Abstract
Agricultural field experiments are costly and time-consuming, and often struggling to capture spatial and temporal variability. Mechanistic crop growth models offer a solution to understand intricate crop-soil-weather system, aiding farm-level management decisions throughout the growing season. The objective of this study was to calibrate and the Crop Environment Resource Synthesis CERES-Maize (DSSAT v 4.8) model to simulate crop growth, yield, and nitrogen dynamics in a long-term conservation agriculture (CA) based maize system. The model was also used to investigate the relationship between, temperature, nitrate and ammoniacal concentration in soil, and nitrogen uptake by the crop. Additionally, the study explored the impact of contrasting tillage practices and fertilizer nitrogen management options on maize yields. Using field data from 2019 and 2020, the DSSAT-CERES-Maize model was calibrated for plant growth stages, leaf area index-LAI, biomass, and yield. Data from 2021 were used to evaluate the model's performance. The treatments consisted of four nitrogen management options, viz., N0 (without nitrogen), N150 (150 kg N/ha through urea), GS (Green seeker-based urea application) and USG (urea super granules @150kg N/ha) in two contrasting tillage systems, i.e., CA-based zero tillage-ZT and conventional tillage-CT. The model accurately simulated maize cultivar’s anthesis and physiological maturity, with observed value falling within 5% of the model’s predictions range. LAI predictions by the model aligned well with measured values (RMSE 0.57 and nRMSE 10.33%), with a 14.6% prediction error at 60 days. The simulated grain yields generally matched with measured values (with prediction error ranging from 0 to 3%), except for plots without nitrogen application, where the model overestimated yields by 9–16%. The study also demonstrated the model's ability to accurately capture soil nitrate–N levels (RMSE 12.63 kg/ha and nRMSE 12.84%). The study concludes that the DSSAT-CERES-Maize model accurately assessed the impacts of tillage and nitrogen management practices on maize crop’s growth, yield, and soil nitrogen dynamics. By providing reliable simulations during the growing season, this modelling approach can facilitate better planning and more efficient resource management. Future research should focus on expanding the model's capabilities and improving its predictions further.
Similar content being viewed by others
Efficient use of agricultural inputs, particularly fertilizers, is crucial for modern agriculture. Among the agricultural inputs, nitrogen (N) fertilizers play a crucial role in increasing the soil supply of N to crops, particularly cereal crops, which are highly responsive to N1. The use of nitrogen fertilizers in the production of food and fibre crops has grown significantly in recent decades, but the nitrogen use efficiency (NUE) remains low in cereal-based agro-ecosystems, with cereals typically taking up only 40–60% of applied nitrogen2,3,4. To maintain or increase grain yield, farmers often apply more N, leading to its excessive losses and resulting in negative environmental and socio-economic impacts such as groundwater contamination and greenhouse gas emissions5,6. Therefore, use of the slow-release N fertilizer source and urea deep placement (UDP) technology may be effective for reducing N losses and improving NUE in field crops. The UDP technology involves using large-sized fertilizer particles, known as urea supergranules or briquettes, which are placed at a depth of 7–10 cm near the root zone of the crop7. Previous studies have shown that these technologies can lead to higher crop yields and lower fertilizer use under conventional crop management conditions8 including the traditional intensive tillage etc., but for conservation agriculture (CA) based farming systems, the performance of these technologies need to be investigated.
In recent years the increasing demand for food and the need to reduce environmental impact has led to a growing interest in identifying economical and environmentally-friendly N management practices which increase the NUE while ensuring sustainable crop production1. Conservation agriculture, which includes three basic principle i.e., minimal soil disturbance, efficient residue cover/retention, and crop diversification9,10, has been shown to improve soil health and increased crop productivity. The past research evidences describe CA as a more sustainable form of agriculture in cereal-based cropping systems, in terms of resource use efficiency (water, fertilizers, and energy, etc.), profitability, soil quality, and agri-ecosystem resilience to climate change11,12. One potential alternative to diversify the traditional rice–wheat (RW) systems in North-West India is the integration of C4 crops like maize in this system9,12. The maize-wheat (MW) system is the third most important cropping system in the region and has the potential to expand in the face of emerging water crisis in the Indo-Gangetic Plains (IGP)9,12. Adopting CA-based practices in the MW system can provide dual benefits of superior food and fodder supply as well as improved soil health and crop productivity. The addition of crop residues has been shown to have improved soil quality by increasing soil organic carbon (SOC) content13, cation exchange capacity, aggregate stability14, and N mineralization15,16. However, the short-term immobilization of N by crop residues must be considered in the long-term strategy to enhance nutrient availability17. Therefore, determining the most appropriate way to manage N in these systems can be challenging due to the complexity of the soil N cycle and the influence of weather. One way to gain insight about these mechanisms is to use of simulation models, such as the Decision Support System for Agrotechnology Transfer (DSSAT). These models have the potential to provide valuable insights into N management in CA systems by simulating the effects of weather variables on soil N transformations, crop growth, and development18. Accurate simulation models are integral for resource management in agriculture, guiding judicious resource use by simulating growth and yield parameters. These models offer real-time decision-making insights, aiding in optimal choices for fertilizers, irrigation, and other inputs. Moreover, they facilitate comprehensive agricultural planning by considering soil conditions, weather variations, and input factors. Through this, simulation models contribute to a more efficient and sustainable farming approach, ensuring the judicious allocation of resources and enhancing productivity in harmony with environmental variables. Previous studies have evaluated the performance of the DSSAT-CSM and CENTURY-based soil module using data from cropping system experiments. For example, researchers have used these models to predict long-term trends in rice and wheat yields19, the effects of different N application rates on corn production20 and crop yields and N dynamics in a 50-year continuous maize production experiment21. These studies have shown that the DSSAT models can provide accurate predictions under a variety of conditions. In addition to their use in predicting crop yields, DSSAT models have also been used for yield forecasting22,23,24. These forecasts can be conducted prior to planting or during the growing season and can be used by farmers to manage expected crop production or by governments for agricultural planning25. However, it is important to note that the CERES-Maize model has not yet been well studied under contrasting tillage (CA vs CT) and N management options. Against this background, the present study was carried out with the following objectives: (1) To evaluate the CERES-Maize model for simulating the growth behavior of maize and soil nitrogen dynamics under emerging CA practices and application of novel fertilizer products such as urea supergranules (USG); (2) To assess the capabilities of DSSAT in evaluating the N application options and doses under different soil conditions (CA vs CT), and (3) To understand the effects of N management on crop growth and soil N dynamics under long-term CA-based maize-wheat system. This research will contribute to the ongoing effort to identify economical and environment-friendly nitrogen management practices that increases nutrient use efficiency (NUE) while ensuring sustainable crop production under CA-based systems.
Results
Temporal distribution of weather variables
The amount of rainfall received during kharif 2019–2021 were 512.5, 587.5 and 1288.7 mm, respectively. The graphical representation of daily rainfall distribution, minimum and maximum temperature and solar radiation during simulation period have been presented in Fig. 1. The rainfall distribution during the experiment seasons i.e., in 2019 (512.3 mm) and 2020 (587.5 mm) were almost comparable with distribution pattern almost similar, whereas 2021 (1288.7 mm) had higher rainfall, with higher amount of rainfall occurred during active growth stage of the cropping season.
Model calibration
The six critical genetic coefficients for the model have been finally calibrated as presented in Table 1.
The performance statistics and goodness of fit are illustrated in Figs. 2a and 3a, c, and e. In the calibration years (2019 and 2020), the model showed strong performance with lower values of RMSE (293 and 1439 kg per hectare for grain and biomass, respectively) and nRMSE (5.83 and 9.21% for grain and biomass yield, respectively), indicating accurate simulation of crop yield. Similarly, for other parameters, RMSE and nRMSE values during the calibration years were approximately 0.56 and 10.76% for LAI, 13.85 and 8.77% for nitrogen uptake, and 16.03 and 16.51% for soil nitrate, respectively (Fig. 3a,c,e).
Model evaluation
The genetic coefficients calibrated were used to simulate the yield and biomass of year 2021. The DSSAT-CERES-Maize model simulated maize cultivar anthesis, and physiological maturity at 54–57 and 90–92 DAP, respectively in all 3-years. (2019, 2020, 2021). The observed days to maturity and anthesis are given in Fig. 4 in which simulated days of the phenological stages lie within the 5% of those observed. Measured leaf area, nitrogen fluxes such as observations on nitrogen uptake and residual soil nitrate were also compared with those from simulation using DSSAT for all three years and statistical indicators suggest a good fit with acceptable RMSE and nRMSE (Fig.3b, d and f).
Crop phenology
The results indicate that the model accurately predicted the time to anthesis of the crop, with an error of only 1–2 days (Fig. 4a), demonstrating good agreement between observed and simulated values (Fig. 4b). It's evident from the model that the timing of anthesis and physiological maturity is mostly determined by the variety of the crop, as treatments had minimal effect on these growth stages. However, under field conditions, slight variations were observed, particularly between treatments without nitrogen and those with nitrogen application. Treatments without nitrogen tended to delay anthesis but accelerated physiological maturity, supporting the idea that stress before flowering prolongs the period before anthesis but shortens it after. The simulated values for anthesis and physiological maturity closely matched the observed values, indicating the model's effectiveness. Treatments with nitrogen application required a longer time to reach physiological maturity, primarily due to the extended availability of nitrogen, which prolonged the post-anthesis duration of the crop.
Leaf area index
The plant leaf area index (LAI) at any growth stage indicates the extent of the assimilatory system, which contributes to dry matter accumulation and partitioning. To validate the model using the LAI, we used data from the year 2021 tillage and nitrogen treatments in maize crop. Simulated LAI ranged from 3.0 to 6.0 across different treatments. During the first few days of the growing season, the simulation of LAI was nearly identical to the observed LAI for all the treatments except for the CT-N0 where model over-predicted the leaf area. At 60 days, the stage which corresponds to the crop’s maximum leaf area index, the model slightly under-predicted the LAI. Except for CT-N0, there were no significant differences in observed leaf area among treatments. The model accurately simulated the trend of LAI for all treatments, showing a rise in leaf area until grain formation, followed by a decrease as the plant transferred photosynthates to sink. The model closely simulated this trend for all treatments in 2021, with an RMSE of 0.57 and nRMSE of 10.3% (4b).
Grain and biomass yield
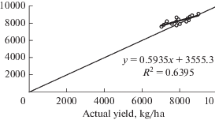
In the evaluation year (2021), the root mean square error (RMSE) for grain and biomass yield was 233 and 1755 kg/ha, respectively (Fig. 2b). The nRMSE for measured and simulated grain yield and biomass yield were 4.69 and 10.60%, respectively. Maize yields varied significantly depending on tillage practices and fertilizer N levels (see Figs. 2b and 5). Generally, the simulated grain yields of maize closely matched the measured values across most treatments, with prediction errors ranging from 0 to 3 percent (Table 2). However, for N0 treatments in both conventional tillage (CT) and zero tillage (ZT), the model tended to over-predict grain yield, with prediction errors ranging from 9 to 16 percent (Table 2). Despite this discrepancy, the model performed well in predicting grain and biomass yields for the majority of treatments, demonstrating its effectiveness in simulating maize crop productivity.
Nitrogen dynamics and model’s sensitivity analysis
Both ammonium N and nitrate N are major forms of mineral N in soil. However, compared to ammonium N, nitrate N is generally available in principal form. In both the CT and ZT conditions, simulations showed that NH4+-N concentrations were similar and increased after adding fertilizer. However, it converted quickly to nitrate, and after a while, its concentration was very low (1–4 kg/ha). Model suggested that ammonia volatilization increased just after the fertilizer N application.
Soil nitrate
Unlike NH4+-N, not only nitrate–N rapidly diffused in soil but also steadily transported toward roots through mass flow via plant transpiration. The model accurately predicted soil nitrate levels, with RMSE and nRMSE values of 12.63 and 12.84%, respectively (Fig. 3f). The soil NO3−-N concentration varied significantly with fertilizer application and crop growth stages and constantly rose with fertilizer application. Consistently rising with nitrogen fertilization. Overall, nitrate levels increased notably with fertilizer use, especially in warmer conditions where nitrifying bacteria activity and nitrification rates are higher (Fig. 6). Warmer temperatures increased nitrate levels as they stimulate nitrifying bacteria and nitrification rates (Fig. 6). Generally, zero tillage (ZT) plots had more nitrate than conventional tillage (CT) plots. Among nitrogen management options, soil nitrate was highest in USG plots, followed by GS and 150N, while plots without nitrogen (N0) had the lowest levels. In 2021, all fertilizer application scenarios showed an extended period of nitrate stress.
Nitrogen uptake
The observed and simulated nitrogen uptake showed a good match for evaluation year (2021), with an RMSE = 16.21 kg/ha and nRMSE = 10.82% (Fig. 3d). Nitrate–N concentration during active plant growth was low and highly dynamic, indicating rapid consumption matching supply rates. At 37 DAP and 65 DAP, despite fertilizer application, soil nitrate levels didn't accumulate much, as nitrate availability synchronized with peak crop growth and nitrogen demand (Fig. 6). The N uptake sharply increased after fertilizer application across all treatments. In this regard model’s prediction was in harmony with the findings of Jackson et al.26,27. It's also notable that higher nitrogen accumulation after basal nitrogen application was due to initial crop uptake absence. Nitrogen uptake patterns remained similar across three years, with maximum uptake in ZT-USG and lowest in CT-N0, despite higher rainfall in 2021 compared to 2019 and 2020 (Fig. 7). But clustered behavior was observed among nitrogen management options; for instance, CT-USG/ ZT-USG behaved similarly, as did CT-GS, CT-150N, ZT-GS, ZT-150N showed similar characteristics in the N uptake pattern. CT-N0 and ZT-N0 showed the similar N uptake capacity. However, on average ZT consistently outperformed CT across all N application options.
Ammonia volatilization
In this and following section, we only looked at simulated data and the discussion is based on comparing it with previous findings. The model showed that ammonia volatilization increased when NH4+-N levels rose after applying fertilizer, as NH4+-N acts as substrate for ammonia volatilization (Fig. 8). Ammonia volatilization depends on temperature and precipitation, increasing with higher temperatures. Heavy rainfall right after the basal nitrogen application in 2021 led to a big increase in volatilization across all N application options. However, the USG and zero tillage (ZT) reduced volatilization compared to conventional tillage (CT) and other N management options. USG fertilizer helped offset volatilization better in 2019 and 2020 compared to other nitrogen management options. The high temperatures in 2020 worsened ammonia loss through volatilization, especially with late heavy rainfall.
Nitrate leaching
Leaching in three crop seasons was mainly influenced by rainfall amount and its distribution throughout the cropping season. The model showed that leaching was highest in 2021, followed by 2019, and lowest in 2020. In 2019, consistent but not very heavy rainfall led to steady leaching, causing more leaching compared to 2020 (Fig. 9). However, in 2021, frequent rainfall events continuously wetted the soil, resulting in higher leaching rates.
Zero tillage (ZT) systems across all nitrogen application categories showed high nitrogen loss due to the soil's high porosity. High rainfall at the late stage of the crop triggered faster leaching. The peak NO3−-N leaching coincided with precipitation and a fair conclusion can be drawn that rainfall triggers leaching and loss of nitrate into the deeper soil layers depended on water flux during each drainage event as stated by Godwin and Jones28. However, considering grain yield, nitrate leaching didn't seem to have much effect. The reason lies in the fact that leached nitrate can rise again with the capillary action to the root zone as it is water soluble, and model also takes into account this fact considering the water flux going upward with capillary action29. The information is important for improving our understanding of the crop–soil N dynamic processes.
Discussion
The distinct behavior of soil and crop residue decomposition in conventional and conservation agriculture (CA) significantly impact crop growth and nitrogen dynamics. The successful understanding of these complex dynamics and underlying processes has been achieved through the effective utilization of simulation models30,31,32.
Phenology
Understanding the different growth stages of maize, like when it flowers and when it's fully grown, is really important for how much corn we can harvest33. In this study, the DSSAT-CERES-Maize model successfully simulated the phenological stages of maize cultivars, showing good agreement with observed values. The model also demonstrated the impact of nitrogen treatments on anthesis and physiological maturity, with no nitrogen delaying anthesis but advancing physiological maturity (Fig. 4). Additionally, the model effectively simulated the leaf area index (LAI) and grain yield (Figs. 3a, b and 5). This effectiveness of the model was also observed by Chisanga et al.34, DeJonge et al.35 and Timsina and Humphreys19. In our study, the LAI simulation closely matched the observed values, indicating the model's ability to capture the assimilatory system's contribution to dry matter accumulation and partitioning18.
Grain yield
Although the model slightly over-predicted grain yield in treatments without nitrogen (N0), it generally matched the measured values well, particularly in treatments with fertilizer nitrogen application. Similar DSSAT simulation findings were reported by Timsina and Humphreys18, Liu et al.21 and Yang et al.36 which showed a poor response of DSSAT for N0 treatment than the treatments with fertilizer N and they attributed this to the higher sensitivity of model to the fertilizer N application. Jones and Kiniry37 also demonstrated the sensitivity of the DSSAT model to the accurate specification of initial mineral N to the simulation response to yield and found to have reported a similar outcome. If initial mineral N is not measured accurately, then the reliability of the simulated response to N must be questioned. Thus, the DSSAT model demonstrated “good to excellent” agreements in simulating the maize yield for the treatments with fertilizer N application. In all the ZT treatments, increase in the grain yield was fairly captured by the model and it was primarily due to higher nitrogen availability and uptake due to more favourable soil conditions in terms of higher organic carbon content, lower bulk density, high water retention and moderation of the soil temperature38.
Nitrogen dynamics
Ammonium and nitrate are the two major forms of mineral nitrogen in soil, with nitrate being the more readily available form. Ammonium is quickly nitrified to nitrate upon addition to the soil. The diffusion rate of nitrate and ammonium in the rhizosphere differs significantly, with ammonium being adsorbed to the soil solid phase and having a much lower effective diffusion coefficient than nitrate39. Our model simulations showed that NH4+-N concentrations increased following fertilizer application but quickly nitrified into nitrate, becoming negligible. Previous studies have shown that soil ammonium N has no correlation with wheat yield40,41. Consistent with experimental results of Li et al.42 and Zhao et al.43, our simulations revealed higher nitrate content in zero tillage plots compared to conventional tillage plots, attributed largely to initial nitrate content and favorable nitrification conditions. Nitrate content was highest in plots with USG nitrogen management and lowest in N0 plots. Despite excessive leaching and volatilization in 2021 causing extended period of nitrate stress, the USG nitrogen application helped alleviate this stress. Nitrate accumulation in maize had multiple benefits, including ensuring smooth & consistent nitrogen nutrition, promoting vigorous growth, and increasing yield. Our DSSAT model provided dynamic soil nitrogen data and indicated a minor influence of nitrogen dose on native nitrogen release and leaching loss of nitrate. The model's ability to simulate NO3−-N dynamics and estimate leaching loss and soil N balance under various management practices and weather conditions improves our understanding of crop-soil nitrogen dynamics. Further, our simulations showed that the ammonia volatilization and aboveground biomass nitrogen uptake were 21 and 67 percent of the total nitrogen available to the crops (applied fertilizer nitrogen and mineralized nitrogen). Assimilation of nitrate in plant tissues, especially when soil nitrate levels are high, is common across all plants and doesn’t show usually any detrimental effect44,45. In fact, nitrate serves as a safe nitrogen source, unlike ammonium, which showed inhibited/poor vegetable growth44,45. Such high accumulation of nitrate–N in maize can offer multiple beneficial effects. Firstly, it ensures a smooth N nutrition and when the external N falls short of supply, the nitrate stored in vacuoles can be released to cytoplasm for plant, contributing to prolonged growth periods and increased photosynthesis 46,47. Secondly, a high accumulation of nitrate–N in plants promotes vigorous growth. With continuous growth, the stored nitrate–N decreases significantly, and at flowering stage, plants with high nitrate–N accumulation still contain nitrate, while those with low nitrate accumulation have nearly all nitrate utilized. Consequently, plants with higher nitrate–N accumulation have a longer growth period and can produce more photosynthetic products. This likely explains why maize plants with higher soil nitrate and fertilizer application took longer to reach physiological maturity compared to those with lower soil nitrate and less/no fertilizer application (Fig. 3). Thus, nitrate accumulation in plants ensures abundant growth and higher yields46,47.
Conclusions
Financial and time constraints coupled with drudgery in field experiments can be alleviated with use of crop models such as DSSAT in field crop investigations. The goal of using DSSAT is simulation of crop growth and nutrient dynamics in a wide range of environmental conditions. In our study, we calibrated the genetic coefficients of a maize cultivar (PMH1) using CSM-CERES-Maize model and demonstrated how its calibrations brought about a close simulation of crop growth and dynamic processes involving the fate of nitrogen. The Model satisfactorily simulated grain yield (RMSE = 233 kg/ha) and biomass yield (RMSE = 1755 kg/ha). The simulated LAI at 60 DAP was in accordance with the measured leaf area index, with an nRMSE of 10.3%. Simulated maximum LAI ranged from 3.0 to 6.0 across different treatments. All nitrogen levels in ZT treatments recorded higher LAI and grain yield than CT treatments. The model also successfully simulated different nitrogen levels in zero tillage (ZT) and conventional tillage (CT) plots, demonstrating higher LAI and grain yield in ZT treatments. It effectively simulated nitrogen dynamics, including inorganic nitrogen application, native nitrogen mineralization, crop nitrogen uptake, nitrate leaching, and ammonia volatilization losses under varying nitrogen levels. Given the importance of maize and the cultivar used in our study, generated genetic coefficients of cultivar in combination with the validated model will help growers/producers and researchers in making site specific and real time decisions. Additionally, it can also help understand complex interactions between genotype, environment, and management (G × E × M) and predict optimal tillage and nitrogen strategies for maximizing maize yield under different climatic conditions, facilitating the timely transfer of agricultural technology.
Methods
Description of experiment site
The experimental data was collected from maize in a long-term ongoing conservation agriculture-based maize-wheat system experiment (started since kharif 2012) for a period from 2019 to 2021. The field site (block ‘9B’) is located in the research farm premises of ICAR-Indian Agricultural Research Institute (IARI), New Delhi (28° 40′N latitude, 77° 11′E longitude and an altitude of 228 m above sea level), representing Indian Trans-Gangetic Plains Zone (Agro Climatic Zone-VI) with a sub-tropical and semi-arid climate. The average annual rainfall of the site is 735 mm with hot dry summer, wet monsoon and cold winters. The site receives about 80% of its total rainfall during the monsoon season (July–September). At the start of the experiment, physico-chemical soil properties were determined using standard procedure as outlined by Parihar et al.11. The detailed initial soil properties are as presented in Supplementary Table 1.
Description of imposed treatments and adopted agrotechniques
In this study, we investigated the growth behavior of maize crops and soil nitrogen dynamics under different tillage and nitrogen management practices. The experiment involved a split plot design with three replications. We compared four different nitrogen sources/method and nitrogen rates in sub plots viz., N0 (no nitrogen), N150 (150 kg N/ha through urea), GS (Green seeker-based urea application) and USG (urea super granules @150kg N/ha). These were tested in two different tillage methods in main plots: CA based zero tillage (ZT) and conventional tillage (CT). The resulting eight treatments are described in detail in Table 3.
In all CT plots, 2.91–2.94 ton/ha crop residue of preceding crop was incorporated into the soil during the study years (2019–2021). Conversely, in the ZT plots, the preceding crop residue was retained on the soil surface. ZT plots were prepared in June 2012 and remained undisturbed throughout the years. During the experimentation period (2019–2021), maize variety PMH1 was manually sown in the first week of July during the rainy season and was harvested in the second week of October. A recommended dose of 60 kg P2O5, 40 kg K2O/ha was applied as basal directly at the time of seeding. The control plot (without N applied plot) received only P2O5 and K2O, while the recommended dose of N (RDN) plots received 150 kg N/ha with 50 kg/ha as a basal dose and the remaining N was split into two equal portions and point-placed at 7–10 cm depth near the root zone at 36 days after planting (DAP) and 61 DAP. Irrigation was applied at critical stages according to the crop water requirement. The water application and measurement procedures followed those outlined by Parihar et al.48.
Measurement of leaf area index, crop yields and plant N uptake
In the present study, crop yields and nitrogen dynamics were measured to evaluate the impact of different tillage and nitrogen management practices. Leaf area was measured periodically at 25 days interval using a leaf area meter (Model LI-COR-3100). In this paper, LAI at 60 DAP is discussed which corresponds to the stage at which the crop attains its maximum LAI values. The leaf area index (LAI) was calculated as follows:
Biomass and yields of the maize crop were recorded at harvest as per the standard protocol11. Crop harvesting was done manually, and maize grain samples were collected at harvest, oven-dried at 65–70 °C for 48 h and weighed. The nitrogen (N) content in both grain and stover samples was determined using a CHNS analyzer, which employs the “Dumas method” involving dry, rapid and complete sample combustion. N uptake was then calculated using the formula:
The total N uptake (kg/ha) was calculated as the sum of N uptake in grain and N uptake in stover using equation as follows:
Determination of mineral-N (NH4 +-N and NO3 −-N)
Soil samples were collected randomly at 25 days interval from three places in each plot at six different depths (0–15, 15–30, 30–45, 45–60, 60–75, and 75–100 cm) using a soil sampler with a 5 cm internal diameter. The samples were then composited for each plot and mineral-N fractions were determined using standard methods49,50,51. To determine the mineral-N (NH4+ and NO3–-N) content, moist soil samples were extracted with a 2M KCl solution (soil:solution ratio of 1:5) by shaking for 1 h using a mechanical shaker50,51. Soil mineral-N content was estimated using steam distillation methods50, first with MgO for NH4+-N and then with MgO and Devarda’s alloy separately for NO3–-N reduction. Ammonium in the extract was estimated from the NH3 liberated with distillation of the extract with MgO, creating the necessary alkaline condition for ammonia formation. In the same extract, a reducing agent (Devarda’s alloy) was added to reduce the NO3−-N to ammonia. The mineral-N content of soil samples was expressed on a dry weight basis. While the soil nitrate was assessed periodically, this paper’s primary emphasis lies in the examination of both observed and simulated soil nitrate levels at the 25 DAP. This stage is used for simulation because at this stage nitrate uptake by the crop is rapid, which presents a challenging scenario for the model to accurately replicate the soil nitrate dynamics.
Crop growth simulation using CERES-Maize
Model description
In this study, simulations were conducted using the CERES-Maize model, which is incorporated within the input modules of the Decision Support System for Agro-technology Transfer (DSSAT) model version 4.8, with a CENTURY soil organic matter module for modeling complex decomposition dynamics of soil organic matter and surface residue layer. A detailed description of the CSM-CERES-Maize model can be found in Ritchie et al.52. The CERES-Maize model is a dynamic mechanistic model that simulates the phenological development and growth of maize on a daily time step in response to environmental factors, such as soil and climate, and management factors, such as crop variety, fertilization, planting conditions, and irrigation (Fig. 10). The model has been extensively tested for different soil types, climatic conditions, and maize hybrids53,54. Crop growth in the CSM-CERES-Maize model is controlled by phenologically defined growth stages driven by energy input in the form of growing degree-days (GDD)55. The CERES-Maize model uses growing degree days (GDD) to calculate the developmental stage of the crop, with GDD being a measure of heat accumulation during the growing season. The model also simulates the uptake of nitrogen by the crop, taking into account factors such as soil properties, weather conditions, and management practices55. The DSSAT model is a collection of independent programs, including crop, weather, soil, and water modules, which are closely coupled with the cropping system model as the core, simplifying the simulation of crop rotations. The model simulates crop growth and development from planting to maturity on a time step based on physiological processes describing the response of a crop to soil and aerial environmental conditions. Potential growth is dependent on photosynthetically active radiation and its interception, whereas actual biomass production on any day is constrained by suboptimal temperatures, soil water deficits, and nitrogen deficiencies. The input data required for the DSSAT models include daily weather data, soil characterization data, cultivar coefficients, and crop management information (Fig. 10). The soil water balance is determined on a daily basis as a function of precipitation, irrigation, transpiration, soil evaporation, runoff, and drainage from the bottom of the profile.
Model calibration and evaluation
To ensure accurate simulations in DSSAT, cultivar coefficients tailored to local conditions and crop varieties must be calibrated under optimum conditions21,59. Six cultivar coefficients (P1, P2, P5, G2, G3, and PHINT)55, three representing crop duration (P1, P2 and P5), two representing grain filling (i.e., G2 and G3), and one representing leaf area and leaf area duration by phylochron interval between successive leaf tip appearances (PHINT), were used in the CSM-CERES-Maize model, and these were calibrated for the PMH1 cultivar using the DSSAT-GLUE tool to match simulated and measured biometric variables and phenological stages. When the day length is less than 14 h, the silking rate is governed by P1 as the second variable P2 has no role in governing the phenology of the maize. P2 is used when the day length is more than 14 h. The P1 was adjusted till the simulated value matched with observed value. Similarly, P5 was adjusted to calibrate the duration between silking to physiological maturity. In a nut shell, first three variables (P1, P2 and P5) govern the crop duration; the other two (G2 and G3) control the grain yield and the last one (PHINT) controls the leaf area and leaf area duration.
For calibration, average values of key crop parameter such as days to 50% anthesis, and physiological maturity, maximum LAI, soil nitrate content, nitrogen uptake and, grain and biomass yield was obtained from nitrogen and water stress free plots in the 2019 and 2020 growing seasons. Subsequently, the calibrated coefficients' performance was validated against an independent dataset from a 2021 field experiment exploring tillage and nitrogen interactions, utilizing the same parameters as in calibration.
Model performance evaluation
Comprehensive evaluation of the DSSAT model performance was carried out by comparing the observed and model-simulated data using deviation statistics, such as prediction error (PE), root mean square error (RMSE), normalized RMSE (nRMSE), and coefficient of determination (R2). Additionally, regression analysis was performed to compare the simulated and measured grain yields. The prediction error, PE is defined as follows:
where Pi is predicted/simulated value, Oi is observed value.
Prediction is considered to be excellent if PE value is close to zero.
The root mean square error (RMSE)60 was used to calculate the fitness between the simulated and measured results. The RMSE summarizes the average difference between observed and predicted values and is expressed as follows:
The normalized RMSE is expressed as RMSE as percent over the mean observed value.
where Pi is predicted/simulated value, Oi is observed value, Ō is observed mean and n is the number of samples. The nRMSE (%) shows the relative difference between the predicted/simulated and observed data. The prediction is considered excellent, good, fair and poor for the nRMSE < 10%, 10–20%, 20–30% and > 30%, respectively60,61.
Permit statement
This study was conducted at IARI, and the authorship and acknowledgements have been appropriately attributed to each contributor based on their respective contributions.
The Experimental research and field studies on maize plants (cultivated), including the collection of plant material, were complied with relevant institutional, national, and international guidelines and legislation.
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- ANOVA:
-
Analysis of variance
- CA:
-
Conservation agriculture
- CERES:
-
Crop Environment Resource Synthesis
- CSM:
-
Cropping system model
- CT:
-
Conventional tillage
- DAP:
-
Days after planting
- DAS:
-
Days after sowing
- Fig.:
-
Figure
- GDD:
-
Growing degree days
- GS:
-
Green seeker-based urea application
- /ha:
-
Per hectare
- IGP:
-
Indo-Gangetic Plains
- LAI:
-
Leaf area index
- nRMSE:
-
Normalized root mean square error
- PE:
-
Prediction error
- PHINT:
-
Phyllochron interval
- R2 :
-
Coefficient of determination
- RMSE:
-
Root mean square error
- SOM:
-
Soil organic carbon
- UDP:
-
Urea deep placement
- USG:
-
Urea super granules
- ZT:
-
Zero tillage
References
Nayak, H. S. et al. Point placement of late vegetative stage nitrogen splits increase the productivity, N-use efficiency, and profitability of tropical maize under decade long conservation agriculture. Eur. J. Agron. 133, 126417 (2022).
Herrera, J. M., Noulas, C., Stamp, P. & Pellet, D. Little potential of spring wheat genotypes as a strategy to reduce nitrogen leaching in Central Europe. Agronomy 6, 29–3847 (2016).
Norton, R., Davidson, E., Roberts, T., Crosby, G. & Erisman, J. W. Position Paper—Nitrogen Use Efficiency and Nutrient Performance Indicators. A Publication of the Global Partnership on Nutrient Management Contributors About the Global Partnership on Nutrient Management [online]. Available: https://wedocs.unep.org/20.500.11822/10750. Accessed 7 Jan 2023 (2015).
Taulemesse, F., Le Gouis, J., Gouache, D., Gibon, Y. & Allard, V. Post-flowering nitrate uptake in wheat is controlled by N status at flowering, with a putative major role of root nitrate transporter NRT2.1. PLoS One 10, e0120291 (2015).
Hawkesford, M. J. Genetic variation in traits for nitrogen use efficiency in wheat. J. Exp. Bot. 68(10), 2627–2632 (2017).
Huang, T., Ju, X. & Yang, H. Nitrate leaching in a winter wheat-summer maize rotation on a calcareous soil as affected by nitrogen and straw management. Sci. Rep. 7, 42247 (2017).
IFDC (International Fertilizer Development Center). Benefits of the fertilizer deep placement technology for smallholder rice farmers. IFDC Q. Mag. 40(4) (2015).
Dhakal, K. et al. Deep placement of briquette urea increases agronomic and economic efficiency of maize in sandy loam soil. AGRIVITA J. Agric. Sci. 42(3), 499–508 (2020).
Jat, S. L. et al. Differential response from nitrogen sources with and without residue management under conservation agriculture on crop yields, water-use and economics in maize-based rotations. Field Crop Res. 236, 96–110 (2019).
Sidhu, H. S. et al. Sub-surface drip fertigation with conservation agriculture in a rice-wheat system: A breakthrough for addressing water and nitrogen use efficiency. Agric. Water Manag. 216, 273–283. https://doi.org/10.1016/j.agwat.2019.02.019 (2019).
Parihar, C. M. et al. Soil quality and carbon sequestration under conservation agriculture with balanced nutrition in intensive cereal-based system. Soil Tillage Res. 202, 104653 (2020).
Gathala, M. K. et al. Optimizing intensive cereal-based cropping systems addressing current and future drivers of agricultural change in the northwestern Indo-Gangetic Plains of India. Agric. Ecosyst. Environ. 177, 85–97 (2013).
Yadav, R. K., Purakayastha, T. J., Khan, M. A. & Kaushik, S. C. Long-term impact of manuring and fertilization on enrichment, stability and quality of organic carbon in Inceptisol under two potato-based cropping systems. Sci. Total Environ. 609, 1535–1543 (2017).
Zuber, S. M., Behnke, G. D., Nafziger, E. D. & Villamil, M. B. Crop rotation and tillage effects on soil physical and chemical properties in Illinois. Agron. J. 107(3), 971–978 (2015).
Sanchez, J. E., Willson, T. C., Kizilkaya, K., Parker, E. & Harwood, R. R. Enhancing the mineralizable nitrogen pool through substrate diversity in long term cropping systems. Soil Sci. Soc. Am. J. 65(5), 1442–1447 (2001).
Tonitto, C., David, M. B. & Drinkwater, L. E. Replacing bare fallows with cover crops in fertilizer-intensive cropping systems: A meta-analysis of crop yield and N dynamics. Agric. Ecosyst. Environ. 112(1), 58–72 (2006).
Singh, Y., Ladha, J. K., Khind, C. S., Khera, T. S. & Bueno, C. S. Effects of residue decomposition on productivity and soil fertility in rice–wheat rotation. Soil Sci. Soc. Am. J. 68, 854–864 (2004).
Ahmed, M. & Hassan, F.U. APSIM and DSSAT models as decision support tools. In MODSIM2011, 19th International Congress on Modelling and Simulation. Modelling and Simulation Society of Australia and New Zealand, December 2011 (eds Chan, F., Marinova, D. & Anderssen, R. S) 1652–1658. www.mssanz.org.au/modsim2011/D10/wongsosaputro.pdf (2011).
Timsina, J. & Humphreys, E. J. A. S. Performance of CERES-Rice and CERES-Wheat models in rice–wheat systems: A review. Agric. Syst. 90(1–3), 5–31 (2006).
Thorp, K. R., Malone, R. W. & Jaynes, D. B. Simulating long-term effects of nitrogen fertilizer application rates on corn yield and nitrogen dynamics. Trans. ASABE 50(4), 1287–1303 (2007).
Liu, H. L. et al. Using the DSSAT-CERES-Maize model to simulate crop yield and nitrogen cycling in fields under long-term continuous maize production. Nutr. Cycl. Agroecosyst. 89, 313–328 (2011).
Duchon, C. E. Corn yield prediction using climatology. J. Appl. Meteorol. Climatol. 25(5), 581–590 (1986).
Thornton, P. K. et al. Estimating millet production for famine early warning: an application of crop simulation modelling using satellite and ground-based data in Burkina Faso. Agric. For. Meteorol. 83, 95–112 (1997).
Bannayan, M., Crout, N. M. J. & Hoogenboom, G. Application of the CERES-wheat model for within-season prediction of winter wheat yield in the United Kingdom. Agron J. 95(1), 114–125 (2003).
Hoogenboom, G. Contribution of agrometeorology to the simulation of crop production and its applications. Agric. For. Meteorol. 103(1–2), 137–157 (2000).
Jackson, L. E., Schimel, J. P. & Firestone, M. K. Short-term partitioning of ammonium and nitrate between plants and microbes in an annual grassland. Soil Biol. Biochem. 21(3), 409–415 (1989).
Jackson, L. E., Burger, M. & Cavagnaro, T. R. Roots, nitrogen transformations, and ecosystem services. Annu. Rev. Plant Biol. 59, 341–363 (2008).
Godwin, D. C. Nitrogen dynamics in soil–plant systems. In Modeling Plant and Soil Systems. Agron. Monogr (eds Hanks, J. & Ritchie, J.) 287–321 (ASA, CSSA, SSSA, 1991).
Godwin, D. C. & Singh, U. Nitrogen balance and crop response to nitrogen in upland and lowland cropping systems. In Understanding Options for Agricultural Production. Systems Approaches for Sustainable Agricultural Development Vol. 7 (eds Tsuji, G. Y. et al.) (Springer, 1998).
Sanyal, S. K. & Majumdar, K. Nutrient dynamics in soils. J. Indian Soc. Soil Sci. 57(4), 477–493 (2009).
Kushwaha, C. P., Tripathi, S. K. & Singh, K. P. Variations in soil microbial biomass and N availability due to residue and tillage management in a dryland rice agroecosystem. Soil Tillage Res. 56, 153–166 (2000).
IFDC (International Fertilizer Development Center). Fertilizer deep placement. IFDC solutions (IFDC, 2013). https://issuu.com/ifdcinfo/docs/fdp_8pg_final_web?e=1773260/1756718.
Ahmad, M., Khaliq, A., Ahmad, R. & Ranjha, A. M. Allometery and productivity of autumn planted maize hybrids under narrow row spacing. Int. J. Agric. Biol. 12(5), 661–667 (2010).
Chisanga, C. B., Phiri, E., Chizumba, S. & Sichingabula, H. Evaluating CERES-maize model using planting dates and nitrogen fertilizer in Zambia. J. Agric. Sci. 7, 79–97 (2015).
DeJonge, K. C. et al. Improving evapotranspiration simulations in the CERES-Maize model under limited irrigation. Agric. Water Manag. 115, 92–103 (2012).
Yang, J. M., Yang, J. Y., Dou, S., Yang, X. M. & Hoogenboom, G. Simulating the effect of long-term fertilization on maize yield and soil C/N dynamics in northeastern China using DSSAT and CENTURY-based soil model. Nutr. Cycl. Agroecosyst. 95(3), 287–303 (2013).
Jones, C. A. & Kiniry, J. R. (eds) CERES-Maize: A Simulation Model of Maize Growth and Development (Texas A&M Univ. Press, 1986).
Parihar, C. M. et al. Soil water dynamics, water productivity and radiation use efficiency of maize under multi-year conservation agriculture during contrasting rainfall events. Field Crops Res. 241, 107570 (2019).
Barber, S. A. Soil Nutrient Bioavailability: A Mechanistic Approach 2nd edn, 180–181 (JohnWiley, 1995).
Hu, T. T. & Li, S. X. A reliable soil N availability index, Initial NO3−-N in soil profile. Agric. Res. Arid Areas 11, 74–82 (1993).
Miao, Y. F., Wang, Z. H. & Li, S. X. Relation of nitrate N accumulation in drylandsoil with wheat response to N fertilizer. Field Crops Res. 170, 119–130 (2015).
Li, S. X., Wang, Z. H., Hu, T. T., Gao, Y. J. & Stewart, B. A. Nitrogen in dryland soils of China and its management. Adv. Agron. 101, 123–181 (2009).
Zhao, R. F. et al. Fertilization and nitrogen balance in a wheat–maize rotation system in North China. Agron. J. 98(4), 938–945 (2006).
Wang, Z. H. & Li, S. X. Relationships between nitrate contents and water, total N as well as total P in different organs of vegetable plants. Plant Nutri. Fertil. Sci. 2(2), 144–152 (1996).
Gruda, N. Impact of environmental factors on product quality of greenhouse vegetables for fresh consumption. Crit. Rev. Plant Sci. 24(3), 227–247 (2005).
van der Leij, M., Smith, S. J. & Miller, A. J. Remobilisation of vacuolar stored nitrate in barley root cells. Planta 205, 64–72 (1998).
Li, S. & Wang, Z. H. Nitrate accumulation in plants and its relation to crop yields. In Third International Nitrogen Conference, Abstracts—Concurrent Oral Sessions Vol. 72 (2004).
Parihar, C. M. et al. Effects of precision conservation agriculture in a maize-wheat-mungbean rotation on crop yield, water-use and radiation conversion under a semiarid agro-ecosystem. Agric. Water Manag. 192, 306–319 (2017).
Kjeldahl, J. A new method for the determination of nitrogen in organic matter. Fresenius Z. Anal. Chem. 22, 366–382 (1983).
Bremner, J. M. Inorganic forms of nitrogen. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties. Agron. Monogr. 9 (ed. Norman, A. G.) 1179–1237 (ASA and SSSA, 1965).
Keeney, D. R. & Nelson, D. W. Nitrogen in organic forms. In Methods of Soil Analysis, Agronomy Monograph 9 Part 2 2nd edn (ed. Page, A. L.) 643–698 (ASA, SSSA, 1982).
Ritchie, J. T. Soil water balance and plant water stress. In Understanding Options for Agricultural Production. Systems Approaches for Sustainable Agricultural Development Vol. 7 (eds Tsuji, G. Y. et al.) 200–300 (Springer, 1998).
Hodges, T., Botner, D., Sakamoto, C. & Haug, J. H. Using the CERES-Maize model to estimate production for the US Cornbelt. Agric. For. Meteorol. 40(4), 293–303 (1987).
Carberry, P. S., Muchow, R. C. & McCown, R. L. Testing the CERES-Maize simulation model in a semi-arid tropical environment. Field Crops Res. 20(4), 297–315 (1989).
Jones, J. W. et al. The DSSAT cropping system model. Eur. J. Agron. 18(3–4), 235–265. https://doi.org/10.1016/S1161-0301(02)00107-7 (2003).
Hoogenboom, G. et al. Decision Support System for Agrotechnology Transfer (DSSAT) Version 4.8 (DSSAT.net). DSSAT Foundation, Gainesville. https://dssat.net/4800/ (2021).
Hunt, L. A., White, J. W. & Hoogenboom, G. Agronomic data: Advances in documentation and protocols for exchange and use. Agric. Syst. 70(2–3), 477–492 (2001).
Yang, H. S. et al. Hybrid-maize—a maize simulation model that combines two crop modeling approaches. Field Crops Res. 87, 131–154 (2004).
Boote, K. J. Concepts for calibrating crop growth models. In DSSAT v3 (eds Hoogenboom, G. et al.) 179–199 (University of Hawaii, 1999).
Willmott, C. J. et al. Statistics for the evaluation and comparison of models. J. Geophys. Res. 90, 8995–9005 (1985).
Jamieson, P. D., Porter, J. R. & Wilson, D. R. A test of the computer simulation model ARCWHEAT1 on wheat crops grown in New Zealand. Field Crops Res. 27(4), 337–350 (1991).
Acknowledgements
We sincerely acknowledge Indian Council of Agricultural Research (ICAR), ICAR-Indian Agricultural Research Institute (IARI), for providing the facilities. The first author also acknowledges ICAR-IIFSR for granting him study leave for Ph.D. The support received from Divisions of Agronomy, Plant Physiology, Agricultural Physics, and Soil Science & Agricultural Chemistry of ICAR-IARI, New Delhi and ICAR-Indian Institute of Maize Research (IIMR) is also acknowledged. The technical inputs of CGIAR Transforming Agrifood Systems in South Asia (TAFSSA) initiative and support received from CIMMYT is also thankfully acknowledged. Special thanks to Dr. Raj Singh, Former Head (Acting), Division of Agronomy, ICAR-IARI, Dr. Renu Pandey, Pr. Scientist, ICAR-IARI, Dr. V.K. Sharma, Pr. Scientist, ICAR-IARI, and Mr. Sanjeev Kumar of ICAR-IIMR for assistance in data management and analysis work.
Author information
Authors and Affiliations
Contributions
Kamlesh, C.M.P., H.S.N., Kiranmoy, K.S.R., and S.L.J. participated in conducting the research, data analysis and writing of the manuscript and editing; Y.S., U.S., M.L.J, D.R.S. & M.K.G. conceptualized the research and contributed to revision of the manuscript and other support; T.K.D., P.C.G., A.L.M., R.D., D.K.S., A.S., S.B. and S.G. participated research design.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kumar, K., Parihar, C.M., Nayak, H.S. et al. Modeling maize growth and nitrogen dynamics using CERES-Maize (DSSAT) under diverse nitrogen management options in a conservation agriculture-based maize-wheat system. Sci Rep 14, 11743 (2024). https://doi.org/10.1038/s41598-024-61976-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-61976-6
- Springer Nature Limited