Abstract
The association between the composite dietary antioxidant index (CDAI) and hyperlipidemia remains unclear. Therefore, this study aimed to investigate the relationship between CDAI and hyperlipidemia. The data used in this study were obtained from the National Health and Nutrition Examination Survey (NHANES) dataset spanning from 2005 to 2020. Based on 24-h dietary recall interviews, the CDAI was calculated using the intake of six dietary antioxidants. Univariate and multivariate logistic regression models were employed to investigate the relationship between CDAI and the occurrence of hyperlipidemia. Additionally, restricted cubic spline (RCS) analysis was utilized to investigate potential non-linear relationships between the CDAI and risk of hyperlipidemia. The final analysis included 30,788 adults in the United States, among whom 25,525 (82.91%) were diagnosed with hyperlipidemia. A significant negative correlation was observed between the CDAI and hyperlipidemia in the unadjusted (Odds ratio [OR] 0.97 [95% CI 0.96, 0.98]) and multi-variable adjusted (OR 0.98 [95% CI 0.97, 0.99]) models. When the CDAI values were analyzed as a categorical variable, individuals in the highest quartile (OR 0.82 [95% CI 0.73, 0.92]) exhibited a nearly one fifth decreased risk of hyperlipidemia compared to those in the lowest quartile. Additionally, RCS analysis revealed a linear relationship between CDAI and hyperlipidemia (P for nonlinearity = 0.124). The results remained consistent across subgroups except for individuals under the age of 60 or those with diabetes mellitus. There was a significant negative correlation between the CDAI and risk of hyperlipidemia, indicating that maintaining an optimal CDAI level could effectively reduce the incidence of hyperlipidemia.
Similar content being viewed by others
Introduction
Hyperlipidemia is a metabolic disorder characterized by excessively elevated serum lipid levels1, which can be classified into different clinical types including hypercholesterolemia, hypertriglyceridemia, mixed hyperlipidemia, and high-density lipoproteinemia2. Hyperlipidemia has emerged as an important public health problem with increased prevalence worldwide. In the US, it is estimated that around 25 million adults had total cholesterol (TC) levels exceeding 200 mg/dL between 2017 and 20203. Hyperlipidemia was not only a major modifiable risk factor for cardiovascular and cerebrovascular diseases4 but also a leading cause of death5. Beyond pharmaceutical treatments, emerging studies emphasize the impact of non-pharmacological interventions in managing hyperlipidemia6.
Previous evidence suggested that hyperlipidemia was primarily associated with increased oxidative stress throughout the body7,8,9. Oxidative stress is a physiological condition characterized by an imbalance in the oxidation–reduction status within the cells or entire body, leading to over production of reactive oxygen species (ROS) such as oxygen free radicals10. The ROS cause damage to biomolecules within cells, such as lipids, proteins, and DNA, resulting in cell injury and abnormal inflammation11. Studies have shown that increasing antioxidant intake in the diet could reduce oxidative stress levels in the body12. Therefore, we speculated that modifying the dietary structure might treat hyperlipidemia by reducing oxidative stress. This hypothesis was first confirmed in a cross-sectional study from Korea, where they found that dietary total antioxidant capacity (TAC) was associated with dyslipidemia (hypercholesterolemia, hypertriglyceridemia, and hypoHDL-cholesterolemia)13. The Composite Dietary Antioxidant Index (CDAI) was a nutritional tool developed to assess the overall antioxidant capacity of an individual's daily food intake14,15,16,17. It took into account six specific antioxidants, namely vitamin A, vitamin C, vitamin E, carotenoids, selenium, and zinc14,15,16,17, with a higher CDAI score indicating a dietary pattern rich in antioxidants. Though previous studies have confirmed the relationship between CDAI and several chronic diseases, hypertension18, diabetes mellitus19, coronary heart disease20. However, the association between CDAI and hyperlipidemia has not been elucidated.
This study intended to determine the relationship between CDAI and hyperlipidemia by using data from the National Health and Nutrition Examination Survey (NHANES), thus, to provide better dietary guidance for hyperlipidemia management.
Methods
Data source and population
The data used in this study were obtained from the NHANES 2005–2020, a comprehensive cross-sectional survey that assessed the nutritional status and health of the US population21,22. Detailed information about the design methods and contents of NHANES could be found at http://www.cdc.gov/nchs/nhanes.htm. The study protocols were approved by the Ethics Review Board of the National Center for Health Statistics, and written informed consent was obtained from all participants prior to their participation in the survey. To reduce sampling and recall bias, this study gathered data for all participants (n = 85,750) who had undergone two dietary recalls from the NHANES datasets. This study initially excluded participants (n = 33,914) who were below the age of 18. Subsequently, participants lacking dietary information (n = 6261) and those without hyperlipidemia data (n = 8497) were also excluded. Moreover, this study also excluded participants with special diet (n = 5914) and implausible energy intake (< 500 kcal/day or ≥ 5000 kcal/day) (n = 376)13. The process of participant selection is depicted in Fig. 1, and the final analysis included 30,788 eligible participants.
Definition of CDAI
The information regarding dietary antioxidant intake was obtained through two separate 24-h dietary recall interviews23. Trained dietary surveyors recorded detailed information about the participants' dietary consumption in the 24 h prior to the interview. The first dietary recall was conducted through face-to-face interviews, while the second recall was carried out via telephone consultation 3–10 days later. Utilizing the average dietary intake data from two non-consecutive days was considered more accurate than relying solely on data from a single day16,24. Six antioxidants (zinc, selenium, total carotenoids, vitamins A, C, and E) were standardized by subtracting the mean and dividing by the standard deviation (SD). The CDAI, developed by Wright et al.25, was based on the sum of these standardized consumptions17,26. It should be noted that the calculation of dietary antioxidant intake in this study did not include antioxidants obtained from supplements, medications, or other additional sources. The detailed calculation formula is presented below:
Definition of hyperlipidemia
The hyperlipidemia status was evaluated based on the Adult Treatment Panel III (ATP 3) guidelines of the National Cholesterol Education Program (NCEP) for adults27. A diagnosis of hyperlipidemia was confirmed if any of the following five conditions were met: triglycerides (TG) ≥ 150 mg/dL, total cholesterol (TC) ≥ 200 mg/dL, low-density lipoprotein (LDL) ≥ 130 mg/dL, or high-density lipoprotein (HDL) ≤ 40 mg/dL in males and ≤ 50 mg/dL in females27. Additionally, participants who reported using cholesterol-lowering medications were also classified as having hyperlipidemia28,29.
Covariates
To evaluate the potential impact of confounding factors, we carefully selected several covariates, including age, body mass index (BMI), gender, race, education level, poverty income ratio (PIR), marital status, alcohol consumption, smoking status, diabetes mellitus, hypertension, physical activity and energy intake. The age of the participants selected for this study refers to their age at the time of screening. BMI was calculated by dividing an individual's weight (in kilograms) by the square of their height (in square meters). Gender was categorized as male or female. Race was classified as non-Hispanic White, non-Hispanic Black, Mexican American, other Hispanic, and other race. Education level was grouped as less than high school, high school, or college and above. The household socioeconomic status, estimated using the PIR index, was categorized into three groups: low (< 1.5), middle (1.5–3.5), and high (> 3.5)30. The marital status categories were defined as single, which included individuals who were separated, widowed, never married, or divorced, and married31. Alcohol consumption was classified into three categories: mild, moderate, and heavy. Heavy alcohol consumption was defined as consuming ≥ 3 drinks per day for females or ≥ 4 drinks per day for males, or engaging in binge drinking on five or more days per month. Moderate alcohol consumption was defined as consuming ≥ 2 drinks per day for females and ≥ 3 drinks per day for males, or engaging in binge drinking on ≥ 2 days per month. Mild alcohol consumption was considered as other forms of alcohol intake17,28,32. Smoking status was classified into three categories: never, former, and current. Never smokers were individuals who had smoked fewer than 100 cigarettes in their lifetime. Former smokers had smoked more than 100 cigarettes in their lifetime but currently did not smoke at all. Current smokers had smoked more than 100 cigarettes in their lifetime and currently smoked either occasionally or daily16,28. Diabetes mellitus was defined as individuals diagnosed with diabetes, those using anti-diabetes drugs or insulin. Hypertension was defined as individuals with an average blood pressure exceeding 140/90 mmHg, self-reporting a physician-diagnosed hypertension, or currently taking medication for hypertension. Physical activity was defined as total MET minutes per week, calculated by multiplying the weekly volume of physical activity (duration × frequency) for each activity by its corresponding MET value16,33. Energy intake was calculated by averaging the dietary recall data collected over a span of two days.
Statistical analysis
Participants characteristics were compared between participants with and without hyperlipidemia. Continuous data were presented as means (± SD) or interquartile range [IQR], and compared using either t test or non-parametric test. Categorical variables were presented as percentages (%) and compared using the chi-square test. Multiple imputation was used to handle missing values in the dataset. Univariate and multivariate logistic regression models were used to examine the relationship between CDAI (including both continuous variables and quartile groups) and hyperlipidemia, with the covariates listed above. In Model 1, no covariate was adjusted. In Model 2, adjustments were made for age and gender. Model 3 included additional covariates such as BMI, race, education level, PIR, marital status, alcohol consumption, smoking status, diabetes mellitus, hypertension, physical activity, and energy intake. Restricted cubic splines (RCS) were also used to explore potential nonlinearity. Identical analysis approach was employed to examine specific subgroups, including age, gender, BMI, hypertension and diabetes mellitus. All statistical analyses were performed using SPSS 27 and R 4.2.2 software. Results with a P < 0.05 were considered statistically significant.
Ethics statement
The studies were approved by The Research Ethics Review Board at the National Center for Health Statistics (NCHS). The participants provided their written informed consent to participate in this study.
Results
Baseline characteristics
In the final analysis, a total of 30,388 individuals from the NHANES datasets were included, with an average age of 49.17 (± 18.54) years, of whom 50.85% were female. There were several characteristics of significant differences between hyperlipidemic and non-hyperlipidemic population (Table 1). Compared to individuals without hyperlipidemia, those with hyperlipidemia exhibited several characteristics: older age, higher BMI levels, greater proportion of female gender, non-Hispanic white ethnicity, college education or above, married status, mild alcohol consumption, non-smoking history, absence of diabetes or hypertension, lower energy intake and physical activity levels, and higher CDAI levels (all P < 0.05).
Associations between CDAI levels and risks of hyperlipidemia
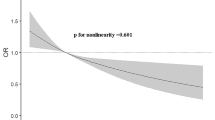
Logistic regression models were used to explore the associations between CDAI and hyperlipidemia (Table 2). In Model 1 (unadjusted), a negative association was observed between continuous CDAI and hyperlipidemia (OR 0.97 [95% CI 0.96, 0.98]). This relationship remained consistent after adjusting for covariates in Model 2 (OR 0.98 [95% CI 0.97, 0.98]) and Model 3 (OR 0.98 [95% CI 0.97, 0.99]). Compared to the CDAI of the lowest quartiles, participants in the third quartile (Q3) (OR 0.89 [95% CI 0.80, 0.98]) and fourth quartile (Q4) (OR 0.82 [95% CI 0.73, 0.92]) of CDAI had significantly decreased risk of hyperlipidemia, with a significant trend of the quartiles (P for trend: 0.01). The RCS analysis confirmed a linear relationship between CDAI and the risk of hyperlipidemia after full adjustment (P for nonlinearity: 0.124, Fig. 2).
The dose–response relationship between CDAI and the prevalence of hyperlipidemia. The blue line represents AOR, and the blue transparent area represents 95% CI. AOR results are adjusted based on Model 3. Abbreviations: AOR, adjusted odds ratio; CI, confidence interval, CDAI, composite dietary antioxidant index.
Association between six antioxidants (components of CDAI) and hyperlipidemia
Table 3 indicates the relationship between individual dietary antioxidant and hyperlipidemia. After adjustment, it was observed that dietary intake of vitamin E (OR 0.91 [95% CI 0.88, 0.94]), selenium (OR 0.95 [95% CI 0.91, 0.99]), and carotenoids (OR 0.96 [95% CI 0.93, 0.98]) exhibited a negative association with the risk of hyperlipidemia. However, a higher intake of dietary vitamin A, vitamin C and zinc did not show a decreased risk of hyperlipidemia.
Subgroup analysis
In all the subgroups (Fig. 3), consistently negative associations were observed between CDAI and hyperlipidemia after adjustment, though the odds in the subgroups of age and diabetes mellitus were insignificant. The P values for interaction were significant only in subgroups stratified by age (P for interaction: < 0.001) and gender (P for interaction: 0.047). Significant association was found only in subgroup populations with age greater than 60 years (OR 0.98 [95% CI 0.96, 0.99]), and the effect of CDAI was greater in the female population (OR 0.97 [95% CI 0.95, 0.98]) than the males (OR 0.99 [95% CI 0.98, 1.00]).
Discussion
In this study, involving 30,788 participants in the final analysis, we observed that elevated CDAI was significantly associated with a lower risk of hyperlipidemia, suggesting that improving CDAI might play a protective role in the development of hyperlipidemia. This study also showed that higher levels of vitamin E, selenium, and carotenoids were associated with a lower incidence of hyperlipidemia, while no significant correlation was observed in vitamin A, vitamin C, and zinc. The results remained consistent across most prespecified subgroups, except for individuals under the age of 60 or those with diabetes mellitus.
The CDAI, a widely recognized scoring system for total dietary antioxidants, has been extensively utilized in clinical research. For example, several cross-sectional studies have investigated and established the correlation between CDAI and various health conditions, such as coronary heart disease20, hypertension18, stroke15, diabetes mellitus19. While research on the association between CDAI and hyperlipidemia is still relatively limited, the exploration of using specific dietary antioxidants for managing hyperlipidemia has become a highly debated topic13. The mechanisms responsible for the negative correlation between CDAI and risk of hyperlipidemia are not yet well understood. It is known that oxidative stress plays a crucial role in the development of hyperlipidemia. In reverse, high levels of blood lipid typically result in increased levels of oxidative stress, which in turn, can worsen the progression of hyperlipidemia34.
The CDAI consists of six antioxidant components including antioxidant vitamins (vitamin A, vitamin C, and vitamin E) and micronutrients (zinc, selenium, and carotenoids), all of which play a crucial role in combating oxidative stress35,36. Vitamins play an essential role in lipid metabolism reactions37. Marguerite et al. conducted a randomized controlled study demonstrating that supplementation with antioxidant vitamins A, C and E could restore endothelial function in individuals with hyperlipidemia, thereby improving their long-term cardiovascular health38. However, this study found no association between vitamins A, C and hyperlipidemia. The difference in results may be attributed to variations in dietary habits across different populations. Consistent with our findings, a systematic review by Li et al. confirmed that serum zinc was not irrelevant with hyperlipidemia39,40. A randomized trial found that taking selenium supplementation could decrease non-high-density lipoprotein cholesterol levels41. However, there were also epidemiological studies indicating a positive association between selenium and lipid levels37,42,43. This difference could be attributed to several reasons. The definition of hyperlipidemia in this study was comprehensive and considered different types of blood lipids, including TC, TG, HDL, and LDL44. Furthermore, variances in the regions, races, and eating habits of the study population could also account for the diverse results. For carotenoids, many studies found a protective effect of carotenoids on cardiovascular diseases40,45, which was consistent with our findings. Vitamins E play a crucial role in alleviating oxidative stress and protecting cells from oxidative damage44. Selenium binds to selenoproteins, thereby preventing lipid peroxidation and reducing oxidative damage to cells46. Carotenoids exert antioxidative roles through both direct interactions with free radicals via electron or hydrogen atom transfer47 and by activating the hormetic-dependent activation of vitagenes48. Hence, we hypothesized that CDAI could potentially prevent the onset of hyperlipidemia induced by oxidative stress.
Subgroup analysis revealed that the negative association between CDAI and hyperlipidemia was robust across gender, BMI, and hypertension, proving the reliability and applicability of our findings. Notably, no association was found between CDAI and hyperlipidemia among individuals under 60 years old or those with diabetes mellitus. It was speculated that the beneficial effect of CDAI on hyperlipidemia may have been weakened in diabetic mellitus patients due to the control of a diabetic diet as well as the administration of insulin and hypoglycemic drugs. However, the exact mechanism remained unclear and required further investigation. Additionally, this study suggested that the association between CDAI and risk of hyperlipidemia was influenced by gender and age. Further research was warranted to investigate these mechanisms and gain a deeper understanding of this intricate relationship.
The CDAI has served as a gauge for a food's capacity to combat oxidative stress and has garnered widespread usage. By evaluating the quantity of antioxidants within food and their effectiveness in neutralizing oxidative substances, it assists in appraising the nutritional worth of various food items. Consequently, it empowers individuals to make healthier dietary selections, thereby preserving optimal health. The results of this study provide important support for the development of healthier dietary guidelines, assisting people in making better food choices to maintain cardiovascular health. Additionally, it may also contribute to deepening our understanding of antioxidants and their mechanisms of action in the human body, offering further insights for future research and clinical practice.
Strengths and limitations
The strengths of this study are as follows: Firstly, the data utilized in this study were derived from the NHANES dataset, which is of high quality and has garnered widespread recognition and application. Secondly, this study was the first to investigate the relationship between CDAI and hyperlipidemia in such a large population, as well as in specific subgroups. The limitations of our study can be summarized as follows: Firstly, although confounding factors have been extensively adjusted, there are still other potential factors that cannot be completely ruled out. Secondly, given the lack of direct correlation between the intake of nutrients of this study and the intake of other nutrients in this study, we did not adjust for them as confounding factors. More research is needed in the future to elucidate whether other nutrients may act as confounders in exploring the association between CDAI and risk of hyperlipidemia. Thirdly, the cross-sectional design in this study limited our ability to explore the causal relationships.
Conclusions
This cross-sectional study demonstrated that higher CDAI was significantly associated with a lower risk of hyperlipidemia. Moreover, the association was also found in individual components as vitamin E, selenium, and carotenoids. Improving dietary antioxidants consumption could be regarded as a practical strategy in hyperlipidemia prevention and lipid management.
Data availability
The detailed datasets about the surveys are available at www.cdc.gov/nchs/nhanes.
References
Su, X., Peng, H., Chen, X., Wu, X. & Wang, B. Hyperlipidemia and hypothyroidism. Clin. Chim. Acta. 527, 61–70. https://doi.org/10.1016/j.cca.2022.01.006 (2022).
Karr, S. Epidemiology and management of hyperlipidemia. Am. J. Manag. Care. 23(9 Suppl), S139–S148 (2017).
Tsao, C. W., Aday, A. W., Almarzooq, Z. I. et al. Heart disease and stroke statistics-2023 update: A report from the American Heart Association. Circulation. 147(8), e93–e621. https://doi.org/10.1161/CIR.0000000000001123 (2023).
Nelson, R. H. Hyperlipidemia as a risk factor for cardiovascular disease. Prim. Care. 40(1), 195–211. https://doi.org/10.1016/j.pop.2012.11.003 (2013).
Mozaffarian, D., Benjamin, E. J., Go, A. S. et al. Heart disease and stroke statistics–2015 update: A report from the American Heart Association. Circulation. 131(4), e29–322. https://doi.org/10.1161/CIR.0000000000000152 (2015).
Houston, M. C., Fazio, S., Chilton F. H. et al. Nonpharmacologic treatment of dyslipidemia. Prog. Cardiovasc. Dis. 52(2), 61–94. https://doi.org/10.1016/j.pcad.2009.02.002 (2009).
Yang, R-L., Shi, Y-H., Hao, G., Li, W., Le G-W. Increasing oxidative stress with progressive hyperlipidemia in human: Relation between malondialdehyde and atherogenic index. J. Clin. Biochem. Nutr. 43(3), 154–8. https://doi.org/10.3164/jcbn.2008044 (2008).
Devaki, M., Nirupama, R, Yajurvedi, H. N. Chronic stress-induced oxidative damage and hyperlipidemia are accompanied by atherosclerotic development in rats. Stress. 6(2), 233–43. https://doi.org/10.3109/10253890.2012.719052 (2013).
Bai, J., Zheng, S,, Jiang, D. et al. Oxidative stress contributes to abnormal glucose metabolism and insuli n sensitivity in two hyperlipidemia models. Int. J. Clin. Exp. Pathol. 8(10), 13193–200 (2015).
Sies, H., Berndt, C., Jones, D. P. Oxidative stress. Annu. Rev. Biochem. 86, 715–748. https://doi.org/10.1146/annurev-biochem-061516-045037 (2017).
Meng, S-J., Yu, L-J. Oxidative stress, molecular inflammation and sarcopenia. Int. J. Mol. Sci. 11(4), 1509–26. https://doi.org/10.3390/ijms11041509 (2010).
Daneshzad, E., Keshavarz, S-A., Qorbani, M., Larijani, B., Azadbakht, L. Dietary total antioxidant capacity and its association with sleep, stress, anxiety, and depression score: A cross-sectional study among diabetic women. Clin. Nutr. ESPEN. 37, 187–194. https://doi.org/10.1016/j.clnesp.2020.03.002 (2020).
Kim, S-A., Joung, H., Shin, S. Dietary pattern, dietary total antioxidant capacity, and dyslipidemia in Korean adults. Nutr. J. 18(1), 37. https://doi.org/10.1186/s12937-019-0459-x (2019).
Hu, W., Ye, Z., Li, T, Shi, Z. Associations between composite dietary antioxidant index and gout: National Health and Nutrition Examination Survey 2007–2018. Biol. Res. Nurs. 26(1), 150–159. https://doi.org/10.1177/10998004231198166 (2024).
Wang. T., Liu, H., Wei, X. Association between the composite dietary antioxidant index and stroke: A cross-sectional study. Biol. Trace Elem. Res. https://doi.org/10.1007/s12011-023-04011-5 (2023).
Wu, D., Wang, H., Wang, W, et al. Association between composite dietary antioxidant index and handgrip strength in American adults: Data from National Health and Nutrition Examination Survey (NHANES, 2011–2014). Front. Nutr. 10, 1147869. https://doi.org/10.3389/fnut.2023.1147869 (2023).
Zhang. J, et al. Associations between composite dietary antioxidant index and estimated 10-year atherosclerotic cardiovascular disease risk among U.S. adults. Front. Nutr. 10, 1214875. https://doi.org/10.3389/fnut.2023.1214875 (2023).
Wu, M., Si, J., Liu, Y., Kang, L., Xu, B. Association between composite dietary antioxidant index and hypertension: Insights from NHANES. Clin. Exp. Hypertens. 45(1), 2233712.https://doi.org/10.1080/10641963.2023.2233712 (2023).
Chen, X., Lu, H., Chen, Y., Sang, H., Tang, Y., Zhao, Y. Composite dietary antioxidant index was negatively associated with the prevalence of diabetes independent of cardiovascular diseases. Diabetol. Metab. Syndr. 15(1), 183. https://doi.org/10.1186/s13098-023-01150-6 (2023).
Ma, R. et al. Association between composite dietary antioxidant index and coronary heart disease among US adults: A cross-sectional analysis. BMC Public Health. 23(1), 2426. https://doi.org/10.1186/s12889-023-17373-1 (2023).
Fain, J. A. NHANES. Diabetes Educ. 43(2), 151. https://doi.org/10.1177/0145721717698651 (2017).
Curtin, L. R., et al. The National Health and Nutrition Examination Survey: Sample design, 1999–2006. Vital Health Stat 2(155), 1–39 (2012).
Pannucci, T. E. et al. Comparing reported dietary supplement intakes between two 24-hour recall methods: The automated self-administered 24-hour dietary assessment tool and the interview-administered automated multiple pass method. J. Acad. Nutr. Diet. 118(6), 1080–1086. https://doi.org/10.1016/j.jand.2018.02.013 (2018).
Yang, T. et al. Associations of dietary fats with all-cause mortality and cardiovascular disease mortality among patients with cardiometabolic disease. Nutrients. 14(17), 3608. https://doi.org/10.3390/nu14173608 (2022).
Maugeri, A. et al. Dietary antioxidant intake decreases carotid intima media thickness in women but not in men: A cross-sectional assessment in the Kardiovize study. Free Radic. Biol. Med. 131, 274–281. https://doi.org/10.1016/j.freeradbiomed.2018.12.018 (2019).
Liu, C., Lai, W., Zhao, M., Zhang, Y., Hu, Y. Association between the composite dietary antioxidant index and atherosclerotic cardiovascular disease in postmenopausal women: A cross-sectional study of NHANES data, 2013–2018. Antioxidants (Basel). 12(9), 1740. https://doi.org/10.3390/antiox12091740 (2023).
Lipsy, R. J. The national cholesterol education program adult treatment panel III guidelines. J. Manag. Care Pharm. 9(1 Suppl), 2–5. https://doi.org/10.18553/jmcp.2003.9.s1.2 (2003).
Mahemuti, N. et al. Association between systemic immunity-inflammation index and hyperlipidemia: A population-based study from the NHANES (2015–2020). Nutrients. 15(5), 1177. https://doi.org/10.3390/nu15051177 (2023).
Merkin, S. S, et al. Education differentials by race and ethnicity in the diagnosis and management of hypercholesterolemia: A national sample of U.S. adults (NHA NES 1999–2002). Int. J. Public Health. 54(3), 166–74. https://doi.org/10.1007/s00038-008-7030-4 (2009).
Fang, Y, et al. The role of socioeconomic status and oxidative balance score in erectile dysfunction: A cross-sectional study. Heliyon. 9(11), e22233. https://doi.org/10.1016/j.heliyon.2023.e22233.
Han, Y., Jiang, X., Qin, Y., Zhao, Y., Zhang, G, Liu, C. A cross-sectional study exploring the relationship between the dietary inflammatory index and hyperlipidemia based on the National Health and Nutrition Examination Survey (2005–2018). Lipids Health Dis. 22(1), 140. https://doi.org/10.1186/s12944-023-01908-x (2023).
Phillips, J. A. Dietary guidelines for Americans, 2020–2025. Workplace Health Saf. 69(8), 395. https://doi.org/10.1177/21650799211026980 (2021).
Tucker, J. M., Welk, G. J. & Beyler, N. K. Physical activity in U.S.: Adults compliance with the Physical Activity Guidelines for Americans. Am. J. Prev. Med. 40(4), 454–61. https://doi.org/10.1016/j.amepre.2010.12.016 (2011).
Ito, F., Sono, Y., Ito T. Measurement and clinical significance of lipid peroxidation as a biomarker of oxidative stress: Oxidative stress in diabetes, atherosclerosis, and chronic inflammation. Antioxidants (Basel). 8(3), 72. https://doi.org/10.3390/antiox8030072 (2019).
Hajam, Y. A, et al. Oxidative stress in human pathology and aging: Molecular mechanisms and perspectives. Cells. 11(3), 552. https://doi.org/10.3390/cells11030552 (2022).
Klotz, L-O., Kröncke, K-D., Buchczyk, DP., Sies, H. Role of copper, zinc, selenium and tellurium in the cellular defense against oxidative and nitrosative stress. J. Nutr. 133(5 Suppl 1), 1448S–51S. https://doi.org/10.1093/jn/133.5.1448S (2003).
Engler, M. M. et al. Antioxidant vitamins C and E improve endothelial function in children with hyperlipidemia: Endothelial Assessment of Risk from Lipids in Youth (EARLY) trial. Circulation. 108(9), 1059–63. https://doi.org/10.1161/01.CIR.0000086345.09861.A0 (2003).
Li, C-P., Song, Y-X., Lin, Z-J., Ma, M-L., He, L-P. Essential trace elements in patients with dyslipidemia: A meta-analysis. Curr. Med. Chem. https://doi.org/10.2174/0929867330666230428161653 (2024).
Rayman, M. P., Stranges, S., Griffin, B. A., Pastor-Barriuso, R., Guallar, E. Effect of supplementation with high-selenium yeast on plasma lipids: A randomized trial. Ann. Intern. Med. 154(10), 656–65. https://doi.org/10.7326/0003-4819-154-10-201105170-00005 (2011).
Stranges. S, et al. Selenium status and blood lipids: the cardiovascular risk in Young Fin ns study. J. Intern. Med. 270(5), 469–77. https://doi.org/10.1111/j.1365-2796.2011.02398.x (2011).
Fülöp, P., Seres, I., Jenei, Z., Juhász, .I, Paragh G. Increased hair selenium concentration in hyperlipidemic patients. J. Cell Mol Med. 17(3), 350–5. https://doi.org/10.1111/jcmm.12013 (2013).
Stranges, S. et al. Higher selenium status is associated with adverse blood lipid profile in British adults. J. Nutr. 140(1), 81–7. https://doi.org/10.3945/jn.109.111252 (2010).
Morris, D. L., Kritchevsky, S. B., Davis, C. E. Serum carotenoids and coronary heart disease. The lipid research clinics coronary primary prevention trial and follow-up study. JAMA. 272(18), 1439–41. https://doi.org/10.1001/jama.272.18.1439 (1994).
Yao, Y., Goh, H. M., Kim, J. E. The roles of carotenoid consumption and bioavailability in cardiovascular health. Antioxidants (Basel). 10(12), 1978. https://doi.org/10.3390/antiox10121978 (2021).
Barcarolo D, et al. Effect of parenteral supplementation of minerals and vitamins on oxidative stress biomarkers and hepatic fatty acid metabolism in dairy cows during the transition period. Biol. Trace Elem. Res. https://doi.org/10.1007/s12011-023-03776-z (2023).
Ramakrishnan, M. et al. Selenium: A potent regulator of ferroptosis and biomass production. Chemosphere. 306, 135531. https://doi.org/10.1016/j.chemosphere.2022.135531 (2022).
Zhuang, C. et al. Effects of oral carotenoids on oxidative stress: A systematic review and meta-analysis of studies in the recent 20 years. Front. Nutr. 9, 754707. https://doi.org/10.3389/fnut.2022.754707 (2022).
Calabrese, V., Cornelius, C., Dinkova-Kostova, A. T., Calabrese, E. J., Mattson, M. P. Cellular stress responses, the hormesis paradigm, and vitagenes: Novel targets for therapeutic intervention in neurodegenerative disorders. Antioxid. Redox Signal. 13(11), 1763–811. https://doi.org/10.1089/ars.2009.3074 (2010).
Acknowledgements
All authors sincerely thank NHANES for its open-access data.
Author information
Authors and Affiliations
Contributions
C.H. and Z.M.L. designed the study and wrote the manuscript. Z.M.L. and Z.D.W. analyzed the data. Z.Q.P. and L.Y. critically revised the manuscript. All authors have read and agreed to submit the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, M., Zhang, D., Zhang, Q. et al. Association between composite dietary antioxidant index and hyperlipidemia: a cross-sectional study from NHANES (2005–2020). Sci Rep 14, 15935 (2024). https://doi.org/10.1038/s41598-024-66922-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-66922-0
- Springer Nature Limited