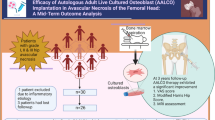
Abstract
This study was desinged to evaluate the efficacy and safety of activated allograft combined with the induced membrane technique for reconstruction of infected segment bone defects of lower limbs. A retrospective analysis was conducted on 19 patients from May 2015 to February 2017. After debridements, the bone defects were filled with antibiotic bone cement to form the induced membrane. Autologous mesenchymal stem cells were seeded onto allografts to construct activated allograft, which was implanted into the induced membrane after infection was controlled. The clinical efficacy and complications were observed. 19 patients with 20 infected segment bone defect were evaluated. The average deficit size was 11.08 (4–17) cm in length. After a mean follow-up of 71.84 (61–82) months, bone union was achieved in 16 patients (17 sites), resulting in a final union rate of 84.21% (16/19 patients). The average bone union time was 10.18 (5–28) months. There were 2 patients with recurrence of infection, 3 patients with graft absorption, and 1 patient with malunion due to implant breakage. There were no graft-related complications. This study provides clinical significance for the treatment of patients with insufficient autologous bone.
Similar content being viewed by others
Introduction
Although significant progress has been made in bone defect treatments, the clinical treatment efficacy of large bone defects remains an area that requires further optimized. Induced membrane technique is a widely used strategy for bone defects due to its simple operation process and few complications1,2,3. Although induced membrane provide a variety of osteogenic and vascular factors for autologous cancellous bone, which can promote the rapid healing of bone defects4,5. One of the difficulties of the induced membrane technique is that the total amount of autologous cancellous bone in patients cannot meet the needs of large segmental bone defects6. The use of allogeneic for the treatment of bone defects is not very effective in patients with insufficient autologous bone mass or unwillingness to accept complications in the autologous bone donor area. Activated allograft, which simulates the structure and function of autologous bone through seeding autologous osteogenesis precursor cell on a scaffold, is expected to solve this problem. Although significant progress has been made in basic research on activated allograft, it has not been widely used in clinical practice. In this study, we report a series of patients with infected segment bone defects of lower limbs treated with activated allograft combined with the induced membrane technique. Our aim was to investigate the outcomes of this new method and to provide a reference for clinicians when applying this method.
Patients and methods
We restrospectively analysed medical records of patients with large segmental bone defects caused by bone infection in our deparment between May 2015 and February 2017. The Ethics Committee of the First Affiliated Hospital of the Army Medical University approved this retrospective investigation, and all methods were performed in accordance with the relevant guidelines and regulations. The need of informed consent was waived by The Ethics Committee of the First Affiliated Hospital of the Army Medical University due to retrospective nature of the study. The inclusion criteria were: (1) Lower extremity bone infection patients admitted to our department; (2) Segment bone defects > 3 cm; (3) Patients with insufficient autologous cancellous bone; (4) Treatment with activated allograft combined with induced membrane technique; (5) Age < 60 years; (6) Follow-up time > 5 years. The exclusion criteria were: (1) Infections without curative treatment (palliative care); (2) Patients with malignant diseases; (3) Serious vascular and nerve injuries with no possibility of limb salvage.
Surgical technique
In the first stage, the bone defects were filled with antibiotic bone cement in order to eradicate infection and form induced membrane after thorough debridement. In the second stage, activated allograft bone was implanted in the induced membrane after the removal of antibiotic bone cement to reconstruct bone defects (Fig. 1).
(A) Preoperative photo; (B) Infected soft tissue and bone tissue; (C) After debridement; (D) Antibiotic bone cement filled with the defects and wrapped the fixation (locking plate); (E,F) Activated allograft; (G) Intramedullary nails are used to fix the bone ends in the bone reconstruction stage; (H) Implantation of Activated allograft.
Debridement
The infection site and range of debridement were determined by imaging results of magnetic resonance and bone scanning preoperatively. The sinus, infected bone and surrounding necrotic tissue were debrided and samples were taken for bacterial culture and pathological examination. After debridement, antibiotic bone cement (5 g vancomycin per 40 g bone cement) were implanted to fill the remained bone defects (Fig. 1D). Sensitive antibiotics were given for 2 weeks according to bacterial culture results and drug sensitivity results.
Preparation of activated allograft bone
Assessment of systemic or local infection eradication: (1) no swelling, pain, redness and pus at the infection site; (2) serum inflammation index declined to normal, (3) no active infection in the whole body; (4) the imaging examination did not indicate infection. A total of 50 ml of autologous bone marrow was taken from multi-site of the anterior or posterior iliac crests and bone marrow derived MSCs (BMSCs) were isolated. The surface markers of BMSCs were determined by flow cytometry (Fig. 2). Antibodies including FITC-labeled CD90, APC-labeled CD73, PE-labeled CD44, percp-cy5.5-labeled CD105 and PE-labeled CD19 + /CD11b + /CD34 + /CDHLa-DR + mixed antibodies (all purchased from BD) were used. The differentiation capacity of the BMSCs were further determined by specific staining (Fig. 3). Allogeneic bone (Beijing Daxing Biotechnology Co., Ltd) was cut to a diameter of 0.3 cm × 0.3 cm × 0.3 cm, then immersed in the BMSCs culture medium. 1 ml BMSCs suspension in a density of 1 × 107/ml were inoculated into 4 cm3 of bone tissue, then cultured for 3 days. Morphology of the stem cells in scaffolds were observed under a microscope (Fig. 4). To ensure their safety, the scaffold underwent testing for the presence of bacteria, endotoxin, and mycoplasma, and was only implanted following negative results. The bone graft material was washed with sterilized saline and implanted into the bone defects.
Bone grafting
Before bone grafting, the length and diameter of the bone defects were measured by CT scan, and then estimate the volume of the bone defects (V = πR2 × H according to cylinder, π = 3.14, R is the radius of the bone defects, H is the length of the bone defect), plus 30% of the calculated volume for the actual implantation. The bone cements were removed after 8 weeks. The surrounding induced membrane will be protected during the secondary operation. After performing a rapid pathological examination intraoperatively in frozen sections, only patients who were determined to not have suppurative inflammation were eligible for grafting. Sensitive antibiotics were used prophylactively in the second stage according to the results of intraoperative bacterial culture. During the bone reconstruction stage, intramedullary nails are used to fix the bone ends (Fig. 1G). The defects were fully filled with activated allograft bone. The induced membrane was sutured carefully, drainage was left and incision was closed after grafting.
Postoperative follow-up
Follow-up was performed every month after the first stage and every 3 to 6 months after the second stage. The review included clinical symptoms and signs of infection (such as swelling, fever, pain and sinus formation), laboratory examinations (WBC, ESR, CRP). The recurrence of infection, bone healing and complications were observed and recorded. Infection control was defined as the absence of both clinical symptoms and normalisation of laboratory inflammatory markers throughout the visits. The bone union time was obtained by X ray. Bone union was defined as three-sided cortical bridging on two perpendicular X-rays of the defect zone7. After X-ray examination reveals the formation of callus, weight-bearing exercises can be initiated, and the weight can be gradually increased.
Results
19 patients with 20 infected segment bone defects were enrolled in this study, including 14 males and 5 females with an average age of 28 (5–52) years. According to the aetiological calssification proposed by Waldvogel, there were 4 cases of haematogenous osteomylelitis and 15 cases of post-traumatic osteomyelitis. The duration of bone infection before admission ranged from 1 to 250 months, and an average of 4.79 surgical procedures were performed.
After thorough debridement, there were residual segmental bone defects in 13 femurs and 7 tibias, with one patient involving both the femur and tibia. The average length of bone defects was 11.08 (4–17) cm. Among the 19 patients, 15 (78.95%) had positive bacterial isolations. Among these cases, 7 (46.7%) cases infected with Staphylococcus aureus (including 5 with methicillin-resistant Staphylococcus aureus), 5 with Escherichia coli, 3 with Pseudomonas aeruginosa, 1 with Enterococcus faecalis, and 3 other types. In 4 remaining cases, no bacteria were isolated (data shown in Table 1). The average bone graft volume was 154 (50–350) cm3.
After a mean follow-up of 71.84 (61–82) months, bone union was achieved in 16 patients (17 sites), resulting a final union rate of 84.21% (16/19 patients). The average time to bone union was 10.18 (5–28) months. There was recurrence of infection in 2 (12.5%) patients. One patients refused further treatment and opted for amputation, the other was treated with bone transport technique. Additionally, three cases with graft absorption required further autografting. Malunion due to implant breakage occurred in one case. During the follow-up period, there were no graft-related complications. (Typical case is presented in Fig. 5).
Discussions
Treatment of infected bone defects is challenging, and the ultimate goal is to clear the infection and restore limb function. Cierny-Mader type IV osteomyelitis requires thorough debridement and often results in bone defects when the infection is controlled. To solve this problem, various techniques have been developed, such as the Illizarov technique. Since its report in 2000, induced membrane technology has gradually gained popularity and been gradually promoted in clinical applications in recent years8. Induced membrane technique has been used to repair infected bone defects in extremities in our department since 2012, and the early clinical effect is good9,10. However, the lack of autologous bone graft is still one of the difficulties in repairing large segmental bone defects with membrane induction technique.
Autogenous cancellous bone graft has long been considered the gold standard for repairing bone defects due to its fast repair and low revision rate compared with other materials. However, the limited availability of autogenous bone and potential donor site complications limit its use in clinical treatment11,12. Therefore, researchers began to search for materials that could replace autologous cancellous bone.
Tissue-engineered grafts have shown promise as an alternative to autografts13. In 2001, Quarto reported successful treatment of 3 cases of bone defects using tissue-engineered grafts constructed with MSCs and macroporous hydroxyapatite scaffolds14. In 2010, Eric successfully repaired a 7.2 cm bone defect using a novel multiple disc graft constructed with autologous mesenchymal stem cells and calf bone in a 58-year-old patient15. Although these cases pioneered the use of tissue engineered grafts to treat bone defects, they are more commonly used at nonbearing sites, such as facial and cranial bones, and the extent of bone defects is modest16,17. In 2010, Maik reported that allogenic cancellous bone seeded with human mesenchymal stromal cells has the potential to repair bone defects, but it has not been widely used in clinical practice18. In this study, the induced membrane technique combined with activated allogeneic bone was applied to the clinical treatment of infectious bone defects. We followed up with patients for an average of more than 5 years and found that a final union rate of 84.21% (16/19 patients).
Osteogenesis, osteoinduction and osteoconduction of the graft are particularly important for bone defects repair13,19. Although autograft has long been considered the gold standard, they are limited to small bone defects20. For large segmental bone defects, autologous bone is prone to bone resorption, suggesting special requirements for osteogenic environment. Induced membrane technology provides an osteogenic microenvironment rich in stem cells and vascular cells for the graft, which is an effective method to repair bone defects1,5. Activated allogeneic bone is one of the popular alternative materials in recent years, but its clinical application is limited due to the difficulty of vascularized construction in vivo. The process of bone healing is controlled by engineering adult stem cells to express genes like bone morphogenetic proteins, core binding factor α1 (Cbfa1), vascular endothelial growth factor (VEGF)21. Induced membranes are constructed in vivo using perfusion bioreactors and serve as a mechanical barrier to prevent reabsorption of the contained graft. Most importantly, the induced membrane provided a large amount of osteoinductive factors, growth factors, stem cells, and vascular cells that can promote revascularization and osteogenesis22. In this study, we used the induced membrane technique combined with activated allograft bone seeded with autologous stem cells to repair large segmental infected bone defects. The bone union rate is 84.21% (16/19 patients) and the overall efficacy was reliable. It suggests that the induced membrane plays an important role in the early stage of bone defects reconstrution, possibly due to vascularization of the graft. However, the average healing time was 10.18 (5–28) months in this group, which was longer than that in autogenous bone group10. These results suggest the importance of graft materials in osseointegration and osteogenesis in repairing large segmental bone defects.
The goal of a successful bone graft is to restore the anatomical, physiological and functional status of the bone23. Activated allograft bone involves the use of osteoconductive biomaterial scaffolds with osteogenic cell populations and osteoinductive bioactive factors24. The scaffolds should have appropriate shape, size, and mechanical competence25 and serve as a template for cell interactions and the formation of bone extra cellular matrix to provide structural support to the newly formed tissue24,26. While various synthetic biomaterials like inorganic ceramics metals, and synthetic biodegradable polymer composites have been investigated for their potential as bone scaffold materials24, their common drawback is inherent brittleness27. In this study, we selected allogeneic bone as scaffold material, which is structurally closest to autogenous bone and has high osteoinductive potential and remodeling characteristics28,29, making it an ideal tissue-engineered scaffold material. Bone marrow is a multipotent stem cell reservoir of mesenchymal tissue that can differentiate into fibroblasts, osteoblasts, adipocytes and reticular cells30. In the present study, we isolated autologous BMSCs and seeded them in scaffolds to construct individual tissue-engineered bone, which has certain advantages, such as accessibility, lack of ethical controversies, painless for donors.
In the present study, we observed graft absorption in three patients. To prevent this, we recommend that the bone graft volume should be at least 120% greater than or equeal to the bone defect volume. Although activated allograft has shown promise for reconstructing large segmental bone defects, it showed longer union time compared to autografts. Therefore, in future research, we should focus on how to improve the activity of seeded cells to accelerate bone uniou, as well as determine the appropriate age, site, and clinical application indications.
Our study highlights the importance of graft materials in the reconstruction of large segmental bone defects and the potential of activated allografts as alternatives to autografts. However, there are still some limitations in this study. First, this is a preliminary clinical study, and the mechanisms behind the observed effect needs further in-depth investigation and discussion. For example, there is a lack of data on the biomechanical stability of the created bone materials. Second, our activated allogeneic bone simply seeded stem cells on the scaffold without adding growth factors to improve its clinical efficacy, which may contribute to the prolonged bone healing time. Third, the source of allogeneic bone is limited and the technical requirements are high. This limits its widespread clinical application and can only be applied in some qualified hospitals. Fourth, there is a lack of comparison with other methods.
Conclusion
Activated allograft combined with the induced membrane technique is a safe and reliable method for the treatment of large infected segmental bone defects in the lower extremity.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References:
Taylor, B. C., French, B. G., Fowler, T. T., Russell, J. & Poka, A. Induced membrane technique for reconstruction to manage bone loss. J. Am. Acad. Orthop. Surg. 20, 142–150. https://doi.org/10.5435/JAAOS-20-03-142 (2012).
Wang, X., Luo, F., Huang, K. & Xie, Z. Induced membrane technique for the treatment of bone defects due to post-traumatic osteomyelitis. Bone Jt. Res. 5, 101–105. https://doi.org/10.1302/2046-3758.53.2000487 (2016).
Olesen, U. K. et al. The Masquelet technique of induced membrane for healing of bone defects. A review of 8 cases. Injury 46, S44–S47. https://doi.org/10.1016/s0020-1383(15)30054-1 (2015).
Cuthbert, R. J. et al. Induced periosteum a complex cellular scaffold for the treatment of large bone defects. Bone 57, 484–492. https://doi.org/10.1016/j.bone.2013.08.009 (2013).
Aho, O. M. et al. The mechanism of action of induced membranes in bone repair. J. Bone Jt. Surg. Am. 95, 597–604. https://doi.org/10.2106/JBJS.L.00310 (2013).
Masquelet, A. C. & Begue, T. The concept of induced membrane for reconstruction of long bone defects. Orthop. Clin. 41, 27–37. https://doi.org/10.1016/j.ocl.2009.07.011 (2010).
Blum, A. L., BongioVanni, J. C., Morgan, S. J., Flierl, M. A. & dos Reis, F. B. Complications associated with distraction osteogenesis for infected nonunion of the femoral shaft in the presence of a bone defect: A retrospective series. J. Bone Jt. Surg. Br. Vol. 92, 565–570. https://doi.org/10.1302/0301-620X.92B4.23475 (2010).
Masquelet, A. C., Fitoussi, F., Begue, T. & Muller, G. P. Reconstruction of the long bones by the induced membrane and spongy autograft. Ann. Chir. Plast. Esthet. 45, 346–353 (2000).
Wang, X. et al. Current data on extremities chronic osteomyelitis in southwest China: Epidemiology, microbiology and therapeutic consequences. Sci. Rep. 7, 16251. https://doi.org/10.1038/s41598-017-16337-x (2017).
Yu, X., Wu, H., Li, J. & Xie, Z. Antibiotic cement-coated locking plate as a temporary internal fixator for femoral osteomyelitis defects. Int. Orthop. 41, 1851–1857. https://doi.org/10.1007/s00264-016-3258-4 (2017).
Flierl, M. A. et al. Outcomes and complication rates of different bone grafting modalities in long bone fracture nonunions: A retrospective cohort study in 182 patients. J. Orthop. Surg. Res. 8, 33. https://doi.org/10.1186/1749-799X-8-33 (2013).
Horch, R. E., Beier, J. P., Kneser, U. & Arkudas, A. Successful human long-term application of in situ bone tissue engineering. J. Cell. Mol. Med. 18, 1478–1485. https://doi.org/10.1111/jcmm.12296 (2014).
Zhang, M. et al. Recent developments in biomaterials for long-bone segmental defect reconstruction: A narrative overview. J. Orthop. Transl. 22, 26–33. https://doi.org/10.1016/j.jot.2019.09.005 (2020).
Quarto, R. et al. Repair of large bone defects with the use of autologous bone marrow stromal cells. N. Engl. J. Med. 344, 385–386. https://doi.org/10.1056/NEJM200102013440516 (2001).
Hesse, E. et al. Repair of a segmental long bone defect in human by implantation of a novel multiple disc graft. Bone 46, 1457–1463. https://doi.org/10.1016/j.bone.2010.02.011 (2010).
Bhumiratana, S. et al. Tissue-engineered autologous grafts for facial bone reconstruction. Sci. Transl. Med. 8, 343ra83. https://doi.org/10.1126/scitranslmed.aad5904 (2016).
Schimming, R. & Schmelzeisen, R. Tissue-engineered bone for maxillary sinus augmentation. J. Oral Maxillofac. Surg. 62, 724–729. https://doi.org/10.1016/j.joms.2004.01.009 (2004).
Stiehler, M. et al. Cancellous bone allograft seeded with human mesenchymal stromal cells: A potential good manufacturing practice-grade tool for the regeneration of bone defects. Cytotherapy 12, 658–668. https://doi.org/10.3109/14653241003774052 (2010).
Moghaddam, A., Zietzschmann, S., Bruckner, T. & Schmidmaier, G. Treatment of atrophic tibia non-unions according to ‘diamond concept’: Results of one- and two-step treatment. Injury 46(Suppl 4), S39–S50. https://doi.org/10.1016/s0020-1383(15)30017-6 (2015).
Woon, C. Y., Chong, K. W. & Wong, M. K. Induced membranes—a staged technique of bone-grafting for segmental bone loss: A report of two cases and a literature review. J. Bone Jt. Surg. Am. 92, 196–201. https://doi.org/10.2106/JBJS.I.00273 (2010).
Fleming, J. E. Jr., Cornell, C. N. & Muschler, G. F. Bone cells and matrices in orthopedic tissue engineering. Orthop. Clin. 31, 357–374. https://doi.org/10.1016/s0030-5898(05)70156-5 (2000).
Wu, H. et al. Discovery of multipotent progenitor cells from human induced membrane: Equivalent to periosteum-derived stem cells in bone regeneration. J. Orthop. Transl. 42, 82–93. https://doi.org/10.1016/j.jot.2023.07.004 (2023).
Hofmann, S. et al. Remodeling of tissue-engineered bone structures in vivo. Eur. J. Pharm. Biopharm. 85, 119–129. https://doi.org/10.1016/j.ejpb.2013.02.011 (2013).
Khan, W. S., Rayan, F., Dhinsa, B. S. & Marsh, D. An osteoconductive, osteoinductive, and osteogenic tissue-engineered product for trauma and orthopaedic surgery: How far are we?. Stem Cells Int. 2012, 236231. https://doi.org/10.1155/2012/236231 (2012).
Kruyt, M. C. et al. Bone tissue engineering and spinal fusion: the potential of hybrid constructs by combining osteoprogenitor cells and scaffolds. Biomaterials 25, 1463–1473. https://doi.org/10.1016/s0142-9612(03)00490-3 (2004).
Langer, R. & Vacanti, J. P. Tissue engineering. Science 260, 920–926. https://doi.org/10.1126/science.8493529 (1993).
Rezwan, K., Chen, Q. Z., Blaker, J. J. & Boccaccini, A. R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 27, 3413–3431. https://doi.org/10.1016/j.biomaterials.2006.01.039 (2006).
Pou, A. M. Update on new biomaterials and their use in reconstructive surgery. Curr. Opin. Otolaryngol. Head Neck Surg. 11, 240–244. https://doi.org/10.1097/00020840-200308000-00004 (2003).
Yang, S., Leong, K. F., Du, Z. & Chua, C. K. The design of scaffolds for use in tissue engineering. Part I. Traditional factors. Tissue Eng. 7, 679–689. https://doi.org/10.1089/107632701753337645 (2001).
Bianco, P., Riminucci, M., Gronthos, S. & Robey, P. G. Bone marrow stromal stem cells: Nature, biology, and potential applications. Stem Cells 19, 180–192. https://doi.org/10.1634/stemcells.19-3-180 (2001).
Acknowledgements
The author thank all participants in this study.
Funding
This work was supported by the National Natural Science Foundation of China (82202707); Natural Science Foundation of Chongqing, cstc2021jcyj-msxmX0541.
Author information
Authors and Affiliations
Contributions
X.W. and C.J. contributed equally to this work and should be considered as co-first authors, and they contributed to data collection, paper writing, data analysis, and performed the surgeries. H.W. and T.H. contributed to laboratory construction of activated allograft. F.L. and G.L. contributed to data analysis and article revision. S.L. contributed to overall planning and article revision. Z.X. contributed to overall planning, article revision and performed surgeries.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, X., Jia, C., Wu, H. et al. Activated allograft combined with induced membrane technique for the reconstruction of infected segmental bone defects. Sci Rep 14, 12587 (2024). https://doi.org/10.1038/s41598-024-63202-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-63202-9
- Springer Nature Limited