Abstract
Suspected central nervous system (CNS) infections may pose a diagnostic challenge, and often concern severely ill patients. We aim to identify predictors of unfavourable outcome to prioritize diagnostics and treatment improvements. Unfavourable outcome was assessed on the Glasgow Outcome Scale at hospital discharge, defined by a score of 1 to 4. Of the 1152 episodes with suspected CNS infection, from two Dutch prospective cohorts, the median age was 54 (IQR 37–67), and 563 episodes (49%) occurred in women. The final diagnoses were categorized as CNS infection (N = 358 episodes, 31%), CNS inflammatory disease (N = 113, 10%), non-infectious non-inflammatory neurological disorder (N = 388, 34%), non-neurological infection (N = 252, 22%), and other systemic disorder (N = 41, 4%). Unfavourable outcome occurred in 412 of 1152 (36%), and 99 died (9%). Predictors for unfavourable outcomes included advanced age, absence of headache, tachycardia, altered mental state, focal cerebral deficits, cranial nerve palsies, low thrombocytes, high CSF protein, and the final diagnosis of CNS inflammatory disease (odds ratio 4.5 [95% confidence interval 1.5–12.6]). Episodes suspected of having a CNS infection face high risk of experiencing unfavourable outcome, stressing the urgent need for rapid and accurate diagnostics. Amongst the suspected CNS infection group, those diagnosed with CNS inflammatory disease have the highest risk.
Similar content being viewed by others
Introduction
Patients suspected of a central nervous system (CNS) infection often present with severe illness, including decreased consciousness, neurological deficits and hemodynamic instability1. Diagnosing these patients frequently poses a challenge due to the wide range of possible conditions, ranging from life-threatening diseases such as bacterial meningitis or septic encephalopathy to more benign and sometimes self-limiting diseases such as migraine or systemic viral infections1. Previous studies have reported an overall mortality of 10% and incomplete recovery in an additional 17% in this population1,2. Prompt diagnostic work-up, identification of the cause-specific diagnosis, and early targeted treatment have been shown to be crucial in improving outcome, particularly in patients with bacterial meningitis2,3,4,5,6,7. However, clinical characteristics and ancillary investigations often lack sensitivity and/or specificity to differentiate between these various causes, although cerebrospinal fluid (CSF) leukocyte count differentiated best between bacterial meningitis and other diagnoses in this population1. Difficulty in making the diagnosis may lead to delayed or unnecessary treatment with antibiotics and antiviral drugs. To improve outcome in this patient population, it is essential to recognize high-risk categories for unfavourable outcome. This prospective study aims to determine predictors for an unfavourable outcome to identify subgroups for enhancing diagnostics and treatment.
Methods
Patient inclusion and data collection
We included episodes from two prospective cohort studies performed between 2012 and 2015 and between 2017 and 2022. The first study (PACEM—Paediatrics and Adult Causes of Encephalitis and Meningitis) was a single-centre study, and a pilot study for the second study (I-PACE—Improving Prognosis by using innovative methods to diAgnose Causes of Encephalitis), which is an ongoing multi-centre study running in 11 Dutch hospitals1. Both studies included adult patients aged 16 years or older with suspected CNS infection presenting to the emergency department or inpatients who underwent CSF examination. Episodes were identified during morning rounds or reported to the investigators by the treating physician. Physicians could contact the investigators 24/7 to include patients. Episodes of suspected CNS infections within three months after head trauma or neurosurgery, and those with a neurosurgical device in situ, were excluded.
Data on patient characteristics, medical history, symptoms and signs on admission, laboratory results, radiological examination, treatment and outcome were collected in online case record forms. All patients and/or their legal representatives have given written informed consent for this study after receiving written information about the study. All patient data were rendered anonymous, and the study was carried out in accordance with Dutch privacy legislation.
Procedures and definitions
Episodes were classified as suspected nosocomial CNS infection if the suspicion occurred during hospital admission (> 48 h after presentation) or within one week after discharge8,9. All other episodes were classified as community-acquired. Neurological examination was performed upon admission and at discharge. The level of consciousness was scored using the Glasgow Coma Scale (GCS)10. An altered mental state was defined as a GCS score of < 14 and coma as a GCS score of ≤ 8. Patients were considered immunocompromised if they were using immunosuppressive drugs or had a medical history of diabetes mellitus, alcoholism, HIV infection or a splenectomy.
Outcome at discharge was scored according to the Glasgow Outcome Scale (GOS), a well-validated scale ranging from 1 to 5. A score of 1 indicates death, 2 vegetative survival, 3 severe disability, 4 moderate disability, and 5 indicates mild or no disability10. A score of 5 was considered a favourable outcome. If pre-existing conditions were the cause of the outcome score below 5 on the GOS, and the patient’s condition did not worsen due to the current episode, we classified the outcome as favourable.
Diagnostic categorization
The final diagnosis of all included episodes was classified into five categories, as previously described1. The categories were; (1) CNS infection, (2) CNS inflammatory disease, (3) non-infectious non-inflammatory neurological disorder, (4) non-neurological infection, and (5) other systemic disorders. Two clinicians independently classified the final diagnoses in the five categories based on all available clinical, laboratory and follow-up data. If there was no consensus, a third investigator was consulted. Inter-rater agreement between the first assessors was assessed by calculating the kappa coefficient, which was 0.76 in cohort 1 and 0.64 in cohort 2.
Statistical analysis
Statistical analyses were conducted using SPSS statistical software, version 28 (SPSS Inc.) and R studio version 4.0.3. We used descriptive statistics for baseline characteristics, with medians and interquartile range (IQR, describing their 25th to 75th percentile). Comparisons were made with the Mann–Whitney U test used for continuous data, and the Fisher exact test was used for categorical data. All tests were 2-tailed, and P < 0.05 was considered significant. We chose possible predictors of an unfavourable outcome based on previous research and availability to examine the predictor early upon disease presentation11. We investigated the association between these predictors and outcomes with logistic regression, providing odds ratios (ORs) and 95% CIs. Univariable and multivariable binary logistic regression models assessed prognostic factors for discharge outcomes. For these multivariable logistic models, missing values in the selected prognostic factors were imputed (median 2.1% per prognostic factor [IQR 0.33–8.8%]). Non-normally distributed continuous variables were transformed into categorical variables.
Standard protocol approvals, registrations and patient consents
The two studies were approved by the Biobank Ethics Assessment Committee of the Amsterdam UMC; number AMC 2014_290. Written informed consent was obtained from all participants or their representatives. All methods were performed in accordance with this approval.
Results
A total of 1165 episodes were included: 363 episodes in the PACEM study and 802 in the I-PACE study. Of these, 13 episodes (1%) were excluded based on exclusion criteria or missing outcome data (Fig. 1), resulting in 1152 episodes in 1127 patients. Patients were evaluated at the emergency department in 861 of 1140 episodes (76%), at the intensive care in 59 (5%), and 220 (19%) at other clinical departments. The episode was classified as nosocomial in 106 of 1137 (9%)8,9.
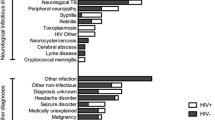
The median age was 54 years (IQR 37–67), and 563 episodes (49%) occurred in women (Table 1). An immunocompromising condition was present in 450 of 1151 episodes (39%), which was due to HIV infection in 74 of 1150 (6%), the use of immunosuppressive drugs in 208 of 1149 (18%), and diabetes mellitus in 188 of 1151 (16%; Table 1). In 417 of 1111 episodes (38%), symptoms were present for less than 24 h. The most common symptoms included headache in 639 of 998 episodes (64%), fever in 466 of 1051 episodes (44%), and neck stiffness in 188 of 892 episodes (21%). An altered mental state was present in 364 of 1143 episodes (32%) and neurological deficits in 347 of 754 (46%).
A lumbar puncture was performed in all episodes, and CSF examination showed an elevated leukocyte count (≥ 4 cells/mm3) in 622 of 1139 episodes (55%). The CSF leukocyte count was between 4 and 99 cells/mm3 in 378 (33%) episodes, between 100 and 999 cells/mm in 147 (13%), and more than 1000 cells/mm3 in 97 (9%) episodes. During the clinical course, antibiotics according to bacterial meningitis regime or antiviral treatment were started in 695 of 1150 episodes (60%).
A final clinical diagnosis was available for all episodes. CNS infection was diagnosed in 358 (31%), CNS inflammatory disease in 113 (10%), non-infectious non-inflammatory neurological disorder in 388 (34%), non-neurological infection in 252 (22%), and other systemic disorder in 41 (4%, Table 2). Of the 358 CNS infections, the diagnosis was microbiologically confirmed in 236 episodes (66%). CSF culture was positive in 79 of 236 (33%) episodes, CSF PCR in 117 (50%), CSF antigen testing in 19 (5%), blood culture in 92 episodes (39%), and blood PCR in 17 (7%).
The outcome was unfavourable in 412 episodes (36%), and in 99 of 1152 episodes (9%), the patient died (Table 3). Neurological sequelae were present in 352 of 1015 (35%) surviving patients. The rate of unfavourable outcome varied per disease category and was 118 out of 358 episodes (33%) diagnosed with CNS infections, 74 out of 113 (65%) with CNS inflammatory diseases, 150 out of 388 (39%) with non-infectious non-inflammatory neurological disorders, 54 out of 252 (21%) with non-neurological infections, and in 16 out of 41 (39%) with other systemic disorders (Table 2). The mortality rate was 36 of 358 (10%) in episodes with CNS infections, eight out of 113 (7%) with CNS inflammatory disease, 28 out of 388 (7%) with non-infectious non-inflammatory neurological disorders, 25 out of 252 (10%) with non-neurological infections two of 41 (5%) with other systemic disorders. To analyse changes in time period and outcome between cohort 1 and cohort 2, we found an unfavourable outcome in 91 of 363 episodes (25%) in cohort 1 versus 321 of 793 episodes (41%) in cohort 2, P < 0.001.
In the multivariable analysis, predictors for unfavourable outcome were advanced age, the absence of headache, tachycardia, GCS score < 14, focal cerebral deficits (aphasia or paresis), cranial nerve palsies, thrombocyte count < 150 × 1012/L, CSF protein count > 0.60 g/L, and the final diagnosis of a CNS inflammatory disease (Table 4).
Predictors for death were advanced age (> 70 years old), an immunocompromised state, GCS score < 14, the absence of headache, diastolic blood pressure < 60 mm Hg, thrombocyte count < 150 × 1012/L, CRP of 40 to 150 mg/dL, and CSF protein concentration > 0.60 g/L (Table 5).
The group of CNS inflammatory diseases consisted of 113 of 1152 episodes (10%). The rate of unfavourable outcome differed between the definitive diagnoses included in this category. Eight out of ten (80%) episodes with confirmed autoimmune encephalitis (AE) had an unfavourable outcome, 18 out of 26 (69%) with possible AE of unknown cause, eight out of nine (89%) with myelitis, and 32 out of 43 (74%) with other neurological autoimmune disorders (Table 2). Unfavourable outcome was due to residual neurological sequelae in 62 out of 74 (84%).Twenty-three of 113 episodes (20%) with CNS inflammatory disease were initially treated with antibiotics consisting of amoxicillin and ceftriaxone according to bacterial meningitis protocol. Aciclovir was given in 35 episodes (31%). When probable CNS inflammation was diagnosed, first line immunosuppressive therapy (e.g., methylprednisolone (MPS), prednisone, intravenous immunoglobulins [IVIg]), was started in 87 of 113 episodes (77%) and escalation to second-line therapy (e.g., plasma exchange [PLEX], azathioprine, rituximab, cyclophosphamide, and mycophenolate mofetil [MMF], methotrexate) was required in 31 of 87 episodes (36%). First line therapy was commenced during initial admission, in 71 of 87 episodes (82%), with escalation to 2nd line therapy during this admission in 19 of 71 episodes (27%). Escalation to 2nd line therapy at a later point in the outpatient clinic or when readmitted was done in 10 of 71 episodes (14%). For 16 of 87 episodes (18%), first line treatment was only started after admission with escalation to 2nd line immunosuppressive therapy in 2 of 16 episodes (13%).
The time between presentation to immunosuppressive treatment was known in 84 of 87 (96%), with a median time to treatment of 5 days (IQR 1- 30). A univariate analysis for time to treatment and outcome showed no association (odds ratio 0.83 [0.51–1.35], P = 0.45). Immunosuppressive treatment was not administered in the remaining 26 episodes for various reasons, including spontaneous recovery occurred in 6 episodes (26%), mild symptoms well-manageable with symptom relief medication in 6 episodes (26%), a self-limiting disorder in 4 (15%), and one patient died before commencing immunosuppressants (4%).
Discussion
Our study shows that patients presenting with an episode of suspected CNS infection have a high risk (36%) of experiencing an unfavourable outcome. Consistent with previous studies, advanced age was found to be an independent predictor of unfavourable outcome12,13,14. The association between outcome and focal cerebral deficits, an altered mental state, and elevated CSF protein count is likely to reflect the severity of neurological damage, while thrombocytopenia and tachycardia are associated with sepsis15,16,17,18,19.
Patients who were eventually diagnosed with CNS inflammatory disease showed the poorest prognosis. This association can be explained by various factors, including the severity of the conditions. Unfavourable outcome was most prevalent in confirmed cases with autoimmune encephalitis (80%) or suspected autoimmune encephalitis (69%). These rates are relatively high compared to previous studies on autoimmune encephalitis, which reported rates ranging from 13 to 80%, depending on the follow-up duration, associated antibodies, and aetiology of the autoimmune encephalitis episode20,21,22,23,24,25,26. The difference in outcome between our cohort and the literature may be due to the limited follow-up time in our study, as most studies provided an extensive follow-up time of up to 33 months, with outcomes that continued to improve for up to 18 months after symptom onset21,22,25,27. Moreover, our cohort consisted of a relatively small group of autoimmune encephalitis cases, most of whom were admitted to a tertiary hospital. Furthermore, our observation that other inflammatory conditions, like inflammatory myelitis, vasculitis, Guillain Barre syndrome, neurosarcoidosis, are associated with an unfavourable outcome aligns with existing literature28,29,30,31.
Contrary to previous studies on predictors for unfavourable outcome in CNS infections, the presence of seizures or an immunocompromised state, e.g., diabetes mellitus, did not show an association in our cohort32,33. This can be explained due to the heterogeneity in diagnoses in the cohort. Notably, for these variables, the odds ratios shifted from indicating a higher likelihood to suggesting a lower likelihood of an unfavourable outcome between the univariate and the multivariate analyses. This change could be caused by interactions with a covariate, such as final diagnosis associated with diabetes or an immunocompromised state, although this is speculative.
In CNS inflammatory diseases, treatment choice frequently rely on expert opinions rather than on randomized controlled trials for comparing treatments. Although our study did not find an association between treatment delay and outcome in CNS inflammatory episodes, it is generally accepted that time to treatment is a modifiable risk factor for poor outcome. Moreover, accumulating evidence and recent guidelines point to the beneficial effects of early diagnosis and treatment on outcome34,35,36,37,38,39.
Currently, diagnostic methods only establish the etiologic cause in 50% of encephalitis cases, with at least 10% being diagnosed as autoimmune encephalitis, of which causative anti-neuronal antibodies could only be detected in 35%1,26,40. The median time to treatment initiation for a CNS inflammatory disease was 5 days, and treatment was started only after 30 days in 25% of the cases. This can be attributed to an insidious onset of the disease, as well as the lengthy duration of diagnostic tests for autoimmune encephalitis, such as anti-neuronal antibody testing. Such episodes can initially be suspected of infectious meningoencephalitis, but after microbiological tests return negative, diagnostic tests for autoimmune encephalitis are ordered and generally take several weeks to generate results. Unfortunately, empirical treatment for autoimmune disorders is often not initiated while waiting for these tests41.
Prompt immunotherapy has been associated with a favourable outcome for all types of autoimmune encephalitis, as spontaneous clinical improvement is infrequent21. Various treatment options are available, including corticosteroids, TPE, IVIG, and immunosuppressant drugs. Treatment choice depends on the pathophysiology of the specific type of autoimmune encephalitis and the patients’ comorbidity26,42. A recent study concluded that more aggressive treatment regimens in autoimmune encephalitis patients improved the 2-year outcome. However, a comment on this study suggested that first-line immunotherapy's effect was underestimated while second-line immunotherapy's effect was overestimated26,43. Based on our study, early treatment with anti-inflammatory drugs should be considered to minimize the risk of an unfavourable outcome in cases of CNS inflammatory diseases.
Our study had several limitations. First, episodes could only be included when a lumbar puncture was performed, and the researchers identified the patients. This may have resulted in missed inclusions. Second, in some episodes, the final diagnosis was based on the clinical picture rather than microbiological evidence, demonstrated antibodies or radiological features, and thus may have led to misclassification. To solve this, we scored the final clinical diagnoses with two independent investigators and a third to solve discrepancies representing a proper classification process. Third, patients were predominantly admitted to a tertiary hospital and were inherently more complex than those in a general hospital, potentially causing selection bias. However, the majority of patients presented at the emergency department, reducing this risk of bias. Fourth, we did not analyse predictors for outcome for each diagnostic category separately. Instead, our focus was on evaluating all adults presenting with a suspected CNS infection, aiming to aid physicians in the acute setting, particularly when patients are still undifferentiated. This approach allowed us to gain insights into which patient subgroup requires more targeted investigation on diagnostics and treatment in future research.
In conclusion, patients suspected of having a CNS infection are at high risk of experiencing an unfavourable outcome, stressing the urgent need for improving rapid and accurate diagnostics. Amongst this suspected CNS infection group, those eventually diagnosed with CNS inflammatory disease have the highest risk of an unfavourable outcome. Our findings underscore the importance of prioritizing diagnostic and treatment improvements in this population. Based on our study, early treatment with immunosuppressive drugs may be considered to reduce the risk of an unfavourable outcome in cases of CNS inflammatory diseases.
Data availability
Anonymized data not published within this article will be made available by request from any qualified investigator. Proposals can be directed to the corresponding author, Matthijs Brouwer, by sending an email to ipace@amc.nl.
References
Khatib, U., van de Beek, D., Lees, J. A. & Brouwer, M. C. Adults with suspected central nervous system infection: A prospective study of diagnostic accuracy. J. Infect. 74, 1–9. https://doi.org/10.1016/j.jinf.2016.09.007 (2017).
Kollef, M. H., Sherman, G., Ward, S. & Fraser, V. J. Inadequate antimicrobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest 115, 462–474. https://doi.org/10.1378/chest.115.2.462 (1999).
van de Beek, D. et al. Community-acquired bacterial meningitis. Nat. Rev. Dis. Primers 2, 16074. https://doi.org/10.1038/nrdp.2016.74 (2016).
Rivers, E. et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N. E. J. Med. 345, 1368–1377. https://doi.org/10.1056/NEJMoa010307 (2001).
van de Beek, D. et al. ESCMID guideline: diagnosis and treatment of acute bacterial meningitis. Clin. Microbiol. Infect. Off. Public. Eur. Soc. Clin. Microbiol. Infect. Dis. 22(Suppl 3), S37-62. https://doi.org/10.1016/j.cmi.2016.01.007 (2016).
Bradshaw, M. J. & Venkatesan, A. Herpes simplex virus-1 encephalitis in adults: pathophysiology, diagnosis, and management. Neurotherapeutics 13, 493–508. https://doi.org/10.1007/s13311-016-0433-7 (2016).
Shin, Y. W. et al. Treatment strategies for autoimmune encephalitis. Ther. Adv. Neurol. Disord. 11, 1756285617722347. https://doi.org/10.1177/1756285617722347 (2018).
van de Beek, D., Drake, J. M. & Tunkel, A. R. Nosocomial bacterial meningitis. N. Eng. J. Med. 362, 146–154. https://doi.org/10.1056/NEJMra0804573 (2010).
Bijlsma, M. W. et al. Community-acquired bacterial meningitis in adults in the Netherlands, 2006–14: a prospective cohort study. Lancet Infect. Dis. 16, 339–347. https://doi.org/10.1016/s1473-3099(15)00430-2 (2016).
Jennett, B., Teasdale, G., Braakman, R., Minderhoud, J. & Knill-Jones, R. Predicting outcome in individual patients after severe head injury. Lancet (London, England) 1, 1031–1034. https://doi.org/10.1016/s0140-6736(76)92215-7 (1976).
Bijlsma, M. W. et al. Community-acquired bacterial meningitis in adults in the Netherlands, 2006–14: a prospective cohort study. Lancet Infect. Dis. 16, 339–347. https://doi.org/10.1016/s1473-3099(15)00430-2 (2016).
van Soest, T. M., Chekrouni, N., van Sorge, N. M., Brouwer, M. C. & van de Beek, D. Community-acquired bacterial meningitis in patients of 80 years and older. J. Am. Geriatr. Soc. 70, 2060–2069. https://doi.org/10.1111/jgs.17766 (2022).
Ter Avest, E., de Jong, M., Brűmmer, I., Wietasch, G. J. & Ter Maaten, J. C. Outcome predictors of uncomplicated sepsis. Int. J. Emerg. Med. 6, 9. https://doi.org/10.1186/1865-1380-6-9 (2013).
van de Beek, D. et al. Clinical features and prognostic factors in adults with bacterial meningitis. N. Eng. J. Med. 351, 1849–1859. https://doi.org/10.1056/NEJMoa040845 (2004).
Santacruz, C. A. et al. Association of cerebrospinal fluid protein biomarkers with outcomes in patients with traumatic and non-traumatic acute brain injury: systematic review of the literature. Crit. Care 25, 278. https://doi.org/10.1186/s13054-021-03698-z (2021).
van Zeggeren, I. E. et al. Neurofilament light chain in central nervous system infections: a prospective study of diagnostic accuracy. Sci. Rep. 12, 14140. https://doi.org/10.1038/s41598-022-17643-9 (2022).
Mariotto, S. et al. Serum and CSF neurofilament light chain levels in antibody-mediated encephalitis. J. Neurol. 266, 1643–1648. https://doi.org/10.1007/s00415-019-09306-z (2019).
Vardon-Bounes, F., Ruiz, S., Gratacap, M. P., Garcia, C., Payrastre, B. & Minville, V. Platelets Are Critical Key Players in Sepsis. Int. J. Mol. Sci. 20 (2019). https://doi.org/10.3390/ijms20143494
Guclu, E., Durmaz, Y. & Karabay, O. Effect of severe sepsis on platelet count and their indices. Afr. Health Sci. 13, 333–338. https://doi.org/10.4314/ahs.v13i2.19 (2013).
Iizuka, T. et al. Association of progressive cerebellar atrophy with long-term outcome in patients with Anti-N-Methyl-d-Aspartate receptor encephalitis. JAMA Neurol. 73, 706–713. https://doi.org/10.1001/jamaneurol.2016.0232 (2016).
Dalmau, J. & Graus, F. Antibody-mediated encephalitis. N. Eng. J. Med. 378, 840–851. https://doi.org/10.1056/NEJMra1708712 (2018).
Titulaer, M. J. et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 12, 157–165. https://doi.org/10.1016/s1474-4422(12)70310-1 (2013).
Swayne, A. et al. An australian state-based cohort study of autoimmune encephalitis cases detailing clinical presentation, investigation results, and response to therapy. Front. Neurol. 12, 607773. https://doi.org/10.3389/fneur.2021.607773 (2021).
Qiao, S. et al. Characteristics and prognosis of autoimmune encephalitis in the east of China: a multi-center study. Front. Neurol. 12, 642078. https://doi.org/10.3389/fneur.2021.642078 (2021).
Hayden, Z. et al. Clinical characteristics and outcome of neuronal surface antibody-mediated autoimmune encephalitis patients in a national cohort. Front. Neurol. 12, 611597. https://doi.org/10.3389/fneur.2021.611597 (2021).
Lee, W. J. et al. Seronegative autoimmune encephalitis: clinical characteristics and factors associated with outcomes. Brain 145, 3509–3521. https://doi.org/10.1093/brain/awac166 (2022).
van Sonderen, A., Petit-Pedrol, M., Dalmau, J. & Titulaer, M. J. The value of LGI1, Caspr2 and voltage-gated potassium channel antibodies in encephalitis. Nat. Rev. Neurol. 13, 290–301. https://doi.org/10.1038/nrneurol.2017.43 (2017).
Ali, A. et al. Assessment of clinical outcomes in patients presenting with transverse myelitis: a tertiary care experience from a developing country. Cureus 11, e4342. https://doi.org/10.7759/cureus.4342 (2019).
Rajabally, Y. A. & Uncini, A. Outcome and its predictors in Guillain-Barre syndrome. J. Neurol. Neurosurg. Psychiat. 83, 711–718. https://doi.org/10.1136/jnnp-2011-301882 (2012).
Fritz, D., van de Beek, D. & Brouwer, M. C. Clinical features, treatment and outcome in neurosarcoidosis: systematic review and meta-analysis. BMC Neurol. 16, 220. https://doi.org/10.1186/s12883-016-0741-x (2016).
Moffett, K. S. & Berkowitz, F. E. Quadriplegia complicating Escherichia coli meningitis in a newborn infant: case report and review of 22 cases of spinal cord dysfunction in patients with acute bacterial meningitis. Clin. Infect. Dis. 25, 211–214. https://doi.org/10.1086/514534 (1997).
van Veen, K. E., Brouwer, M. C., van der Ende, A. & van de Beek, D. Bacterial meningitis in diabetes patients: a population-based prospective study. Sci. Rep. 6, 36996. https://doi.org/10.1038/srep36996 (2016).
Alam, A. M. et al. Increased volume of cerebral oedema is associated with risk of acute seizure activity and adverse neurological outcomes in encephalitis - regional and volumetric analysis in a multi-centre cohort. BMC Neurol. 22, 412. https://doi.org/10.1186/s12883-022-02926-5 (2022).
Bodilsen, J., Dalager-Pedersen, M., Schønheyder, H. C. & Nielsen, H. Time to antibiotic therapy and outcome in bacterial meningitis: a Danish population-based cohort study. BMC Infect. Dis. 16, 392. https://doi.org/10.1186/s12879-016-1711-z (2016).
Costerus, J. M., Lemmens, C. M. C., van de Beek, D. & Brouwer, M. C. Cranial imaging and lumbar puncture in patients with suspected central nervous system infection. Clin. Infect. Dis. 70, 2469–2475. https://doi.org/10.1093/cid/ciz694 (2020).
Granillo, A. et al. Development and validation of a risk score to differentiate viral and autoimmune encephalitis in adults. Clin. Infect. Dis. 76, e1294–e1301. https://doi.org/10.1093/cid/ciac711 (2023).
Leonhard, S. E., Fritz, D., Eftimov, F., van der Kooi, A. J., van de Beek, D. & Brouwer, M. C. Neurosarcoidosis in a Tertiary Referral Center: A Cross-Sectional Cohort Study. Medicine (Baltimore) 95, e3277 (2016). https://doi.org/10.1097/md.0000000000003277
Kenan, G. et al. Reasons for delayed treatment initiation in Guillain-Barre syndrome. J. Neurol. Sci. 434, 120179. https://doi.org/10.1016/j.jns.2022.120179 (2022).
Sreih, A. G. et al. Diagnostic delays in vasculitis and factors associated with time to diagnosis. Orphanet. J. Rare Dis. 16, 184. https://doi.org/10.1186/s13023-021-01794-5 (2021).
Glaser, C. A. et al. In search of encephalitis etiologies: diagnostic challenges in the California Encephalitis Project, 1998–2000. Clin. Infect. Dis. 36, 731–742. https://doi.org/10.1086/367841 (2003).
Graus, F. et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 15, 391–404. https://doi.org/10.1016/s1474-4422(15)00401-9 (2016).
Jiao, L. & Guo, S. Anti-IL-6 therapies in central nervous system inflammatory demyelinating diseases. Front. Immunol. 13, 966766. https://doi.org/10.3389/fimmu.2022.966766 (2022).
van Steenhoven, R. W. & Titulaer, M. J. Seronegative autoimmune encephalitis: exploring the unknown. Brain 145, 3339–3340. https://doi.org/10.1093/brain/awac338 (2022).
Acknowledgements
Supported by grants from the European Research Council (ERC Consolidator grant 101001237 to MB), and the Netherlands Organization for Health Research and Development (Vici grant number 918.19.627 to DvdB; Vidi grant number 917.17.308 to MB). Dr. Jan Meerwaldt Foundation (travel grant to LtH); Remmert Adriaan Laan Foundation (travel grant to LtH); Amsterdam University Fund (travel grant to LtH), and the Royal Netherlands Academy of Arts & Sciences (KNAW Van Leersum Grant to LtH).
Author information
Authors and Affiliations
Consortia
Contributions
LtH: methodology, data collection, data analysis, data interpretation, and writing the original draft of the manuscript; IvZ; study design, data collection, review and editing of the report; SO: data collection, review and editing of the report; DvdB: review, editing, and supervision of the report; MB: methodology, study design, data interpretation, review, editing and supervision of the report, and funding acquisition. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
ter Horst, L., van Zeggeren, I.E., Olie, S.E. et al. Predictors of unfavourable outcome in adults with suspected central nervous system infections: a prospective cohort study. Sci Rep 13, 21250 (2023). https://doi.org/10.1038/s41598-023-48472-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48472-z
- Springer Nature Limited