Abstract
Subthreshold social anxiety (SSA) is a condition in which individuals experience social anxiety that does not reach the threshold required for a clinical diagnosis of a social anxiety disorder (SAD). Although SSA may not impair lives as severely as SAD, it can affect social functioning. However, only a few studies focused on structural neural correlates of SSA. We recruited 65 individuals with SSA and used the Leibowitz Social Anxiety Scale to assess their social and performance anxiety levels and other relevant measures of social anxiety. Voxel-wise whole-brain correlational analyses showed a positive association between the cortical thickness (CT) of the superior frontal gyrus (SFG) and social anxiety levels and a negative correlation between the CT of the fusiform gyrus (FG) and performance anxiety levels in individuals with SSA. Exploratory Pearson’s correlation analyses showed significant positive correlations between the CT of the SFG and Generalized Anxiety Disorder-7 total scores and negative associations between the CT of the FG and Beck Anxiety Inventory total scores. Our study provides insight into the neural basis of SSA, particularly performance anxiety, by highlighting the association between CT in specific brain regions and SSA characteristics.
Similar content being viewed by others
Introduction
Social anxiety, or intense fear and avoidance of social or performance situations, is related to distress, which ranges from low or moderate nervousness to severe and incapacitating dread1,2. People with extreme levels of social anxiety and persistent avoidant behaviors for at least six months may meet the diagnostic criteria for social anxiety disorder (SAD)1. Social anxiety symptoms typically lead to substantial distress or impairment in various areas of life, such as social activities, work, school, or relationships3,4,5.
Performance anxiety is the fear of speaking or performing in public among individuals who must speak or perform in front of an audience. It is also regarded as a subtype (performance-only subtype) of social anxiety in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5)4. It is prevalent in various fields, including the performing arts, sports, and public speaking6,7. People with high levels of performance anxiety tend to have high expectations of themselves and desire to perform flawlessly without any mistakes8. Fear rooted in concern about making mistakes and being negatively evaluated by others can be intense9,10. The fear of negative evaluation and pressure to perform perfectly can impact a person’s self-esteem and confidence considerably, which can have detrimental effects on their professional career10,11.
Subthreshold social anxiety (SSA) is a condition in which individuals experience social anxiety similar to that seen in SAD, but the symptoms do not reach the threshold required for a clinical diagnosis of SAD12. A previous study reported that the prevalence of SSA was higher than that of SAD13. The one-year prevalence of SSA was 3.0%, while SAD's was 2.0%13. SSA might impair both interpersonal relationships and social life, although to a somewhat lesser extent than SAD and could lead to the progression of SAD14,15. Despite the potential seriousness of its effects, the neurobiological mechanisms underlying SSA remain unclear. Furthermore, a few studies have examined the relationship between SSA-related brain regions and other psychological characteristics, including excessive worry or anxiety.
In recent years, the construct of social anxiety has existed on a continuum rather than as a discrete, dichotomous concept. This shift aligns with the Research Domain Criteria (RDoC) framework proposed by the National Institute of Mental Health16,17. Factor analysis findings suggest that the latent structure of social anxiety appears to be similar for people with and without SAD18. Neuropsychologically, attentional biases related to social anxiety at clinical levels of severity have also been found to play a role at non-clinical levels of severity19,20. This overlap may implicate the continuity in the underlying neural mechanisms related to social anxiety across the spectrum of severity20,21.
Meanwhile, functional magnetic resonance imaging (MRI) studies have revealed that the amygdala, insular cortex, and hippocampus play a substantial role in social anxiety22,23,24,25. A meta-analysis of structural MRI found larger gray matter volume (GMV) in the cortical regions, such as the superior frontal gyrus (SFG) and fusiform gyrus (FG), and smaller GMV in the subcortical regions, including the putamen and thalamus, of patients with SAD26. Furthermore, voxel-wise analyses revealed increased cortical thickness (CT) in the frontoparietal cortex of patients with SAD27. Patients with SAD show increased cortical surface area (SA) in the prefrontal cortex, lateral orbitofrontal cortex, and superior temporal gyrus28. Interestingly, a previous study demonstrated that dissociated morphological alterations were prominent in patients with SAD, as demonstrated by decreased CT and increased SA in the prefrontal cortex28.
However, in individuals with SSA, only a few studies have focused on structural brain alterations29,30. One study found significant positive correlations between social anxiety levels and the amygdala and striatal volume in a small sample of healthy female individuals29. Another study also showed that CT or SA in the FG was significant and negatively related to social anxiety levels measured via a self-report questionnaire, demonstrating a lower number of brain regions with decreased CT among individuals with SSA when compared to those in patients with SAD30. From a clinical perspective, as there are individuals with SSA, who do not reach the diagnosis of SAD, but suffer from social anxiety symptoms along a continuum of social anxiety dimensions, it may be crucial to alleviate their discomfort and to understand the pathophysiology of SSA.
Therefore, this study aimed to identify the structural differences in the brain based on the degree of social anxiety (or performance anxiety), particularly within individuals with SSA. Because the findings of previous studies on the association between social anxiety (or performance anxiety) and structural alterations are discrepant and inconsistent concerning individuals with SSA, we conducted voxel-wise whole-brain analyses instead of a regions of interest study. Furthermore, we investigated whether the dissociations between CT and SA in patients with SAD are also prominent in individuals with SSA. We hypothesized that (1) individuals with SSA (or performance anxiety) would show altered GMV, CT, and SA (i.e., a lower number of regions compared to SAD) in the above-mentioned regions, which are known to be related to social anxiety; (2) dissociated morphological alterations between CT and SA would be present in the prefrontal regions of individuals with SSA; and (3) there would be significant correlations between the brain regions implicated in SSA (or performance anxiety) and psychological measures of individuals with SSA.
Results
Sociodemographics and clinical characteristics
Table 1 summarizes the participants’ sociodemographic and clinical characteristics. Among the 65 participants (37 women and 28 men), most reported having a bachelor’s degree or higher. The social anxiety level (mean LSAS score) was 21.35, and the performance anxiety level (performance anxiety subscale mean scores) was 12.36 in individuals with SSA.
Pearson’s correlation analyses among clinical symptom measurements
Table 2 shows Pearson’s correlation analysis results among clinical symptom measurements, including social anxiety (and performance anxiety) levels, Generalized Anxiety Disorder-7 (GAD-7), Beck Anxiety Inventory (BAI), and Beck Depression Inventory (BDI-II) in individuals with SSA. There are significant positive correlations between social anxiety (and performance anxiety) levels and symptom severity levels in other clinical measurements (e.g., GAD-7, BAI, or BDI-II).
Voxel-wise correlational analyses between the social anxiety (or performance anxiety) levels and the brain structures in individuals with SSA
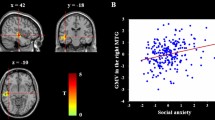
We performed voxel-wise correlation analyses between social anxiety scores and structural brain measures, including the brain regions’ GMV, CT, and SA. A significant positive correlation was observed between the Liebowitz Social Anxiety Scale (LSAS) total scores and the CT of the SFG in individuals with SSA [cluster-wise probability (CWP) = 0.0026]. This association remained significant after adjusting for age at the time of the MRI scan, gender, and intracranial volume (ICV) (CWP = 0.0056; Fig. 1). However, both SA and GMV were not significantly correlated.
Result of voxel-wise analysis for social anxiety levels. The CT of the SFG is significantly positively correlated with the LSAS total scores in individuals with SSA. Note MNI coordinate X = -23.8, Y = 16.5, Z = 39.1. Monte Carlo simulations correction (CWP < 0.05). The color bar shows a logarithmic scale of p values (− log10). CT, cortical thickness; SFG, superior frontal gyrus; LSAS, Leibowitz Social Anxiety Scale; SSA, subthreshold social anxiety; MNI, Montreal Neurological Institute, CWP: cluster-wise probability.
The LSAS performance anxiety scores showed significant correlations with the CT of several brain regions in individuals with SSA (Fig. 2). They were significantly and positively correlated with the CT of the SFG (CWP = 0.0006), even after controlling for age, gender, and ICV (CWP = 0.0024). Furthermore, the LSAS performance anxiety scores were significantly negatively correlated with the CT of the FG (CWP = 0.0006), even after controlling for age, gender, and ICV (CWP = 0.0046). No significant correlations were found with other structural brain measures, including GMV and SA.
Result of voxel-wise analysis for performance anxiety levels. (a) The CT of the SFG is significantly positively correlated with performance anxiety levels in individuals with SSA (MNI coordinate X =—23.4, Y = 17.8, Z = 38.3; Monte Carlo simulations correction, CWP < 0.05). (b) The CT of the FG is significantly negatively correlated with the LSAS performance anxiety scores in individuals with SSA (MNI coordinate X = 34.4, Y = -48.9, Z = -11.4; Monte Carlo simulations correction, CWP < 0.05). Note: The color bar shows a logarithmic scale of p values (− log10). CT, cortical thickness; SFG, superior frontal gyrus; SSA, subthreshold social anxiety; MNI, Montreal Neurological Institute; CWP, cluster-wise probability; FG, fusiform gyrus; LSAS, Leibowitz Social Anxiety Scale.
Exploratory Pearson’s correlation analyses between the cortical thickness of the significant brain regions and social anxiety-relevant psychological scores in individuals with SSA
We performed Pearson’s correlation analyses between the CT of significant brain regions related to levels of social anxiety (or performance anxiety) and the total scores regarding the GAD-7, BAI, and BDI-II for individuals with SSA (Supplementary Table 1). Figure 3 shows significant positive correlations between the CT of the SFG and the total scores of the GAD-7 (r = 0.317, p = 0.010). Additionally, the significance was maintained after Spearman’s correlation analyses between the CT of significant brain regions and the total GAD-7 scores in individuals with SSA.
Exploratory Pearson’s correlation analyses showed significant positive associations between the CT of the SFG related to social anxiety levels and the GAD-7 total scores in the individuals with SSA. CT, cortical thickness; SFG, superior frontal gyrus; GAD-7, Generalized Anxiety Disorder-7; SSA, subthreshold social anxiety.
The thinner the CT of the FG, the higher the total BAI score in individuals with SSA (r = -0.451, p < 0.001; Fig. 4). Additionally, the significance was maintained after Spearman’s correlation analyses between the CT of significant brain regions and the total BAI scores in individuals with SSA. The CT of the FG did not show significant correlations with other clinical variables.
Exploratory Pearson’s correlation analyses showed significant negative associations between the CT of the FG related to performance anxiety levels and the BAI total scores in individuals with SSA. CT, cortical thickness; FG, fusiform gyrus; BAI, Beck Anxiety Inventory; SSA, subthreshold social anxiety.
Discussion
Our study investigated structural brain alterations, such as the CT of the SFG and FG, and their relations with the clinical characteristics of individuals with SSA. We found significant positive correlations between the CT of the SFG and excessive worry levels. The thinner the CT of the FG, the higher the anxiety level in individuals with SSA. The CT of the SFG and FG may thus be associated with social anxiety and anxiety-related behaviors in individuals with SSA.
This study suggests that individuals with SSA exhibit positive associations between social anxiety and the CT of the SFG, which is located in the anterior portion of the dorsomedial prefrontal cortex. This brain region is involved in cognitive functions such as working memory and problem-solving31,32,33. Although little research has directly linked cognitive functions to social anxiety, structural MRI research has shown a negative correlation between social anxiety levels and CT in the SFG28. Interestingly, the increased CT in the dorsolateral prefrontal and parietal cortices of patients with SAD could be a compensatory response to coping efforts in emotion regulation27. Similarly, the increased CT observed in the SFG of individuals with SSA in the current study may be interpreted as a compensatory response to the challenges posed by social anxiety. However, we did not observe significant changes in the SA of the SFG among individuals with SSA compared to a previous SAD neuroimaging study28 showing increased CT and decreased SA in the SFG region. This suggests that the structural alterations of the SA in the SFG in SSA may differ from those in SAD.
Our study showed significant positive correlations between the CT of the SFG and excessive worry levels in individuals with SSA. Excessive worry is a psychological trait associated with dysfunction of the prefrontal cortex, including the SFG34. The positive correlations between the CT of the SFG and excessive worry scores suggest that alterations in SFG morphology may contribute to the cognitive and emotional processes underlying excessive worry.
Meanwhile, the negative correlation between the CT of the FG and performance anxiety levels in individuals with SSA suggests that those with elevated performance anxiety may exhibit thinner cortical tissue in this region. The FG is a key region involved in processing emotional faces and is considered a central node within the brain’s facial processing circuits35,36. Previous research has shown significant negative correlations between the CT or SA of the FG and social anxiety levels in patients with SAD30. This aligns with prior research suggesting a link between FG thinning and elevated levels of performance anxiety, potentially reflecting difficulties in processing social cues and emotional facial expressions37. This thinning in the FG may indicate aberrant social information processing, specifically related to facial expressions.
Our study revealed that a thinner CT in the FG correlated with higher anxiety levels in individuals with SSA. The FG is involved in processing emotional facial information and receiving direct input from the amygdala38. Recent research has demonstrated a strong relationship between the amygdala and visual cortical activation, such as facial expressions, during emotional stimulant processing39. Our findings confirm that thinning of the FG is associated with abnormal social information processing, particularly in individuals with SAD40. Therefore, cortical thinning of the FG influences anxiety regulation by affecting the amygdala in individuals with SSA. Structural differences in the FG may affect the processing and regulation of emotional stimuli, potentially leading to dysregulated emotional responses and heightened anxiety levels in social situations.
As mentioned previously, the brain regions associated with SAD are the amygdala, hippocampus, insular cortex, SFG, FG, putamen, thalamus, and frontoparietal cortex. However, our study found the lower numbers of the brain regions (e.g., two regions: SFG and FG) correlated with social anxiety in individuals with SSA when compared to those with SAD. The reason is unclear; however, we can assume that the severity of social anxiety may have played a role in the observed differences. This heightened severity could have led to more pronounced structural alterations in brain regions associated with social anxiety. Secondly, differences in the study design, such as analysis methods and sample sizes between our study and those focused on SAD, may have contributed to the differences in results.
Our findings showed the following clinical significance: even if individuals have SSA, they show morphological and structural alterations in the SFG or FG that are relevant to their psychological distress. These changes may lead to significant functional impairments in social relationships and related changes in cognitive and behavioral manifestations41. Therefore, consideration of individuals seeking SSA treatment is needed, even if they are not diagnosed with SAD.
This study has several limitations. First, the sample size was relatively small and limited to SSA without comparison groups—such as among patients with SAD and healthy controls without SSA. Therefore, further studies should investigate the association between social anxiety levels and structural brain alterations in larger and more diverse groups. Second, because a self-report questionnaire was used to assess psychological characteristics, a neuroimaging study using clinician-administered measures will be needed in the future. Nevertheless, all the assessments in our study were valid and objective indicators that had been repeatedly verified. Third, because we used a correlation analysis to determine the neural correlates of SSA, the neural correlates do not necessarily indicate the cause of SSA. In addition, as there are changes in the autonomic nervous, immune, and endocrine systems as well as in the brain, it was difficult to prove causality by controlling for these changes, which may be involved in SSA. Fourth, our study showed that the correlation between levels of social anxiety and performance anxiety is statistically significant. This may suggest that it is difficult to statistically separate the constructs of social anxiety severity and performance anxiety severity. Therefore, the study results need to be interpreted carefully, and well-designed integrative research is required in the future.
In conclusion, heightened levels of social anxiety, including performance anxiety, in individuals with SSA are associated with specific structural changes in the brain—specifically, cortical thickening in the SFG and thinning in the FG. Cortical thickening in the SFG may be a compensatory response to manage social anxiety, while the observed thinning of the FG may indicate difficulties with facial-emotional processing among individuals with SSA.
Materials and methods
Participants
We recruited 65 right-handed Korean individuals aged between 19 and 54 years from the Department of Psychiatry at the CHA Bundang Medical Center of CHA University (Gyenoggi-do, Republic of Korea) through advertisements between October 2014 and November 2021. Participants were excluded if they met any of the following criteria: (1) history of head trauma; (2) neurological disorders; (3) major medical illness; (4) current or past substance abuse and dependence; (5) current or past other major psychiatric disorders (e.g., schizophrenia spectrum disorders, major depressive disorders, or bipolar disorders); (6) current or past anxiety disorders, including SAD, panic disorder, agoraphobia, generalized anxiety disorder, and specific phobia; (7) ongoing psychological treatment or recently started pharmacological treatment; (8) MRI contraindications (e.g., metallic implants, claustrophobia); and (9) pregnancy. We used the Structured Clinical Interview for the DSM-5 to assess the presence of anxiety disorders and other psychiatric conditions, ensuring that only individuals without these disorders were included in the study42,43.
All study procedures were approved and reviewed by the Institutional Review Board of the CHA Bundang Medical Center (IRB no. 2019-05-030). Participants were provided with a detailed explanation of the study, and their written informed consent was obtained. The study was conducted in accordance with the Declaration of Helsinki. Furthermore, the principles of Good Clinical Practice were followed to ensure the study’s integrity, reliability, and ethical conduct.
Social and performance anxiety assessments
The LSAS is a well-validated assessment tool for measuring symptom severity of social and performance anxiety by assessing fear levels and avoidance of social situations44. The clinical utility of the LSAS has been supported for use with individuals with SSA41,45. One study demonstrated the effectiveness of the LSAS in detecting changes in SSA levels among a sample of college students45. In addition, the LSAS was useful in identifying alterations in functional connectivity in individuals with SSA in a neuroimaging study41. The questionnaire consists of 24 items assessing fear and avoidance in different social situations on a 4-point scale (ranging from 0 = none or never to 4 = severe fear or usually, 68–100%)44. This scale can be divided into two subscales: 11 items for social interaction anxiety and 13 items for public performance anxiety. Our study included only clinical participants who complained of SSA levels but who were not diagnosed with SAD through experienced psychiatrist interviews. Their total LSAS score was below 68, which was consistent with previous research findings. To assess the levels of social and performance anxiety, all participants completed the LSAS. The Cronbach’s α coefficients for the LSAS total score were high (0.94), and those for the subscales ranged from 0.71 to 0.9146.
Other clinical characteristics assessments
To investigate other clinical characteristics, we used the GAD-747, BAI48, and BDI-II49. The GAD-7 is a self-report questionnaire designed to assess an individual’s level of excessive worry in the previous two weeks50, consisting of seven questions. The Korean versions of the BAI and BDI-II were used to evaluate psychological distress, including depression and anxiety. The BAI has 21 self-reported inventories for assessing the severity of clinical anxiety on a scale of 0–348. Similar to the BAI, the BDI-II consists of 21 self-administered items ranging from 0 to 63,49. The total BAI and BDI-II scores reflect the severity of anxiety and depression.
Neuroimaging data acquisition and analyses
The GMV, CT, and SA of all participants’ whole brains were analyzed by T1-weighted MRI using FreeSurfer’s (v7.4.1; https://surfer.nmr.mgh.harvard.edu/) surface-based pipeline. All study participants underwent brain MRI through a 3.0 Tesla GE Signa HDxt scanner (GE Healthcare, Milwaukee, WI, USA). High-resolution structural images were acquired using a three-dimensional, T1-weighted, fast-spoiled, gradient-recalled echo sequence (repetition time = 6.3 ms; echo time = 2.1 ms; flip angle, 12°; field of view, 256 mm; matrix, 256 × 256; and voxel size, 1 × 1 × 1 mm3). Appropriate registration, cortical parcellation, and subcortical segmentation were performed during the data preprocessing. The cortex was automatically parcellated into 34 gyral regions per hemisphere using FreeSurfer. The Desikan-Killiany atlas was used for this parcellation51. The results were visually inspected for quality control, and no manual editing was required.
After a quality check, cortical and subcortical volumes were measured for each individual, along with the total GMV and total ICV. In addition, the CT for each surface vertex was computed as the shortest distance between the white matter and the pial surface. The cortical maps were then smoothed using a Gaussian kernel with a full width at half maximum of 10 mm. The SA, which represents the area of the vertex on the gray matter surface, was measured at the pial level. It was calculated as the mean of the areas of the tessellated triangles touching that vertex.
Statistical analyses
We used descriptive statistics of mean values and standard deviation to analyze the demographic information of individuals with SSA.
To determine the association between the level of social anxiety and structural neural correlates, we performed voxel-wise correlational analyses using FreeSurfer between the LSAS total scores and the GMV, CT, and SA of whole brain regions. We further analyzed the correlation between the LSAS performance anxiety scores and structural brain measures similarly. Age, sex, and ICV were used as covariates in the model; by controlling for these variables, we conducted a Monte Carlo simulation to correct for multiple comparisons using CWP and assessed the unique contributions of social anxiety levels (or performance anxiety levels) and brain regions to the outcome measures.
According to the results of the voxel-wise analysis, the mean GMV, CT, and SA values were extracted from regions with a significant correlation with the LSAS total scores or the LSAS performance anxiety scores. We performed exploratory Pearson’s correlation analyses to assess the linear relationships between the mean GMV, CT, and SA values in brain regions related to social anxiety level (or performance anxiety level) and other clinical features (e.g., GAD-7, BAI, and BDI-II) among individuals with SSA. The significance level of the α coefficient was set to 0.05 for statistical significance. Furthermore, we performed a Bonferroni correction in the exploratory correlation analyses (corrected p-value = 0.05/3 = 0.0167). We conducted additional statistical analyses using Statistical Package for the Social Sciences (SPSS) Windows version 27.0.
Data availability
All data used or analyzed during this study were included in this article. And further requests can be directed with a detailed description to the corresponding authors.
References
Morrison, A. S. & Heimberg, R. G. Social anxiety and social anxiety disorder. Annu. Rev. Clin. Psychol. 9, 249–274 (2013).
Rapee, R. M. & Heimberg, R. G. A cognitive-behavioral model of anxiety in social phobia. Behav. Res. Ther. 35, 741–756 (1997).
Moitra, E., Beard, C., Weisberg, R. B. & Keller, M. B. Occupational impairment and social anxiety disorder in a sample of primary care patients. J. Affect. Disord. 130, 209–212 (2011).
APA. Diagnostic and Statistical Manual of Mental Disorders (The American Psychiatric Association, 2013).
Lépine, J. P. & Pelissolo, A. Why take social anxiety disorder seriously?. Depress. Anxiety 11, 87–92 (2000).
Schwartz, C. E., Snidman, N. & Kagan, J. Adolescent social anxiety as an outcome of inhibited temperament in childhood. J. Am. Acad. Child Adolesc. Psychiatry 38, 1008–1015 (1999).
Bascomb, J. S. Performing Arts and Performance Anxiety (2019).
Powell, D. H. Treating individuals with debilitating performance anxiety: An introduction. J. Clin. Psychol. 60, 801–808 (2004).
Wesner, R. B., Noyes, R. Jr. & Davis, T. L. The occurence of performance anxiety among musicians. J. Affect. Disord. 18, 177–185 (1990).
McPherson, G. & Parncutt, R. The Science & Psychology of Music Performance: Creative Strategies for Teaching and Learning (Oxford University Press, 2002).
Cox, W. J. & Kenardy, J. Performance anxiety, social phobia, and setting effects in instrumental music students. J. Anxiety Disord. 7, 49–60 (1993).
Karsten, J., Penninx, B. W., Verboom, C. E., Nolen, W. A. & Hartman, C. A. Course and risk factors of functional impairment in subthreshold depression and anxiety. Depress. Anxiety 30, 386–394 (2013).
Fehm, L., Beesdo, K., Jacobi, F. & Fiedler, A. Social anxiety disorder above and below the diagnostic threshold: prevalence, comorbidity and impairment in the general population. Soc. Psychiatry Psychiatr. Epidemiol. 43, 257–265 (2008).
Bosman, R. C. et al. Prevalence and course of subthreshold anxiety disorder in the general population: a three-year follow-up study. J. Affect. Disord. 247, 105–113 (2019).
Marsh, B. et al. Shyness, alcohol use disorders and ‘hangxiety’: A naturalistic study of social drinkers. Personal. Ind. Differ. 139, 13–18 (2019).
Hershenberg, R. & Goldfried, M. R. Implications of RDoC for the research and practice of psychotherapy. Behav. Therapy 46, 156–165 (2015).
Lilienfeld, S. O. The Research Domain Criteria (RDoC): An analysis of methodological and conceptual challenges. Behav. Res. Ther. 62, 129–139 (2014).
Iza, M. et al. Latent structure of social fears and social anxiety disorders. Psychol. Med. 44, 361–370 (2014).
Maidenberg, E., Chen, E., Craske, M., Bohn, P. & Bystritsky, A. Specificity of attentional bias in panic disorder and social phobia. J. Anxiety Disord. 10, 529–541 (1996).
Laeger, I. et al. Amygdala responsiveness to emotional words is modulated by subclinical anxiety and depression. Behav. Brain Res. 233, 508–516 (2012).
Bruehl, A. B., Delsignore, A., Komossa, K. & Weidt, S. Neuroimaging in social anxiety disorder: A meta-analytic review resulting in a new neurofunctional model. Neurosci. Biobehav. Rev. 47, 260–280 (2014).
Phan, K. L., Fitzgerald, D. A., Nathan, P. J. & Tancer, M. E. Association between amygdala hyperactivity to harsh faces and severity of social anxiety in generalized social phobia. Biol. Psychiatry 59, 424–429 (2006).
Amir, N. et al. Increased activation of the anterior cingulate cortex during processing of disgust faces in individuals with social phobia. Biol. Psychiatry 57, 975–981 (2005).
Stein, M. B., Goldin, P. R., Sareen, J., Zorrilla, L. T. E. & Brown, G. G. Increased amygdala activation to angry and contemptuous faces in generalized social phobia. Arch. Gener. Psychiatry 59, 1027–1034 (2002).
Freitas-Ferrari, M. C. et al. Neuroimaging in social anxiety disorder: a systematic review of the literature. Progress Neuro-Psychopharmacol. Biol. Psychiatry 34, 565–580 (2010).
Wang, X., Cheng, B., Luo, Q., Qiu, L. & Wang, S. Gray matter structural alterations in social anxiety disorder: A voxel-based meta-analysis. Front. Psychiatry 9, 449 (2018).
Brühl, A. B. et al. Increased cortical thickness in a frontoparietal network in social anxiety disorder. Hum. Brain Mapp. 35, 2966–2977 (2014).
Zhang, X. et al. Dissociations in cortical thickness and surface area in non-comorbid never-treated patients with social anxiety disorder. EBioMedicine 58 (2020).
Günther, V. et al. Volumetric associations between amygdala, nucleus accumbens, and socially anxious tendencies in healthy women. Neuroscience 374, 25–32 (2018).
Bas-Hoogendam, J. M. et al. Subcortical brain volumes, cortical thickness and cortical surface area in families genetically enriched for social anxiety disorder: A multiplex multigenerational neuroimaging study. EBioMedicine 36, 410–428 (2018).
Koechlin, E., Basso, G., Pietrini, P., Panzer, S. & Grafman, J. The role of the anterior prefrontal cortex in human cognition. Nature 399, 148–151 (1999).
Braver, T. S. & Bongiolatti, S. R. The role of frontopolar cortex in subgoal processing during working memory. Neuroimage 15, 523–536 (2002).
Burgess, P. W., Veitch, E., de Lacy Costello, A. & Shallice, T. The cognitive and neuroanatomical correlates of multitasking. Neuropsychologia 38, 848–863 (2000).
Baxter, M. G. & Croxson, P. L. Facing the role of the amygdala in emotional information processing. Proc. Natl. Acad. Sci. 109, 21180–21181 (2012).
Fusar-Poli, P. et al. Functional atlas of emotional faces processing: a voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. J. Psychiatry Neurosci. 34, 418–432 (2009).
Haxby, J. V., Hoffman, E. A. & Gobbini, M. I. Human neural systems for face recognition and social communication. Biol. Psychiatry 51, 59–67 (2002).
Frick, A. et al. Cortical thickness alterations in social anxiety disorder. Neurosci. Lett. 536, 52–55 (2013).
Herrington, J. D., Taylor, J. M., Grupe, D. W., Curby, K. M. & Schultz, R. T. Bidirectional communication between amygdala and fusiform gyrus during facial recognition. Neuroimage 56, 2348–2355 (2011).
Sabatinelli, D., Bradley, M. M., Fitzsimmons, J. R. & Lang, P. J. Parallel amygdala and inferotemporal activation reflect emotional intensity and fear relevance. Neuroimage 24, 1265–1270. https://doi.org/10.1016/j.neuroimage.2004.12.015 (2005).
Syal, S. et al. Grey matter abnormalities in social anxiety disorder: A pilot study. Metab. Brain Dis. 27, 299–309 (2012).
Carré, A. et al. Linear association between social anxiety symptoms and neural activations to angry faces: From subclinical to clinical levels. Soc. Cognit. Affect. Neurosci. 9, 880–886 (2014).
First, M. B. Structured clinical interview for the DSM (SCID). Encycl. Clin. Psychol. 25, 1–6 (2014).
APA. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV-TR. 4 edn (American Psychiatric Association, 2000).
Rytwinski, N. K. et al. Screening for social anxiety disorder with the self-report version of the Liebowitz Social Anxiety Scale. Depress. Anxiety 26, 34–38 (2009).
Ștefan, C. A., Căpraru, C. & Szilágyi, M. Investigating effects and mechanisms of a mindfulness-based stress reduction intervention in a sample of college students at risk for social anxiety. Mindfulness 9, 1509–1521 (2018).
Fresco, D. et al. The Liebowitz Social Anxiety Scale: a comparison of the psychometric properties of self-report and clinician-administered formats. Psychol. Med. 31, 1025–1035 (2001).
Spitzer, R. L., Kroenke, K., Williams, J. B. & Löwe, B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch. Intern. Med. 166, 1092–1097 (2006).
Beck, A. T., Epstein, N., Brown, G. & Steer, R. Beck anxiety inventory. J. Consult. Clin. Psychol. (1993).
Beck, A. T., Gary Brown, N. E. & Steer, R. A. An inventory for measuring clinical anxiety_psychometric properties. J. Consult. Clin. Psychol. 56, 893–897. https://doi.org/10.1037/0022-006X.56.6.893 (1988).
Williams, N. The GAD-7 questionnaire. Occup. Med. 64, 224–224 (2014).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Funding
This research was supported by the Basic Science Research Program of the National Research Foundation of Korea, funded by the Ministry of Science and ICT (Grant Numbers NRF-2019M3C7A1032262 and NRF-2021M3E5D9025026). This study was also funded in part by the Healthcare AI Convergence Research & Development Program through the National IT Industry Promotion Agency of Korea (NIPA), funded by the Ministry of Science and ICT (Grant Number S0102-23-1008). S. H. Lee secured both sets of funding. This study was partially funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number HI23C0297000023). C. I. Park secured funding. This study was also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, and Future Planning (Grant Number NRF-2021R1C1C1012901). M. J. Bang also secured funding.
Author information
Authors and Affiliations
Contributions
B.K.: Conceptualization, Methodology, Data curation, Formal analysis, and Writing—original draft/review and editing. S.P.: Conceptualization, Methodology, Data curation, Formal analysis, and Writing—original draft/review and editing. C.I.P.: Conceptualization, Data interpretation, Supervision, Validation, and Funding Acquisition. M.B.: Conceptualization, Supervision, Validation, and Funding Acquisition. H.-J.K.: Conceptualization, Methodology, Formal analysis, Investigation, Writing—original draft/review and editing, Supervision, and Validation. S.-H.L.: Conceptualization, Data curation, Formal analysis, Writing—review and editing, Supervision, Validation, Project Administration, and Funding Acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, BH., Park, SY., Park, C.I. et al. Altered cortical thickness of the superior frontal gyrus and fusiform gyrus in individuals with subthreshold social anxiety. Sci Rep 13, 21822 (2023). https://doi.org/10.1038/s41598-023-49288-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-49288-7
- Springer Nature Limited