Abstract
Lassa fever (LF) is prevalent in many West African countries, including Nigeria. Efforts to combat LF have primarily focused on rural areas where interactions between rodents and humans are common. However, recent studies indicate a shift in its occurrence from rural to urban areas. We analysed secondary data of reported LF outbreaks from 2017 to 2021 in Ondo State, Nigeria to identify the distribution pattern, ecological variations, and other determinants of disease spread from the ward level using nearest neighbour statistics and regression analysis. Data utilised include LF incidence, ecological variables involving population, nighttime light intensity, vegetation, temperature, market presence, road length, and building area coverage. ArcGIS Pro 3.0 software was employed for spatial analysis. Results revealed spatio-temporal clustering of LF incidents between 2017 and 2021, with an increasing trend followed by a decline in 2021. All wards in Owo Local Government Area were identified as LF hotspots. The ecological variables exhibited significant correlations with the number of LF cases in the wards, except for maximum temperature. Notably, these variables varied significantly between wards with confirmed LF and those without. Therefore, it is important to prioritise strategies for mitigating LF outbreaks in urban areas of Nigeria and other LF-endemic countries.
Similar content being viewed by others
Introduction
Lassa fever (LF) is an acute and sometimes severe viral haemorrhagic illness transmitted by rodents1. It is endemic in West Africa, particularly rural Nigeria and the Mano River Union countries (Liberia, Guinea, and Sierra Leone)1,2. Discovered in 1969, LF is a threat to human health, accounting for between 300,000 and 500,000 cases and 5000 deaths annually in West Africa, with case fatality rate (CFR) of 15–25% among hospitalised patients2,3. The disease presents with various clinical manifestations, including several cases thought to be mild or asymptomatic4. Unspecific fever and malaise are the symptoms associated with most cases. However, occasional progression to haemorrhagic symptoms and fatalities in about 20% of severe cases are often observed4.
Lassa fever is caused by a single-stranded RNA virus belonging to the Arenaviridae family5. There are currently six main Lassa virus (LASV) clades distributed across different West African countries: clades I–III (Nigeria), clade IV (Sierra Leone, Guinea, and Liberia), clade V (southern Mali) and clade VI (recently reported from Togo). The sustenance of LASV transmission has been attributed to its ability to mutate over time6. Lassa fever poses significant public health challenge with recurrent yearly seasonal outbreaks in West Africa 4. Although the disease is geographically associated with some specific countries in West Africa, it is one of the most exported viral haemorrhagic fevers (VHFs)7. In this sub-region alone, it is estimated that LF affects about 2 million persons, resulting in about 5000–10,000 deaths annually8. Two major epicentres of the disease have been identified in West Africa, the first in Nigeria and the second localised around Sierra Leone and Liberia8. However, recent sporadic outbreaks have been widespread around most countries in the sub-region, particularly in Nigeria5.
Several factors have been identified as responsible for exacerbating and sustaining the transmission of LF in Nigeria. Some of these include food exposure to droppings or urine of infected rodents that normally occur during open drying of grains and other food items9. Other factors are the consumption of infected rodents, inappropriate practices such as unhygienic waste disposal and poor environmental sanitation9,10,11. It has been reported that the burden of LF is more associated with the rural areas owing to the preponderance of associated risk factors in these settings, some of which include cultural practices (hunting, processing and consumption of rodents as protein source, and drying of food items by the roadside) and poverty12,13,14,15. However, recent studies have reported a probable shift in LF occurrence from rural to main urban or urban slum settings12,14.
Currently, in Nigeria, 5083 suspected cases of LF were reported between January and May 2023, with 1067 (13.0%) confirmed and 156 deaths (CFR = 17.7%)16. Overall, 112 Local Government Areas (LGAs) across 27 States of the country reported at least one confirmed case over this period in 2022. Furthermore, three states accounted for about 72% of the cases reported in 2022 (Ondo: 33%, Edo: 25% and Bauchi: 14%)17. Thus, for the first time since 2017, Ondo State surpassed Edo State as the epicentre of LF in Nigeria18. The steady increase in the number of cases of LF in Ondo State underscores the importance of the state in the overall epidemiology of LF in Nigeria, and this poses huge concerns for public health.
Additionally, this increase highlights the need for further investigation of the dynamics of occurrence and spread of LF in community settings in Ondo State, Nigeria. Although previous workers studied the effects of seasonal variations19 as well as climatic parameters such as rainfall and temperature8,20 on the incidence of LF, little has been done to assess the impact of multiple ecological indicators on the incidence of the disease at the smallest administrative units (i.e., ward level) in a hotspot location such as Ondo State. This study was therefore designed to determine the ecological correlates and predictors of LF in Ondo State over five years (2017 to 2021) with a view to examining the distribution pattern of persons diagnosed with LF, as well as the correlates and determinants of LF incidence and occurrence at the ward level in the state.
Results
Spatio-temporal distribution of Lassa fever incidents between 2017 and 2021
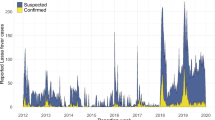
A list of 4315 suspected LF cases from 2017 to 2021 was obtained, including 1057 laboratory-confirmed cases (24.50% of all cases) (Fig. 1). The remaining cases were negative or classified as probable. Furthermore, out of the individuals included in the data, 821 individuals (77.62%) were still alive at the time of data collection.
The number of confirmed LF incidents increased significantly from 61 in 2017 to 147 in 2018 (140.98% increase). Subsequently, there were increases of 82.31% in 2019 and 54.48% in 2020. However, there was a notable decline of 59.66% between 2020 and 2021 (Supplementary Fig. 1). Regarding temporal incidence, the highest number of confirmed cases occurred in 2020 (39.17%), followed by 2019, 2021, 2018, and 2017. These findings align with a study by Dalhat et al. (2022)9 that analysed LF surveillance data from all states in Nigeria between January 2018 and December 2021.
Notably, the temporal analysis of LF incidents in Ondo State from 2017 to 2021 revealed significant spatial clustering. The Average Nearest Neighbour Statistic values (ranging from 0.161168 to 0.376471) indicated clustering in specific locations rather than random dispersion across the state (Table 1). Kernel Density Estimation (KDE) results showed that the majority of LF clusters were concentrated in Owo town, while Akure town had a notable number of cases but no detected clusters. This suggests a random distribution in Akure town.
Spatiotemporal pattern of Lassa fever incidents among wards between 2017 and 2021
Out of the 203 wards in Ondo State, 70 wards (34.48%) experienced LF incidents between 2017 and 2021 (Supplementary Table 1). The distribution of incidents varied across the years, with an increase in the number of affected wards. In 2017, there were 26 wards with LF incidents, which increased to 28 in 2018, 45 in 2019, 55 in 2020, and 35 in 2021. Among the wards with the highest number of incidents, Ehinogbe ward had the most cases in 2017, while Igboroko 2 had the highest in 2018. In 2019, both Ijebu 1 and Ijebu 2 wards had the highest number of incidents. Igboroko 2 ward consistently had the highest number of new cases in subsequent years, followed by Ijebu 2 and Ijebu 1 (Supplementary Table 1). These three wards, Igboroko 2, Ijebu 2, and Ijebu 1, were identified as the major hotspots of LF in Ondo State from 2018 to 2021.
LF was reported in 12 (66.7%) out of 18 LGAs in Ondo State. Owo LGA had the highest number of confirmed LF (651) incidents and was followed by Akoko Southwest (83) and Ose (77) LGAs (Table 2). Owo and Akoko Southwest LGAs had the largest numbers of wards with confirmed LF, while Ose and Akure South LGAs had nine wards with confirmed LF (Table 2). Figure 2 shows the temporal increase in the number of wards with confirmed LF between 2017 and 2021.
In 2017, 2019, 2020, and 2021, wards with similar LF incidents showed clustering, supported by statistical measures. For 2017, the z-score was 4.798255 with a Moran's Index of 0.862321; for 2019, a z-score of 2.238864 and Moran's Index of 0.24755; for 2020, a z-score of 2.396517 and Moran's Index of 0.20656; for 2021, a z-score of 2.810033 and Moran's Index of 0.322375; and for the years 2017 to 2021, a z-score of 5.131246 and Moran's Index of 0.405904 (Table 3). These measures indicate that the observed clustering patterns were not likely due to chance. However, in 2018, the LF incidents among wards followed a random pattern without statistical significance. The high z-score and low pseudo p-value provided strong evidence of the statistical significance of the calculated Global Moran's I, confirming that wards with similar LF incidents exhibited significant spatial contiguity in Ondo State.
Hotspot of Lassa fever among wards in Ondo State
According to Fichet-Calvet et al. (2014) 8 and Redding et al. (2021)19, LF prevalence in rodents can vary significantly at small geographic scales, such as neighbouring villages, suggesting a localised and discontinuous LF risk. To identify these localised patterns, Local Indicators of spatial autocorrelation were used. In Ondo State, the northeastern section, particularly in Owo town, was identified as the main hotspot for LF incidents. In 2017, eight wards were identified as hotspots, which reduced to six wards in 2018, with new additions and exclusions. Overall, 11 wards consistently appeared as hotspots between 2017 and 2021 (Table 4). The hotspot maps from 2017 to 2021 display the concentration of LF incidents in these areas (Supplementary Figs. 2A–F). The hotspots were predominantly located in Owo LGA, except for Ogbese ward in Akure North LGA. This pattern persisted, with all wards in Owo LGA (except Uso/Emure-Ile) and Ogbese ward in Akure South LGA identified as significant LF hotspots when considering the entire dataset from 2017 to 2021.
Correlates and variations in the LF incidents across ecological and demographical factors in Ondo State
The study utilised the urban and rural classification of wards by Grid3.org to analyse the difference in the number of confirmed LF cases in Ondo State. Between 2017 and 2021, the average number of LF incidents in rural wards (3.85 ± 14.068) was significantly lower than in urban wards (16.95 ± 36.411) (t(201) = 4.289, p = 0.000). The area occupied by buildings in wards with LF incidents (856,774.6609 ± 1,352,574.833) was significantly higher than in wards without LF incidents (284,858.9932 ± 390,088.3419) (t(201) = 4.550, p = 0.000), showing a positive correlation between LF cases and building area (r = 0.467, p = 0.000). Similarly, the number of buildings in wards with LF incidents (5469.94 ± 8119.98) was significantly higher than in wards without LF incidents (2164.84 ± 2676.94) (t(201 = 4.289, p = 0.000), correlating positively with LF cases (r = 0.409, p = 0.000). Additionally, the population in wards with LF incidents (26,771.84 ± 37,788.64) was significantly higher than in wards without LF incidents (11,294.58 ± 14,107.78) (t(201) = 4.213, p = 0.000), with a positive correlation between LF incidents and ward population (r = 0.400, p = 0.000). These findings indicate that higher building area, number of buildings, and population are associated with a higher number of confirmed LF cases.
The average elevation in wards with confirmed LF incidents (299.77 ± 99.69 m) was significantly higher than in wards without LF incidents (213.65 ± 166.87 m) (t(201) = 4.289, p = 0.000), and there was a positive correlation between LF cases and elevation (r = 0.232, p = 0.000). Furthermore, the average Normalised Difference Vegetation Index (NDVI) in wards with confirmed LF incidents (0.328425 ± 0.105014) was significantly lower compared to wards without LF incidents (0.378117 ± 0.056094) (t(201) = 4.399, p = 0.000), with a negative correlation between LF cases and NDVI (r = − 0.254, p = 0.000). Additionally, the average Nighttime Light (NTL) in wards with confirmed LF incidents (0.715723 ± 0.778167) was significantly higher than in wards without LF incidents (0.321713 ± 0.138504) (t(201) = 5.701, p = 0.000), and there was a positive correlation between LF cases and NTL (r = 0.343, p = 0.000). These findings indicate that higher elevation and NTL, as well as lower NDVI, are associated with a higher number of confirmed LF cases.
The average length of roads in wards with confirmed LF incidents (92.96 ± 109.53 km) was significantly higher than in wards without LF incidents (43.19 ± 45.73 km) (t(201) = 4.553, p = 0.000), and there was a positive correlation between LF cases and road length (r = 0.300, p = 0.000). Moreover, the average number of markets in wards with confirmed LF incidents (1.03 ± 1.32) was significantly higher than in wards without LF incidents (0.57 ± 0.78) (t(201) = 3.134, p = 0.001), and there was a positive correlation between LF cases and the number of markets (r = 0.191, p = 0.003). This suggests that as the number of markets increases, there is a slight increase in LF cases. Similarly, the average distance to markets in wards with confirmed LF incidents (4279.62 ± 3176.54 m) was significantly lower than in wards without LF incidents (6537.40 ± 5867.84 m) (t(201) = 2.977, p = 0.001), and there was a negative correlation between LF cases and distance to markets (r = − 0.223, p = 0.001).
The average minimum temperature in wards with confirmed LF incidents (22.05 ± 0.59 °C) was significantly lower compared to wards without LF incidents (22.47 ± 1.08 °C) (t(201) = 3.053, p = 0.000). There was a negative and significant correlation between LF cases and minimum temperature (r = − 0.189, p = 0.003). However, the average maximum temperature did not significantly differ between wards with confirmed LF incidents (31.58 ± 0.33 °C) and wards without LF incidents (31.51 ± 0.35 °C) (t(201) = 1.367, p = 0.506), and there was no significant relationship between LF cases and maximum temperature (r = − 0.064, p = 0.183). On the other hand, the average precipitation in wards with confirmed LF incidents (1546.83 ± 174.27 mm) was significantly lower compared to wards without LF incidents (1833.99 ± 388.64 mm) (t(201) = 5.838, p = 0.000), and there was a negative and significant correlation between LF cases and precipitation (r = − 0.324, p = 0.000).
Figure 3 presents a summary of the relationship between LF cases and number of markets, population, mean elevation, number of buildings, nighttime exposure, minimum temperature, precipitation and vegetation.
Spatial predictors of Lassa fever incidents among wards in Ondo state
Principal Component Analysis (PCA) was used to address high multicollinearity (VIF > 10) among predictor variables. Three uncorrelated factors were extracted, explaining 86.18% of the total variance. The first factor, labeled "Human factor," included variables like buildings, area occupied by buildings, population, and length of roads. The second factor, "Physical factor," comprised average elevation, temperature, and precipitation. The third factor, "Environmental factor," included NDVI and NTL. These factors collectively captured 86.18% of the original predictors' information, with the first factor explaining 34.53%, the second factor 32.78%, and the third factor 17.08% of the variance.
The relationship between the number of confirmed LF cases and the extracted factor scores was examined using Ordinary Least Square (OLS) regression. The three factors accounted for only 15.4% of the variation in LF incidents, assuming constant relationships across the study area. To relax this assumption, Geographically Weighted Regression (GWR) with an optimal bandwidth of 50 nearest neighbors was employed. Multiscale Geographical Weighted Regression (MGWR) was then used to identify LF predictors in each ward.
Supplementary Table 2 presents the results, showing that the R-Square increased from 15.4% in OLS to 67.34% in GWR, and further to 71.90% in MGWR. The Adjusted R-Square coefficient was highest for MGWR, indicating a better model fit. Additionally, MGWR had the lowest Akaikes Information Criterion (AICc) value, suggesting its effectiveness in modeling the spatially varying relationship.
The coefficient estimates from Table 5 reveal that human factors and environmental factors are positively correlated with LF incidences, while physical factors show a negative correlation. Human factors exhibit the strongest positive association with LF incidence and display more spatial variations compared to environmental or physical factors.
Supplementary Table 3 which is model diagnostic coefficients showed that MGWR had slightly lower R-Squared coefficients compared to GWR, but MGWR had a lower AICc (165.17) and higher Adjusted R-Squared (0.5054) than GWR (171.00 and 0.4875), respectively. Therefore, MGWR was selected to model the association between human, physical, and environmental factors and LF incidence across wards in Ondo State.
Table 6 which is summary of explanatory variables and neighborhoods reveals that none of the independent variables operated locally. Human and environmental factors operated regionally, while physical factors operated globally. Human factors had a broader association with LF incidence across wards compared to environmental factors.
In Supplementary Fig. 3A, wards shaded deep brown displayed a strong, significant positive relationship with the number of LF incidents, while some orange-colored wards also had a positive relationship. None of the wards showed a negative relationship with human-related factors. A total of 34 wards exhibited a positive association, with 25 wards having a strong positive relationship. In Supplementary Fig. 3B, no significant association was found between LF incidents and physical factors. Supplementary Fig. 3C showed that environmental factors had a strong, positive association with LF incidents in 23 wards and a significant association in six wards, while Supplementary Fig. 3D displayed a similar pattern for the intercept.
There were 35 wards where a strong, significant positive relationship existed between the number of confirmed LF incidents and human and environmental factors (Supplementary Fig. 3E). This indicates that as the human and environmental factors increased, the number of confirmed LF incidents also increased among the wards in Ondo State.
Predictors of the likelihood of confirmed LF cases among wards in Ondo State
The analysis in Table 7 revealed significant associations between various factors and the number of LF incidents in wards. Nighttime light intensity showed a positive relationship, with each unit increase corresponding to an increase of 3.150 wards with confirmed LF incidents. Wards with high nighttime light intensity had 23.335 times higher odds of having confirmed LF incidents compared to those with low intensity. Longer road lengths were also positively associated with LF incidents, with each unit increase resulting in an increase of 0.012 wards. Wards with longer road lengths had 1.012 times higher odds of having confirmed LF incidents. Higher maximum temperatures were positively correlated with LF incidents, with each unit increase associated with an increase of 2.773 wards. Wards with higher temperatures had 16.006 times higher odds of having confirmed LF incidents. Conversely, higher precipitation was negatively associated with LF incidents, with each unit increase leading to a decrease of 0.006 wards. Wards with higher precipitation had 0.994 lower odds of having confirmed LF incidents.
Discussion
This study addresses the gap in research on LF by focusing on micro-scale analysis at the ward level in Nigeria. Previous studies have predominantly examined LF at regional or national scales, potentially overlooking the intricate interplay of human, physical, and environmental factors that sustain LF incidents. By conducting a comprehensive spatial analysis, this study determined the spatial patterns of LF incidents and identified significant differences in ecological factors between wards with and without LF cases. Furthermore, the study identified the correlates and predictors of LF across wards in Ondo State, Nigeria. Unlike previous research, which primarily considered rainfall, temperature, vegetation, and altitude, this study incorporates additional factors such as population distribution, market numbers, distance from markets, and NTL intensity to assess the likelihood of LF incidents in the study area, expanding the understanding of LF dynamics.
During the study period, there was a notable increase in confirmed LF cases in Ondo State, Nigeria perhaps due to improved surveillance and disease reporting. However, the possible role of poor sanitation, insufficient healthcare infrastructure, limited access to diagnostics, inadequate infection prevention and control practices, and low level of awareness among the general population cannot be discountenanced21,22. With its substantial rural population engaged in activities like farming and hunting, Ondo State faces an elevated risk of LF due to continuous exposure to rodents. Moreover, healthcare facilities in certain areas of Ondo State, like Owo and Ose LGAs, lack essential resources for infection prevention and control23. Media campaigns targeting LF awareness have had limited impact in rural regions, resulting in insufficient knowledge of preventive measures and curative practices24. Similar challenges have been observed in Akwa Ibom, indicating the need for improved health behaviour modifications following health campaigns25.
The occurrence of LF showed significant temporal and spatial clustering in Ondo State, Nigeria. The highest LF incidence was observed in Owo LGA, followed by Akoko Southwest LGA, located northward and adjacent to Owo LGA. Additionally, Ose LGA, situated eastward of Owo LGA, reported many LF cases. These findings support previous studies and suggest the possibility of human-to-human transmission in the contiguous LGAs25,26. LF diagnoses were concentrated in specific localities and wards, particularly in indigenous areas of towns, indicating factors such as poor hygiene, inadequate waste management, limited healthcare access, population density, and poor ventilation as potential contributors to LF epidemics in Ondo State. The maximum clustering of LF cases occurred within a distance of 20,800 m, highlighting the area with the most pronounced spatial processes promoting clustering (Supplementary Plates 1 and 1).
The clustering of LF in traditional areas of cities can be attributed to various factors, including environmental, social, and cultural influences. Environmental factors such as land use and vegetation cover affect the ecology of rodents, which serve as the primary reservoirs for the Lassa virus. Traditional areas often have open spaces used as waste dumpsites, buildings that provide access points for rodents, and less developed infrastructure. Poor household waste management practices and close-knit communities in these areas can contribute to LF clustering. Limited access to healthcare services in traditional areas may lead residents to rely on traditional healers or self-medication. Addressing LF clustering in traditional areas requires a comprehensive understanding of the complex interactions between environmental, social, and cultural factors. Urban ecological factors such as population density, land use patterns, and sanitation conditions play a role in LF incidents. High population density and poor sanitation can create favorable conditions for rodent populations, increasing the risk of LF outbreaks (12, 13, 14, 15). Housing characteristics and domestic practices also influence rodent density, potentially affecting LF transmission to humans27.
Between 2017 and 2021, 10 wards in Owo LGA (Ehinogbe, Idasen, Igboroko 1, Igboroko 2, Ijebu 1, Ijebu 2, Iloro, Ipele, Isaipen, and Iyere) and one ward in Akure North LGA (Ogbese) were identified as LF hotspots. LF incidents in other wards in Akure appeared as outliers without significant hotspots. The observed hotspots in specific wards may be attributed to several factors. Favorable environmental conditions in wards with suitable rodent habitats can facilitate virus transmission. Poor sanitation and waste management practices can serve as breeding grounds for rodents, increasing the risk of human exposure9,10,11. Social and cultural factors, such as traditional food storage practices and communal living arrangements, may also contribute to the clustering of cases in certain areas12,13,14,15. Limited access to healthcare services and delays in diagnosis and treatment can worsen the impact of the disease. Variations in surveillance and reporting practices across communities can affect LF case detection and reporting, leading to apparent hotspots in wards with more rigorous surveillance and reporting systems, particularly in areas with a history of LF prevalence23.
Lassa fever is an endemic viral illness in Nigeria, with a growing prevalence in urban areas due to the expansion of cities, changes in land use, and shifts in population demographics28,29. Higher population densities in urban areas may facilitate human-to-human transmission and impact the distribution of rodent reservoirs. Additionally, environmental conditions in urban areas differ from rural areas, influencing the abundance of virus-carrying rodents29. This study reveals that urban wards in Ondo State had a higher number of LF cases compared to rural wards, in contrast to a study by Gomerep et al. (2022) conducted in Plateau State. These findings suggest a complex relationship between residence and LF incidence, emphasising the need for further investigation12.
Urban indicators, including the number of buildings, population size, presence of markets, proximity to markets, NTL, road length, and lower vegetation (NDVI), are closely linked to higher incidents of LF across wards8,30,31,32,33. These highlight LF as a growing urban health concern in Ondo State12. Redding et al. (2021) similarly found a positive association between LF and urbanisation, specifically measured by built-up areas, which may impact human-rodent interactions19.
Lassa fever incidents were found to be more common in wards with higher elevation, minimal rainfall, and lower minimum temperature19,20. The significant predictors of LF incidents in Ondo State's wards were human factors such as the number of buildings, population size, and road length, along with environmental factors like NDVI and NTL intensity. However, physical factors like average elevation, temperature, and precipitation did not play a significant role in explaining the number of LF incidents. These align with Fichet-Calvet and Rogers' (2009) previous findings, which suggested that neither vegetation nor elevation had a strong predictive value for LF8.
The occurrence of LF incidents in Ondo State's wards is influenced by factors such as NTL intensity, road length, maximum temperature, and precipitation. These findings are consistent with previous studies8 that have shown a negative association between LF cases and minimum temperature. There is evidence suggesting that NTL exposure may affect rodent abundance. Artificial lighting can disrupt rodent behavior, attract them to lit areas for foraging and nesting, and potentially make them more susceptible to Lassa virus infection. Additionally, light pollution's impact on melatonin production, a hormone regulating circadian rhythms and immune response, may further contribute to rodent vulnerability34,35.
The increasing prevalence of exposure to light at night has significant social, ecological, behavioral, and health consequences that are only now becoming apparent36. The presence of artificial light at night disrupts the natural behavior of rodents, attracting them to illuminated areas for foraging and nesting34. Higher levels of NTL exposure in urban areas are associated with increased LF incidence, potentially due to increased rodent abundance and altered behavior patterns34,35. This suggests that NTL exposure may contribute to the urbanisation of LF. However, further research is needed to fully understand the relationship between NTL exposure, rodent abundance, and LF transmission. Road length is believed to be associated with LF incidence, as it influences factors such as changes in land use patterns, human activity, and rodent reservoir distribution27. Roads also facilitate the movement of humans, potentially aiding the spread of the virus across regions and populations.
Lassa fever is a virus that is sensitive to changes in temperature and humidity, and outbreaks have been linked to these conditions. For instance, droughts or extreme weather events can cause rodents to move into urban areas, thereby increasing the risk of human exposure37. The relationship between temperature and LF incidence is complex and may be influenced by other factors, such as rainfall, humidity, and land use patterns. In Sierra Leone, LF incidence shows seasonal fluctuations, with peaks during the dry season and smaller peaks during the rainy season, indicating that climatological factors and agricultural labor patterns may impact rodent-human contact38. Fichet-Calvet and Rogers (2009) demonstrated that recorded LF outbreaks in human populations occurred in areas receiving annual rainfall between 1500 and 3000 mm8. Changes in temperature can affect the immune response of humans and rodents, potentially influencing the severity and outcomes of LF infections39. Temperature plays a crucial role in the transmission and incidence of LF, which is primarily carried by rodents and transmitted to humans through contact with contaminated excreta or urine. There is evidence suggesting that higher temperatures may be associated with increased LF incidence, with the highest incidence observed during the hottest months of the year40. Higher temperatures can enhance the reproductive rates of rodent populations, leading to an increase in the number of infected animals and potentially raising the risk of transmission to humans39. Understanding these temperature-related dynamics can help shape strategies aimed at reducing the urbanisation of LF and mitigating the risk of outbreaks in urban areas.
The major limitation of this study is the use of secondary data which may have limited the scope of interpretation of results. Nevertheless, the remotely sensed secondary data used were the most adequate in a data-scarce environment like Nigeria. Moreover, our findings provided valuable insight into the spatial factors that contributed to the increased incidence of urban LF cases in the study area.
Conclusion
Lassa fever incidence in Ondo State, Nigeria has been on the increase since 2017 with significant clusters noticed within urban rather than rural wards. The clusters were most prominent in the city core where hygiene and access to healthcare facilities are poor, thereby providing a favourable habitat where Mastomys rodents may thrive. Ecological factors such as population density, land use patterns, and sanitation conditions favour increased LF incidence in urban compared to rural wards. Human and environmental factors such as nighttime light intensity and length of roads positively influenced the number of confirmed LF cases in wards in Ondo State. Hence, strategies to prevent and control LF outbreaks in urban areas such as improved sanitation, proper waste management, implementation of effective rodent control measures and public health education are advocated.
Methods
Study area
Ondo State, situated in southwestern Nigeria, shares borders with Ekiti, Kogi, Osun, Ogun, and Edo States. It has an estimated population of approximately 4.6 million people. The people in Ondo State are engaged in diverse occupations, with significant number involved in agriculture, particularly farming and fishing, due to the State's coastal location. The economy of Ondo State is primarily agrarian, with agriculture playing a pivotal role. Moreover, the State is renowned for producing crops such as cocoa, palm oil, yam, maize, and cassava. Ondo State experiences a tropical climate with two distinct seasons. The rainy season spans from April to October, characterised by heavy rainfall, while the dry season lasts from November to March, featuring reduced rainfall and higher temperatures. The State has a well-developed road network that connects its cities, towns, and rural areas. Major roads include the Akure-Owo Road, Akure-Ondo Road, and the Akure-Ilesha Expressway. These roads facilitate transportation within the state and connect it to other states in Nigeria.
Data sources
Hospital records from two specialised healthcare facilities, covering 2017 to 2021, were utilised. However, these records did not include coordinates of patients' house addresses; instead, descriptive addresses were provided. The study assumed the patient's home as the point of contracting and spreading the infection. Google Earth was used to georeference the addresses in the patient’s files because of its comprehensive road and street address data on Ondo State, allowing a near precise location of individual addresses using street names. This approach facilitated accurate mapping and ensured confidentiality of patient identity. The coordinates of all individuals were extracted from Google Earth and mapped using ArcGIS Pro 3.0 software. Since the raw data included the number of individuals with LF symptoms, laboratory confirmed cases were filtered for analysis. All spatial data used in the analysis were projected to the Universal Transverse Mercator (UTM) Zone 31 for precise computation of parameters.
Previous research on LF has primarily focused on national or Local Government Area units, with limited attention to ward-level patterns19,41. In Nigeria, the ward represents the smallest administrative unit. However, the lack of accessible and available ward boundary data has hindered spatial analysis. To address this, the digital administrative ward boundary for Ondo State was downloaded from https://grid3.org/ in ArcGIS Pro compatible shape-file format. The downloaded ward boundary file included additional attributes such as ward area, classification (urban/rural), and unique identifiers. The ward boundary served as the basis for aggregating and summarising all other data related to LF analysis in Ondo State. Consequently, the ward boundary was adopted as the analysis and reporting unit for this study.
In previous studies, researchers have explored the impact of elevation on the distribution of LF in the West African Sub-region8. For this study, the 30-m digital elevation data for Ondo State was downloaded from https://dwtkns.com/srtm30m/ in the form of four tiles. These tiles were then combined to create a seamless elevation dataset (Fig. 4A). Using ArcGIS Pro software, the average elevation for each ward in Ondo State was extracted through the Zonal Statistic as Table function. The extracted mean elevation values were subsequently linked to their respective wards. Additionally, road network data for Ondo State was obtained from https://www.openstreetmap.org/.
(A) Shuttle Radar Topographical Mapping Data Showing Elevation in Ondo State. (B) Population Distribution Data for Ondo State (Worldpop). (C) Normalised Difference Vegetation Index Map of Ondo State. (D) Nighttime Light Image of Ondo State. (E) CHIRPS Precipitation (mm) Data of Ondo State. (F) Minimum Temperature (0c) Distribution in Ondo State. (G) Maximum Temperature (0c) Distribution in Ondo State. (H) Distribution of Road Networks and Markets in Ondo State. (I) Buildings and Road Network in Ondo State. (J) Distance from Market in Ondo State.
Due to the absence of recent census data in Nigeria, the 2021 gridded population data for Ondo State, acquired from the Worldpop database (worldpop.org), was used to analyse the impact of population dynamics on LF incidence. This raster population database provided estimated population figures for each ward, enabling the assessment of population contributions to LF (Fig. 4B).
Mastomys rodents inhabit not only homes but also bushy areas and farmland, which serve as suitable habitats for them. Consequently, more Mastomys are expected to be found in these bushy areas. The Normalised Difference Vegetation Index (NDVI) has been used to assess vegetation levels in various environments30,33. In this study, the NDVI image for Ondo State was obtained from https://land.copernicus.vgt.vito.be/, providing information about the greenness of the environment, scaled between − 1 and ± 1 (Fig. 4C). Additionally, the study explored the potential impact of Nighttime Light (NTL) on Mastomys' nocturnal activities. While NTL has been utilised in previous investigations42,43, its use in understanding the dynamics of LF is still being established. NTL imagery of Ondo State was obtained from https://eogdata.mines.edu/nighttime_light/annual/v20/2021/, based on Version 2.1 produced from monthly cloud-free average radiance44 (Fig. 4D).
The distribution of LF in endemic areas is influenced by climatic variables such as temperature and rainfall. However, integrating climatic data into LF modeling in Nigeria is challenging due to the scarcity and resolution of meteorological stations. To address this, rainfall data from the Climate Hazards Group InfraRed Precipitation with Station data (CHIRPS) was obtained. CHIRPS is a 35-year quasi-global rainfall dataset that combines satellite imagery and in-situ station data to create gridded rainfall time series. Average annual precipitation raster data (Fig. 4E) was downloaded from the CHIRPS website. Additionally, TerraClimate data, which combines high-spatial resolution climatological normals from the WorldClim dataset with time-varying data, was used for maximum (Fig. 4F) and minimum temperatures (Fig. 4G) analysis8,45,46.
Road network data for Ondo State was downloaded from the OpenStreetMap.org (Fig. 4H). Mastomys rodents also frequently inhabit residential houses, where they feed on various food residues and household wastes47,48. Housing data, including building footprints, were downloaded from https://minedbuildings.blob.core.windows.net/africa/nigeria.geojsonl.zip and clipped using the administrative boundary of Ondo State (Fig. 4I). The building footprints were generated from satellite imagery using machine learning and are licensed by Microsoft under the Open Data Commons Open Database License (ODbL).
Mastomys rodents are commonly found in unhygienic food markets due to poor environmental conditions and food residues32,33. Their faecal droppings and urine are often observed in food stores during early mornings. To analyse their spatial distribution, the coordinate locations of verifiable markets in Ondo State were obtained from https://grid3.org/ as a shape-file, and this was transformed to a distance surface to be able to assess the effect of distance from markets on LF incident (Fig. 4J).
Data analysis
LF incidence analysis in this study specifically considered confirmed cases from hospitals and excluded suspected cases. Using ArcGIS Pro 3.0 software, laboratory confirmed LF cases were plotted to visualise the distribution of incidents from 2017 to 2021. Graphical and statistical inference methods were employed to examine LF incident distribution among Ondo State wards. The number of LF cases were aggregated based on the ward boundaries, providing data on the occurrences within each ward between 2017 and 2021.
The temporal change in confirmed LF incidents was analysed using a regression trend line in Microsoft Excel. The spatial pattern of LF incidents from 2017 to 2021 was explored using Average Nearest Neighbour (Rn) analysis. Rn values close to zero indicate clustering, 1 represents randomness, and > 1 represents regularity. Kernel Density Estimation (KDE) was used to identify LF clusters, which is a mathematical process to estimate the probability density of a variable. The KDE of LF incidents was implemented in ArcGIS Pro 3.0.
The study involved computing the number of LF incidents in each administrative ward in Ondo State by overlaying the ward boundary on the LF incidents map. This approach was also applied to extract data from multiple layers, such as elevation, population, precipitation, nighttime light image (NLT), vegetation (NDVI), temperature, market, distance from market, number of buildings, length of roads, and building area coverage. To address the issue of high collinearity among these variables, Principal Component Analysis (PCA) was utilised to reduce the dimensionality and generate uncorrelated variables. The PCA resulted in three factor scores, which were then employed in subsequent regression models including Ordinary Least Square (OLS), Geographical Weighted Regression (GWR), and Multiscale Geographical Weighted Regression (MGWR). GWR was calculated within the MGWR framework to identify the most suitable model for the data. The study acknowledges the potential for multicollinearity in local statistical models and effectively manages it through the implementation of PCA49,50,51.
The study employed the global Moran's I index to examine the spatial pattern of LF among wards in Ondo State. The Moran's I index is a widely used indicator of spatial autocorrelation that assesses the relationship between feature locations (wards) and attribute values (number of LF incidents). The contiguity edges corners option with row standardisation was utilised to investigate the influence of ward contiguity on LF incidents. The Moran's I statistic coefficient ranges from − 1 to + 1, where positive and significant coefficients indicate clustering of similar values, negative and significant coefficients indicate dispersion, and a zero coefficient suggests a random process. The statistical significance of the global Moran's I index was determined using the conditional permutation method with 999 permutations. The Moran's I statistic z-score and p-value provide the statistical significance of the calculated index21,52,53,54.
The study examined the relationship between LF incidents at the ward level and 11 predictor variables using Pearson product-moment correlation. To identify predictors of LF in Ondo State, a multiscale geographical weighted regression (MGWR) was employed, which allows flexibility in modeling spatial phenomena. The preferred model was selected based on highest R-Square and Adjusted R-Square values and lowest Akaike's Information Criteria (AIC). Initially, an Ordinary Least Square (OLS) regression (global model) was developed, assuming constant predictor variables across wards. Subsequently, an MGWR model was created using optimal bandwidths obtained through a golden section search routine. The variables were standardised for comparison, and MGWR maps were generated to visualise predictors of LF across wards49.
The independent sample t-test method was employed to compare variables between wards that reported LF incidents and those that did not. The aim was to determine if there were significant differences in ecological and environmental variables. Subsequently, a binary logistic regression analysis was conducted to identify which urban ecological and environmental variables predict LF incidents in all wards of Ondo State19,25.
Ethical consideration
Approval was received from the Surveillance and Epidemiology Unit of Ondo State Ministry of Health and the University of Ibadan/University College Hospital Institutional Review Board (UI/UCH/22/0305). This study involved secondary data analysis and there was no contact with the patients. Access was granted to the secondary data after all forms of identifiers that could reveal the patient's identity were removed. Appropriate steps were taken to ensure confidentiality and privacy as stated in the Helsinki Declaration55.
Data availability
All the data for this study will be made available on request. Contact for request of data from this study: simeonc5@gmail.com; Brainbox_taiwo@yahoo.com.
References
Richmond, J. K. & Baglole, D. J. Lassa fever: epidemiology, clinical features, and social consequences. BMJ 327(7426), 1271–1275 (2003).
Geisbert, T. W. Predicting outcome and improving treatment for Lassa fever. Lancet Infect Dis. 18, 594–595 (2018).
Mba, S. et al. A description of Lassa Fever mortality during the 2019 outbreak in Nigeria. Int. J Infect. Dis. 101(Suppl 1), 409–410 (2020).
Gibb, R., Moses, L. M., Redding, D. W. & Jones, K. E. Understanding the cryptic nature of Lassa fever in West Africa. Pathog. Glob. Health. 111, 276–288 (2017).
Tambo, E., Adetunde, O.T., Olalubi, O.A. Re-emerging Lassa fever outbreaks in Nigeria: Re-enforcing “One Health” community surveillance and emergency response practice. Infect. Dis. Poverty. 2018.
Asogun, D. A., Günther, S., Akpede, G. O., Ihekweazu, C. & Zumla, A. Lassa fever: Epidemiology, clinical features, diagnosis, management and prevention. Infect. Dis. Clin. North Am. 33, 933–951 (2019).
Kofman, A., Choi, M. J. & Rollin, P. E. Lassa fever in travelers from West Africa, 1969–2016. Emerg. Infect. Dis. 25(2), 236 (2019).
Fichet-Calvet, E. & Rogers, D. J. Risk maps of Lassa fever in West Africa. PLoS Negl. Trop. Dis. 3, e388 (2009).
Dalhat, M. M. et al. Epidemiological trends of Lassa fever in Nigeria, 2018–2021. PLoS One. 17, e0279467 (2022).
Abdullahi, I.N., Anka, A.U., Ghamba, P.E., Onukegbe, N.B., Amadu, D.O., Salami, M.O. Need for preventive and control measures for Lassa fever through the One Health strategic approach. In Proc. Singapore Healthc. SAGE Publications Inc. 190–4 (2020).
Ter Meulen, J. et al. Hunting of peridomestic rodents and consumption of their meat as possible risk factors for rodent-to-human transmission of Lassa virus in the Republic of Guinea. Am. J. Trop. Med. Hyg. https://doi.org/10.4269/ajtmh.1996.55.661 (1996).
Gomerep, S. et al. Epidemiological review of confirmed Lassa fever cases during 2016–2018, in Plateau State, North Central Nigeria. PLOS Glob Public Health 2(6), e0000290. https://doi.org/10.1371/journal.pgph.0000290 (2022).
Gobir, A.A., Ejembi, C.L., Alhaji, A.A., Garba, M.B., Igboanusi, C.J.C., Usman, B., et al. Knowledge of Lassa fever disease and its risk factors among rural people in a Nigerian community. In Proceedings of the 5th African Conference on Emerging Infectious Diseases; 2019 August 7–9; Abuja, Nigeria. 2021. Doi: https://doi.org/10.3390/proceedings2020045009.
Usuwa, I. S. et al. Knowledge and risk perception towards Lassa fever infection among residents of affected communities in Ebonyi State, Nigeria: Implications for risk communication. BMC Public Health. 20(1), 217 (2020).
Mariën, J. et al. Households as hotspots of Lassa fever? Assessing the spatial distribution of Lassa virus-infected rodents in rural villages of Guinea. Emerg. Microbes. Infect. 9(1), 1055–1064. https://doi.org/10.1080/22221751.2020.1766381 (2020).
NCDC. Lassa fever Situation Report Epi Week 21, 22–28. (2023). https://ncdc.gov.ng/themes/common/files/sitreps/c0487b15689b6bc1bc3beddaffd34c30.pdf
NCDC. Lassa fever Situation Report Week 52, 2022. Niger Cent Dis Control Lassa fever Situat Rep. 1, 1–7 (2022). https://ncdc.gov.ng/themes/common/files/sitreps/2fd923c3f16ae99e0ddbfa1ced8a0ae9.pdf
NCDC. Lassa fever Situation Report Week 52, 2021. Lassa fever Situat Rep. 52, 1–6 (2021). https://reliefweb.int/attachments/536d34c9-1173-3a93-a769-ba089f121e18/An%20update%20of%20Lassa%20fever%20outbreak%20in%20Nigeria_251221_52.pdf
Redding, D. W. et al. Geographical drivers and climate-linked dynamics of Lassa fever in Nigeria. Nat. Commun. 12(1), 5759 (2021).
Clegg, J. C. Influence of climate change on the incidence and impact of arenavirus diseases: A speculative assessment. Clin. Microbiol. Infect. 15(6), 504–509 (2009).
Balogun, O. O., Akande, O. W. & Hamer, D. H. Lassa fever: An evolving emergency in West Africa. Am. J. Trop. Med. Hyg. 104(2), 466 (2021).
Abalkhail, A. & Alslamah, T. Institutional factors associated with infection prevention and control practices globally during the infectious pandemics in resource-limited settings. Vaccines 10(11), 1811 (2022).
Ijarotimi, I. T., Ilesanmi, O. S., Aderinwale, A., Abiodun-Adewusi, O. & Okon, I. M. Knowledge of Lassa fever and use of infection prevention and control facilities among health care workers during Lassa fever outbreak in Ondo State, Nigeria. Pan Afr Med J. https://doi.org/10.11604/pamj.2018.30.56.13125 (2018).
Wogu, J. O. Mass media awareness campaign and the prevention of the spread of Lassa fever in the rural communities of Ebonyi State, Nigeria: Impact evaluation. J. Public Health Afr. 9(3) (2018)
Okokhere, P. et al. Clinical and laboratory predictors of Lassa fever outcome in a dedicated treatment facility in Nigeria: A retrospective, observational cohort study. Lancet Infect. Dis. 18(6), 684–695 (2018).
Ilori, E. A. et al. Epidemiologic and clinical features of Lassa fever outbreak in Nigeria, January 1–May 6, 2018. Emerg. Infect. Dis. 25(6), 1066 (2019).
Bonwitt, J. et al. At home with Mastomys and Rattus: Human-rodent interactions and potential for primary transmission of Lassa virus in domestic spaces. Am. J. Trop. Med. Hyg. 96(4), 935 (2017).
Longet, S. et al. Influence of landscape patterns on exposure to Lassa fever virus, Guinea. Emerg. Infect. Dis. 29(2), 304 (2023).
Klitting, R. et al. Predicting the evolution of the Lassa virus endemic area and population at risk over the next decades. Nat. Commun. 13(1), 5596 (2022).
Johansen, B. & Tømmervik, H. The relationship between phytomass, NDVI and vegetation communities on Svalbard. Int. J. Appl. Earth Observ. Geoinf. 27, 20–30 (2014).
Xu, Y., Yang, J. & Chen, Y. NDVI-based vegetation responses to climate change in an arid area of China. Theor. Appl. Climatol. 126, 213–222 (2016).
Karan, L.S., Makenov, M.T., Korneev, M.G., Sacko, N., Boumbaly, S., Bayandin, R.B., et al. Lassa virus in the host rodent mastomys natalensis within urban areas of N’zerekore, Guinea. BioRxiv, 616466 (2019).
Adeke, A. S., Opayele, A. V., Umeokonkwo, C. D., Okedo-Alex, I. N., Usman, A. B., & Sufiyan, M. B. Knowledge and practices of foodstuff traders on Lassa fever and rodent control in Izzi Local Government Area, Ebonyi State, Nigeria. J. Intervent. Epidemiol. Public Health. 4(3), (2021).
Zhang, F. S. et al. Effects of artificial light at night on foraging behavior and vigilance in a nocturnal rodent. Sci. Total Environ. 724, 138271 (2020).
McLay, L. K., Hopkins, J. P., Wong, B. B. M., Candolin, U. & Jones, T. M. What is the available evidence that artificial light at night affects animal behaviour? A systematic map protocol. Envtal. Evid. 8(1), 1–5 (2019).
Navara, K. J. & Nelson, R. J. The dark side of light at night: physiological, epidemiological, and ecological consequences. J. Pineal Res. 43(3), 215–224 (2007).
Patz, J. A. & Olson, S. H. Climate change and health: Global to local influences on disease risk. Ann. Trop. Med. Parasitol. 100(5–6), 535–549 (2006).
Leach, M. et al. Local disease–ecosystem–livelihood dynamics: Reflections from comparative case studies in Africa. Philos. Trans. R. Soc. B Biol. Sci. 372(1725), 20160163 (2017).
Nchom, J. I., Abubakar, A. S., Arimoro, F. O. & Mohammed, B. Y. The role of weather in the spread of Lassa fever in parts of Northern Nigeria. Int. J. Trop Dis. Health 42(23), 33–40 (2021).
Ndayakalah, K. B., Williams, J. J. & Gwade, W. J. An Assessment of rainfall and temperature on seasonal outbreak of Lassa fever in Nigeria for the year 2020. Int. J. Res. Innov. Soc. Sci. 6(4), 629–638 (2022).
Akhmetzhanov, A. R., Asai, Y. & Nishiura, H. Quantifying the seasonal drivers of transmission for Lassa fever in Nigeria. Philos. Trans. R. Soc. B 374(1775), 20180268 (2019).
Huang, Q., Yang, X., Gao, B., Yang, Y. & Zhao, Y. Application of DMSP/OLS nighttime light images: A meta-analysis and a systematic literature review. Remote Sens. 6(8), 6844–6866 (2014).
Jasiński, T. Modeling electricity consumption using nighttime light images and artificial neural networks. Energy. 179, 831–842 (2019).
Elvidge, C. D., Zhizhin, M., Ghosh, T., Fichet-Calvet, E. & Rogers, D. J. Risk maps of Lassa fever in West Africa. PLoS Negl. Trop. Dis. 3(3), e388 (2009).
Mohammed, B. Y., Arimoro, F. O., Abubakar, A. S., & Nchom, J. I. The role of weather in the spread of Lassa fever in parts of Northern Nigeria. Int. J. Trop. Dis. Health. 33–40 (2021).
Bausch, D. G. et al. Lassa fever in Guinea: I. Epidemiology of human disease and clinical observations. Vector Borne Zoonotic Dis. 1(4), 269–281 (2001).
Bonner, P. C. et al. Poor housing quality increases risk of rodent infestation and Lassa fever in refugee camps of Sierra Leone. Am. J. Trop. Med. Hyg. 77(1), 169–175 (2007).
Ilesanmi, O., Omotoso, B., Alele, F. & Adewuyi, P. Awareness of Lassa fever in a rural community in South West Nigeria. J. Commun. Health Res. 4(1), 1–10 (2015).
Oshan, T. M., Smith, J. P. & Fotheringham, A. S. (Targeting the spatial context of obesity determinants via multiscale geographically weighted regression. Int. J. Health Geogr. 19(1), 1–17 (2020).
Davino, C., Romano, R. & Vistocco, D. Handling multicollinearity in quantile regression through the use of principal component regression. METRON 80(2), 153–174 (2022).
Jolliffe, I. T. & Cadima, J. Principal component analysis: a review and recent developments. Philos. Trans. Royal Soc. 374(2065), 20150202 (2016).
Cliff, A. D. & Ord, J. K. Spatial processes: Models and applications (Pion Limited, 1981).
Moran, P. A. Notes on continuous stochastic phenomena. Biometrika 37(1/2), 17–23 (1950).
Ijumulana, J., Ligate, F., Bhattacharya, P., Mtalo, F. & Zhang, C. Spatial analysis and GIS mapping of regional hotspots and potential health risk of fluoride concentrations in groundwater of northern Tanzania. Sci. Total Environ. 735, 139584 (2020).
World Medical Association. World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. Bull World Health Org. 79(4), 373–374 (2001).
Acknowledgements
We acknowledge the support of the West Africa One-Health Consortium supported by the International Development Research Centre (IDRC) Canada.
Author information
Authors and Affiliations
Contributions
S.C. conceived the idea of the study. S.C. and O.J.T. designed the study. T.O., S.C., V.A., E.C., G.F., S.F., and R.A. wrote the initial draft of the manuscript. O.J.T. and A.A. carried out the visualisation and data analyses. S.C., T.O., V.A., E.C., A.O., A.A., D.O., S.O. and O.T. carried out the critical review of the manuscript. All authors approved the final draft of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cadmus, S., Taiwo, O.J., Akinseye, V. et al. Ecological correlates and predictors of Lassa fever incidence in Ondo State, Nigeria 2017–2021: an emerging urban trend. Sci Rep 13, 20855 (2023). https://doi.org/10.1038/s41598-023-47820-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-47820-3
- Springer Nature Limited