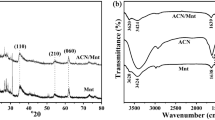
Abstract
Flavylium cations serve as models for the chemical and photochemical reactivity of anthocyanins, the natural plant pigment responsible for many of the red, blue and purple colors of fruits and flowers. Likewise, pyranoflavylium cations serve as models of the fundamental chromophoric moiety of pyra-noanthocyanins, molecules that can form from reactions of grape anthocyanins in red wines during their maturation. In the present work, hybrid pigments are prepared by the adsorption of a series of five synthetic flavylium cations (FL) and five synthetic pyranoflavylium cations (PFL) on sepiolite clay (SEP). The FL are smaller in size than the PFL, but both can in principle fit into the tunnels and/or external grooves (with dimensions of 3.7 × 10.6 Å) of SEP. Measurements of the fluorescence quantum yields of the adsorbed dyes indicate that they are at least as fluorescent as in acidic acetonitrile solution, and in a few cases substantially more fluorescent. The observation of biexponential fluorescence decays is consistent with emission from dye molecules adsorbed at two distinct sites, presumably tunnels and grooves. These hybrid materials also have improved properties in terms of stability of the color in contact with pH 10 aqueous solution and resistance to thermal degradation of the dye. SEP thus appears to be a promising substrate for the development of highly fluorescent flavylium or pyranoflavylium cation-derived hybrid pigments with improved color and thermal stability.
Similar content being viewed by others
References
M. F. Brigatti, E. Galan and B. K. G. Theng, Developments in Clay Science, in, Chapter 2, Handbook of Clay Science, 2006, vol. 1, pp. 19–86.
E. Galan, Properties and applications of palygorskite-sepiolite clays, Clay Miner., 1996, 31, 443–453.
V. Martínez-Martínez, C. Corcóstegui, J. Bañuelos Prieto, L. Gartzia, S. Salleres and I. López Arbeloa, Distribution and orientation study of dyes intercalated into single sepiolite fibers. A confocal fluorescence microscopy approach, J. Mater. Chem., 2011, 21, 269–276.
E. Ruiz-Hitzky, Molecular access to intracrystalline tunnels of sepiolite, J. Mater. Chem., 2001, 11, 86–91.
D. Karataş, A. Tekin and M. S. Çelik, Density functional theory computation of organic compound penetration into sepiolite tunnels, Clays Clay Miner., 2017, 65, 1–13.
S. Ovarlez, F. Giulieri, A. Chaze and F. Delamare, The Incorporation of Indigo Molecules in Sepiolite Tunnels, Chem. –, Eur. J., 2009, 15, 11326–11332.
M. Sánchez Del Río and P. Martinetto, Synthesis and Acid Resistance of Maya Blue Pigment, Archaeometry, 2006, 48, 115–130.
E. Arnold and J. Branden, The first direct evidence for the production of Maya Blue: rediscovery of a technology, Antiquity, 2008, 82, 151–164.
G. Chiari, R. Giustetto, J. Druzik, E. Doehne and G. Ricchiardi, Pre-columbian nanotechnology: Reconciling the mysteries of the maya blue pigment, Appl. Phys. A: Mater. Sci. Process., 2008, 90, 3–7.
A. Tilocca and E. Fois, The color and stability of maya blue: TDDFT calculations, J. Phys. Chem. C, 2009, 113, 8683–8687.
R. Giustetto, F. X. Llabrés I Xamena, G. Ricchiardi, S. Bordiga, A. Damin, R. Gobetto and M. R. Chierotti, Maya blue: A computational and spectroscopic study, J. Phys. Chem. B, 2005, 109, 19360–19368.
R. Giustetto, D. Levy and G. Chiari, Crystal structure refinement of Maya Blue pigment prepared with deuterated indigo, using neutron powder diffraction, Eur. J. Mineral., 2006, 18, 629–640.
R. Giustetto, D. Levy, O. Wahyudi, G. Ricchiardi and J. G. Vitillo, Crystal structure refinement of a sepiolite/indigo Maya Blue pigment using molecular modelling and synchrotron diffraction, Eur. J. Mineral., 2011, 23, 449–466.
E. Lima, A. Guzmán, M. Vera, J. L. Rivera and J. Fraissard, Aged natural and synthetic Maya Blue-like pigments: What difference does it make?, J. Phys. Chem. C, 2012, 116, 4556–4563.
R. Giustetto, K. Seenivasan, F. Bonino, G. Ricchiardi, S. Bordiga, M. R. Chierotti and R. Gobetto, Host/guest interactions in a sepiolite-based maya blue pigment: A spectroscopic study, J. Phys. Chem. C, 2011, 115, 16764–16776.
A. Doménech, M. T. Doménech-Carbó and H. G. M. Edwards, On the interpretation of the Raman spectra of Maya Blue: A review on the literature data, J. Raman Spectrosc., 2011, 42, 86–96.
C. Tsiantos, M. Tsampodimou, G. H. Kacandes, M. Sánchez Del Río, V. Gionis and G. D. Chryssikos, Vibrational investigation of indigo-palygorskite association (s) in synthetic Maya blue, J. Mater. Sci., 2012, 47, 3415–3428.
A. Doménech-Carbó, M. T. Doménech-Carbó, F. M. Valle-Algarra, M. E. Domine and L. Osete-Cortina, On the dehydroindigo contribution to Maya Blue, J. Mater. Sci., 2013, 48, 7171–7183.
G. Chiari, R. Giustetto and G. Ricchiardi, Crystal structure refinements of palygorskite and Maya Blue from molecular modelling and powder synchrotron diffraction, Eur. J. Mineral., 2003, 15, 21–33.
R. Giustetto, O. Wahyudi, I. Corazzari and F. Turci, Chemical stability and dehydration behavior of a sepiolite/indigo Maya Blue pigment, Appl. Clay Sci., 2011, 52, 41–50.
Y. Zhang, J. Dong, H. Sun, B. Yu, Z. Zhu, J. Zhang and A. Wang, Solvatochromic Coatings with Self-Cleaning Property from Palygorskite@Polysiloxane/Crystal Violet Lactone, ACS Appl. Mater. Interfaces, 2016, 8, 27346–27352.
Y. Zhang, J. Zhang and A. Wang, From Maya blue to biomimetic pigments: durable biomimetic pigments with self-cleaning property, J. Mater. Chem. A, 2016, 4, 901–907.
S. Wu, J. Huang, H. Cui, T. Ye, F. Hao, W. Xiong, P. Liu and H. Luo, Preparation of organic–inorganic hybrid methylene blue polymerized organosilane/sepiolite pigments with superhydrophobic and self-cleaning properties, Text. Res. J., DOI: 10.1177/0040517519829924.
Y. Zhang, J. Zhang and A. Wang, Facile preparation of stable palygorskite/methyl violet@SiO2 “Maya Violet” pigment, J. Colloid Interface Sci., 2015, 457, 254–263.
L. Fan, Y. Zhang, J. Zhang and A. Wang, Facile preparation of stable palygorskite/cationic red X-GRL@SiO 2 “Maya Red” pigments, RSC Adv., 2014, 4, 63485–63493.
Y. Zhang, W. Wang, B. Mu, Q. Wang and A. Wang, Effect of grinding time on fabricating a stable methylene blue/palygorskite hybrid nanocomposite, Powder Technol., 2015, 280, 173–179.
Q. Wang, B. Mu, Y. Zhang, J. Zhang and A. Wang, Palygorskite-based hybrid fluorescent pigment: Preparation, spectroscopic characterization and environmental stability, Microporous Mesoporous Mater., 2016, 224, 107–115.
Y. Zhang, L. Fan, H. Chen, J. Zhang, Y. Zhang and A. Wang, Learning from ancient Maya: Preparation of stable palygorskite/methylene blue@SiO2 Maya Blue-like pigment, Microporous Mesoporous Mater., 2015, 211, 124–133.
R. Giustetto, J. G. Vitillo, I. Corazzari and F. Turci, Evolution and reversibility of host/guest interactions with temperature changes in a methyl red@palygorskite polyfunctional hybrid nanocomposite, J. Phys. Chem. C, 2014, 118, 19322–19337.
R. Giustetto and O. Wahyudi, Sorption of red dyes on palygorskite: Synthesis and stability of red/purple Mayan nanocomposites, Microporous Mesoporous Mater., 2011, 142, 221–235.
S. Wu, Z. Duan, F. Hao, S. Xiong, W. Xiong, Y. Lv, P. Liu and H. Luo, Preparation of acid-activated sepiolite/Rhodamine B@SiO2 hybrid fluorescent pigments with high stability, Dyes Pigm., 2017, 137, 395–402.
S. Wu, H. Cui, C. Wang, F. Hao, P. Liu and W. Xiong, In situ self-assembled preparation of the hybrid nanopigment from raw sepiolite with excellent stability and optical performance, Appl. Clay Sci., 2018, 163, 1–9.
G. Tian, W. Wang, D. Wang, Q. Wang and A. Wang, Novel environment friendly inorganic red pigments based on attapulgite, Powder Technol., 2017, 315, 60–67.
Q. Wang, B. Mu, W. Wang, A. Wang and G. Tian, Costefficient, vivid and stable red hybrid pigments derived from naturally available sepiolite and halloysite, Ceram. Int., 2016, 43, 1862–1869.
H. Li, A. Zhang, A. Wang, B. Mu and X. An, Cobalt blue hybrid pigment doped with magnesium derived from sepiolite, Appl. Clay Sci., 2018, 157, 111–120.
L. F. Casassa and J. F. Harbertson, Extraction, Evolution, and Sensory Impact of Phenolic Compounds During Red Wine Maceration, Annu. Rev. Food Sci. Technol., 2014, 5, 83–109.
J. Heras-Roger, C. Díaz-Romero and J. Darias-Martín, What Gives a Wine Its Strong Red Color? Main Correlations Affecting Copigmentation, J. Agric. Food Chem., 2016, 64, 6567–6574.
K. Tang, T. Liu, Y. Han, Y. Xu and J. M. Li, The Importance of Monomeric Anthocyanins in the Definition of Wine Colour Properties, S. Afr. J. Enol. Vitic., 2017, 38, 1–10.
R. Brouillard, S. Chassaing and A. Fougerousse, Why are grape/fresh wine anthocyanins so simple and why is it that red wine color lasts so long?, Phytochemistry, 2003, 64, 1179–1186.
M. Schwarz, T. C. Wabnitz and P. Winterhalter, Pathway Leading to the Formation of Anthocyanin−Vinylphenol Adducts and Related Pigments in Red Wines, J. Agric. Food Chem., 2003, 51, 3682–3687.
H. Fulcrand, M. Dueñas, E. Salas and V. Cheynier, Phenolic reactions during winemaking and aging, Am. J. Enol. Vitic., 2006, 57, 289–297.
A. Marquez, M. P. Serratosa and J. Merida, Pyranoanthocyanin Derived Pigments in Wine: Structure and Formation during Winemaking, J. Chem., 2013, 2013, 1–15.
J. Oliveira, N. Mateus and V. de Freitas, Previous and recent advances in pyranoanthocyanins equilibria in aqueous solution, Dyes Pigm., 2014, 100, 190–200.
M. Figueiredo-González, B. Cancho-Grande, J. Simal-Gándara, N. Teixeira, N. Mateus and V. De Freitas, The phenolic chemistry and spectrochemistry of red sweet winemaking and oak-aging, Food Chem., 2014, 152, 522–530.
G. T. M. Silva, C. P. Silva, M. H. Gehlen, J. Oake, C. Bohne and F. H. Quina, Organic/inorganic hybrid pigments from flavylium cations and palygorskite, Appl. Clay Sci., 2018, 162, 478–486.
A. A. Freitas, A. A. L. Maçanita and F. H. Quina, Improved analysis of excited state proton transfer kinetics by the combination of standard and convolution methods. Photochem. Photobiol. Sci., 2013, 12, 902–910.
B. Held, H. Tang, P. Natarajan, C. P. Silva, V. O. Silva, C. Bohne and F. H. Quina, Cucurbit[7]uril inclusion complexation as a supramolecular strategy for color stabilization of anthocyanin model compounds, Photochem. Photobiol. Sci., 2016, 15, 752–757.
C. P. Silva, R. M. Pioli, L. Liu, S. Zheng, M. Zhang, G. T. M. Silva, V. M. T. Carneiro and F. H. Quina, Improved Synthesis of Analogues of Red Wine Pyranoanthocyanin Pigments, ACS Omega, 2018, 3, 954–960.
J. Madejová, W. P. Gates and S. Petit, Developments in Clay Science, in, Chapter 5, Handbook of Clay Science, 2017, vol. 8, pp. 107–149.
C. He, E. Makovicky and B. Osbæck, Thermal treatment and pozzolanic activity of sepiolite, Appl. Clay Sci., 1996, 10, 337–349.
M. K.Wang, P. C. Tseng, S. S. Chang, D. T. Ray, Y. H. Shau, Y. W. Shen, R. C. Chen and P. N. Chiang, Origin and mineralogy of sepiolite and palygorskite from the tuluanshan formation, eastern taiwan, Clays Clay Miner., 2009, 57, 521–530.
V. Gionis, G. H. Kacandes, I. D. Kastritis and G. D. Chryssikos, Combined near-infrared and x-ray diffraction investigation of the octahedral sheet composition of palygorskite, Clays Clay Miner., 2007, 55, 543–553.
C. Vogt, J. Lauterjung and R. X. Fischer, Investigation of the Clay Fraction (<2 μm) of the Clay Minerals Society Reference Clays, Clays Clay Miner., 2002, 50, 388–400.
M. R. Weir, W. Kuang, G. A. Facey and C. Detellier, Solidstate nuclear magnetic resonance study of sepiolite and partially dehydrated sepiolite, Clays Clay Miner., 2002, 50, 240–247.
B. Hubbard, W. Kuang, A. Moser, G. A. Facey and C. Detellier, Structural study of Maya Blue: Textural, thermal and solid-state multinuclear magnetic resonance characterization of the palygorskite-indigo and sepioliteindigo adducts, Clays Clay Miner., 2003, 51, 318–326.
Z. Li, C. A. Willms and K. Kniola, Removal of anionic contaminants using surfactant-modified palygorskite and sepiolite, Clays Clay Miner., 2003, 51, 445–451.
A. Gilchrist and J. Nobbs, Encycl. Spectrosc. Spectrom, 2nd edn, 1999, pp. 380–385.
E. P. Tomasini, E. S. Román and S. E. Braslavsky, Validation of fluorescence quantum yields for light-scattering powdered samples by laser-induced optoacoustic spectroscopy, Langmuir, 2009, 25, 5861–5868.
S. Tunç, O. Duman and R. Uysal, Electrokinetic and Rheological Behaviors of Sepiolite Suspensions in the Presence of Poly(acrylic acid sodium salt)s, Polyacrylamides, and Poly(ethylene glycol)s of Different Molecular Weights, J. Appl. Polym. Sci., 2008, 109, 1850–1860.
H. Shariatmadari, A. R. Mermut and M. B. Benke, Sorption of selected cationic and neutral organic molecules on palygorskite and sepiolite, Clays Clay Miner., 1999, 47, 44–53.
F. L. Arbeloa, T. L. Arbeloa and I. L. Arbeloa, Spectroscopy of Rhodamine 6G Adsorbed on Sepiolite Aqueous Suspensions, J. Colloid Interface Sci., 2002, 187, 105–112.
A. A. Freitas, C. P. Silva, G. T. M. Silva, A. L. Maçanita and F. H. Quina, Ground- and Excited-State Acidity of Analogs of Red Wine Pyranoanthocyanins, Photochem. Photobiol., 2018, 94, 1086–1091.
M. S. Çelik, in, Clay Surfaces: Fundamentals and Applications, 2004, vol. 1, pp. 57–89.
M. Alkan, Ö. Demirbaş and M. Doğan, Electrokinetic properties of sepiolite suspensions in different electrolyte media, J. Colloid Interface Sci., 2005, 281, 240–248.
F. Siddique, C. P. Silva, G. T. M. Silva, H. Lischka, F. H. Quina and A. J. A. Aquino, The electronic transitions of analogs of red wine pyranoanthocyanin pigments, Photochem. Photobiol. Sci., 2019, 18, 45–53.
Y. Kohno, Y. Shibata, N. Oyaizu, K. Yoda, M. Shibata and R. Matsushima, Stabilization of flavylium dye by incorporation into the pore of protonated zeolites, Microporous Mesoporous Mater., 2008, 114, 373–379.
Y. Kohno, R. Kinoshita, S. Ikoma, K. Yoda, M. Shibata, R. Matsushima, Y. Tomita, Y. Maeda and K. Kobayashi, Stabilization of natural anthocyanin by intercalation into montmorillonite, Appl. Clay Sci., 2009, 42, 519–523.
Y. Kohno, S. Tsubota, Y. Shibata, K. Nozawa, K. Yoda, M. Shibata and R. Matsushima, Enhancement of the photostability of flavylium dye adsorbed on mesoporous silicate, Microporous Mesoporous Mater., 2008, 116, 70–76.
G. T. M. Silva, S. S. Thomas, C. P. Silva, J. C. Schlothauer, M. S. Baptista, A. A. Freitas, C. Bohne and F. H. Quina, Triplet Excited States and Singlet Oxygen Production by Analogs of Red Wine Pyranoanthocyanins, Photochem. Photobiol., 2018, 95, 176–182.
A. A. Freitas, C. P. Silva, G. T. M. Silva, A. L. Maçanita and F. H. Quina, From vine to wine: photophysics of a pyranoflavylium analog of red wine pyranoanthocyanins, Pure Appl. Chem., 2017, 89, 1761–1767.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available. See DOI: 10.1039/c9pp00141g
Rights and permissions
About this article
Cite this article
Silva, G.T.M., da Silva, K.M., Silva, C.P. et al. Highly fluorescent hybrid pigments from anthocyanin- and red wine pyranoanthocyanin-analogs adsorbed on sepiolite clay. Photochem Photobiol Sci 18, 1750–1760 (2019). https://doi.org/10.1039/c9pp00141g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00141g