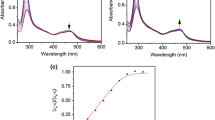
Abstract
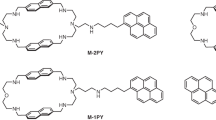
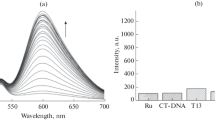
(E)-2-[1'-((Diphenylamino)styryl)quinolizinium (3a) and 2,2'-{(phenylimino)-bis[(E)-1″,1″′-styryl]}-bis[quin-olizinium] (3b) were synthesized, and their interactions with duplex DNA and quadruplex DNA were investigated with a particular focus on their ability to operate as DNA-sensitive fluorescent probes. Due to the significantly different size and steric demand of these quinolizinium derivatives they exhibit different binding modes. Thus, 3a intercalates into duplex DNA and binds through n stacking to quadruplex DNA, whereas 3b favours groove binding to both DNA forms. The emission intensity of these compounds is very low in aqueous solution, but it increases drastically upon association with duplex DNA by a factor of 11 (3a) and >100 (3b) and with quadruplex DNA by a factor of >100 (3a) and 10 (3b), with emission bands between 600 and 750 nm.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. B. Dervan, Molecular recognition of DNA by small molecules, Bioorg. Med. Chem., 2001, 9, 2215–2235
J. Sheng, J. Gan and Z. Huang, Structure-based DNA-targeting strategies with small molecule ligands for drug discovery, Med. Res. Rev., 2013, 33, 1119–1173
J. Wang, Survey and summary: From DNA biosensors to gene chips, Nucleic Acids Res., 2000, 28, 3011–3016
L. H. Hurley, DNA and its associated processes as targets for cancer therapy, Nat. Rev. Cancer, 2002, 2, 188–200
P. G. Baraldi, A. Bovero, F. Fruttarolo, D. Preti, M. A. Tabrizi, M. G. Pavani and R. Romagnoli, DNA minor groove binders as potential antitumor and antimicrobial agents, Med. Res. Rev., 2004, 24, 475–528.
N. C. Seeman and H. F. Sleiman, DNA nanotechnology, Nat. Rev. Mater., 2017, 3, 17068.
Y. Jung and S. J. Lippard, Direct Cellular Responses to Platinum-Induced DNA Damage, Chem. Rev., 2007, 107, 1387–1407
J. Wu, Y. Zou, C. Li, W. Sicking, I. Piantanida, T. Yi and C. Schmuck, A Molecular Peptide Beacon for the Ratiometric Sensing of Nucleic Acids, J. Am. Chem. Soc., 2012, 134, 1958–1961
H. Song, J. T. Kaiser and J. K. Barton, Crystal structure of A-[Ru(bpy)2dppz]2+ bound to mismatched DNA reveals side-by-side metalloinsertion and intercalation, Nat. Chem., 2012, 4, 615–620.
H. Kobayashi, M. Ogawa, R. Alford, P. L. Choyke and Y. Urano, New Strategies for Fluorescent Probe Design in Medical Diagnostic Imaging, Chem. Rev., 2010, 110, 2620–2640
D.-L. Ma, H.-Z. He, K.-H. Leung, H.-J. Zhong, D. S.-H. Chan and C.-H. Leung, Label-free luminescent oligonucleotide-based probes, Chem. Soc. Rev., 2013, 42, 3427–3440.
Y. V. Suseela, N. Narayanaswamy, S. Pratihar and T. Govindaraju, Far-red fluorescent probes for canonical and non-canonical nucleic acid structures: current progress and future implications, Chem. Soc. Rev., 2018, 47, 1098–1131
S. J. Smith, C. R. Nemr and S. O. Kelley, Chemistry-Driven Approaches for Ultrasensitive Nucleic Acid Detection, J. Am. Chem. Soc., 2017, 139, 1020–1028
S. Manna and S. G. Srivatsan, Fluorescence-based tools to probe G-quadruplexes in cell-free and cellular environments, RSC Adv., 2018, 8, 25673–25694
D. Wu, A. C. Sedgwick, T. Gunnlaugsson, E. U. Akkaya, J. Yoon and T. D. James, Fluorescent chemosensors: the past, present and future, Chem. Soc. Rev., 2017, 46, 7105–7123.
P. Prento, A contribution to the theory of biological staining based on the principles for structural organization of biological macromolecules, Biotech. Histochem., 2001, 76, 137–161
J. B. LePecq and C. A. Paoletti, A fluorescent complex between ethidium bromide and nucleic acids. Physical-chemical characterization, J. Mol. Biol., 1967, 27, 87–106
L. S. Lermann, Structural considerations in the interaction of DNA and acridines, J. Mol. Biol., 1961, 3, 18–30
A. Slama-Schwok, J. Jazwinski, A. Bere, T. Montenay-Garestier, M. Rougée, C. Hélène and J. M. Lehn, Interactions of the Dimethyldiazaperopyrenium Dication with Nucleic Acids. 1. Binding to Nucleic Acid Components and to Single-Stranded Polynucleotides and Photocleavage of Single-Stranded Oligonucleotides, Biochemistry, 1989, 28, 3227–3234.
S. Neidle, Quadruplex nucleic acids as targets for anticancer therapeutics, Nat. Rev. Chem., 2017, 1, UNSP 0041
D.-L. Ma, Z. Zhang, M. Wang, L. Lu, H. J. Zhong and C. -H. Leung, Recent Developments in G-Quadruplex Probes, Chem. Biol., 2015, 22, 812–828
S. Neidle, Quadruplex Nucleic Acids as Novel Therapeutic Targets, J. Med. Chem., 2016, 59, 5987–6011.
Y. Xu, Chemistry in human telomere biology: structure, function and targeting of telomere DNA/RNA, Chem. Soc. Rev., 2011, 40, 2719–2740
A. Ali and S. Bhattacharya, DNA binders in clinical trials and chemotherapy, Bioorg. Med. Chem., 2014, 22, 4506–4521
P. Murat, Y. Singh and E. Defrancq, Methods for investigating G-quadruplex DNA/ ligand interactions, Chem. Soc. Rev., 2011, 40, 5293–5307
E. Largy, A. Granzhan, F. Hamon, D. Verga and M. P. Teulade-Fichou, Visualizing the quadruplex: from fluorescent ligands to light-up probes, Top. Curr. Chem., 2013, 330, 111–177.
R. W. Sinkeldam, N. J. Greco and Y. Tor, Fluorescent Analogs of Biomolecular Building Blocks: Design, Properties, and Applications, Chem. Rev., 2010, 110, 2579–2619
T. Deligeorgiev, A. Vasilev, S. Kaloyanova and J. J. Vaquero, Styryl dyes - synthesis and applications during the last 15 years, Color. Technol., 2010, 126, 55–80
G. R. Rosania, J. W. Lee, L. Ding, H. S. Yoon and Y. T. Chang, Combinatorial Approach to Organelle-Targeted Fluorescent Library Based on the Styryl Scaffold, J. Am. Chem. Soc., 2003, 125, 1130–1131.
H. Özhalici-Ünal, C. L. Pow, S. A. Marks, L. D. Jesper, G. L. Silva, N. I. Shank, E. W. Jones, J. M. Burnette, P. B. Berget and B. A. Armitage, A Rainbow of Fluoromodules: A Promiscuous scFv Protein Binds to and Activates a Diverse Set of Fluorogenic Cyanine Dyes, J. Am. Chem. Soc., 2008, 130, 12620–12621
A. Ajayaghosh, E. Arunkumar and J. Daub, A Highly Specific Ca2+-Ion Sensor: Signaling by Exciton Interaction in a Rigid-Flexible-Rigid Bichromophoric “H” Foldamer, Angew. Chem., Int. Ed., 2002, 41, 1766–1769.
R. W. Dirks and H. J. Tanke, Styryl Molecules Light-Up RNAs, Chem. Biol., 2006, 13, 559–561
C. V. Kumar, R. S. Turner and E. H. Asuncion, Groove binding of a styryl cyanine dye to the DNA double helix: the salt effect, J. Photochem. Photobiol., A, 1993, 74, 231–238
N. Akbay, M. Y. Losytskyy, V. B. Kovalska, A. O. Balanda and S. M. Yarmoluk, The Mechanism of Benzothiazole Styryl cyanine Dyes Binding with dsDNA: Studies by Spectral-Luminescent Methods, J. Fluoresc., 2008, 18, 139–147
N. Nizomov, E. N. Kurtaliev, S. N. Nizamov and G. Khodjayev, Spectral-luminescent study of the interaction of some styrylcyanine dyes with bovine serum albumin and DNA in aqueous solutions, J. Mol. Struct., 2009, 936, 199–205.
V. B. Kovalska, D. V. Kryvorotenko, A. O. Balanda, M. Y. Losytskyy, V. P. Tokar and S. M. Yarmoluk, Fluorescent homodimer styrylcyanines: synthesis and spectral-luminescent studies in nucleic acids and protein complexes, Dyes Pigm., 2005, 67, 47–54
J.-S. Lee, Y. K. Kim, M. Vendrel and Y.-T. Chang, Diversity-oriented fluorescence library approach for the discovery of sensors and probes, Mol. BioSyst., 2009, 5, 411–421
D. V. Berdnikova, O. A. Fedorova, E. V. Tulyakova, H. Li, S. Kölsch and H. Ihmels, Interaction of Crown Ether-Annelated Styryl Dyes with Double-Stranded DNA, Photochem. Photobiol., 2015, 91, 723–731
A. Mazzoli, B. Carlotti, G. Consiglio, C. G. Fortuna, G. Miolo and A. Spalletti, Photobehaviour of methyl-pyridinium and quinolinium iodide derivatives, free and complexed with DNA. A case of bisintercalation, Photochem. Photobiol. Sci., 2014, 13, 939–950
M.-Q. Wang, J. Xu, L. Zhang, Y. Liao, H. Wei, Y.-Y. Yin, Q. Liu and Y. Zhang, Tuning the selectivity of N-alkylated styrylquinolinium dyes for sensing of G-quadruplex DNA, Bioorg. Med. Chem., 2019, 27, 552–559.
Q. Li, Y. Kim, J. Namm, A. Kulkarni, G. R. Rosania, Y. H. Ahn and Y. T. Chang, RNA-selective, live cell imaging probes for studying nuclear structure and function, Chem. Biol., 2006, 13, 615–623
M. Q. Wang, S. Liu, C. P. Tang, A. Raza, S. Li, L. X. Gao, J. Sun and S. P. Guo, Flexible amine-functionalized triphenylamine derivative as a fluorescent “light-up” probe for G-quadruplex DNA, Dyes Pigm., 2017, 136, 78–84
A. Manna and S. Chakravorti, Modification of a Styryl Dye Binding Mode with Calf Thymus DNA in Vesicular Medium: From Minor Groove to Intercalative, J. Phys. Chem. B, 2012, 116, 5226–5233
Z.-Q. Liu, S.-T. Zhuo, J.-H. Tan, T.-M. Ou, D. Li, L.-Q. Gu and Z.-S. Huang, Facile syntheses of disubstituted bis(vinyl-quinolinium)benzene derivatives as G-quadruplex DNA binders, Tetrahedron, 2013, 69, 4922–4932
Y.-J. Lu, D. -P. Hu, K. Zhang, W.-L. Wong and C.-F. Chow, New pyridinium-based fluorescent dyes: A comparison of symmetry and side-group effects on G-Quadruplex DNA binding selectivity and application in live cell imaging, Biosens. Bioelectron., 2016, 81, 373–381.
A. Granzhan and H. Ihmels, Playing Around with the Size and Shape of Quinolizinium-Derivatives: Versatile Ligands for Duplex, Triplex, Quadruplex and Abasic Site-Containing DNA, Synlett, 2016, 27, 1775–1793.
A. Granzhan, H. Ihmels and M. Tian, The benzo[b]quinol-izinium ion as a water-soluble platform for the fluorimetric detection of biologically relevant analytes, ARKIVOC, 2015, vi, 494.
L. Chang, C. Liu, S. He, Y. Lu, S. Zhang, L. Zhao and X. Zeng, Novel styryldehydropyridocolinium derivative as turn-on fluorescent probe for DNA detection, Sens. Actuators, B, 2014, 202, 483–488
H. Yao, L. Chang, C. Liu, X. Jiao, S. He, H. Liu and X. Zeng, A Novel Styryldehydropyridocolinium Homodimer: Synthesis and Fluorescence Properties Upon Interaction with DNA, J. Fluoresc., 2015, 25, 1637–1643
E. Zacharioudakis, T. Caňeque, R. Custodio, S. Müller, A. M. Cuadro, J. J. Vaquero and R. Rodriguez, Quinolizinium as a new fluorescent lysosomotropic probe, Bioorg. Med. Chem. Lett., 2017, 27, 203–207
M. A. Martín, B. del Castillo, J. Ezquerra and J. Alvarez-Builla, Quinolizinium Salts as Fluorescent Probes for N-Nucleophiles, Anal. Chim. Acta, 1985, 170, 89–94
P. Martin, M. A. Martin, B. del Castillo and I. Cayre, Polarity effect on fluorescence of styryl derivatives of quinolizinium salts in micellar media, Anal. Chim. Acta, 1988, 205, 129–137.
Z. J. Ning and H. Tian, Triarylamine: a promising core unit for efficient photovoltaic materials, Chem. Commun., 2009, 37, 5483–5495
R. Chennoufi, H. Bougherara, N. Gagey-Eilstein, B. Dumat, E. Henry, F. Subra, S. Bury-Moné, F. Mahuteau-Betzer, P. Tauc, M. P. Teulade-Fichou and E. Deprez, Mitochondria-targeted Triphenylamine Derivatives Activatable by Two-Photon Excitation for Triggering and Imaging Cell Apoptosis, Sci. Rep., 2016, 6, 21458.
M. Q. Wang, W.-X. Zhu, Z.-Z. Song, S. Li and Y.-Z. Zhang, A triphenylamine-based colorimetric and fluorescent probe with donor-bridge-acceptor structure for detection of G-quadruplex DNA, Bioorg. Med. Chem. Lett., 2015, 25, 5672–5676.
R. S. Juang, H. W. Wen, M. T. Chen and P. C. Yang, Enhanced sensing ability of fluorescent chemosensors with triphenylamine-functionalized conjugated polyfluorene, Sens. Actuators, B, 2016, 231, 399–411
S. Koersten and G. J. Mohr, Star-Shaped Tripodal Chemosensors for the Detection of Aliphatic Amines, Chem. - Eur. J., 2011, 17, 969–975
T. Liu, F. Huo, C. Yin, J. Li, J. Chao and Y. Zhang, A triphenylamine as a fluorophore and male-imide as a bonding group selective turn-on fluorescent imaging probe for thiols, Dyes Pigm., 2016, 128, 209–214
M. Q. Wang, L. X. Gao, Y. F. Yang, X. N. Xiong, Z. Y. Zheng, S. Li, Y. Wu and L. L. Ma, A triphenylamine derivative as a naked-eye and light-up fluorescent probe for G-quadruplex DNA, Tetrahedron Lett., 2016, 57, 5042–5046
I. Pont, J. González-García, M. Inclán, M. Reynolds, E. Delgado-Pinar, M. T. Albelda, R. Vilar and E. García-Espaňa, Aza-Macrocyclic Triphenylamine Ligands for G-Quadruplex Recognition, Chem. - Eur. J., 2018, 24, 10850–10858.
L. F. Tietze and U. Beifuss, in Comprehensive Organic Synthesis, ed. B.M. Trost, Pergamon, Oxford, 1991.
A. Richards and T. S. Stevens, Synthesis and properties of dehydropyridocolinium salts, J. Chem. Soc., 1958, 3067–3073
O. F. Beumel Jr., W. N. Smith and B. Rybalka, Preparation of 2- and 4-Picolyllithium, Synthesis, 1974, 43–45.
G. Marcelo, S. Pinto, T. Caňeque, I. F. A. Mariz, A. M. Cuadro, J. J. Vaquero, J. M. G. Martinho and E. M. S. Maçôas, Nonlinear Emission of Quinolizinium- Based Dyes with Application in Fluorescence Lifetime Imaging, J. Phys. Chem. A, 2015, 119, 2351–2362.
F. Arbeloa, P. R. Ojeda and I. Arbeloa, Flourescence selfquenching of the molecular forms of Rhodamine B in aqueous and ethanolic solutions, J. Lumin., 1989, 44, 105–112.
M. A. Martin, M. Ballesteros and B. D. Castillo, The influence of solvent polarity and viscosity on fluorescence of quinolizinium salts, Anal. Chim. Acta, 1985, 170, 95–100.
M. Shortreed, R. Kopelman, M. Kuhn and B. Hoyland, Fluorescent Fiber-Optic Calcium Sensor for Physiological Measurements, Anal. Chem., 1996, 68, 1414–1418
W. Lin, L. Yuan, Z. Cao, Y. Feng and L. Long, A Sensitive and Selective Fluorescent Thiol Probe in Water Based on the Conjugate 1,4-Addition of Thiols to a, ß-Unsaturated Ketones, Chem. - Eur. J., 2009, 15, 5096–5103.
F. H. Stootman, D. M. Fisher, A. Rodger and J. R. Aldrich-Wright, Analyst, 2006, 131, 1145.
E. M. Rezler, J. Seenisamy, S. Bashyam, M. Y. Kim, E. White, D. Wilson and L. H. Hurley, Telomestatin and Diseleno Sapphyrin Bind Selectively to Two Different Forms of the Human Telomeric G-Quadruplex Structure, J. Am. Chem. Soc., 2005, 127, 9439–9447.
O. van den Berg, W. F. Jager and S. J. Picken, 7-Dialkylamino-1-alkylquinolinium Salts: Highly Versatile and Stable Fluorescent Probes, J. Org. Chem., 2006, 71, 2666–2676
A. Granzhan, H. Ihmels and G. Viola, 9-Donor-Substituted Acridizinium Salts: Versatile Environment-Sensitive Fluorophores for the Detection of Biomacromolecules, J. Am. Chem. Soc., 2007, 129, 1254–1267.
B. Heyne, Self-assembly of organic dyes in supramolecular aggregates, Photochem. Photobiol. Sci., 2016, 15, 1103–1114.
M. A. Haidekker and E. A. Theodorakis, Molecular rotors—fluorescent biosensors for viscosity and flow, Org. Biomol. Chem., 2007, 5, 1669–1678
A. K. Chibisov, G. V. Zakharova and H. Görner, Effects of substituents in the polymethine chain on the photoprocesses in indodicarbocyanine dyes, J. Chem. Soc., Faraday Trans., 1996, 92, 4917–4925
M. S. A. Abdel-Mottaleb, R. O. Loutfy and R. Lapouyade, Non-radiative deactivation channels of molecular rotors, J. Photochem. Photobiol., A, 1989, 48, 87–93.
Y. Hong, J. W. Y. Lam and B. Z. Tang, Aggregation-induced emission, Chem. Soc. Rev., 2011, 40, 5361–5388.
W. Sbliwa, G. Matusiak and B. Bachowska, An Overview of the Optical and Electrochemical Methods for Detection of DNA - Drug Interactions, Croat. Chem. Acta, 2006, 79, 513.
B. Norden, A. Rodger and T. Dafforn, Linear Dichroism and Circular Dichroism, RSC Publishing, Cambridge, 2010
T. Šmidlehner, I. Piantanida and G. Pescitelli, Polarization spectroscopy methods in the determination of interactions of small molecules with nucleic acids - tutorial, Beilstein J. Org. Chem., 2018, 14, 84–105.
B. Norden and T. Kurucsev, Analysing DNA complexes by circular and linear dichroism, J. Mol. Recognit., 1994, 7, 141–156.
N. Narayanaswamy, S. Das, P. K. Samanta, K. Banu, G. P. Sharma, N. Mondal, S. K. Dhar, S. K. Pati and T. Govindaraju, Sequence-specific recognition of DNA minor groove by an NIR-fluorescence switch-on probe and its potential applications, Nucleic Acids Res., 2015, 43, 8651–8663
X. B. Fu, D. D. Liu, Y. Lin, W. Hu, Z. W. Mao and X. Y. Le, Water-soluble DNA minor groove binders as potential chemotherapeutic agents: synthesis, characterization, DNA binding and cleavage, antioxidation, cytotoxicity and HSA interactions, Dalton Trans., 2014, 43, 8721–8737.
S. Murphy, S. A. Bright, F. E. Poynton, T. McCabe, J. A. Kitchen, E. B. Veale, D. C. Williams and T. Gunnlaugsson, Synthesis, photophysical and cytotoxicity evaluations of DNA targeting agents based on 3-amino- I, 8-naphthalimide derived Tröger's bases, Org. Biomol. Chem., 2014, 12, 6610–6623.
H. Ihmels and L. Thomas, in Materials Science of DNA Chemistry, ed. J.I. Jin, CRC Press, Boca Raton, 2011, p. 49
Y. Xie, V. K. Tam and Y. Tor, in The chemical biology of nucleic acids, ed. G. Mayer, John Wiley & Sons, Chichester, 2010, p. 115
I. Haq, in Nucleic acids in chemistry and biology, ed. G.M. Blackburn, M.J. Gait, D. Loakes and D.M. Williams, Royal Society of Chemistry, Cambridge, 2006, p. 341.
S. Neidle, DNA minor-groove recognition by small molecules, Nat. Prod. Rep., 2001, 18, 291–309
Y. Liu, A. Kumar, S. Depauw, R. Nhili, M. H. David-Cordonnier, M. P. Lee, M. A. Ismail, A. A. Farahat, M. Say, S. Chackal-Catoen, A. Batista-Parra, S. Neidle, D. W. Boykin and W. D. Wilson, Water-Mediated Binding of Agents that Target the DNA Minor Groove, J. Am. Chem. Soc., 2011, 133, 10171–10183.
A. Tawani, S. K. Mishra and A. Kumar, Structural insight for the recognition of G-quadruplex structure at human c-myc promoter sequence by flavonoid Quercetin, Sci. Rep., 2017, 7, 3600.
N. Ranjan, K. F. Andreasen, S. Kumar, D. H. Volpe and D. P. Arya, Aminoglycoside binding to Oxytricha nova telomeric DNA, Biochemistry, 2010, 49, 9891–9903.
I. Manet, F. Manoli, B. Zambelli, G. Andreano, A. Masi, L. Cellai and S. Monti, Affinity of the anthracycline antitumor drugs Doxorubicin and Sabarubicin for human telomeric G-quadruplex structures, Phys. Chem. Chem. Phys., 2011, 13, 540–551
W.-B. Wu, S.-H. Chen, J.-Q. Hou, J. -H. Tan, T.-M. Ou, S.-L. Huang, D. Li, L.-Q. Gu and Z.-S. Huang, Disubstituted 2-phenyl-benzopyranopyr-imidine derivatives as a new type of highly selective ligands for telomeric G-quadruplex DNA, Org. Biomol. Chem., 2011, 9, 2975–2986
Y. J. Lu, T. M. Ou, J. H. Tan, J. Q. Hou, W. Y. Shao, D. Peng, N. Sun, X. D. Wang, W. B. Wu, X. Z. Bu, Z. S. Huang, D. L. Ma, K. Y. Wong and L. Q. Gu, 5-N-Methylated Quindoline Derivatives as Telomeric G-Quadruplex Stabilizing Ligands: Effects of 5-N Positive Charge on Quadruplex Binding Affinity and Cell Proliferation, J. Med. Chem., 2008, 51, 6381–6392.
H. Sun, Y. Tang, J. Xiang, G. Xu, Y. Zhang, H. Zhang and L. Xu, Spectroscopic studies of the interaction between quercetin and G-quadruplex DNA, Bioorg. Med. Chem. Lett., 2006, 16, 3586–3589
T. Yamashita, T. Uno and Y. Ishikawa, Stabilization of guanine quadruplex DNA by the binding of porphyrins with cationic side arms, Bioorg. Med. Chem., 2005, 13, 2423–2430.
A. K. Jain and S. Bhattacharya, Interaction of G-Quadruplexes with Nonintercalating Duplex-DNA Minor Groove Binding Ligands, Bioconjugate Chem., 2011, 22, 2355–2368.
R. N. Dsouza, U. Pischel and W. M. Nau, Fluorescent Dyes and Their Supramolecular Host/Guest Complexes with Macrocycles in Aqueous Solution, Chem. Rev., 2011, 111, 7941–7980.
K. Faulhaber, A. Granzhan, H. Ihmels, D. Otto, L. Thomas and S. Wells, Studies of the fluorescence light-up effect of amino-substituted benzo[b]quinolizinium derivatives in the presence of biomacromolecules, Photochem. Photobiol. Sci., 2011, 10, 1535–1545
R. Bortolozzi, H. Ihmels, L. Thomas, M. Tian and G. Viola, 9-(4 Dimethylaminophenyl) benzo[b]quinol-izinium: A Near-Infrared Fluorophore for the Multicolor Analysis of Proteins and Nucleic Acids in Living Cells, Chem. - Eur.J., 2013, 19, 8736–8741.
T. Caneque, A. M. Cuadro, J. Alvarez-Builla, J. Pérez-Moreno, K. Clays, O. Castano, J. L. Andrés and J. J. Vaquero, Novel charged NLO chromophores based on quinolizinium acceptor units, Dyes Pigm., 2014, 101, 116–121
B. Dumat, G. Bordeau, E. Faurel-Paul, F. Mahuteau-Betzer, N. Saettel, G. Metge, C. Fiorini-Debuisschert, F. Charra and M. P. Teulade-Fichou, DNA Switches on the Two-Photon Efficiency of an Ultrabright Triphenylamine Fluorescent Probe Specific of AT Regions, J. Am. Chem. Soc., 2013, 135, 12697–12706
G. Saielli, G. Scorrano, A. Bagno and A. Wakisaka, Solvation of Tetraalkylammonium Chlorides in Acetonitrile-Water Mixtures: Mass Spectrometry and Molecular Dynamics Simulations, ChemPhysChem, 2005, 6, 1307–1315
J. Li, K. Guo, J. Shen, W. Yang and M. Yin, A Difunctional Squarylium Indocyanine Dye Distinguishes Dead Cells through Diverse Staining of the Cell Nuclei/Membranes, Small, 2014, 10, 1351–1360.
Acknowledgements
We thank the University of Siegen and the Alexander von Humboldt Foundation (postdoctoral research fellowship for A. K. D.) for financial support. We thank Ms Jennifer Hermann and Ms Sandra Uebach for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 3.0 License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/3.0/.
About this article
Cite this article
Das, A.K., Ihmels, H. & Kölsch, S. Diphenylaminostyryl-substituted quinolizinium derivatives as fluorescent light-up probes for duplex and quadruplex DNA. Photochem Photobiol Sci 18, 1373–1381 (2019). https://doi.org/10.1039/c9pp00096h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00096h