Abstract
Cardiorespiratory fitness is an established predictor of metabolic disease and mortality. Fitness is directly measured as maximal oxygen consumption (VO2max), or indirectly assessed using heart rate responses to standard exercise tests. However, such testing is costly and burdensome because it requires specialized equipment such as treadmills and oxygen masks, limiting its utility. Modern wearables capture dynamic real-world data which could improve fitness prediction. In this work, we design algorithms and models that convert raw wearable sensor data into cardiorespiratory fitness estimates. We validate these estimates’ ability to capture fitness profiles in free-living conditions using the Fenland Study (N=11,059), along with its longitudinal cohort (N = 2675), and a third external cohort using the UK Biobank Validation Study (N = 181) who underwent maximal VO2max testing, the gold standard measurement of fitness. Our results show that the combination of wearables and other biomarkers as inputs to neural networks yields a strong correlation to ground truth in a holdout sample (r = 0.82, 95CI 0.80–0.83), outperforming other approaches and models and detects fitness change over time (e.g., after 7 years). We also show how the model’s latent space can be used for fitness-aware patient subtyping paving the way to scalable interventions and personalized trial recruitment. These results demonstrate the value of wearables for fitness estimation that today can be measured only with laboratory tests.
Similar content being viewed by others
Introduction
Cardiorespiratory fitness (CRF) is one of the strongest known predictors of cardiovascular disease (CVD) risk and is inversely associated with many other health outcomes1. CRF is also a potentially stronger predictor of CVD outcomes when compared to other risk factors like hypertension, type 2 diabetes, high cholesterol, and smoking. Despite its prognostic value, routine CRF assessment remains uncommon in clinical settings because maximal oxygen consumption (VO2max), the gold-standard measure of CRF, is challenging to directly measure. A computerised gas analysis system is needed to monitor ventilation and expired gas fractions during exhaustive exercise on a treadmill or cycle ergometer. Additional equipment may be needed to monitor other biosignals, such as heart rate (HR). These equipment require trained research personnel to operate, and an attending physician is a requisite for exercise testing in some scenarios. Several criteria must also be achieved to verify that exhaustion has been reached, including leveling off of VO2, achieving a percentage of age-predicted maximal HR, and surpassing a peak respiratory exchange ratio threshold2. The costs of VO2 measurement and risks of exhaustive exercise not only limit direct CRF assessment in clinical settings, but also restrict research of CRF at the population level. Thus, our understanding of differences in CRF within populations, across geographic regions, and over time is lacking.
Non-exercise prediction models of VO2max are an alternative to exercise testing in clinical settings. These models are usually regression-based and incorporate variables like sex, age, body mass index (BMI), resting heart rate (RHR), and self-reported physical activity3. We have recently shown that RHR alone can be used to estimate VO2max4, however, the validity of estimates from this approach are considerably lower than those achieved with exercise testing5,6. Also, the response of heart rate to activity has been shown to be predictive of VO2max, in coarse-grained data7. Wearable devices such as activity trackers and smartwatches can monitor not only RHR and physical activity but other biosignals in free-living conditions8, potentially enabling more precise estimation of VO2max without exercise testing. Recent attempts to use wearable devices to estimate VO2max are difficult to externally evaluate, however, because their estimation methods tend to be non-transparent9 and lack scientific validation9,10. Although certain wearable devices show promise, they tend to rely on detailed physical activity intensity measurements, GPS-based speed monitoring, and require users to reach near-maximal HR values, which limits their use to fitter individuals11. Some studies attempt to estimate VO2max from data collected during free-living conditions, but these are typically from small-scale cohorts and use contextual data from treadmill activity, which restricts their application in population settings12,13.
Here, we use data from the largest study of its kind, by over two orders of magnitude, and use purely free-living data to predict VO2max, with no requirement for context-awareness. This work substantially advances previous non-exercise models for predicting CRF by introducing an adaptive representation learning approach to physiological signals derived from wearable sensors in a large-scale population with free-living condition data. We employ a deep neural network model that utilizes feedforward non-linear layers to learn personalized fitness representations. We demonstrate that these models yield better performance than traditional and state-of-the-art non-exercise models. We illustrate how these models can be used to predict the magnitude and direction of change in CRF. Moreover, we show that they can adapt in time given behavioural changes by showcasing strong performance in a subset of the same population who were retested seven years later. This has implications for the estimation of population fitness levels and lifestyle trends, including the identification of sub-populations or areas in particular need of intervention. Such models can improve both population health and personalized medicine applications. For instance, the ability of the patient to undergo certain treatments like surgery14 or chemotherapy15 can be assessed through wearables before the procedure takes place and therefore reduce postoperative complications.
Results
Baseline measurements were collected from 12,435 healthy adults from the Fenland study in the United Kingdom16, where all required data for the present analysis were available in 11,059 participants (Fenland I, baseline timepoint referred to as “current” in our evaluation). A subset of 2675 participants was assessed again after a median (interquantile range) of 7 (5–8) years (Fenland II, referred to as “future” in our evaluations). Descriptive characteristics of the two analysis samples are presented in Fig. 1. We present the characteristics of the longitudinal cohort in both temporal snapshots in Fig. 1 (“present” and “future”). Mean and standard deviations for each characteristic are presented in this table. An overview of the study design and the three experimental tasks is provided in Fig. 2.
Top: The first task trains a model to predict fitness using the large cohort (Fenland I), the second task is using the smaller cohort of repeats in Fenland I (called Fenland II) and trains further models to predict fitness now and in the future (and their delta). The third task evaluates the original model trained in Task 1 by feeding new sensor data to assess the adaptability of the model to pick up change. (*Training set is 90% of the 80% remaining dataset after splitting to testing set. Validation set is 10% of the training set). Bottom: Dataset statistics breakdown by sex and features [data is in mean (std)]. Values with asterisk(*) indicate that this variable comes from Fenland II sensor data which is a smaller cohort (N = 2071) due to data filtering (see Top panel, Task 3). The values in FII (future) cohort correspond to the second assessment (7 years later).
We use a cohort study of 11,059 participants with laboratory and wearable sensor data and a longitudinal subsample of 2675 participants who repeated the protocol 7 years later. Using the free-living sensors as input data, we train machine learning models to predict lab-measured cardio-respiratory fitness (VO2max).
Fine-grained fitness prediction from wearable sensors
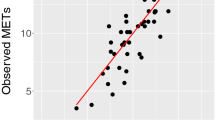
We first developed and externally validated several non-exercise VO2max estimation models as a regression task using features commonly measured by wearable devices (anthropometry, resting heart rate (RHR), physical activity (PA); see Table 1). Here our goal was to explore how conventional non-exercise approaches to VO2max estimation could be enhanced by features from free-living PA data. We split participant data into independent training and test sets. The training set (n = 8384, participants with baseline data only) was used for model development. The test set (n = 2675, participants with baseline and followup data) was used to externally validate each model. Starting with linear regression models, we used anthropometry or RHR alone which yielded poor external validity (R2 of 0.35–37), but validity improved when combined in the same model (R2 of 0.61). The best performance (R2 of 0.67) was attained using a deep neural network model combining wearable sensors, RHR, and anthropometric data (Fig. 3). For reference, we compare these results to traditional non-model equations, which rely on Body Mass, RHR, and Age. Using a popular equation (as proposed in refs. 17,18) we obtained poor validity (R2 of −3.2 and Correlation of 0.389), a performance lower than using anthropometrics only in our setup (see Methods for details). This motivates the use of machine learning which captures better covariate interactions.
Comparing the predicted and true VO2max coming from the best performing comprehensive model (Sensors + RHR + Anthro.) trained with Fenland I. a, c Linear and dense models produce accurate predictions with correlation of predicted and true VO2max up to r = 0.82, p < 0.005 (see Table 1)b, d The plot combines a kernel density estimate and histogram, while the gray line denotes a linear regression fit. Transparency has been applied to the datapoints to combat crowding.
To understand the limits of the models, we conducted a number of post-hoc sensitivity analyses by investigating subgroup performance in terms of sex (male/female), age, weight, BMI, and height (Table 3), as well as investigating model errors on Bland-Altman agreement plots (Fig. Suppl. 2). We found that the comprehensive model is robust to most subgroups, showing minimal differences in most cases, with exceptions in weight and age. In particular, we found no difference between male and female participants even though the performance was lower for each group compared to the mixed set (R2 of 0.59). Further, the models perform better on participants of lower age (R2 of 0.68), higher weight (R2 of 0.69). No effects were observed on height or BMI differences (overlapping CIs). The best performing subgroup is “higher weight" and the worst performing “sex-male". Last, Bland-Altman plots showed that the Dense model has better upper difference compared to the linear model, where the lower and mean difference were similar (Fig. Suppl. 2).
Deep neural networks can learn feature representations that are suitable for clustering tasks, such as population stratification by implicit health status, but are difficult to reveal using linear dimension-reduction techniques19. We used t-distributed stochastic neighbor embedding (tSNE), a nonlinear dimension-reduction technique, to visualise learned feature representations from our model and their relationship to participant VO2max (Fig. 4). Clustering and coloring by VO2max was shown to be inversely related and more apparent in the learned latent space compared to the original feature space. Further, we show how this latent space can be used for patient subtyping through embedding neighbours. Starting from an initial patient (query), we retrieved the five nearest neighbours in the latent and original space. In a case study with three randomly selected participants, we found that the total euclidean distance of the query to all neighbours is higher in the original than the latent space, pointing to better semantic clustering (Fig. 4, bottom panel).
tSNE projection of the original feature vector (Fenland I testing set, Sensors + RHR + Anthro.) and the latent space of the Dense model after training. Top: The original data presents some clusters but the outcome is not clearly linearly separable. The model activations on the penultimate layer of the neural network capture the continuum of low-high VO2max both locally and globally. Bottom: Starting from a query participant (+) we retrieve the five nearest neighbours in the original and latent space and list their details on the tables on the right. The total distance of each query to each neighbour is listed in each subplot. Transparency has been applied to combat crowding and the colorbar is centered on the median value to illustrate extreme cases. The VO2max label is used only for color-coding purposes (the projection is label-agnostic). Every participant is a dot.
Predicting magnitude and direction of fitness change in the future
The second group of tasks evaluated our model on the subset of participants who returned for Fenland II ≈ 7 years later (referred to as future in our evaluations). For these experiments, we carried out three evaluations. Following the process described earlier, we re-trained a model to predict future VO2max using only information from the present as input (Table 2). This model yielded a slightly lower accuracy than Fenland I, achieving an R2 of 0.49 and a correlation of 0.72. This lower performance is expected since the model has no information on the behavior of the individuals 7 years later. We also trained a model to directly predict the difference (or delta) of current-future VO2max, which reached a correlation of 0.23.
Motivated by the moderate predictability of the fine-grained delta of VO2max, we formulated this problem as a classification task. A visual representation of this task can be found in Fig. 5a. By inspecting the distribution of the difference (delta) of current-future VO2max on the training set, we split it into two halves (50% quantiles) of equally balanced data and set these as prediction outcomes. The purpose of this task is to assess the direction of individual change of fitness. We report an area under the curve (AUC) of 0.61 in predicting the direction of change (N = 2675). We also investigated equal numbers of participants on the tails of the change distribution which indicates participants who underwent substantial and dramatic change in fitness over the period of time between Fenland I and Fenland II (≈7 years). In this case, we picked participants from 80%/20% (substantial) and 90%/10% (dramatic) quantiles of the outcome distribution. The results from these experiments show that the models can distinguish between substantial fitness change with an AUC of 0.72 (N = 1068) and between dramatic fitness change with an AUC of 0.74 (N = 535). All AUC curves can be found in Fig. 5b.
a Distribution of the Δ of VO2max in the present and the future. The shaded areas represent different binary bins that are used as outcomes, increasingly focusing on the extremes of this distribution. b ROC AUC performance in predicting the three Δ outcomes as shown on the left-hand side. Brackets represent 95% CIs.
Enabling adaptive cardiorespiratory fitness inferences
For the final task, we assessed whether the trained models can pick up change using new sensor data from Fenland II, considering that obtaining new wearable data is relatively easy since these devices are becoming increasingly pervasive. The intuition behind this task is to evaluate the generalizability of the models over time. We first matched the populations that provided sensor data for both cohorts (N = 2042) and applied the trained model from Task 1 in order to produce VO2max inferences. We then compared the predictions with the respective ground truth (current and future VO2max). The true and predictive distributions are shown in Fig. 6c, d. Through this procedure, we found that the model achieves an r = 0.84 for VO2max future prediction and an r = 0.82 for VO2max current prediction (validating our Task 1 results). In other words, if we have access to wearable sensor data and other information from the future time, we can reuse the already trained model from Fenland I to accurately infer fitness with minimal loss of accuracy over time, even though this is new sensor data from a completely separate (future) week.
By matching the populations who provided sensor data for both cohorts (N = 2042) we passed them through the trained model from Task 1. a, b Calculating the difference (Δ) of the predictions juxtaposed with the true difference of fitness over the years with a correlation of Δ of predicted and true VO2max (r = 0.57, p < 0.005). c, d Comparison of predicted and true VO2max using FI and FII covariates (sensors, RHR, anthro.), respectively.
Last, we calculated the delta of the predictions and compared it to the actual delta of fitness over the years. This task showed that the models tend to focus mostly on positive change and under-predict when participants’ fitness deteriorates over the years (Fig. 6a, b). The overall correlation between the delta of the predictions with the ground truth is significant (r = 0.57, p < 0.005).
Discussion
Cardiorespiratory fitness declines with age independently of changes to body composition, and low cardiorespiratory fitness is associated with poor health outcomes1,20,21,22. As such, having the capacity to predict whether CRF would decline in excess of natural aging could be valuable to clinicians when tailoring therapeutic interventions. Here we have developed a deep learning framework for predicting CRF and changes in CRF over time. Our framework estimates VO2max by combining learned features from heart rate and accelerometer free-living data extracted from wearable sensors with anthropometric measures. To evaluate our framework’s performance, VO2max estimates were compared with VO2max values derived from a submaximal exercise test5. Free-living and exercise test data were collected at a baseline investigation in 11,059 participants (Fenland I). A subset of those participants (n = 2675) completed another exercise test at a follow-up investigation approximately seven years later (Fenland II). This study design allowed us to address three questions: 1) Do baseline estimates of VO2max from the deep learning framework agree with VO2max values measured from exercise testing at baseline?, 2) Can the framework learn features from heart rate and accelerometer free-living data collected at baseline that predict VO2max measured at follow-up?, and 3) Can the framework be used to predict the magnitude of change in VO2max from baseline to follow-up?
In the VO2max estimation tasks, our model demonstrated strong agreement with VO2max measured from the submaximal exercise test at baseline (r: 0.82) as well as for the longitudinal, follow-up visit (r: 0.72). We were also able to distinguish between substantial and dramatic changes in CRF (AUCs 0.72 and 0.74, respectively). Finally, we further evaluated the initial model on new input data by feeding Fenland II free-living data along with updated heart rate and anthropometrics to the model, showing that it is able to adapt and monitor change over time. We evaluated the inference capabilities of the model in the difference (delta) between the current (Fenland I) and future (Fenland II) VO2max for those participants that came back ~7 years later. For this last task, the model produced outcomes that translated to a 0.57 correlation between the delta of predicted and delta of true VO2max.
The application of our work to other cohort and longitudinal studies is of particular importance as serial measurement of cardiorespiratory fitness has significant prognostic value in clinical practice. Small increases in fitness are associated with reduced cardiovascular disease mortality risk and better clinical outcomes in patients with heart failure and type 2 diabetes23. Nevertheless, routine measurement of fitness in clinical practice is rare due to the costs and risks of exercise testing. Non-exercise-based regression models can be used to estimate changes in fitness in lieu of serial exercise testing. It is unclear, however, the extent to which changes in fitness detected with such models reflect true changes in exercise capacity. Here, we relied on the relationship between CRF and heart rate responses to different levels of physical activity at submaximal, real-life conditions captured through wearable sensors. Using deep learning techniques, we have developed a non-exercise-based fitness estimation approach that can be used not only to accurately infer current VO2max, but also to do so in the settings of a future cohort, where the model did not require any retraining, just influx of new data. Further, we show that the model can also be used to infer the changes in CRF that occurred during the ≈7 year time span between Fenland I and II.
Our proposed deep learning approach outperforms traditional non-exercise models, which are the state-of-the-art in the field and rely on simple variables inputted to a linear model. Importantly, our model is able to take week-level information from each participant and combine it with various anthropometrics and bio-markers such as the RHR, providing a truly personalized approach for CRF inference generation. The approach we present here outperforms traditional non-exercise models, which are considered state-of-the-art methods for longitudinal monitoring and highlights the potential of wearable sensing technologies for digital health monitoring. An additional application of our work is the potential routine estimation of VO2max in clinical settings, given the strong association between estimated CRF levels and CVD health outcomes24.
This study has several limitations worthy of recognition. First, the validity of the deep learning framework was assessed by comparing estimated VO2max values with VO2max values derived from a submaximal exercise test. Ideally, one would use VO2max values directly measured during a maximal exercise test to establish the ground truth for cardiorespiratory fitness comparisons. Maximal exercise tests, however, are problematic when used in large population-based studies because they may be unsafe for some participants and, consequently, induce selection bias. The submaximal exercise test used in the Fenland Study was well tolerated by study participants and demonstrated acceptable validity against direct VO2max measurements5. Submaximal tests are also utilized to validate popular wearable devices such as the Apple Watch25. We are therefore confident that VO2max values estimated from the deep learning framework reflect true cardiorespiratory fitness levels.
To investigate this limitation, we validate our models with 181 participants from the UK Biobank Validation Study (BBVS) who were recruited from the Fenland study. These participants completed an independent maximal exercise test, where VO2max was directly measured. Taking into account that the BBVS cohort is less fit (VO2max = 32.9 ± 7) compared to the cohort the model was trained on (Fenland I, VO2max = 39.5 ± 5), we observe that the model over-predicts with a mean prediction of VO2max = 39.9, RMSE = 8.998. This is expected because the range of VO2max seen during training did not include participants with VO2max below 25. Even when looking at women in isolation—who perform lower than men in these tests-, they had a VO2max = 37.4 ± 4.7 in Fenland I (see Fig. 1), which is still significantly higher than the average participant of BBVS. This is a common distribution/label shift issue where the model encounters an outcome which is out of its training data range and is still an open problem in statistical modelling26. To partially mitigate this issue, we match BBVS’s fitness to have similar statistics to the training set of original model (Fenland I: VO2max = 39 ± 5, matched BBVS: VO2max = 39 ± 4) and observe an RMSE = 5.19 (see Fig. Suppl. 1). This result is still not on par with our main results in Fenland but shows the impact of including very low-fitness participants in this validation study. To improve future CRF models, we believe that population-scale studies should focus on including low-fitness participants along with the general population.
Putting our results in context, we are confident that the level of accuracy of our methods is acceptable for use in population scale or even commercial wearables. For example, in Table 3, we observe that the MAE of all subgroups is between 2.1 and 2.3. Future directions to improve these results include transfer learning and domain adaptation, particularly tailored to the problem of distribution shift, as discussed in the previous paragraph. Compared to smaller studies such as the Apple Watch study which was conducted in more constrained environments that required users to log their workouts25, we see that the reported MAE is 1.4. We believe this difference is reasonable due to the completely free-living data we incorporate, bringing our study’s evaluation setup closer to the real world.
In this paper, we developed deep learning models utilising wearable data and other bio-markers to predict the gold standard of fitness (VO2max) and achieved strong performance compared to other traditional approaches. Cardio-respiratory fitness is a well-established predictor of metabolic disease and mortality and our premise is that modern wearables capture non-standardised dynamic data which could improve fitness prediction. Our findings on a population of 11,059 participants showed that the combination of all modalities reached an r = 0.82, when compared to the ground truth in a holdout sample. Additionally, we show the adaptability and applicability of this approach for detecting fitness change over time in a longitudinal subsample (n = 2675) who repeated measurements after 7 years. Last, the latent representations that arise from this model pave the way for fitness-aware monitoring and interventions at scale. It is often said that "If you cannot measure it, you cannot improve it". Cardio-fitness is such an important health marker, but until now we did not have the means to measure it at scale. Our findings could have significant implications for population health policies, finally moving beyond weaker health proxies such as the BMI.
Methods
Study description
The Fenland study is a population-based cohort study designed to investigate the independent and interacting effects of environmental, lifestyle, and genetic influences on the development of obesity, type 2 diabetes, and related metabolic disorders. Exclusion criteria included prevalent diabetes, pregnancy or lactation, inability to walk unaided, psychosis or terminal illness (life expectancy of ≤1 year at the time of recruitment).
The Fenland study has two distinct phases. Phase I, during which baseline data was collected from 12,435 participants, took place between 2005 and 2015. Phase II was launched in 2014 and involved repeating the measurements collected during Phase I, alongside the collection of new measures. All participants who had consented to be re-contacted after their Phase I assessment were invited to participate in Phase II. At least 4 years must have elapsed between visits. As a result of this stipulation, recruitment to Phase II is ongoing. A flowchart of the analytical sample by each one of the study tasks is provided in Fig. 1.
After a baseline clinic visit, participants were asked to wear a combined heart rate and movement chest sensor Actiheart, CamNtech, Cambridgeshire, UK) for 6 complete days. For this study, data from 11,059 participants were included after excluding participants with insufficient or corrupt data or missing covariates as shown in Fig. 1. A subset of 2675 of the study participants returned for the second phase of the study, after a median (interquartile range) of 7 (5–8) years, and underwent a similar set of tests and protocols, including wearing the combined heart rate and movement sensing for 6 days. All participants provided written informed consent and the study was approved by the University of Cambridge, NRES Committee—East of England Cambridge Central committee. All experiments and data collected were done in accordance with the declaration of Helsinki.
Study procedure
Participants wore the Actiheart wearable ECG which measured heart rate and movement recording at 60-s intervals27. The Actiheart device was attached to the chest at the base of the sternum by two standard ECG electrodes. Participants were told to wear the monitor continuously for 6 complete days and were advised that these were waterproof and could be worn during showering, sleeping, or exercising. During a lab visit, all participants performed a treadmill test that was used to establish their individual response to a submaximal test, informing their VO2max (maximum rate of oxygen consumption measured during incremental exercise)28. RHR was measured with the participant in a supine position using the Actiheart device. HR was recorded for 15 min and RHR was calculated as the mean heart rate measured during the last 3 min. Our RHR is a combination of the Sleeping HR measured by the ECG over the free-living phase and the RHR as described above.
Cardiorespiratory fitness assessment
VO2max was predicted in study participants using a previously validated submaximal treadmill test5. Participants exercised while treadmill grade and speed were progressively increased across several stages of level walking, inclined walking, and level running. The test was terminated if one of the following criteria were met: 1) the participant wanted to stop, 2) the participant reached 90% of age-predicted maximal heart rate (208-0.7*age)18, 3) the participants exercised at or above 80% of age-predicted maximal heart rate for 2 min. Details about the fitness characteristics of the cohort and the validation of the submaximal test are provided elsewhere29. To further validate the models trained on submaximal VO2max, we employ the external cohort UK Biobank Validation Study (BBVS)29. We recruited 105 female (mean age: 54.3y ± 7.3) and 86 male (mean age: 55.0y ± 6.5) validation study participants and VO2max was directly measured during an independent maximal exercise test, which was completed to exhaustion. Some maximal exercise test data were excluded because certain direct measurements were anomalous due to testing conditions (N = 10). BBVS participants completed the same free-living protocol as in Fenland and we collected similar sensor and antrhopometrics data which were processed with the same way as in Fenland (see next section).
Free-living wearable sensor data processing
Participants were excluded from this analysis if they had less than 72 h of concurrent wear data (three full days of recording) or insufficient individual calibration data (treadmill test-based data). All heart rate data collected during free-living conditions underwent pre-processing for noise filtering. Non-wear detection procedures were applied and any of those non-wear periods were excluded from the analyses. This algorithm detected extended periods of non-physiological heart rate concomitantly with extended (>90 min) periods that also registered no movement through the device’s accelerometer. We converted movement these intensities into standard metabolic equivalent units (METs), through the conversion 1 MET = 71 J/min/kg (3.5 ml O2 min− kg−1). These conversions where then used to determine intensity levels with ≤1.5 METs classified as sedentary behaviour, activities between 3 and 6 METs were classified as moderate to vigorous physical activity (MVPA) and those >6 METs were classified as vigorous physical activity (VPA). Since the season can have a big impact on physical activity considering on how it affects workouts, sleeping patterns, and commuting patterns, we encoded the sensor timestamps using cyclical temporal featuresTf. Here we encoded the month of the year as (x, y) coordinates on a circle:
where t is the relevant temporal feature (month). The intuition behind this encoding is that the model will “see” that e.g. December (12th) and January (1st) are 1 month apart (not 11). Considering that the month might change over the course of the week, we use the month of the first time-step only. Additionally, we extracted summary statistics from the following sensor time series: raw acceleration, HR, HRV, Aceleration-derived Euclidean Norm Minus One, and Acceleration-derived Metabolic Equivalents of Task30,31. Then, for every time series we extracted the following variables which cover a diverse set of attributes of their distributions: mean, minimum, maximum, standard deviation, percentiles (25%, 50%, 75%), and the slope of a linear regression fit. The rest of the variables (anthropometrics and RHR) are used as a single measurement.
In total, we derived a comprehensive set of 68 features using the Python libraries Pandas and Numpy. A detailed view of the variables is provided in Table Suppl. 2.
Deep learning models
We developed deep neural network models that are able to capture non-linear relationships between the input data and the respective outcomes. Considering the high-sampling rate of the sensors (1 sample/min) after aligning HR and Acceleration modalities, it is impossible to learn patterns with such long dependencies (a week of sensor data includes more than 10,000 timesteps). Even the most well-tuned recurrent neural networks cannot cope with such sequences and given the size of the training set (7545 samples), the best option was to extract statistical features from the sensors and represent every participant week as a row in a feature vector (see Fig. 2). This feature vector was fed to fully connected neural network layers which were trained with backpropagation. All deep learning models are implemented in Python using Tensorflow/Keras.
Data preparation
For Task 1 (see Fig. 1), we matched the sensor data with the participants who had eligible lab tests. Then we split into disjoint train and test sets, making sure that participants from Fenland I go to the train set, while those from Fenland II go to the test set (see Fig. 7). This would allow to re-use the trained model from Task 1, with different sensor data from Fenland II participants. Intuitively, we train a model on the big population, and we evaluate it with two snapshots of another longitudinal population over time (Task 1 and 3). After splitting, we normalize the training data by applying standard scaling (removing the mean and scaling to unit variance) and then denoise it by applying Principal Components Analysis (PCA), retaining the components that explain 99.99% of the variance. In practice, the original 68 features are reduced to 48. We save the fitted PCA projection and scaler and we apply them individually to the test-set, to avoid information leakage across the sets. The same projection and scaler are applied to all downstream models (Task 2 and 3) to leverage the knowledge of the big cohort (Fenland I).
Model architecture and training
The main neural network (used in Task 1) receives a 2D vector of [users, features] and predicts a real value. For this work, we assume \({{{\mathcal{N}}}}\) users and \({{{\mathcal{F}}}}\) features of an input vector \({\mathbf{X}}\) = (x1,...,xN) \(\in {{\mathbb{R}}}^{N\times F}\) and a target \(VO_{2}max_{\mathbf{y}}\) = (y1,...,yN) \(\in {{\mathbb{R}}}^{N}\). The network consists of two densely-connected feed-forward layers with 128 units each. A dense layer works as follows: output = activation (input ⋅ kernel + bias), where activation is the element-wise activation function (the exponential linear unit in our case), kernel is a learned weights matrix with a Glorot uniform initialization, and bias is a learned bias vector. Each layer is followed by a batch normalization operation, which maintains the mean output close to 0 and the output standard deviation close to 1. Also, dropout of 0.3 probability is applied to every layer, which randomly sets input units to 0 and helps prevent overfitting. Last, the final layer is a single-unit dense layer and the network is trained with the Adam optimizer to minimize the Mean Squared Error (MSE) loss, which is appropriate for continuous outcomes. We use a random 10% subset of the train-set as a validation set. To combat overfitting, we train for 300 epochs with a batch size of 32 and we perform early stopping when the validation loss stops improving after 15 epochs and the learning rate is reduced by 0.1 every 5 epochs. All hyperparameters (# layers, # units, dropout rates, batch size, activations, and early stopping) were found after tuning on the validation set.
Model differences across tasks
Task 1 trains the main neural network of our study (see previous subsection). Task 2 re-trains an identical model to predict VO2max in the future (and the delta present-future). We note that the delta prediction task cannot be comparable with the models predicting the present and future outcomes. Essentially, the delta model predicts the difference between these two timepoints, which results in a range of values roughly from −10 to +10. This distribution is not normally distributed (Shapiro-Wilk test = 0.991, p = 0.002) and hence both linear and neural models cannot approximate the tails, with most of their predictions lying between −3 and +3. The negative/positive signs of this outcome make the error metrics not very interpretable. We do not believe this performance is caused by overfitting because the results of both linear and neural models are similar. This result motivated us to study the delta distribution as a binary problem. When we re-frame this problem as a classification task (see Fig. 5), we use significantly fewer participants when we focus on the tails of the change distribution. Therefore, to combat overfitting, we train a smaller network with one Dense layer of 128 units and a sigmoid output unit, which is appropriate for binary problems. Instead of optimizing the MSE, we now minimize the binary cross-entropy. In all other cases—such as in Task 3 or when visualizing the latent space—, we do not train new models; the model which was trained in Task 1 is used in inference mode (prediction).
Prediction equations
For reference, we compare our models' results to traditional non-model equations, which rely on Body Mass, RHR, and Age. We incorporate the popular equation proposed by Uth et al.17, which corresponds to VO2max = 15.0 (\({m/\!*min}^{-1}\)) * Body Mass (kg) * (HRmax/HRrest), in combination with Tanaka’s equation18 where HRmax = 208 − \(0.7\!*\!age\). Other approaches rely on measurements such as the waist circumference, which however was not recorded in our cohorts.
Linear model
We begin our investigation by establishing a strong baseline with a linear regression model (as seen in Table 1). We compare differenet combinations of input data and finally compare the comprehensive model with the Dense neural network. We use the Python sklearn implementation for linear regression.
Evaluation
To evaluate the performance of the deep learning models which predict continuous values, we computed the root mean squared error (RMSE) \(=\sqrt{\frac{1}{\left\vert {N}_{test}\right\vert }{\sum }_{y\in {{{{\mathscr{D}}}}}_{test}}\mathop{\sum }\nolimits_{t = 1}^{N}{({y}_{t}-{\hat{y}}_{t})}^{2}}\), the coefficient of determination (R2) \(=1-\frac{\mathop{\sum }\nolimits_{t = 1}^{N}{({y}_{t}-{\hat{y}}_{t})}^{2}}{\mathop{\sum }\nolimits_{t = 1}^{N}{({y}_{t}-{\bar{y}}_{t})}^{2}}\), and the Pearson correlation coefficient for the majority of the analyses as they capture different properties of the error distributions in regression tasks. For the subgroup sensitivity analysis, we additionally employed the Mean Squared Error (MSE), Mean Absolute Error (MAE) and its standard deviation (STD of MAE), and the Mean Absolute Percentage Error (MAPE). In most regression metrics, y and \(\hat{y}\) are the measured and predicted VO2max and is the mean. For the binary models, we used the Area under the Receiver Operator Characteristic (AUROC or AUC) which evaluates the probability of a randomly selected positive sample to be ranked higher than a randomly selected negative sample.
Visualizing the latent space
The activations of the trained model allow us to understand the inner workings of the network and explore its latent space. We first pass the test set of Task 1 through the trained model and retrieve the activations of the penultimate layer32. This is a 2D vector of [2675, 128] size, considering that the layer size is 128 and the participants of the test set are 2675. Intuitively, every participant corresponds to a 128-dimensional point. In order to visualize this embedding, we apply tSNE33, an algorithm for dimensionality reduction. For its optimization, we use a perplexity of 50, as it was suggested recently34. We calculated the k-nearest neighbours on both the original and latent spaces (using k = 5) in a case study presented in Fig. 4. We also calculated the total euclidean distance of each query participant across all their neighbours, as a means of quantifying the proximity in the high-dimensional space.
Statistical analyses
We performed a number of sensitivity analyses to investigate potential sources of bias in our results. Full results of these sensitivity analyses are shown in the main text and corresponding Tables. In particular, we use bootstraping with replacement (500 samples) to calculate 95% Confidence Intervals when we report the performance of the models in the hold-out sets. Wherever we report p values, we use the recently suggested threshold of p < 0.005 for human studies35.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All Fenland data used in our analyses is available from the MRC Epidemiology Unit at the University of Cambridge upon reasonable request (http://www.mrc-epid.cam.ac.uk/research/studies/fenland/).
Code availability
Source code, data samples, and pre-trained models of this work are available on Github (https://github.com/sdimi/cardiofitness/).
References
Mandsager, K. et al. Association of cardiorespiratory fitness with long-term mortality among adults undergoing exercise treadmill testing. JAMA Netw. Open 1, e183605–e183605 (2018).
Swain, D. P., Brawner, C. A., of Sports Medicine, A. C. et al. ACSM’s Resource Manual for Guidelines for Exercise Testing and Prescription (Wolters Kluwer Health/Lippincott Williams & Wilkins, 2014).
Cao, Z.-B. et al. Predicting VO2max with an objectively measured physical activity in Japanese women. Med. Sci. Sports Exerc. 42, 179–186 (2010).
Gonzales, T. I. et al. Resting heart rate as a biomarker for tracking change in cardiorespiratory fitness of UK adults: the fenland study. medRxiv (2020).
Gonzales, T. I. et al. Estimating maximal oxygen consumption from heart rate response to submaximal ramped treadmill test. medRxiv (2020).
Nes, B. M. et al. Estimating VO2 peak from a nonexercise prediction model: the hunt study, norway. Med. Sci. Sports Exerc. 43, 2024–2030 (2011).
Spathis, D., Perez-Pozuelo, I., Brage, S., Wareham, N. J. & Mascolo, C. Self-supervised transfer learning of physiological representations from free-living wearable data. In Proceedings of the Conference on Health, Inference, and Learning, 69–78 (2021).
Dunn, J. et al. Wearable sensors enable personalized predictions of clinical laboratory measurements. Nature medicine 27, 1105–1112 (2021).
Shcherbina, A. et al. Accuracy in wrist-worn, sensor-based measurements of heart rate and energy expenditure in a diverse cohort. J. Person. Med. 7, 3 (2017).
Passler, S., Bohrer, J., Blöchinger, L. & Senner, V. Validity of wrist-worn activity trackers for estimating vo2max and energy expenditure. Int. J. Environ. Res. Public Health 16, 3037 (2019).
Cooper, K. D. & Shafer, A. B. Validity and reliability of the polar a300’s fitness test feature to predict vo2max. Int. J. Exerc. Sci. 12, 393 (2019).
Altini, M., Casale, P., Penders, J. & Amft, O. Cardiorespiratory fitness estimation in free-living using wearable sensors. Artif. Intell. Med. 68, 37–46 (2016).
Helgerud, J., Haglo, H. & Hoff, J. et al. Prediction of vo2max from submaximal exercise using the smartphone application myworkout go: Validation study of a digital health method. JMIR Cardio 6, e38570 (2022).
Richardson, K., Levett, D., Jack, S. & Grocott, M. Fit for surgery? perspectives on preoperative exercise testing and training. Br. J. Anaesth. 119, i34–i43 (2017).
Leon-Ferre, R., Ruddy, K. J., Staff, N. P. & Loprinzi, C. L. Fit for chemo: Nerves may thank you. J. Natl Cancer Inst. 109 (2017).
Lindsay, T. et al. Descriptive epidemiology of physical activity energy expenditure in uk adults (the fenland study). Int. J. Behav. Nutr. Phys. Activity 16, 1–13 (2019).
Uth, N., Sørensen, H., Overgaard, K. & Pedersen, P. K. Estimation of vo2max from the ratio between hrmax and hrrest–the heart rate ratio method. Eur. J. Appl. Physiol. 91, 111–115 (2004).
Tanaka, H., Monahan, K. D. & Seals, D. R. Age-predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 37, 153–156 (2001).
Gaspar, H. A. & Breen, G. Probabilistic ancestry maps: a method to assess and visualize population substructures in genetics. BMC Bioinform. 20, 1–11 (2019).
Schmid, D. & Leitzmann, M. Cardiorespiratory fitness as predictor of cancer mortality: a systematic review and meta-analysis. Ann. Oncol. 26, 272–278 (2015).
Schuch, F. B. et al. Are lower levels of cardiorespiratory fitness associated with incident depression? a systematic review of prospective cohort studies. Prev. Med. 93, 159–165 (2016).
Laukkanen, J. A., Kurl, S., Salonen, R., Rauramaa, R. & Salonen, J. T. The predictive value of cardiorespiratory fitness for cardiovascular events in men with various risk profiles: a prospective population-based cohort study. Eur. Heart J. 25, 1428–1437 (2004).
Jakicic, J. M. et al. Four-year change in cardiorespiratory fitness and influence on glycemic control in adults with type 2 diabetes in a randomized trial: the look ahead trial. Diabetes Care 36, 1297–1303 (2013).
Qui, S., Cai, X., Sun, Z., Wu, T. & Schumann, U. Is estimated cardiorespiratory fitness an effective predictor for cardiovascular and all-cause mortality? a meta-analysis. Atherosclerosis 330, 22–28 (2021).
Apple. Using apple watch to estimate cardio fitness with vo2 max (2021). https://www.apple.com/healthcare/docs/site/Using_Apple_Watch_to_Estimate_Cardio_Fitness_with_VO2_max.pdf. [Online; posted 2021, note = https://www.apple.com/healthcare/docs/site/Using_Apple_Watch_to_Estimate_Cardio_Fitness_with_VO2_max.pdf].
Lipton, Z., Wang, Y.-X. & Smola, A. Detecting and correcting for label shift with black box predictors. In Proceedings of International conference on machine learning, 3122–3130 (PMLR, 2018).
Brage, S., Brage, N., Franks, P. W., Ekelund, U. & Wareham, N. J. Reliability and validity of the combined heart rate and movement sensor actiheart. Eur. J. Clin. Nutr. 59, 561–570 (2005).
Brage, S. et al. Hierarchy of individual calibration levels for heart rate and accelerometry to measure physical activity. J. Appl. Physiol. 103, 682–692 (2007).
Gonzales, T. I. et al. Descriptive epidemiology of cardiorespiratory fitness in UK adults: The fenland study. medRxiv (2022).
Faurholt-Jepsen, M., Brage, S., Kessing, L. V. & Munkholm, K. State-related differences in heart rate variability in bipolar disorder. J. Psychiatr. Res. 84, 169–173 (2017).
White, T., Westgate, K., Wareham, N. J. & Brage, S. Estimation of physical activity energy expenditure during free-living from wrist accelerometry in uk adults. PLoS One 11, e0167472 (2016).
Yosinski, J., Clune, J., Nguyen, A., Fuchs, T. & Lipson, H. Understanding neural networks through deep visualization. In Proceedings of ICML Workshop on Deep Learning (2015).
Van der Maaten, L. & Hinton, G. Visualizing data using t-sne. J. Mach. Learn. Res. 9 (2008).
Wattenberg, M., Viégas, F. & Johnson, I. How to use t-sne effectively. Distill 1, e2 (2016).
Benjamin, D. J. et al. Redefine statistical significance. Nat. Hum. Behav. 2, 6–10 (2018).
Acknowledgements
D.S. was supported by the Embiricos Trust Scholarship of Jesus College Cambridge and the Engineering and Physical Sciences Research Council through grant DTP (EP/N509620/1). I.P.-P. was supported by GlaxoSmithKline and Engineering and Physical Sciences Research Council through an iCase fellowship (17100053).
Author information
Authors and Affiliations
Contributions
D.S., I.P.-P., S.B., N.W., C.M. designed the study. S.B., N.W. collected the data on behalf of the Fenland Study. D.S., I.P.-P. are co-first authors and sourced, selected, and pre-processed the data. D.S. developed the models and produced the figures and tables. D.S., I.P.-P. performed the statistical analysis. I.P.-P., D.S. wrote the first draft of the manuscript. All authors (D.S., I.P.-P., T.G., Y.W., S.B., N.W., C.M.) critically reviewed, contributed to the preparation of the manuscript, and approved the final version. All authors vouch for the data, analyses, and interpretations.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Spathis, D., Perez-Pozuelo, I., Gonzales, T.I. et al. Longitudinal cardio-respiratory fitness prediction through wearables in free-living environments. npj Digit. Med. 5, 176 (2022). https://doi.org/10.1038/s41746-022-00719-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-022-00719-1
- Springer Nature Limited