Abstract
Chan-Lam cross coupling allowed efficient synthesis of N,N’-disubstituted ortho-phenylene diamines bearing strong electron donating or withdrawing groups, such as nitro or methoxy groups, with moderate to high yields. These diamines can then be turned into N-heterocyclic carbene precursors after condensation with trimethyl orthoformate. The same strategy can also be utilized for the synthesis of N-monosubstituted aniline derivatives containing a functionalized ortho-aminomethyl group as intermediates for chiral 6-membered ring carbene precursors.
Similar content being viewed by others
Introduction
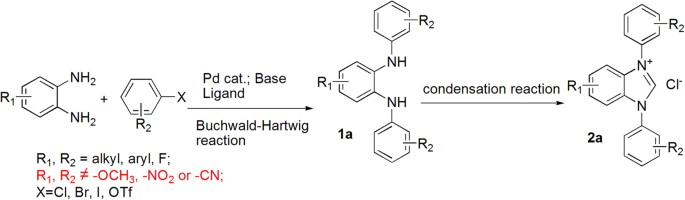
As one of the fastest developing classes of catalyst in organic synthesis, N-heterocyclic carbenes (NHCs) are widely used in aldol condensation1,2,3,4,5, Stetter reaction6,7,8,9, Diels-Alder reaction10, olefin metathesis11,12 and a variety of carbon-heteroatom cross coupling reactions13,14,15,16,17. Commonly used NHCs include an imidazole, imidazoline, benzimidazole, or triazole core for the carbene moiety (Fig. 1)18,19,20. Our group has recently reported several benzimidazole-based NHCs for catalysis in organic transformations21,22. These benzimidazole-based NHCs were traditionally synthesized in a two-step procedure (Fig. 2) mostly using Buchwald-Hartwig cross coupling reaction to construct the key N,N’-disubstituted ortho-phenylene diamines23 before ring closure with an orthoformate24. Although Buchwald-Hartwig reaction has the advantage of using readily available ortho-phenylene diamine and organic halides as starting materials, it is well known from previous studies of the Buchwald group that the reaction conditions (high temperature and use of strong base in conjunction with palladium catalyst) often are not compatible with substrates bearing strong electron effecting groups on aromatic rings such as nitro, nitrile, or alkoxy substituted phenyl halides25. In our hands particularly, reaction of ortho-phenylene diamine with neither nitro nor methoxy substituted phenyl bromide could produce the desired products in sufficient yields (Fig. 2). As our studies venture into the understanding of the effects of various substituents on NHC catalysis, alternative synthetic strategies for the desired NHCs are needed. We sought to investigate if Chan-Lam reaction can be used to replace Buchwald-Hartwig coupling to obtain N,N’-disubstituted phenylene diamines because the mild reaction conditions of Chan-Lam reaction should in theory tolerate a wide range of functional groups26,27,28,29,30,31.
Results and Discussion
We first chose to optimize reaction conditions for the Chan-Lam cross coupling of phenylene diamine 1 and two separate boronic acids 2a (bearing ortho-OMe) and 2c (bearing meta-nitro) to afford the desired diamine intermediates 3a and 3c (Table 1). The investigation covered variations in catalyst loading ratio, amount or identity of base and reaction time, etc. As summarized in Table 1, higher catalyst loading led to shorter reaction time to completely consume the starting materials but not necessarily higher yields (entries 1–6 for synthesizing 3a and entries 7–12 for 3c). Triethylamine as the base for the reaction seemed to be adequate as replacement by pyridine or using a mixture of triethylamine and pyridine did not lead to obvious improvements. Based on these results, we chose the condition in entry 3 of Table 1 for coupling with substrates bearing strong electron donating groups and the condition in entry 7 of Table 1 for substrate containing strong electron withdrawing groups.
Then, we expanded the reaction to larger scope of substrates to prepare the N, N-disubstituted phenylene diamine intermediates, followed by ring closure to form the final carbene precursors. The results are summarized in Table 2 and discussed below.
The 13 entries in Table 2 cover examples of various electron withdrawing or donating groups (NO2, F, CF3, or OMe) either on the phenylene diamine 1 or on the boronic acid 2. These all gave satisfactory results for the synthesis of 3 using the optimized conditions found in Table 1 and yields of 51–84% were obtained. Further ring closures with trimethyl orthoformate to form the carbene precursors 4 were mostly smooth and with high isolated yields of 88–95%. The exceptions are the ring closure reactions for 3g and 3h bearing a nitro group on the phenylene diamine ring and either a CF3 or a F at the ortho position on the N and N’ phenyl rings. Probably the strong electron withdrawing groups ortho to the aniline moiety makes the diamine extremely unreactive, because no product were detected for ring closures of 3g or 3h even when the reactions were carried out at much elevated temperature of 80 °C.
With the above success, we sought to further expand the application of Chan-Lam cross coupling reaction to the preparation of intermediates of another class of carbene precursors. We recently reported a new class of NHCs in which the carbene bearing central cores contain a chiral center and fused aromatic ring32. The original synthesis of the precursors utilized Buchwald-Hartwig reaction to functionalize the two key carbene stabilizing nitrogen atoms. As expected, there are two drawbacks for this original synthetic route: 1) low compatibility to strong electron donating or withdrawing groups; and 2) simultaneous modification of both the aromatic and the aliphatic amines leading to loss of control on differentiating substitutions on the nitrogen atoms of the NHC core. Utilizing the Chan-Lam reaction to firstly derivatize the aromatic amino group could potentially overcome both shortcomings. Our results with various substrates are summarized Table 3. In all the six examples, the optimal conditions found for phenylene diamine in Table 1 were no longer useful because that led to slow reactions. We therefore further increased catalyst loading and changed reaction time. For reactions involving boronic acids 2a, 2g, or 2h (entries 1, 3, 4 and 5 in Table 3), we increased Cu(OAc)2·H2O to 1.0 eq of 1e and 1f and the reaction time was decreased to around 12 h. For reactions of substrate 2c, we increased catalyst loading to 0.5 eq of 1 and decreased reaction time to 3h (entries 2 and 6 in Table 3). These led to yields of 43–76% in the six examples of 3. All reaction products of the Chan-Lam coupling were mono-functionalized derivatives on the aniline nitrogen atom. This opened up region-control opportunities of the intermediates. To demonstrate this, compounds 3n and 3q were selected to be further derivatized using Buchwald-Hartwig reaction to modify the chiral alkyl amine and afforded 3n1 and 3q1 (Table 3), respectively. These were then condensed with trimethyl orthoformate to formed the desired carbene precursors 4n and 4q (Table 3) in excellent yields.
Summary
In summary, we used Chan-Lam cross coupling reaction to successfully incorporate aromatic rings containing strong electron donating or withdrawing groups on phenylene diamines or aminomethyl anilines for the synthesis of NHC precursors. This protocol is a good complementary to the traditional Buchwald-Hartwig coupling reactions. In addition, application of the Chan-Lam reaction can properly distinguish derivatization on aromatic amines over alphatic amines and therefore provides regio-control for the installation of different substitutions on a class of chiral 6-membered ring carbenes.
Methods
General procedure A for synthesis of 3a-s
A mixture of diamine 1 (1.0 mmol), phenylboronic acid 2 (2.1 mmol) was dissolved in dichloromethane (20–30 mL) in a 100 mL round-bottomed flask equipped with a stir bar. Then, Et3N (1.0–2.0 equiv) and Cu(OAc)2·H2O (0.2–0.5 equiv) were added at room temperature. The reaction mixture was stirred for 2–48 h. After completion of the reaction (checked by TLC), the mixture was filtered through Celite and washed with EtOAc. The crude product was purified by silica gel chromatography (hexane : EtOAc = 40:1, 20:1 or 5:1) to give the desired product 3.
General procedure B for synthesis of N-heterocyclic Carbene precursors 4a-s
Compound 3a-s (0.5 mmol) was dissolved in trimethyl orthoformate (5 mL). Then, concentrated hydrochloric acid (0.1 mL) was added. The mixture was reacted at room temperature for 1–12 h. Then most solvent was evaporated under reduced pressure. The crude product was purified by silica gel chromatography (dichloromethane: methanol = 10:1) to give the desired product 4a-s.
Additional Information
How to cite this article: Huang, L. et al. Application of Chan-Lam cross coupling for the synthesis of N-heterocyclic carbene precursors bearing strong electron donating or withdrawing groups. Sci. Rep. 5, 12431; doi: 10.1038/srep12431 (2015).
References
Enders, D. & Kallfass, U. An efficient nucleophilic carbene catalyst for the asymmetric benzoin condensation. Angew. Chem. Int. Ed. 41, 1743–1745 (2002).
Baragwanath, L., Rose, C. A., Zeitler, K. & Connon, S. J. Highly enantioselective benzoin condensation reactions involving a bifunctional protic pentafluorophenyl-substituted triazolium precatalyst. J. Org. Chem. 74, 9214–9217 (2009).
Hachisu, Y., Bode, J. W. & Suzuki, K. Catalytic intramolecular crossed aldehyde-ketone benzoin reactions: A novel synthesis of functionalized preanthraquinones. J. Am. Chem. Soc. 125, 8432–8433 (2003).
Kuhl, N. & Glorius, F. Direct and efficient N-heterocyclic carbene-catalyzed hydroxymethylation of aldehydes. Chem. Commun. 47, 573–575 (2011).
Toole, S. E. Ó., Rose, C. A., Gundala, S., Zeitler, K. & Connon, S. J. Highly chemoselective direct crossed aliphatic-aromatic acyloin condensations with triazolium-derived carbene catalysts. J. Org. Chem. 76, 347–357 (2011).
Mattson, A. E., Bharadwaj, A. R. & Scheidt, K. A. The thiazolium-catalyzed Sila-Stetter reaction: Conjugate addition of acylsilanes to unsaturated esters and ketones. J. Am. Chem. Soc. 126, 2314–2315 (2004).
Enders, D., Han, J. & Henseler, A. Asymmetric intermolecular Stetter reactions catalyzed by a novel triazolium derived N-heterocyclic carbine. Chem. Commun. 34, 3989–3991 (2008).
Jousseaume, T., Wurz, N. E. & Glorius, F. Highly enantioselective synthesis of α-amino acid derivatives by an NHC-catalyzed intermolecular Stetter reaction. Angew. Chem. Int. Ed. 50, 1410–1414 (2011).
Mattson, A. E., Bharadwaj, A. R., Zuhl, A. M. & Scheidt, K. A. Thiazolium-catalyzed additions of acylsilanes: A general strategy for acyl anion addition reactions. J. Org. Chem. 71, 5715–5724 (2006).
He, M., Struble, J. R. & Bode, J. W. Highly Enantioselective Azadiene Diels-Alder Reactions Catalyzed by Chiral N-Heterocyclic Carbenes. J. Am. Chem. Soc. 128, 8418–8420 (2006).
Trnka, T. M. & Grubbs, R. H. The Development of L2 × 2Ru = CHR olefin metathesis catalysts: An organometallic success story. Acc. Chem. Res. 34, 18–29 (2001).
Schrock, R. R. et al. Synthesis of molybdenum imido alkylidene complexes and some reactions involving acyclic olefins. J. Am. Chem. Soc. 112, 3875–3886 (1990).
Lee, S., Beare, N. & Hartwig, J. Palladium-catalyzed α-arylation of esters and protected amino acids. J. Am. Chem. Soc. 123, 8410–8411 (2001).
Viciu, M. S., Germaneau, R. F. & Nolan, S. P. Well-defined, air-stable (NHC) Pd(Allyl) Cl (NHC = N-heterocyclic carbene) catalysts for the arylation of ketones. Org Lett. 4, 4053–4056 (2002).
Navarro, O. et al. Synthesis of novel (NHC) Pd(acac) Cl complexes (acac = acetylacetonate) and their activity in cross-coupling reactions. Tetrahedron. 61, 9716–9722 (2005).
Quallich, G. J., Makowski, T. W., Sanders, A. F., Urban, F. J. & Vazquez, E. Synthesis of 1, 2, 3, 4-tetrahydroisoquinolines containing electron-withdrawing groups. J. Org. Chem. 63, 4116–4119 (1998).
Okuro, K., Furuune, M., Miura, M. & Nomura, M. Copper-catalyzed reaction of aryl iodides with active methylene compounds. J. Org. Chem. 58, 7606–7607 (1993).
Hopkinson, M. N., Richter, C., Schedler, M. & Glorius, F. An overview of N-heterocyclic carbenes. Nature. 510, 485–496 (2014).
Benhamou, L., Chardon, E., Lavigne, G., Bellemin-Laponnaz, S. & César, V. Synthetic routes to N-heterocyclic carbene precursors. Chem. Rev. 111, 2705–2733 (2011).
Enders, D., Niemeier, O. & Henseler, A. Organocatalysis by N-heterocyclic carbenes. Chem. Rev. 107, 5606–5655 (2007).
Wang, H., Xia, Y.-Y., Lv, Sh., Xu, J.–L. & Sun, Zh.-H. Facial and practical synthesis of benzimidazole-based N-heterocyclic carbenes. Tetra. Lett. 54, 2124–2127 (2013).
Xia, Y.-Y., Chen, L.-Y., Lv, Sh., Sun, Zh.-H. & Wang, B. Microwave-assisted or Cu-NHC-catalyzed cycloaddition of azido-disubstituted alkynes: bifurcation of reaction pathways. J. Org. Chem. 79, 9818–9825 (2014).
Charles, M. D., Schultz, P. & Buchwald, S. L. Efficient Pd-Catalyzed Amination of Heteroaryl Halides. Org. Lett. 7, 3965–3968 (2005).
Duan, W.-L., Shi, M. & Rong, G.-B. Synthesis of novel axially chiral Rh-NHC complexes derived from BINAM and application in the enantioselective hydrosilylation of methyl ketones. Chem. Commun. 2916–2917 (2003).
Wolfe, J. P. & Buchwald, S. L. Scope and Limitations of the Pd/BINAP-Catalyzed Amination of Aryl Bromides. J. Org. Chem. 65, 1144–1157 (2000).
Chan, D. M. T., Monaco, K. L., Wang, R. P. & Winters, M. P. New N- and O-arylation with phenylboronic acids and cupric acetate. Tetrahedron Lett. 39, 2933–2936 (1998).
Lam, P. Y. S. et al. New aryl/heteroaryl C-N bond cross-coupling reactions via arylboronic acid/cupric acetate arylation. Tetrahedron Lett. 39, 2941–2944 (1998).
Lam, P. Y. S., Clark, C. G. & Li, R. Structure-based design of novel guanidine/benzamidine mimics: potent and orally bioavailable factor Xa inhibitors as novel anticoagulants. J. Med. Chem. 46, 4405–4418 (2003).
Rao, H. et al. Easy copper-catalyzed synthesis of primary aromatic amines by coupling aromatic boronic acids with aqueous ammonia at room temperature. Angew. Chem. Int. Ed. 48, 1114–1116 (2009).
Zhuang, R. et al. Copper-catalyzed C-P bond construction via direct coupling of phenylboronic acids with H-phosphonate diesters. Org. Lett. 13, 2110–2113 (2011).
Rossi, S. A. et al. Selective formation of secondary amides via the copper-catalyzed cross-coupling of alkylboronic acids with primary amides. Org. Lett. 15, 2314–2317 (2013).
Huang, L.-L., Cao, Y., Zhao, M.-P., Tang, Zh.-F. & Sun, Zh. -H. Asymmetric borylation of α, β-unsaturated esters catalyzed by novel ring expanded N-heterocyclic carbenes based on chiral 3, 4-dihydro-quinazolinium compounds. Org. Biomol. Chem. 12, 6554–6556 (2014).
Acknowledgements
We thank Professor Erkang Fan at the University of Washington for helpful discussions. Financial support by the Shanghai Municipal Education Commission (No.14ZZ159), the Shanghai Municipal Science and Technology Commission (No. 12430501300) and the Program for Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Education are gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
Z.S. designed the experiments. L.H. and C.H. performed the experiments. All authors analyzed the data and contributed to writing the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Huang, L., He, C. & Sun, Z. Application of Chan-Lam cross coupling for the synthesis of N-heterocyclic carbene precursors bearing strong electron donating or withdrawing groups. Sci Rep 5, 12431 (2015). https://doi.org/10.1038/srep12431
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12431
- Springer Nature Limited