Abstract
Intravenous iron is commonly used to treat iron deficiency anemia in non-dialysis chronic kidney disease (ND-CKD). There is a paucity of information on the potential impact of intravenous iron on patient reported outcome measures, functional status and markers of cardiovascular health. As part of the secondary analysis of this double-blind exploratory randomized controlled trial focusing on patients with iron deficiency (+ /− anemia) and ND-CKD (serum ferritin < 200 µg/L or transferrin saturation ≤ 20% and serum ferritin 200–299 µg/L; CKD stages: 3a-5), 26 patients were randomized in a 1:1 ratio to receive ferric derisomaltose or ferric carboxymaltose. Participants received 1000 mg at baseline and 500–1000 mg at one month to achieve iron repletion. Quality of life and fatigue status were assessed using the Short-Form (36) questionnaire and the fatigue severity scale. Functional status was evaluated using the Duke Activity Status Index and the 1-min-sit-to-stand test. Cardiac markers such as NT-proBNP, Troponin T and pulse wave velocity were monitored. Intravenous iron was associated with similar improvements in most domains of the Short-Form (36) questionnaire, fatigue status, and 1-min-sit-to-stand ability increased significantly by the end of the trial in both groups (p < 0.001). Markers of cardiac function remained stable, with no arterial stiffness impact. Longer term studies are required to further evaluate the impact of intravenous iron on quality of life and cardiac safety in patients with ND-CKD.
Similar content being viewed by others
Introduction
Anemia (with/without) iron deficiency is a common complication of non-dialysis dependent chronic kidney disease (ND-CKD). This is associated with an impact on mortality, disease progression and hospitalizations1. Importantly, patients with anemia of CKD have a greater risk for cardiovascular disease2 and frequently report worse health related quality of life outcomes. Indeed as a recent large-scale cross-sectional, prospective cohort study (n = 5,004,957) highlighted, patients with iron deficiency anemia independently correlated with a higher risk of cardiovascular disease3. This has been previously noted in two large cohort studies (n = 1,012,014) focusing on cardiovascular associated hospitalizations in CKD, with a relationship between mortality and hospitalization arising in the presence of functional iron deficiency4,5. This is not surprising as CKD and uremia represents a state of chronic dysregulation of processes including but not restricted to inflammation and oxidative stress, endothelial function, bone metabolism, autonomic balance and volume control6. The degree of injury to these processes can be amplified in the presence of iron deficiency and anemia and may be actually secondary to the extra-hematopoietic functions of iron that include DNA cycle, mitochondrial and non-mitochondrial energy production, neurohormonal abnormalities and decreased muscular oxygen content secondary to reduced myoglobin7. In fact, these extra-hematopoietic properties of iron may be related to the symptoms displayed by patients with iron deficiency anemia in CKD.
Iron deficiency with/without anemia in CKD is associated with a significant burden to the patient. It is characterized by debilitating symptoms such as fatigue, dyspnea, angina, headache and loss of concentration. A recent observational study utilizing data for the CKDOPPS registry (n = 2513, ND-CKD) has aptly demonstrated the association between iron deficiency and physical impact. Focusing primarily on the effect of iron deficiency (transferrin saturation < 20% and ferritin > 300 or < 50 ng/mL) on the mental and physical component of KDQOL-36, they concluded that iron deficiency was associated with worse physical component scores. This scoring disadvantage was maintained irrespective of confounders and was most strongly related to transferrin saturation values8. Moreover, Finkelstein and colleagues in an analysis utilizing the Short -Form (36) questionnaire (SF-36) in patients with CKD and anemia indicated that at baseline the reported scores in all domains had been lower than the normative value, highlighting the impact of the disease process on quality of life9. Iron deficiency has been proposed as key in the symptomatology displayed, as studies suggest that skeletal muscle mitochondrial dysfunction associated with iron depletion may be key in skeletal muscle dysfunction10,11.
Intravenous iron is frequently used in the treatment of iron deficiency anemia in patients with ND-CKD. Despite convincing evidence relative to its efficacy and safety based on meta-analyses, a paucity exists on the impact of intravenous iron on patient-reported outcome measures, functional status and cardiovascular markers12,13. Additionally concerns remain regarding the implications of iron on cardiovascular toxicity, despite the positive results expressed in studies focusing on patients with heart failure in terms of hospitalization, quality of life and functional status14,15,16,17. Given the advent of modern intravenous iron compounds such as ferric carboxymaltose (FCM) and ferric derisomaltose (FDI) which share similar efficacy at equivalent dosing but a distinct differential effect on reductions in serum phosphate, research is required on the topic18,19,20,21.
The “Iron and Phosphaturia – ExplorIRON-CKD” study was primarily designed to explore the differential effect of FDI and FCM on fibroblast growth factor 23 (FGF23) and markers relevant to hypophosphatemia and bone metabolism. Pre-specified secondary outcomes on patient-reported outcome measures, functional status and cardiovascular markers were also collected22. We hypothesized that iron repletion would improve functional status and quality of life, while also investigating any differential effect and cardiac consequences.
Results
Baseline
A total of 26 patients were randomized into the two groups. Fourteen participants received FDI and 12 received FCM. The baseline group results have been previously discussed22. The mean age of the participants was 68 (12.4) years with most of them male. At baseline most patients had absolute iron deficiency (serum ferritin < 100 μg/L and transferrin saturation < 20%) as opposed to functional iron deficiency (61.5% vs. 38.5%). Most participants had advanced CKD with 22 (84.6%) having a CKD stage of 4 or 5 (eGFR < 30 mL/min/1.73 m2). All randomized participants received at least one dose of intravenous iron (1000 mg); 10 patients in the FDI group and 11 in the FCM group received a second infusion. At baseline the two groups were comparable with the exception of age, fatigue severity scale (FSS) and heart failure prevalence, whereby the participants in the FDI group were younger (FDI: 63.4 (12.2) vs. FCM: 73.2 (10.8); p = 0.040), reported a higher FSS score (FDI: 54.5 (47.8–60.3) vs. FCM: 42.0 (24.5–54.5); p = 0.0036) and had a lower prevalence of heart failure (0.026). In addition, a statistically significant difference was noted in terms of QRS interval between the two groups (p = 0.018) (Table 1).
Patient reported outcome measures
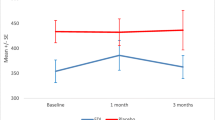
Seven out of eight of the SF-36 domains improved numerically for the total group. Physical function, limitations due to emotional restriction, energy and pain scores demonstrated a significant improvement within the whole cohort (Table 2). No differential effect was noted between the two comparators at each visit, however the significance of the intergroup trends varied. Physical function and energy demonstrated a significant intergroup increase with FDI but not FCM, while for role limitations due to emotional reasons the reverse intergroup trend was observed. No significant difference between the two groups was noted at specific time-points. Fatigue severity scale scores improved in the whole cohort and each group, with a greater but non-significant improvement within the FDI group (supplementary table 1). A significant improvement in fatigue status within the whole cohort and the FDI group was noted based on the FSS Visual Analogue Scale (Baseline: 3.5 (1.8–6.3)/10; 3 months: 6.0 (3.7–7.0)/10; p = 0.027 and Baseline: 3.0 (1.0–7.0)/10; 3 months: 7.0 (3.7–8.7)/10; p = 0.048 respectively) (Fig. 1).
Functional status
There was no trend in Duke Activity Status Index (DASI) score in the whole cohort or individual groups (supplementary table 1). No significant differential effect between the two comparators was detected at any point in the trial. The 1-min-sit-to-stand testscores improved significantly and persistently following intravenous iron (p < 0.001), irrespective of compound administered indicated by the lack of differential significant effect (Fig. 2).
Cardiovascular measurements
Cardiac markers were elevated at baseline compared to reference ranges, both within the whole population and individual groups. NT-proBNP and Troponin T were unchanged in both groups throughout the study (Table 3) (Fig. 3). There was a small increase in NT-proBNP in the FDI group and a small decrease within the FCM group, however these were both non-statistically significant, numerical changes. Electrocardiograms were not significantly affected in either group by intravenous iron, (supplementary table 2). Pulse wave velocity (PWV) and augmentation index improved numerically but not statistically throughout the study with no differential effect noted (Figs. 4 and 5).
Pulse wave velocity. Legend: Line graph summarizing the results of the pulse wave velocity measurements within the study. A numerical improvement was noted in terms of pulse wave velocity in the whole cohort and individual groups, which did not reach statistical significance. No differential effect was noted.
Discussion
The “Iron and Phosphaturia – ExplorIRON-CKD” study has demonstrated a trend for improvement in quality of life and functional status following administration of intravenous iron. A significant improvement in the whole cohort was seen in terms of physical function, energy, emotional role limitation, and pain using the results of the SF-36, alongside a significant improvement in functional status and fatigue as delineated by 1-min-sit-to-stand test and FSS visual analogue scale respectively. Intravenous iron did not lead to any detrimental effect on surrogate markers of cardiac strain and cardiomyocite damage, whilst some beneficial result was noted on vascular function/arterial stiffness.
As suggested by Strauss and Auerbach, fatigue resolution is best monitored through a combination that allows the investigation of a continuum between vitality and fatigue23. The combination of SF-36 and FSS has allowed us to assess this continuum through a fatigue-specific questionnaire alongside a multifaceted assessment of various domains that are associated with elements of fatigue. At baseline, the SF-36 scores noted were below the 50% cut-off value, indicating the impact that CKD has on patients. In addition, it was below the previously reported values by Finkelstein and colleagues, potentially highlighting the implications of iron deficiency anemia in this patient group9. This was also the case in FSS24. Most domains relevant to physical function improved significantly in both the whole-cohort and the FDI subgroup, suggesting that iron repletion (but not necessarily anemia resolution) can be associated with improved physical function. This was also the case for the energy (vitality) domain of the SF-36. The absence of significance within the FCM group may be explained by the recent findings commented in PHOSPHARE-IBD, where a slower, lesser improvement in FACIT-fatigue was seen where FCM was administered compared to FDI despite similar changes in hemoglobin with equivalent doses18. Analysis of this study suggests that this, with analysis suggesting that this could be phosphate dependent, where fatigue and muscle weakness are common symptoms of hyposphatemia18. These trends were replicated in the FSS visual analogue scale, with a significant improvement within the whole cohort and the FDI group. Nonetheless, no significant differential effect was witnessed—this may be partially due to the extent of kidney dysfunction in the randomized population limiting the potential for hyperphosphaturic hypophosphatemia. Indeed no hypophosphatemia as per protocol was detected (serum phosphate concentration < 0.65 mmol/L). However, it is important to aknowledge that the participants receiving FCM were significantly older than those receiving FDI with comorbidities such as heart failure which may have impacted their response to intravenous iron supplementation.
Functional status improved as indicated through the 1-min-sit-to-stand test scores. Similar to quality of life, the baseline scores achieved were lower than the ones previously reported in literature relevant to CKD, potentially noting the effect of iron deficiency anemia25. A statistically significant improvement throughout the trial was seen in all groups, with no differential effect potentially underlining the effect of iron repletion irrespective of compound used. It is also important to note that the scores achieved in both groups and the whole cohort were above the minimal detectable change previously described by Wilkinson and colleagues25. These results appear to complement the previously completed “Iron and the Heart” multicenter randomized controlled trial which noted an increase in the 6-min-walking test distance in participants with ND-CKD and iron deficiency that received FDI26. The trend observed in the aforesaid study was not significant; this could be secondary to the small sample size and the high baseline function of the randomized participants. The DASI did not exhibit any significant trends during the study, but a beneficial signal was seen with FCM. This may be due to the group dynamics. The FDI group was composed by three participants requiring hemodialysis during the study, a procedure associated with worse DASI scores than pre-dialysis27.
Cardiovascular markers including NT-proBNP and Troponin T remained stable throughout the trial and was similar in both groups. Previous studies related to ND-CKD and iron deficiency have noted a non-significant decrease in NT-proBNP following intravenous iron administration (1000 mg FDI) (baseline: 422.5 (881.9) pg/mL to 1-month: 242.5 (209.1) pg/mL)26. Iron sucrose has been previously associated with a significant decrease of NT-proBNP in patients with ND-CKD and heart failure28. In the present study, this appeared to be the case in patients that received FCM but not FDI. This, nonetheless, may be related to the sample of the patients within FDI (greater number of participants in the FDI group requiring dialysis) and not signify a differential effect. Outside interference such as increased diuretic therapy in certain patients cannot be excluded. Troponin T was not affected by the administration of intravenous iron and that potentially suggests a degree of cardiac safety in the use of high dose iron administration. In the absence of comparative/observational in-vivo studies, animal models of kidney injury and iron deficiency have demonstrated improved cardiac mitochondrial function with attenuation of oxidative/nitrosative stress secondary to a pre-conditioned/primed oxidant status existing in iron deficiency and kidney disease11,29,30. Indeed, previous results relevant to FDI have suggested that the use of high dose intravenous iron (1000 mg) is not associated with increased oxidative stress or inflammation, that are pathological processes directly implicated in cardiac toxicity31,32. Any analysis of electrocardiographical parameters and extrapolation of the results is limited by the presence of implantable cardiac devices and arrhythmias in a number of the participants. Similar to the HOMeaFers randomized controlled study, comparing FDI and FCM in individuals with iron deficiency no differential effect was witnessed between the two intravenous iron preparations33.
The results of the present trial are also in agreement with previously published evidence pertaining to vascular function following administration of intravenous iron in patients with ND-CKD. Such trends highlight a potential mechanistic safety despite concerns regarding oxidative stress and endothelial damage. A previous pooled analysis has noted no worsening in PWV(cf) or Augmentation Index following administration of 1000 mg FDI in ND-CKD34. Similarly, intravenous iron sucrose (300 mg) administered in patients on peritoneal dialysis was not associated with increased vascular reactivity35.
This trial had several limitations including a small number of participants with no ethnic minority groups and therefore generalizability of the results is not possible. In addition, the possibility of errors upon completion of the questionnaires provided cannot be excluded. The small numbers may represent the reason behind the lack of any differential effect noted. In addition, certain differences between the two groups at baseline may account for the lack of significant findings of the DASI scores or the differential effect in terms of FSS scores. Indeed participants in the FCM arm were significantly older with prolonged QRS complexes and greater incidence of heart failure. It is also possible that detection of significance within the FCM group was hindered by its small size. Moreover, the trial took place throughout the COVID-19 pandemic which was characterized by higher psychological stress in patients with CKD36,37. Given that secondary outcomes focused on patient reported outcomes, the absence of placebo arm may be also considered as a limitation; placebo response has previously been documented as an important phenomenon in patient reported outcome measures and this is something not addressed in the present study38.
Nevertheless, the “Iron and Phosphaturia – ExplorIRON-CKD” was a double-blind randomized controlled trial, providing valuable results for direct future research. The results are suggestive of improvement in functional status and quality of life following intravenous iron infusion. To our knowledge, this is the first trial to have provided evidence supporting the notion of intravenous iron administration leading to 1-min-sit-to-stand test improvement in patients with ND-CKD and iron deficiency anemia. In addition, this exploratory study is the first to compare to two modern intravenous iron in patients with advanced kidney dysfunction (median eGFR 18 mL/min/1.73 m2), displaying results that complement the efficacy of supplementation beyond hematological markers, into aspects of quality of life. Moreover, the results of cardiovascular variables support the notion of mechanistic safety and potential improvement of certain processes (such as arterial stiffness) following alleviation of iron deficiency. Additionally, it was created based on research priorities and targets delineated both by patient groups and large research groups39,40.
As part of the secondary outcome analysis of the current trial, no significant differential effect was noted in quality of life measures, functional status and cardiovascular biomarkers. An improvement was witnessed in most domains of the Short-Form (36) questionnaire, fatigue status, and 1-min-sit-to-stand ability increased significantly by the end of the trial in whole population. These results provide further evidence on the neutrality of modern intravenous iron compounds in terms of cardio and myotoxicity and potential benefit in terms of functional status. This is important clinically, as CKD and iron deficiency anemia are considered risk factors to cardiovascular disease, with prior concerns rending iron as a pro-oxidant substance. These concerns may explain previous publication results suggesting persistent under-dosing in patients with CKD requiring intravenous iron, therefore potentially limiting the benefit that could be obtained by accurate supplementation41. Some particular signals were noted (larger improvement in aspects relevant to fatigue, physical function) with FDI that may be related to the cascade effects of FCM on the metabolism of FGF23. Further studies are required to elicit whether any differential effect exists secondary to intravenous iron compound used in this patient group, as has been demonstrated in patients with inflammatory bowel disease. Moreover, further research could help identify, via stratification, whether a differential response in terms of functional status and quality of life exists dependent on background kidney function, anemia status, and mode of iron supplementation. The eagerly awaited “Iron and Muscle” trial could provide further answers as to the potential improvement in exercise capacity of patients with iron deficiency and ND-CKD, a signal alluded in the present trial42.
Methods
The “Iron and Phosphaturia – ExplorIRON-CKD” (EudraCT number: 2019-004370-26) took place according to the Declaration of Helsinki and the Good Clinical Practice guidelines. Clinical trial authorization was provided by the Health Research Authority and the Medicines and Healthcare Products Regulatory Agency of the United Kingdom. The trial received the favorable opinion of the Research Ethics Committee Leeds West (20/YH/0005). All participants were enrolled only after full informed consent was taken.
The baseline results and methodology of the present study have been previously published22. “Iron and Phosphaturia – ExplorIRON-CKD” was a double blind randomized exploratory study taking place in a tertiary university hospital in the United Kingdom, primarily investigating the potential differential effect of two modern intravenous iron preparations on FGF23 and markers of bone metabolism. As a secondary outcome, the study explored the impact of high dose intravenous iron supplementation on the quality of life, functional status and markers of cardiovascular status of the participants. Patients with iron deficiency with/without anemia (serum ferritin < 200 μg/L and/or transferrin saturation < 20% and serum ferritin 200–299 μg/L) and stable ND-CKD (3a-5; estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2) were randomized in a 1:1 ratio to receive 1000 mg of FDI or FCM. A repeat infusion of either 500/1000 mg (dependent on baseline hematinic parameters and weight) was administered a month later.
Patient reported outcome measures
Patient reported outcome measures were monitored at baseline, 1 month following first infusion, 2 months following first infusion and 3 months following first infusion. The SF-36 version 1.0 questionnaire and the FSS were used to assess quality of life. The SF-36 is a 36 question based survey that assesses eight variable including vitality, physical functioning, bodily pain, physical role functioning, social functioning, emotional functioning, mental health and general health43. Higher SF-36 questionnaire scores indicate better quality of life. The FSS is a nine-item questionnaire that is scored on a seven-point Likert scale, specifically assessing elements pertaining to fatigue. These include motivation, impact of exercise, fatigability, interference with life, impact on normal function, fatigue frequency, impact on activities and fatigue interference with social life44. The FSS can be scored as a total and on a visual analogue scale. The greater the FSS, the greater the fatigue experienced by the patient, whilst a lower visual analogue scale score is associated with worse fatigue.
Functional status
This was monitored through the DASI and the 1-min-sit-to-stand test. Measurements took place on baseline, 1 month, 2 months and 3 months following initial infusion. The DASI is a 12-item questionnaire developed to non-invasively evaluate the functional capacity of participants (VO2 peak), a surrogate measure of aerobic fitness. The scores were converted and are reported as metabolic cost of task (MET). The DASI score is considered to have high reliability in CKD and advocated by the Renal Association UK for the estimation of functional capacity45. The 1-min-sit-to-stand test was utilized as a measure of lower extremity, exercise capacity and fatigability46. Participants were asked to sit and stand up as many times as possible within one minute from a chair of a standard height (46 cm with no armrests) with arms crossed across chest. Only full motion (complete sitting) was counted and recorded. All participants were informed that they could stop as necessary. The 1-min-sit-to-stand test has been previously demonstrated as valid and reliable in CKD27.
Cardiovascular measurements
N-terminal pro-hormone BNP (NT-proBNP) and Troponin T were measured, in addition to electrocardiography. Cardiac strain was non-invasively monitored at baseline, 1 month, 2 months and 3 months following initial administration through NT-proBNP. The Roche cobas e411 analyzer was used alongside the Elecsys proBNP II immunoassay (Roche Diagnostics, Risch-Rotkreuz, Switzerland). Troponin T, a widely accepted marker of ischemia and myocyte damage, was measured at baseline, prior to first infusion and 30 min following first infusion, 1–2 days following first infusion, 2 weeks following first infusion, 1 month (before and after second administration), 1–2 days following second infusion, 2 months and 3 months following initial infusion. The Elecsys Troponin T assay was used (Roche Diagnostics, Risch-Rotkreuz, Switzerland. Electrocardiography was performed at baseline, 1–2 days following first infusion, 1 month, 2 months and 3 months after initial administration to identify any arrhythmias, with measurements of PR, QRS and QTc intervals. Aortic stiffness assessed using measurement of pulse wave velocity carotid-femoral (PWVcf) and Augmentation Index using the Enverdis® Vascular Explorer (Enverdis GmbH Medical Solutions, Jena, Germany). Measurements took place at baseline, 1 month, 2 months and 3 months following initial intravenous iron infusion. The American Heart Association advocates the use of PWVcf as a surrogate marker of arterial stiffness47.
Statistical analysis
All randomized participants were included in the statistical analysis based on an intention-to-treat approach. Whole cohort and subgroup analysis took place to identify any signals of differential effects. Categorical data is summarized as mean (standard deviation (SD)) or median (interquartile range (IQR)), whilst categorical data is presented as number (%). The Shapiro–Wilk test was used to assess normality of distribution. The independent T-test and Mann–Whitney U tests were used for between group analyses (i.e. FCM vs FDI). The Skillings-Mack test was performed to identify any longitudinal trends within groups. Given the small sample, adjustments for multiple comparisons were not performed. A statistical software package was used for the analysis of data (IBM SPSS Statistics Version 26, IBM Corp. 2019). Statistical significance was deduced at p-value < 0.05.
Data availability
The data associated with the paper are not publicly available but are available from the corresponding author on reasonable request with the relevant permissions and agreement of the Research and Development Department of the Hull University Teaching Hospitals NHS Trust that served as the sponsor for the trial. Further enquiries can be directed to the corresponding author.
References:
Palaka, E., Grandy, S., Van Haalen, H., McEwan, P. & Darlington, O. The impact of CKD anaemia on patients: Incidence, risk factors, and clinical outcomes—A systematic literature review. Int. J. Nephrol. 2020, (2020).
Babitt, J. L. & Lin, H. Y. Mechanisms of anemia in CKD. J. Am. Soc. Nephrol. 23, 1631–1634 (2012).
Farrington, D. K. et al. Anemia prevalence, type, and associated risks in a cohort of 5.0 million insured patients in the United States by level of kidney function. Am. J. Kidney Diseases 81, 201-209.e1 (2022).
Awan, A. et al. Prevalence, correlates and outcomes of absolute and functional iron deficiency anemia in nondialysis-dependent chronic kidney disease. Nephrol. Dial. Transpl. 36, 129–136 (2021).
Cho, M. E. et al. Heart failure hospitalization risk associated with iron status in veterans with CKD. Clin. J. Am. Soc. Nephrol. 16, 522–531 (2021).
Kassianides, X., Hazara, A. & Bhandari, S. Cardiac complications in end-stage renal disease: A shared care challenge. Br. J. Cardiol. https://doi.org/10.5837/bjc.2020.010 (2020).
Wish, J. B. et al. Iron deficiency in CKD without concomitant anemia. Kidney Int. Rep. (2021).
Guedes, M. et al. Serum biomarkers of iron stores are associated with worse physical health-related quality of life in nondialysis-dependent chronic kidney disease patients with or without anemia. Nephrol. Dialysis Transpl. 36, 1694–1703 (2021).
Finkelstein, F. O., van Nooten, F., Wiklund, I., Trundell, D. & Cella, D. Measurement properties of the Short Form-36 (SF-36) and the Functional Assessment of Cancer Therapy - Anemia (FACT-An) in patients with anemia associated with chronic kidney disease. Health Qual. Life. Outcomes 16, (2018).
Gamboa, J. L. et al. Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiol. Rep. 4, 1–12 (2016).
Bhandari, S. Impact of intravenous iron on cardiac and skeletal oxidative stress and cardiac mitochondrial function in experimental uraemia chronic kidney disease. Front. Biosci. - Landmark 26, 442–464 (2021).
Shepshelovich, D., Rozen-Zvi, B., Avni, T., Gafter, U. & Gafter-Gvili, A. Intravenous versus oral iron supplementation for the treatment of anemia in CKD: An updated systematic review and meta-analysis. Am. J. Kidney Diseases 68, 677–690 (2016).
O’Lone, E. L. et al. Parenteral versus oral iron therapy for adults and children with chronic kidney disease. Cochrane Database System. Rev. https://doi.org/10.1002/14651858.CD007857.PUB3 (2019).
Osman, M. et al. A meta-analysis of intravenous iron therapy for patients with iron deficiency and heart failure. Am. J. Cardiol. 141, 152 (2021).
Jankowska, E. A. et al. The effect of intravenous ferric carboxymaltose on health-related quality of life in iron-deficient patients with acute heart failure: The results of the AFFIRM-AHF study. Eur. Heart J. 42, 3011 (2021).
Kalra, P. R. et al. Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN): An investigator-initiated, prospective, randomised, open-label, blinded-endpoint trial. Lancet 17, 2199–2209 (2022).
Bhandari, S. et al. Safety and efficacy of iron isomaltoside 1000/ferric derisomaltose versus iron sucrose in patients with chronic kidney disease: the FERWON-NEPHRO randomized, open-label, comparative trial. Nephrol. Dialysis Transpl. https://doi.org/10.1093/ndt/gfaa011 (2020).
Zoller, H. et al. Hypophosphataemia following ferric derisomaltose and ferric carboxymaltose in patients with iron deficiency anaemia due to inflammatory bowel disease (PHOSPHARE-IBD): A randomised clinical trial. Gut 0, gutjnl-2022-327897 (2022).
Kassianides, X. & Bhandari, S. Hypophosphataemia, fibroblast growth factor 23 and third-generation intravenous iron compounds: A narrative review. Drugs Context 10, 1–29 (2021).
Schaefer, B. et al. Hypophosphatemia after treatment of iron deficiency with intravenous ferric carboxymaltose or iron isomaltoside - A systematic review and meta-analysis. Br. J. Clin. Pharmacol. 87, 2256–2273 (2020).
Wolf, M. et al. Effects of iron isomaltoside vs ferric carboxymaltose on hypophosphatemia in iron-deficiency anemia: Two randomized clinical trials. JAMA – J. Am. Med. Assoc. 323, 432–443 (2020).
Kassianides, X. & Bhandari, S. Methodology and baseline data of a comparative exploratory double-blinded randomized study of intravenous iron on fibroblast growth factor 23 and phosphate in chronic kidney disease. Kidney Blood Press Res. 48, 151–164 (2023).
Strauss, W. E. & Auerbach, M. Health-related quality of life in patients with iron deficiency anemia: impact of treatment with intravenous iron. Patient Relat. Outcome Meas. 9, 285 (2018).
Bonner, A., Caltabiano, M. & Berlund, L. Quality of life, fatigue, and activity in Australians with chronic kidney disease: a longitudinal study. Nurs. Health Sci. 15, 360–367 (2013).
Wilkinson, T. J. et al. Test-retest reliability, validation, and ‘minimal detectable change’ scores for frequently reported tests of objective physical function in patients with non-dialysis chronic kidney disease. Physiother. Theory Pract. 35, 565–576 (2019).
Bhandari, S., Allgar, V., Lamplugh, A., Macdougall, I. & Kalra, P. A. A multicentre prospective double blinded randomised controlled trial of intravenous iron (ferric Derisomaltose (FDI)) in Iron deficient but not anaemic patients with chronic kidney disease on functional status. BMC Nephrol. 22, (2021).
Hadjiioannou, I. et al. Test-retest reliability for physical function measures in patients with chronic kidney disease. J. Ren. Care 46, 25–34 (2020).
Toblli, J. E., Lombraña, A., Duarte, P. & di Gennaro, F. Intravenous iron reduces NT-pro-brain natriuretic peptide in anemic patients with chronic heart failure and renal insufficiency. J. Am. Coll. Cardiol. 50, 1657–1665 (2007).
Zager, R. A., Johnson, A. C. M. & Frostad, K. B. Combined iron sucrose and protoporphyrin treatment protects against ischemic and toxin-mediated acute renal failure. Kidney Int. 90, 67 (2016).
Toblli, J. E., Cao, G., Rivas, C., Giani, J. F. & Dominici, F. P. Intravenous iron sucrose reverses anemia-induced cardiac remodeling, prevents myocardial fibrosis, and improves cardiac function by attenuating oxidative/nitrosative stress and inflammation. Int. J. Cardiol. 212, 84–91 (2016).
Kassianides, X., Gordon, A., Sturmey, R. & Bhandari, S. The comparative effects of intravenous iron on oxidative stress and inflammation in patients with chronic kidney disease and iron deficiency: A randomized controlled pilot study. Kidney Res. Clin. Pract. 40, 89–98 (2021).
Kassianides, X., Allgar, V., Macdougall, I. C., Kalra, P. A. & Bhandari, S. Analysis of oxidative stress, inflammation and endothelial function following intravenous iron in chronic kidney disease in the Iron and Heart Trial. Sci. Rep. 12, 1–8 (2022).
Emrich, I. E. et al. Hypophosphatemia after high-dose iron repletion with ferric carboxymaltose and ferric derisomaltose - The randomized controlled HOMe aFers study. BMC Med. 18, (2020).
Kassianides, X., White, S. & Bhandari, S. Markers of oxidative stress, inflammation and endothelial function following high-dose intravenous iron in patients with non-dialysis-dependent chronic kidney disease-a pooled analysis. Int. J. Mol. Sci. 23, (2022).
Schaller, G. et al. Intravenous iron increases labile serum iron but does not impair forearm blood flow reactivity in dialysis patients. Kidney Int. 68, 2814–2822 (2005).
Lee, K.-M. et al. The higher the CKD stage, the higher the psychological stress in patients with CKD during COVID-19 pandemic. J. Clin. Med. 11, 4776 (2022).
Voorend, C. G. N. et al. Impact of the COVID-19 pandemic on symptoms of anxiety and depression and health-related quality of life in older patients with chronic kidney disease. BMC Geriatr 21, 1–10 (2021).
Kluzek, S., Dean, B. & Wartolowska, K. A. Patient-reported outcome measures (PROMs) as proof of treatment efficacy. BMJ 27, (2022).
Urquhart-Secord, R. et al. Patient and caregiver priorities for outcomes in hemodialysis: An international nominal group technique study. Am. J. Kidney Dis. 68, 444–454 (2016).
Kidney Research UK. Research strategy - Kidney Research UK. https://www.kidneyresearchuk.org/research/research-strategy/ (2022).
Kalra, P. A. et al. NIMO-CKD-UK: A real-world, observational study of iron isomaltoside in patients with iron deficiency anaemia and chronic kidney disease. BMC Nephrol. 21, 1–10 (2020).
Greenwood, S. A. et al. The effect of intravenous iron supplementation on exercise capacity in iron-deficient but not anaemic patients with chronic kidney disease: Study design and baseline data for a multicentre prospective double-blind randomised controlled trial. BMC Nephrol. 23, (2022).
Ware, J. E. & Sherbourne, C. D. The MOS 36-item short-form health survey (Sf-36): I conceptual framework and item selection. Med. Care 30, 473–483 (1992).
Krupp, L. B., Larocca, N. G., Muir Nash, J. & Steinberg, A. D. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch. Neurol. 46, 1121–1123 (1989).
Dukes Activity Status Index (DASI) - The Kidney Quality Improvement Partnership. Think Kidneys https://www.thinkkidneys.nhs.uk/kquip/hub/dukes-activity-status-index-dasi/ (2017).
Bohannon, R. W. & Crouch, R. 1-Minute sit-to-stand test: Systematic review of procedures, performance and clinimetric properties. J. Cardiopulm. Rehabil. Prevent. 39, 2–8. Preprint at https://doi.org/10.1097/HCR.0000000000000336 (2019).
Townsend, R. R. et al. Recommendations for improving and standardizing vascular research on arterial stiffness: A scientific statement from the American Heart Association. Hypertension 66, 698–722 (2015).
Acknowledgements
We would like to thank all the participants to the trial, Vikki Jubb, Monaliza Delizo, and Sylvia Madrid-Wallingham at the Hull University Teaching Hospitals NHS Trust, Victoras Kassianides at the University of Glasgow, and the Research and Development team (James Illingworth) of the Hull University Teaching Hospitals NHS Trust for their help and input throughout the trial.
Funding
The “Iron and Phosphaturia – ExplorIRON-CKD” trial received funding through unrestricted competitive grants from Pharmacosmos A/S and Kidney Research Yorkshire. The funders had no role in trial design, data collection and analysis, decision to publish or preparation of manuscript.
Author information
Authors and Affiliations
Contributions
X.K. (first author) participated in all aspects of the study including funding for the trial, protocol design, trial execution, data collection and analysis, and manuscript preparation. S.B. (senior author) obtained funding for the trial, critically contributed to protocol design, provided supervision and revision of the manuscript. The order of authorship has been a joint decision of the co-authors based on substantial contribution to conception and design, execution, analysis and interpretation of data.
Corresponding author
Ethics declarations
Competing interests
XK has no conflicts of interest. SB has received honorarium for lectures, attended expert opinion committees and received educational funds to attend international nephrology meetings from Pharmacosmos A/S and Vifor Pharma.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kassianides, X., Bhandari, S. Patient reported outcome measures and cardiovascular outcomes following high dose modern intravenous iron in non-dialysis dependent chronic kidney disease: secondary analysis of ExplorIRON-CKD. Sci Rep 13, 18401 (2023). https://doi.org/10.1038/s41598-023-44578-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44578-6
- Springer Nature Limited