Cosa, J. Am. Chem. Soc., 2016, 138, 1215–1225.
H. Chen, J. Tian, W. He and Z. Guo, J. Am. Chem. Soc., 2015, 137, 1539–1547.
R. Lincoln, L. E. Greene, W. Zhang, S. Louisia and G. Cosa, J. Am. Chem. Soc., 2017, 139, 16273–16281.
A. Loudet and K. Burgess, Chem. Rev., 2007, 107, 4891–4932.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16, Revision A.03, Gaussian, Inc., Wallingford CT, 2016.
W. Koch and M. C. Holthausen, A Chemist’s Guide to Density Functional Theory, John Wiley & Sons, 2015.
R. Dennington, T. A. Keith and J. M. Millam, GaussView, Version 6, Semichem Inc., Shawnee Mission, KS, 2016.
F. Wilkinson, W. P. Helman and A. B. Ross, J. Phys. Chem. Ref. Data, 1993, 22, 113–262.
J. Schindelin, I. Arganda-Carreras, E. Frise, V. Kaynig, M. Longair, T. Pietzsch, S. Preibisch, C. Rueden, S. Saalfeld, B. Schmid, J. Y. Tinevez, D. J. White, V. Hartenstein, K. Eliceiri, P. Tomancak and A. Cardona, Nat. Methods, 2012, 9, 676–682.
L. E. Greene, R. Lincoln and G. Cosa, J. Am. Chem. Soc., 2017, 139, 15801–15811.
T. Yogo, Y. Urano, Y. Ishitsuka, F. Maniwa and T. Nagano, J. Am. Chem. Soc., 2005, 127, 12162–12163.
A. Kamkaew, S. H. Lim, H. B. Lee, L. V. Kiew, L. Y. Chung and K. Burgess, Chem. Soc. Rev., 2013, 42, 77–88.
R. Lincoln, A. M. Durantini, L. E. Greene, S. R. Martinez, R. Knox, M. C. Becerra and G. Cosa, Photochem. Photobiol. Sci., 2017, 16, 178–184.
A. Romieu, C. Massif, S. Rihn, G. Ulrich, R. Ziessel and P. Y. Renard, New J. Chem., 2013, 37, 1016–1027.
J. Sangster, J. Phys. Chem. Ref. Data, 1989, 18, 1111–1229.
L. P. Jameson, N. W. Smith, O. Annunziata and S. V. Dzyuba, Phys. Chem. Chem. Phys., 2016, 18, 14182–14185.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: 1H and 13C NMR spectra for BromoAcroB and Br2BEt, TD-DFT optimized structure of BromoAcroB, determination of quantum yield of singlet oxygen, fluorescence emission spectra and lifetimes of BromoAcroB and Br2BEt. See DOI: 10.1039/c9pp00162j
Rights and permissions
About this article
Cite this article
Lincoln, R., Van Kessel, A.T.M., Zhang, W. et al. A dormant BODIPY-acrolein singlet oxygen photosensitizer intracellularly activated upon adduct formation with cysteine residues. Photochem Photobiol Sci 18, 2003–2011 (2019). https://doi.org/10.1039/c9pp00162j
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00162j