Abstract
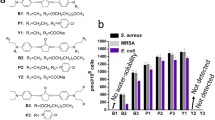
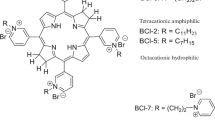
Light-mediated killing of pathogens by cationic photosensitizers (PS) is a promising antimicrobial approach avoiding resistance as being present upon the use of antibiotics. In this study we focused on the impact of the substituents in phenalen-1-one PS. Photodynamic efficacy depending on positively charged moieties including a primary aliphatic, quaternary aliphatic, aromatic ammonium and a guanidinium cation was investigated against Gram-positive and Gram-negative pathogens. Considering the altered steric demand and lipophilicity of these functional groups we deduced a structure–activity relationship. SAGUA was the most potent PS in this series reaching a maximum efficacy of ≥6log10 steps of bacteria killing at a concentration of 10 μM upon irradiation with blue light (20 mW cm−2) for 60 s (1.2 J cm−2) without exhibiting inherent dark toxicity. Its guanidinium moiety may be able to form strong bidentate and directional hydrogen bonds to carboxylate groups of bacterial surfaces in addition to ionic charge attraction. This may supplement fast and effective antimicrobial activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. M. Rossolini, E. Mantengoli, Clin. Microbiol. Infect., 2008, 14Suppl 6, 2–8.
T. Yamamoto, Y. Tamura, T. Yokota, Antimicrob. Agents Chemother., 1988, 32, 932–935.
S. P. Yazdankhah, A. A. Scheie, E. A. Høiby, B.-T. Lunestad, E. Heir, T. Ø. Fotland, K. Naterstad, H. Kruse, Microb. Drug Resist., 2006, 12, 83–90.
G. Jori, G. Roncucci, Adv. Clin. Exp. Med., 2006, 15, 421–426.
E. Alves, M. A. Faustino, M. G. Neves, A. Cunha, J. Tome, A. Almeida, Future Med. Chem., 2014, 6, 141–164.
T. Maisch, Lasers Med. Sci., 2007, 22, 83–91.
T. Maisch, S. Hackbarth, J. Regensburger, A. Felgenträger, W. Bäumler, M. Landthaler, B. Röder, J. Dtsch. Dermatol. Ges., 2011, 9, 360–366.
E. Alves, L. Costa, C. M. Carvalho, J. P. Tomé, M. A. Faustino, M. G. Neves, A. C. Tomé, J. A. Cavaleiro, A. Cunha, A. Almeida, BMC Microbiol., 2009, 9, 70.
M. Wainwright, J. Antimicrob. Chemother., 1998, 42, 13–28.
T. Maisch, J. Baier, B. Franz, M. Maier, M. Landthaler, R.-M. Szeimies, W. Bäumler, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 7223–7228.
A. Späth, C. Leibl, F. Cieplik, K. Lehner, J. Regensburger, K.-A. Hiller, W. Bäumler, G. Schmalz, T. Maisch, J. Med. Chem., 2014, 57, 5157–5168.
F. Cieplik, A. Späth, J. Regensburger, A. Gollmer, L. Tabenski, K.-A. Hiller, W. Bäumler, T. Maisch, G. Schmalz, Free Radical Biol. Med., 2013, 65, 477–487.
F. Cieplik, A. Pummer, J. Regensburger, K. A. Hiller, A. Spath, L. Tabenski, W. Buchalla, T. Maisch, Front. Microbiol., 2015, 6, 706.
T. J. Baker, M. Tomioka, M. Goodman, Org. Synth., 2002, 78, 91.
W. Bäumler, A. Felgenträger, K. Lehner, T. Maisch, J. Regensburger, F. Santarelli and A. Späth, WO Patent, WO/2012/113860, 2012.
A. A. Miles, S. S. Misra, J. O. Irwin, J. Hyg., 1938, 38, 732–749.
J. M. Boyce, D. Pittet, Infect. Control Hosp. Epidemiol., 2002, 23, 3–40.
Z. Rappoport and M. Frankel, CRC handbook of tables for organic compound identification, 1967.
S. E. Boiadjiev, D. A. Lightner, Chirality, 2000, 12, 204–215.
A. Späth, B. König, Tetrahedron, 2010, 66, 1859–1873.
A. Späth, B. König, Tetrahedron, 2010, 66, 6019–6025.
O. M. New, D. Dolphin, Eur. J. Org. Chem., 2009, 2675–2686.
F. Cieplik, A. Späth, C. Leibl, A. Gollmer, J. Regensburger, L. Tabenski, K.-A. Hiller, T. Maisch, G. Schmalz, Clin. Oral Invest., 2014, 18, 1763–1769.
J. Liebmann, M. Born, V. Kolb-Bachofen, J. Invest. Dermatol., 2010, 130, 259–269.
T. J. Spranley, M. Winkler, J. Dagate, D. Oncale, E. Strother, Gen. Dent., 2012, 60, e210–e214.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: Experimental details, materials and methods relating to synthesis and characterization, selected NMR spectra, UV-Vis data concerning aggregation and stability and relative survival rates against all bacteria. See DOI: 10.1039/c5pp00262a
These authors share senior-authorship.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tabenski, I., Cieplik, F., Tabenski, L. et al. The impact of cationic substituents in phenalen-1-one photosensitizers on antimicrobial photodynamic efficacy. Photochem Photobiol Sci 15, 57–68 (2016). https://doi.org/10.1039/c5pp00262a
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c5pp00262a