Abstract
Pre-eclampsia (PE) and gestational diabetes mellitus (GDM) are common complications of pregnancy, but the mechanisms underlying these disorders remain unclear. The aim was to identify the extent of altered gene expression in term placentas from pregnant women with late-onset PE and GDM compared to controls. RNAseq identified few significantly differentially regulated genes in placental biopsies between PE, GDM, or uncomplicated pregnancy (n = 10 each group). Five genes were altered in placentas from PE including 4 non-coding genes and Angiopoietin 2 (ANGPT2). No genes were significantly regulated by GDM. In contrast, many genes were significantly regulated by fetal, maternal and delivery-specific variables, particularly spinal and epidural anesthesia. We selected ANGPT2 and Chemokine (C-X-C motif) ligand 14 (CXCL14) to test with qPCR in a larger set of placentas (n = 475) and found no differences between the groups. However, regression analysis revealed a stronger association between placental ANGPT2 and CXCL14 mRNA expression and fetal, maternal and delivery-specific variables than diagnostic group. To conclude, the gene expression in term placentas are highly affected by fetal, maternal and delivery specific variables. Few regulated genes were found in late-onset PE and GDM placentas, which may suggest that these conditions could be more affected by maternal factors.
Similar content being viewed by others
Introduction
The placenta is a composite structure of embryonic and maternal tissues that supplies nutrients and removes waste products from the developing embryo. It also has critical endocrine and immuno-modulatory properties. Up to 10% of pregnancies suffer from abnormal placental development and, as such, are at high risk for complications such as miscarriage, pre-eclampsia (PE), gestational diabetes mellitus (GDM), fetal growth restriction and preterm birth1.
Pre-eclampsia is characterized by new-onset hypertension and end-organ damage after 20 weeks of gestation and affects 3–5% of all pregnancies2. PE has a multifactorial etiology and includes several subtypes3. Early-onset PE generally involves poor placental development, whereas late-onset PE is believed to arise from the interaction between a normal placenta and a damaged maternal microvasculature due, for example, to chronic hypertension or diabetes4. The late-onset form of PE may thus be more similar to GDM, in which pregnancy serves as a stress test in predisposed women5,6. GDM is defined as glucose intolerance of variable degrees first recognized during pregnancy. It may affect up to 20% of pregnant women depending on ethnicity and the diagnostic criteria used7.
The exact mechanisms behind GDM and PE are unclear. Previously, PE and other placental disease-related complications were considered pregnancy-specific and were thought to resolve hours or days after delivery of the placenta. More recently it has become clear that such diseases may contribute to the development of future cardiovascular disease8,9, providing additional need to understand the role of the placenta in the etiology of these disorders. Thus, identifying genes critical for placenta function may serve as a basis for revealing mechanisms underlying both normal and pathologic pregnancies. Gene expression in PE placentas has been extensively studied using microarrays10,11,12 and RNA sequencing (RNAseq)13,14 and to a lesser extent in placentas from GDM women15,16,17. However, the differentially expressed genes (DEGs) identified in these studies vary greatly, likely due to the phenotypic and etiologic heterogeneity of PE and the different diagnostic criteria used for GDM. Furthermore, some factors that may affect placental gene expression and add further heterogeneity between the studies (such as offspring sex and delivery specific variables)14 are rarely acknowledged or accounted for.
This study investigates the potential of RNAseq to discover novel genes that may encode the prediction and management of PE and GDM. Our aims were: (1) to examine DEGs in placentas from pregnant women with late-onset PE and GDM compared to normal placentas by RNAseq; (2) to identify DEGs in relation to maternal, fetal and delivery-specific variables by RNAseq; and (3) to validate our DEGs identified by RNAseq using RT-qPCR in a larger sample set and to control for the influence of maternal, fetal and delivery-specific variables.
Results
Clinical characteristics
Table 1 shows the characteristics of the study population chosen for RNAseq (n = 30) and validation with RT-qPCR (n = 475). In the RNAseq cohorts, as expected, women with GDM had a significantly higher BMI and placental weight and women with PE had a higher DBP at visit 4. In the validation cohort, GDM and PE women had higher BMI, BP, more frequent induction of labor, and shorter duration of labor. In addition, GDM women were older, had higher birth weight and placental weight, had more cesarean deliveries, and were more often nulliparous. Gestational age was shorter in PE women. Women with PE had no indication of poor placental function based on birth weight and the relatively long gestational age underlining the mildness of the phenotype. Blood pressure in the PE patients were higher than 140/90, but we did not have access to exact values after visit 4, and we could therefore not include severity of PE women based on blood pressure. Using World Health Organization (WHO) as opposed to International Association of Diabetes and Pregnancy Study Groups (IADPSG) criteria for the diagnosis of GDM, the differences with controls were somewhat attenuated (Supplemental Table 1). Supplemental Table 2 compares some maternal, fetal and delivery specific variables between women with and without epidural or spinal anesthesia demonstrating that these groups were comparable for most variables. However, a higher umbilical cord venous base excess and a higher incidence of cesarean deliveries were observed in the spinal group compared to women without anesthesia.
Highest expressed genes in term placentas
The genes were ordered by their median expression level. The highest expressed genes were similar in normal, GDM and PE placentas (Fig. 1). Genes that may regulate placental/fetal growth were highly expressed, including the noncoding RNA, H19, and insulin growth factor 2 (IGF2)18. Many placental-specific genes were also highly expressed, i.e. the placental lactogens (CSH1 and CSH2), placenta specific glycoproteins (PSG1, PSG3 and PSG4, PSG5 and PSG9), pregnancy-associated plasma protein A (PAPPA), hCG alpha subunit (CGA), estrogen synthase (CYP19A1), ADAM metallopeptidase domain 12 (ADAM12), tissue factor pathway inhibitor 2 (TFPI2), and placenta specific 4 (PLAC4). Other highly expressed genes were fibronectin (FN1), NADH dehydrogenase subunit 4 (ND4), nuclear paraspeckle assembly transcript 1 (NEAT1), eukaryotic translation elongation factor 1 alpha 1 (EEF1A1), cytochrome c oxidase genes (COX1, COX2 and COX3), cytochrome b (CYTB), hemoglobin beta (HBB) and microRNA 4485 (MIR4485).
The amount of RNA coming from maternal decidual and nucleated blood cells was examined by looking at the XIST transcript in the XX (median count 10099) and XY (median count 218.5) placenta samples. Median estimate for the fraction of RNA originated from maternal cells was 2.2% range (25th, 75th) 1.7–4.6% (mean, 4.0%). This suggests that the RNAseq data was of good quality both technically and biologically with little contamination from the maternal decidua or blood14.
Effects of maternal, delivery-specific, and fetal variables on placental gene expression by RNAseq
We evaluated potential maternal, delivery-specific, and fetal confounding variables on placental DEGs with FDR < 0.1. Investigating maternal confounding variables using all 30 samples, 16 DEGs were detected between primiparous compared to multiparous women. Of these, 15 were downregulated and 1 was upregulated in the multiparous women (Fig. 2). Some of these genes are involved in placentation and inflammation, i.e. pentraxin 3 (PTX3), chemokine (C-X3-C motif) receptor 1 (CX3CR1), and TSC22 domain family, member 3 (TSC22D3). Nine DEGs were detected between low and high BMI patients at visit 4. Of these, 8 were upregulated and 1 downregulated in the placenta from women with higher BMI, including increased adipsin (CFD). For BMI visit 1 and age of the mother, we did not find any significantly DEGs, FDR < 0.1 (data not shown).
A heat map is shown demonstrating the effect of two maternal variables on placental DEGs: (A) parity (nulliparous vs. multiparous), and (B) BMI (low-median vs. high) with FDR < 0.1. Heatmaps were constructed using log2 expression levels. The numbers to the left shows the log2 fold change value (log2FC) between groups. Samples are shown at the bottom of the figure, controls (yellow [CTR]), GDM (purple), and PE (red).
For the delivery-specific variables (Fig. 3), we found 33 DEGs with epidural anesthesia (22 upregulated and 11 downregulated) and 35 DEGs with spinal analgesia (10 upregulated and 25 downregulated). Women who received general anesthesia (n = 1) and pudendal block (n = 1) were excluded from these analyses. Some DEGs were similar between epidural and spinal anesthesia (e.g., keratins), but there were also specific differences in the gene signature depending on the anesthesia type. In particular, 9 transcripts for heat shock proteins (Hsp) belonging to the Hsp70 and Hsp40 family were abundantly expressed and increased in placentas from women who received epidural anesthesia. In addition, myeloperoxidase (MPO), a marker of neutrophil activation, was enhanced in women who received epidural anesthesia. In women who received spinal anesthesia, genes involved in the regulation of immune responses including several immunoglobins (e.g. IGHG2, IGJ, IGCL2, MARCH1) and chemokines (CXCL9/10) were differentially increased. With regard to differences relating to type of delivery, 1 DEG was downregulated in the women who delivered by cesarean, and 7 DEGs were upregulated in the placentas of women with a longer duration of labor, while no DEGs were associated with induction of labor.
A heat map is shown demonstrating the effect of four delivery-specific variables on placental DEGs: (A) epidural anesthesia (yes vs. no), (B) spinal anesthesia (yes vs. no), (C) labor duration (low-median vs high), and (D) mode of delivery (vaginal vs. cesarean) with FDR < 0.1. Heatmaps were constructed using log2 expression levels. The numbers to the left shows the log2 fold change value (log2FC) between groups. Samples are shown at the bottom of the figure, controls (yellow [CTR]), GDM (purple), and PE (red).
For fetal confounding variables (Fig. 4), 51 DEGs were associated with offspring’s sex. Thirty-seven were upregulated and 14 were downregulated when using males as the reference. Most of these were sex-specific chromosomal genes. We found 4 DEGs between the median high and low placental weights. Three were upregulated and 1 was downregulated in those with the highest placental weight. For birth weight, no significant DEGs were observed (Fig. 4). A list of the 70 most DEGs for the confounding variables can be found in Supplemental Table 3.
A heat map is shown demonstrating the effect of three fetal variables on placental DEGs: (A) Offspring sex (female vs. male), and (B) placenta weight (low-median vs. high) with FDR < 0.1. Heatmaps were constructed using log2 expression levels. The numbers to the left shows the log2 fold change value (log2FC) between groups. Samples are shown at the bottom of the figure, controls (yellow [CTR]), GDM (purple), and PE (red).
Effect of PE and GDM on placental gene expression by RNAseq
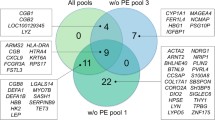
Only 5 DEGs were identified between the control and PE placentas with a FDR < 0.1. Of these, there were 3 downregulated non-coding genes, one downregulated protein-coding gene (Angiopoietin 2 [ANGPT2]), and one upregulated non-protein coding gene. Median counts (25th–75th percentile) for ANGPT2 in control vs. PE placentas were 663 (224–933) and 239 (155–386), respectively. No DEGs were detected between GDM and control placentas with FDR < 0.1. The 70 most DEGs for the diagnostic groups based on FDR and unadjusted p-value are listed in Supplemental Table 4.
Validation of RNAseq data by RT-qPCR
To validate our RNAseq data, we performed RT-qPCR. Samples for RT-qPCR validation included not only the 30 samples included in the RNAseq analysis, but all 475 placental samples from the STORK study with high RNA quality. However, since few genes were differentially expressed, in addition to ANGPT2, we chose to measure CXCL14 which was on the top of the lists for both PE and GDM (unadjusted p-value < 0.0006 and <0.001 respectively), and may be known for potentially regulate trophoblast outgrowth at the maternal-fetal interface19. As seen in Supplemental Fig. 3, differential (decreased) expression of ANGPT2 in PE placentas was confirmed by qPCR in the same 30 samples used in the RNAseq experiment. However, in the larger sample set, although ANGPT2 expression was decreased in PE placentas compared with controls, the difference did not reach statistical significance (P = 0.74). CXCL14 expression was not increased by qPCR in GDM (p = 0.055) and PE (p = 0.085) placentas in the same 30 samples used in the RNAseq experiment or in the larger sample set (p = 0.14 (GDM WHO), p = 0.11 (GDM IADPSG), p = 0.93 (PE)) (Supplemental Fig. 3).
Prediction of ANGPT2 and CXCL14 expression in placenta in the large cohort
To evaluate the influence of maternal, delivery- specific, and fetal characteristics on ANGPT2 and CXCL14 expression, we finally performed multivariable regression analysis, including diagnostic group (i.e. GDM or PE) to identify the most important predictors (Table 2). Stepwise linear regression identified gestational age, parity and offspring sex as predictors of ANGPT2 expression. Gestational age followed by birth weight and parity were the variables that predicted the gene expression of CXCL14.
Discussion
We conducted RNAseq analysis to compare gene expression between late-onset PE, GDM and control placentas at term. Using this approach, few genes were differentially expressed between these groups. In contrast, maternal, delivery-specific, and fetal variables were associated with altered expression of multiple genes in the term placenta. In particular, spinal and epidural anesthesia were unexpectedly associated with a significant upregulation of heat shock protection and immunomodulatory genes. When using qPCR in a larger cohort to validate the most highly differentially regulated genes in late-onset PE and GDM placentas identified in the RNAseq experiment (namely, ANGPT2 and CXCL14), we noted that maternal, delivery-specific, and fetal variables were stronger predictors of gene expression than the diagnostic group. These results suggest that maternal, delivery, and fetal variables substantially contribute to placental gene expression and should be considered when designing and evaluating such studies.
Samples chosen for RNAseq were well matched by clinical criteria and the RNA was of acceptable quality with a small percentage of contamination by maternal decidual and nucleated blood cells (~2%). This is similar to data published by Sõber et al. following a single wash with phosphate buffer saline and stabilization in RNA-later solution14. Furthermore, the most abundantly expressed genes identified in the term placenta were ones known to be involved in placental function, with good overlap with transcripts identified in previous studies14. For the sex specific genes, 15 and 19 of the DEGs identified in the current study corresponded with prior publications by Sõber et al.14 and Buckberry et al.20, respectively. Other DEGs associated with offspring sex in the current study were either non-functional genes expressed at low levels or sex specific genes (i.e. TSIX, TXNLGY, DDX3X, TTTY14, TBL1X, TBL1Y and TTTY10) not reported in the prior manuscripts. Totally from the 51 DEGs, 28 were located on the Y-Chromosome while 12 were located on the X-Chromosome. Taken together, the similarities in gene expression, related to abundance and offspring sex, with previous studies as well as the low maternal contamination supports the validity of our placental RNA samples.
A major finding in this study was the significant impact of epidural and spinal anesthesia on placental gene expression. To our knowledge, this has not been previously reported. Since women receiving epidural may be more likely to experience hyperthermia21, the increased expression of mRNAs coding for members of the HSP family could reflect the heat shock response, a switch in transcription and translation to preferentially express HSP’s that may preserve essential proteins22. Other relevant causes of increased HSP expression due to different kinds of stress could be infection, inflammation, hypoxia, injury and also adrenaline, which is a component in epidural anesthesia23,24. In particular, we identified a higher expression of the genes encoding HSP70, a HSP frequently studied in placental disease that may also confer harmful effects on binding to toll-like receptors25. Of note, increased expression of HSP70 protein has been demonstrated in the placenta of women with PE26,27 and in relation to delivery-specific variables, such as preterm labor28. In contrast, we found no significant association between HSP expression and mode of delivery, induction and duration of labor, or presence of PE. Several studies suggest that underlying maternal inflammation may confer increased risk of fever following epidural anesthesia29,30. However, apart from increased MPO mRNA levels in women receiving epidural, possibly reflecting neutrophil activation during the hyperthermic response31, few inflammatory genes were differentially regulated. In contrast, the use of spinal anesthesia was associated with enhanced mRNA expression of several inflammatory genes, including IGJ, CXCL9 and CXCL10, all chemokines specific for T cell signaling through the CXCR3 receptor. Activation of CXCR3 signaling has been implicated in the onset of labor32. Raman et al. identified enhanced IGJ expression as the most distinguishable feature of chronic placental inflammation in addition to CXCL9 and other inflammatory components from T and B cells33. Activation of opioid receptors increases the expression of CXCL10 in circulating immune cells in vitro34 and in the spinal cord in experimental animal models in vivo35. The use of anesthesia may reflect underlying obstetric conditions and we cannot excluded that the differences in mRNA expression observed for use of epidural and spinal anesthesia may results from longer acting mechanisms and systemic stress responses of the mother.
For maternal confounding variables, the expression of genes involved in inflammation and placentation (i.e., CX3CR136, TSC22D337 and PTX3)38 were associated with parity, indicating that women who have had a prior pregnancy may respond differently to the presence of invading trophoblast compared to nulliparous women. Adipsin expression, which is shown to be abundant in adipose tissue, was increased in the placentas of women with high BMI at term, and placentas from obese women have been shown to secrete higher levels of adipsin39.
Surprisingly few genes were differentially expressed in the placentas of women with late-onset PE at term and none in the placentas of women with GDM. We found only five significantly differentially regulated genes in PE placentas based on FDR < 0.1. The result of two recent meta-analyses found few common genes regulated in PE placentas compared to controls10,12. Using unsupervised clustering of 7 microarray data sets, Leavey et al.3 found a large degree of co-clustering of PE and control samples suggesting no distinct gene signature in PE. Direct comparison with previous studies is difficult since these mainly have focused on early-onset PE, a separate entity from late-onset PE as demonstrated in a recent micro-array study40. Furthermore, Sõber et al. identified an extensive shift in the placental transcriptome profile in late-onset PE using RNAseq14. For one specific DEG, ZDHHC8P1, we found increased expression in late-onset PE patients which were in contrast to their results were it was downregulated in their late-onset PE. However, their patients had a markedly more severe phenotype according to the ACOG 2013 criteria (hypertension with additional symptoms41 compared to the milder PE phenotype in our patients. This could imply that milder forms of preeclampsia that meet diagnostic criteria may in fact involve no underlying placental pathology and therefore may technically represent gestational hypertension with concomitant kidney dysfunction. It’s possible these cases may respond well to standard antihypertensive therapy and not require the termination of pregnancy.
As for GDM, previous studies evaluating gene expression have been performed in mixed cohorts (i.e. women with both PE in addition to GDM)15, in women on insulin therapy15,17, using different diagnostic criteria, or with very small sample numbers16, making comparisons difficult. Since our patients were not on insulin, it may be reasonable to assume that they have a milder GDM phenotype compared to other studies15,17. Importantly, excluding some differences in BMI and placental weights (Table 1), the major confounding maternal, delivery-specific and fetal variables were similar between our three comparison groups (late-onset PE, GDM and controls). Furthermore, when validating ANGPT2 and CXCL14 gene expression in the larger cohort, maternal, fetal and delivery-specific covariates were by far the strongest determinants of their expression. Since the objective of expression analysis is to identify unique expression patterns for distinct disease entities and not necessarily the effect of confounding conditions, our results highlight the importance of well characterized phenotypes to allow for accurate matching and relevant comparison between groups.
Several limitations of this study should be emphasized. The quality of the RNA could have been better. Five out of 30 RNAseq samples had RIN values below 7, and additional 8 samples had values below 8. However, we judged them to be of acceptable quality as judged by electropherograms. The placenta is a heterogeneous tissue and the biopsy was taken from the placental parenchyma. Other parts and specific cells of the placenta may be more important when investigating genes regulated in PE and GDM. Only one biopsy was taken from each placenta and the collection was performed by different individuals. While the technique for placental biopsy was standardized, we cannot exclude the possibility that biopsies were taken at different sites and that regional differences may have accounted for differences in DEGs42. Also, only term placentas were used in our analysis. While this ensures that appropriate control placentas will be available for comparison, it does represent the endpoint of a chronic disease state. It is likely that mRNA expression profiles at an earlier stage will be more reflective of the origin and pathogenesis of the disease and may be more likely to yield a distinctive fingerprint. This is especially true of PE3. Also, as we studied late-onset PE, our results cannot be applied to the spectrum of PE. Finally, we have incomplete information about the precise timing from when the placenta was delivered to when the biopsy was taken and the tissue stored at −80 °C.
In conclusion, the current study shows that confounding variables have a greater influence on gene expression in term placenta than do underlying disease states (such as late-onset PE and GDM). We found few genes significantly differentially regulated between late-onset PE, GDM and control placenta, suggesting that maternal factors may be more important than placental (fetal) factors, in the genesis of these pregnancy-specific conditions.
Methods
Study population
The STORK study was a prospective cohort study with a longitudinal design in which 1031 low-risk women of Scandinavian heritage who planned to deliver at Oslo University Hospital, Rikshospitalet, between 2002 and 2008 were followed throughout their pregnancy. Details about the study have been previously published43. Exclusion criteria included multiple pregnancy, known pre-gestational diabetes, severe chronic medical conditions (such as lung, cardiac, gastrointestinal or renal diseases), and pregnancies complicated by major fetal malformations or aneuploidy. Patients were routinely monitored for the development of pregnancy complications, such as PE, GDM, or preterm labor. Demographic and clinical data (including use of anesthesia in labor, route and mode of delivery, and neonatal outcome) were abstracted from the medical records. The difference in the formula used at our hospital between the epidural and spinal anesthesia is adrenaline in the epidural anesthesia, in addition to opioids, both fentanyl (or sufentanil in spinal) and bupivacaine in both.
Written informed consent was obtained from all participants. All clinical investigations were conducted according to the principles in the Declaration of Helsinki. The study was approved by the Regional Committee for Medical Research Ethics of Southern Norway in Oslo, Norway.
Clinical Diagnosis of GDM
A 75 g oral glucose tolerance test (OGTT) was performed in the morning after an overnight fast on all women at 30–32 weeks of gestation. Venous blood samples collected into tubes containing EDTA were analyzed at point of care using an Accu-Check Sensor glucometer (Roche Diagnostics GmbH, Mannheim, Germany). Additional venous blood samples were allowed to clot for 30 min. The serum was separated by centrifugation for 10 min at 3000 g and stored at −80 °C. Glucose levels were also measured from frozen serum samples collected at 30–32 weeks using the hexokinase method (Hitachi Modular P800, Roche Diagnostics, Mannheim, Germany) at an accredited clinical chemistry laboratory at Oslo University Hospital Rikshospitalet, as previously reported44. GDM was diagnosed on a 75 g OGTT using both the new IADPSG criteria and the old WHO criteria as follows: (1) IADPSG criteria: fasting plasma glucose (FPG) of 5.1–6.9 mmol/L (92–124 mg/dL) and 1 h plasma glucose ≥10.0 mmol/L (≥180 mg/dL) or 2 h plasma glucose 8.5–11.0 mmol/L (153–198 mg/dL); and (2) WHO criteria: 2 h plasma glucose ≥7.8 mmol/L (140 mg/dL)45, as previously reported44.
Clinical Diagnosis of PE
PE was diagnosed by new-onset blood pressure ≥140/90 mmHg and significant proteinuria (urinary total protein/creatinine ratio >30 or +1 on urine dipstick). All cases were diagnosed after 34 weeks gestation (late-onset PE).
Collection, storage and RNA extraction of placental biopsies
Placental biopsies were collected after vaginal or cesarean delivery. One biopsy was taken per placenta. The STORK study set included 475 placental samples, with good RNA quality, in which 19 were from women with PE, 103 with GDM, and 353 controls (overview, Supplemental, Fig. 1). Blocks of 2–4 cm were taken from the placental parenchyma, briefly washed in phosphate buffer saline, snap frozen in liquid nitrogen, and stored at −80 °C until RNA isolation. Half of the biopsy was homogenized in TRIzol reagent (Invitrogen, Life Technologies) on ice with a tissue grinder (Sigma Aldrich). Total RNA was extracted using TRIzol reagent (Invitrogen, Life Technologies) and purified with RNeasy microkit columns (Qiagen, Netherlands). Purity and concentration of isolated total RNA was measured using Nanodrop ND-1000 Spectrophotometer (Thermo Fisher Scientific Inc., USA) and RNA integrity number (RIN) was estimated using Agilent 2100 Bioanalyzer (Agilent Technologies, USA). RNA samples with a RIN <4 were excluded from subsequent RNAseq and qPCR analysis46.
RNAseq
Samples with a RIN greater than 4 were preferred for RNAseq analysis (median RIN 8.6; range (25th, 75th), 7.9–9.2). The RIN values and gel/electrophoresis results from the samples used for RNAseq are shown in Supplemental Fig. 2. The samples chosen for RNAseq were matched (based on mothers age and gestational age) between the groups of normal (control), GDM pregnancy and pre-eclamptic pregnancy (10 samples in each group). Sequencing libraries were prepared from 500 ng of total RNA using the TruSeq RNA sample preparation reagents (Illumina, San Diego, California) according to the manufacturer’s instructions, with fragmentation for 4 minutes at 94 °C. The libraries were sequenced using 125 bp paired-end sequencing on an Ilumina HiSeq 2000. We recorded an average 22.3 million (range, 20.2–24.6 million) paired reads per sample. Fastq files were generated using bcl2fastq (v1.8.4). Sequence reads were mapped to the reference genome (hg19/GRCh38) using TopHat2 (v2.0.13) and Bowtie2 (v.2.2.3.0). Library sizes and standard deviations for input into TopHat were calculated empirically by aligning 1000000 reads to an index built from human cDNA sequence. Sequence alignment was guided using only previously annotated gene models downloaded from Ensembl (www.ensembl.org; Homo_sapiens.GRCh38.79.gtf). On average, there was 73.6% concordant read pair mapping (range 69.6–76.6), with a mean unique mapping of 94.1%. Raw expression counts were calculated per gene using featureCounts (http://bioinf.wehi.edu.au/featureCounts/) and the same gtf file which was used for the read alignment.
Differential expressed genes in RNAseq data was tested using DESeq247 package for R. Outlier detection (Cook distance cutoff) and filtering out low expressed genes was performed using the default method in DESeq2. Example code is provided below:
# DE - control v preeclampsia
sampleTableCvP <- read.csv(“/SampleSheet_control_Preeclampsia.csv”)
countTableCvP <- read.table(“counts_pBs2_control_Preeclampsia.txt”)
countMatrixCvP <- as.matrix(countTableCvP)
ddsCvP <- DESeqDataSetFromMatrix(countData = countMatrixCvP, colData = sampleTableCvP, design = ~ Status)
ddsCvP$Status <- relevel(ddsCvP$Status, “control”)
ddsCvP <- DESeq(ddsCvP)
(resCvP <- results(ddsCvP))
For the highest expressed genes in the control, GDM and PE group we normalized the counts by this example code:
sampleTableAll <- read.csv(“SampleSheet.csv”)
countTableAll <- read.table(“counts_pBs2.txt”)
countMatrixAll <- as.matrix(countTableAll)
ddsAll <- DESeqDataSetFromMatrix(countData = countMatrixAll, colData = sampleTableAll, design = ~ Status)
vsd <- varianceStabilizingTransformation(ddsAll, blind = FALSE)
write.table(assay(vsd), “vsd.txt”, quote = F, sep = “\t”)
Maternal, Delivery-specific and Fetal Confounding factors
The confounding variables investigated in the RNAseq of the 30 placenta samples were dichotomized by median low vs. high or divided by categories (Table 1). Maternal variables: parity (nulliparous vs. multiparous), age (median 31 years, 25-median (low) vs. median-42 (high)), BMI visit 1 (median 24.9 kg/m2, 17.2-median (low) vs. median-34.9 (high)) and 4 (median 28.3 kg/m2, 20.5-median (low) vs. median-38.1 (high). For delivery specific variables, we investigated anesthesia (spinal vs. no anesthesia) and (epidural vs. no anesthesia), delivery mode (vaginal vs. cesarean section), duration of labor (median 5 hours, 2-median (low) vs. median-15 (high) and induction of labor (oxytocin or prostaglandin vs. no induction). For fetal variables we investigated offspring sex (girls vs. boys), placental weight (median 700 g, 530-median (low) vs. median-1100 (high)), and birth weight (median 3610 g, 2910-median (low) vs. median-4760 (high)). Gestational age had a very narrow spectrum (median 39.9 weeks, (37.7- median (low) vs. median-41.1 (high)) to make a meaningful analysis and was excluded from the RNAseq confounding factors analysis.
RT-qPCR
Reverse transcription was performed using a High Capacity cDNA Archive Kit (Applied Biosystems, Foster City, CA) with 1 μg of total RNA. mRNA quantification was performed using Power SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA) and the standard curve method on an ABI Prism 7900 (Applied Biosystems) with duplicate samples and standards. RT-qPCR of select genes was then performed on RNA extracted from all 475 placental samples with acceptable quality in the entire STORK population (n = 475, which included 19 with PE, 103 with GDM, and 353 controls) to validate the RNAseq results. In brief, sequence specific intron spanning oligonucleotide primers were designed using the Primer Express software version 2.0 (Applied Biosystems). ANGPT2 ‘GACACACCACGAATGGCATCTA (forward), GGGTTACCAAATCCCACTTTATATT’ (reverse), and CXCL14 GGACGGGTCCAAATGCAAGT (forward), GGTACCTGGACACGCTCTTG (reverse). Transcript expression levels were normalized to the geometric mean of three reference genes known to have great expression stability in placentas48; YWHAZ GATGACAAGAAAGGGATTGTCGAT (forward), CAGACCCAGTCTGATAGGATGTGT (reverse), TBP GCAGCTGCAAAATATTGTATCCACA (forward), CGTGGTTCGTGGCTCTCTTA (reverse), SDHA TCCTGATGGAGAATGGGGAGT (forward), GACGTGCAGCTGAAGTAGGT (reverse) and expressed as relative mRNA levels (data were log transformed to normal distribution).
Statistical analyses
Statistical analyses were conducted using SPSS for Windows, version 21.0 (Chicago, IL, USA). Data are expressed as mean ± SD when normally distributed and median (25th, 75th percentile) when skewed. Comparison between women with GDM or PE compared to controls was performed using t-test or Mann-Whitney U, depending on distribution, and Chi-square test or Fisher’s exact test (less than five observations) for categorical variables. Univariate and stepwise (variables p < 0.2 in univariate analysis) linear regression analyses were carried out on log transformed variables (if skewed) and results given as standardized regression coefficients. Linear relationship, multivariate normality, multicollinearity tested by variance inflation factors, autocorrelations tested by Durbin-Watson and homoscedasticity were tested and found satisfactory for linear regression. A gene in RNAseq was considered as differently expressed when the false discovery rate (FDR) was <0.1. FDR was calculated according to Benjamini and Hochberg.
Additional Information
How to cite this article: Lekva, T. et al. Gene expression in term placentas is regulated more by spinal or epidural anesthesia than by late-onset preeclampsia or gestational diabetes mellitus. Sci. Rep. 6, 29715; doi: 10.1038/srep29715 (2016).
References
Siddiqui, N. & Hladunewich, M. Understanding the link between the placenta and future cardiovascular disease. 21, 188–193 (2011).
Hernandez-Diaz, S., Toh, S. & Cnattingius, S. Risk of pre-eclampsia in first and subsequent pregnancies: prospective cohort study. 338, b2255 (2009).
Leavey, K., Bainbridge, S. A. & Cox, B. J. Large scale aggregate microarray analysis reveals three distinct molecular subclasses of human preeclampsia. PLoS One. 10, e0116508 (2015).
Oudejans, C. B., van, D. M., Oosterkamp, M., Lachmeijer, A. & Blankenstein, M. A. Genetics of preeclampsia: paradigm shifts. Hum Genet. 120, 607–612 (2007).
Myatt, L. & Roberts, J. M. Preeclampsia: Syndrome or Disease? 17, 83 (2015).
Williams, D. Pregnancy: a stress test for life. Curr Opin Obstet Gynecol. 15, 465–471 (2003).
Cundy, T., Ackermann, E. & Ryan, E. A. Gestational diabetes: new criteria may triple the prevalence but effect on outcomes is unclear. BMJ. 348, g1567 (2014).
Chen, C. W., Jaffe, I. Z. & Karumanchi, S. A. Pre-eclampsia and cardiovascular disease. Cardiovasc Res. 101, 579–586 (2014).
Pruthi, D. et al. Exposure to experimental preeclampsia in mice enhances the vascular response to future injury. Hypertension. 65, 863–870 (2015).
Kleinrouweler, C. E. et al. Differentially expressed genes in the pre-eclamptic placenta: a systematic review and meta-analysis. PLoS One. 8, e68991 (2013).
Louwen, F., Muschol-Steinmetz, C., Reinhard, J., Reitter, A. & Yuan, J. A lesson for cancer research: placental microarray gene analysis in preeclampsia. Oncotarget. 3, 759–773 (2012).
Vaiman, D., Calicchio, R. & Miralles, F. Landscape of transcriptional deregulations in the preeclamptic placenta. PLoS One. 8, e65498 (2013).
Kaartokallio, T., Cervera, A., Kyllonen, A. & Laivuori, K. Gene expression profiling of pre-eclamptic placentae by RNA sequencing. Sci Rep. 5, 14107 (2015).
Sober, S. et al. Extensive shift in placental transcriptome profile in preeclampsia and placental origin of adverse pregnancy outcomes. Sci Rep. 5, 13336 (2015).
Enquobahrie, D. A., Williams, M. A., Qiu, C., Meller, M. & Sorensen, T. K. Global placental gene expression in gestational diabetes mellitus. 200, 206–213 (2009).
Zhao, Y. H. et al. Genomic expression profiles of blood and placenta reveal significant immune-related pathways and categories in Chinese women with gestational diabetes mellitus. 28, 237–246 (2011).
Radaelli, T., Varastehpour, A., Catalano, P. & Hauguel-de, M. S. Gestational diabetes induces placental genes for chronic stress and inflammatory pathways. 52, 2951–2958 (2003).
Nordin, M., Bergman, D., Halje, M., Engstrom, W. & Ward, A. Epigenetic regulation of the Igf2/H19 gene cluster. Cell Prolif. 47, 189–199 (2014).
Kuang, H. et al. CXCL14 inhibits trophoblast outgrowth via a paracrine/autocrine manner during early pregnancy in mice. J Cell Physiol. 221, 448–457 (2009).
Buckberry, S., Bianco-Miotto, T., Bent, S. J., Dekker, G. A. & Roberts, C. T. Integrative transcriptome meta-analysis reveals widespread sex-biased gene expression at the human fetal-maternal interface. Mol Hum Reprod. 20, 810–819 (2014).
Goetzl, L. Epidural analgesia and maternal fever: a clinical and research update. Curr Opin Anaesthesiol. 25, 292–299 (2012).
Singh, I. S. & Hasday, J. D. Fever, hyperthermia and the heat shock response. Int J Hyperthermia. 29, 423–435 (2013).
Zuo, D., Subjeck, J. & Wang, X. Y. Unfolding the Role of Large Heat Shock Proteins: New Insights and Therapeutic Implications. Front Immunol. 7, 75 (2016).
Basu, N. et al. Heat shock protein genes and their functional significance in fish. Gene. 295, 173–183 (2002).
Asea, A. et al. Novel signal transduction pathway utilized by extracellular HSP70: role of toll-like receptor (TLR) 2 and TLR4. J Biol Chem. 277, 15028–15034 (2002).
Sheikhi, A. et al. Higher expression of HSP70 and LOX-1 in the placental tissues of pre-eclampsia pregnancies. Clin Exp Hypertens. 37, 128–135 (2015).
Abdulsid, A., Hanretty, K. & Lyall, F. Heat shock protein 70 expression is spatially distributed in human placenta and selectively upregulated during labor and preeclampsia. PLoS One. 8, e54540 (2013).
Chang, A. et al. Alteration of heat shock protein 70 expression levels in term and preterm delivery. J Matern Fetal Neonatal Med. 26, 1581–1585 (2013).
Goetzl, L., Evans, T., Rivers, J., Suresh, M. S. & Lieberman, E. Elevated maternal and fetal serum interleukin-6 levels are associated with epidural fever. Am J Obstet Gynecol. 187, 834–838 (2002).
Riley, L. E. et al. Association of epidural-related fever and noninfectious inflammation in term labor. Obstet Gynecol. 117, 588–595 (2011).
Evans, S. S., Repasky, E. A. & Fisher, D. T. Fever and the thermal regulation of immunity: the immune system feels the heat. Nat Rev Immunol. 15, 335–349 (2015).
Karjalainen, M. K. et al. CXCR3 Polymorphism and Expression Associate with Spontaneous Preterm Birth. J Immunol. 195, 2187–2198 (2015).
Raman, K. et al. Overlap Chronic Placental Inflammation Is Associated with a Unique Gene Expression Pattern. PLoS One. 10, e0133738 (2015).
Wetzel, M. A. et al. Mu-opioid induction of monocyte chemoattractant protein-1, RANTES, and IFN-gamma-inducible protein-10 expression in human peripheral blood mononuclear cells. J Immunol. 165, 6519–6524 (2000).
Ye, D. et al. Activation of CXCL10/CXCR3 signaling attenuates morphine analgesia: involvement of Gi protein. J Mol Neurosci. 53, 571–579 (2014).
Hannan, N. J. & Salamonsen, L. A. CX3CL1 and CCL14 regulate extracellular matrix and adhesion molecules in the trophoblast: potential roles in human embryo implantation. Biol Reprod. 79, 58–65 (2008).
Patterson, A. L., Squires, E. L., Hansen, T. R., Bouma, G. J. & Bruemmer, J. E. Gene profiling of inflammatory genes in day 18 endometria from pregnant and non-pregnant mares. Mol Reprod Dev. 79, 777–784 (2012).
Garlanda, C., Maina, V., Cotena, A. & Moalli, F. The soluble pattern recognition receptor pentraxin-3 in innate immunity, inflammation and fertility. J Reprod Immunol. 83, 128–133 (2009).
Sivakumar, K. et al. Elevated fetal adipsin/acylation-stimulating protein (ASP) in obese pregnancy: novel placental secretion via Hofbauer cells. J Clin Endocrinol Metab. 98, 4113–4122 (2013).
Junus, K. et al. Gene expression profiling of placentae from women with early- and late-onset pre-eclampsia: down-regulation of the angiogenesis-related genes ACVRL1 and EGFL7 in early-onset disease. Mol Hum Reprod. 18, 146–155 (2012).
Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet Gynecol. 122, 1122–1131 (2013).
Cox, B., Leavey, K., Nosi, U., Wong, F. & Kingdom, J. Placental transcriptome in development and pathology: expression, function, and methods of analysis. 213, S138–S151 (2015).
Roland, M. C. et al. Fetal growth versus birthweight: the role of placenta versus other determinants. PLoS One. 7, e39324 (2012).
Lekva, T. et al. Beta-cell dysfunction in women with previous gestational diabetes is associated with visceral adipose tissue distribution. Eur J Endocrinol. 173, 63–70 (2015).
Diagnostic criteria and classification of hyperglycaemia first detected in pregnancy: a World Health Organization Guideline. Diabetes Res Clin Pract. 103, 341–363 (2014).
Jobarteh, M. L., Moore, S. E., Kennedy, C., Gambling, L. & McArdle, H. J. The effect of delay in collection and processing on RNA integrity in human placenta: experiences from rural Africa. Placenta. 35, 72–74 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Meller, M., Vadachkoria, S., Luthy, D. A. & Williams, M. A. Evaluation of housekeeping genes in placental comparative expression studies. Placenta. 26, 601–607 (2005).
Acknowledgements
The sequencing service was provided by the Norwegian Sequencing Centre (www.sequencing.uio.no), a national technology platform hosted by the University of Oslo and supported by the Functional Genomics and Infrastructure programs of the Research Council of Norway and the Southeastern Regional Health Authorities.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: T.L., R.L., D.W.B., I.Z.J., E.R.N. and T.U. Performed the experiments: T.L. and R.L. Analyzed the data: T.L., R.L. and T.U. Contributed reagents/materials/analysis tools: R.L., M.C.P.R., C.F., D.W.B., I.Z.J., E.R.N., J.B., T.H. and T.U. Wrote the paper: T.L.and T.U. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lekva, T., Lyle, R., Roland, M. et al. Gene expression in term placentas is regulated more by spinal or epidural anesthesia than by late-onset preeclampsia or gestational diabetes mellitus. Sci Rep 6, 29715 (2016). https://doi.org/10.1038/srep29715
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep29715
- Springer Nature Limited
This article is cited by
-
Differential gene expression in two consecutive pregnancies between same sex siblings and implications on maternal constraint
Scientific Reports (2024)
-
Dysregulated non-coding telomerase RNA component and associated exonuclease XRN1 in leucocytes from women developing preeclampsia-possible link to enhanced senescence
Scientific Reports (2021)
-
Low CETP activity and unique composition of large VLDL and small HDL in women giving birth to small-for-gestational age infants
Scientific Reports (2021)