Abstract
Within home ranges, animals repeatedly visit certain areas. Recursive movement patterns are widespread throughout the animal kingdom, but are rarely considered when developing resource selection models. We examined how behavioral state-dependent recursive movements influenced reource selection of eastern wild turkey (Meleagris gallopavo silvestris) broods as they aged from day 1 to 28. Because broods become more plastic in behaviors once they begin roosting off the ground, we separated data into broods that were ground roosting (1–13 days) and tree roosting (14–28 days). We used Hidden Markov Models to identify 2 behavioral states (restricted and mobile). We extracted state-specific recursive movements based on states and specific step lengths, which we integrated into a step selection analysis to evaluate resource selection. We found that in a restricted state, ground roosting broods spent less time in areas of mixed pine-hardwoods and more time in areas with greater vegetation density. Tree roosting broods revisited areas closer to shrub/scrub landcover types, and areas with greater vegetation density. Tree roosting broods also spent less time near mixed pine-hardwoods, while spending more time in areas with greater vegetation density. We found that in a mobile state, ground roosting broods revisited areas closer to secondary roads and mixed pine-hardwoods, but farther from hardwoods. Tree roosting broods revisited areas farther from secondary roads and with greater vegetation density. Tree roosting broods also spent more time in areas closer to pine. Resource selection varied depending on behavioral state and recursive movements. However, revisitation and residence time impacted selection in both ground and tree roosting broods. Our findings highlight the need to consider how behaviors can influence movement decisions and ultimately resource selection.
Similar content being viewed by others
Introduction
Spatial distribution of resources such as forage, water, and shelter influences how animals move across the landscape1,2. Animals collect resources from patches within home ranges through repeated visitation3,4,5. By revisiting areas that contain resources, animals minimize risks associated with navigating unfamiliar areas which may improve survival and fitness6,7,8. Recursive movement patterns are returns to previously visited areas, and are a widespread phenomenon in the animal kingdom9. Understanding how recursive movement strategies influence behavioral processes is important in examining areas for resource acquisition9,10,11,12.
Recursive movement strategies have been documented in a variety of species, typically occurring when individuals are locating resources within a heterogeneous landscape13,14,15. Path recursions, defined as nonrandom movements in which animals repeatedly return to resource rich locations9, is a profitable foraging strategy that enables resources to recover2,13. Although generality of recursive movements is recognized in how animals select habitat, resource selection analyses often associate movements to availability of resources within individual home ranges16,17,18. Including recursion information within a resource selection framework can potentially identify one mechanism driving behavioral decision-making and resource availability within a home range19,20.
Behavioral decisions can influence the fitness of a species via resource selection21,22,23. Changes in behavior and movement patterns may suggest a response to variation in habitat conditions, such as where an individual goes to acquire resources or avoid risk24,25. Identifying behavioral patterns in resource selection could provide insight as to where individuals choose to travel versus forage26,27,28. Failing to incorporate such behavioral patterns in resource selection models can result in biased results, including misidentifying where animals travel and misallocating limits in foraging resources29,30,31.
Wild turkeys (Meleagris gallopavo) are considered habitat generalists; however, habitat requirements of adults differ from their precocial offspring32. During the first 28 days post-hatch, wild turkey poults experience high mortality risk33,34,35 as they are unable to thermoregulate and must find high quality foraging patches rich in arthropods36,37,38,39. Wild turkey poults grow rapidly during the first month, developing an ability to fly within 2 weeks post-hatch40,41,42. Increasing maneuverability facilitates behavioral changes and can alter foraging strategies to exploit areas within their range more efficiently40,41. Hence, resource needs and selection of poults may be more specialized immediately post-hatch and become more generalized as they age35,39.
Wild turkey broods are thought to revisit profitable areas32,43, but whether there are patterns in revisitations remains unknown. Knowledge of revisitation timing to specific sites across the landscape could provide insight to drivers of the selection processes used by broods44. Therefore, we examined how behavioral state-dependent recursive movements influenced resource selection as broods aged. Our objective was to quantify differences in recursive movements and resource selection of ground versus tree-roosting broods. We used movement behaviors to infer a behavioral state (mobile and restricted) to account for differences in resource selection. We hypothesized that broods would exhibit differential resource selection across behavioral states. We predicted that as broods aged, they would become more plastic in resource selection, but continue to exhibit consistency in recursive movements.
Methods
Capture and handling
We used rocket nets to capture wild turkeys from January-March of 2014–2021 (For details on study sites refer to Supplementary Information). We aged captured individuals based on presence of barring on the ninth and tenth primary feathers, and identified sex of each individual by the coloration of the breast feathers42. We banded each bird with an aluminum rivet leg band (National Band and Tag Company, Newport, Kentucky; female size = 8, male size = 9) and radio-tagged each individual with a backpack-style GPS-VHF transmitter45 produced by Biotrack Ltd. (Wareham, Dorset, UK). We programmed transmitters to record 1 GPS location nightly (23:58:58) and hourly GPS locations from 0500 to 2000 (Standard Time according to the appropriate time zones) until the battery died or the unit was recovered46. Each transmitter had a mortality switch that was programmed to activate after > 23 h of no movement. We released turkeys immediately at the capture location after processing.
Nest and brood monitoring
We located wild turkeys ≥ 2 times per week using a 3-element handheld Yagi antenna and receiver to monitor survival based on the presence of a mortality signal, general movements of individuals across their ranges, and nesting activity. We remotely downloaded GPS locations from each turkey ≥ 1 time per week. In ArcGIS 10.8 (Environment Systems Research Institute, Redlands, California, USA), we spatially projected GPS locations to identify nest locations by determining when a female’s locations became concentrated, which represented the onset of incubation47,48. When GPS locations indicated nest termination, we located the nest site to determine if hatching had occurred49,50,51.
Following methods outlined by Chamberlain et al.35, we monitored brooding females until brood failure or 28 days after hatch, as most brood mortality occurs during this period33. We located females that hatched successfully every 2–3 days post-hatch via VHF to conduct brood surveys, and considered a brood to be present if ≥ 1 poult was seen or heard with the female35. If we detected a brooding female on the ground prior to sunrise less than 14 days post-hatch, we assumed she was still with a brood as brooding females typically begin tree roosting with poults 14 days post-hatch32,34,40. Hence, if we were able to detect a brood during the night, we did not disturb them during the day. Likewise, if we detected a brooding female roosted in a tree prior to 14 days post-hatch and could not detect poults, we assumed the brood was lost. We performed brood surveys up to 28 days after hatch or until we failed to detect any poults during 2 consecutive attempts, at which point we assumed the brood was lost. We defined brood success as the proportion of broods with ≥ 1 poult surviving to 28 days post-hatch35.
GPS data processing
We processed and cleaned the GPS data by removing fix locations that had dilution of precision values (DOP) > 7. We excluded from analyses females that successfully hatched a nest but were never visually confirmed to have poults. We then removed roosting locations (1 point collected at midnight, 0500 and 0600) as we expected broods would rarely revisit roost sites35,52, and our interest was in behaviors most likely to be associated with foraging, loafing, or traveling.
Behavioral analysis
We fit a Hidden Markov Model (HMM;53) to define movement trajectories into behavioral states based on step lengths and turning angles (Fig. 1). We modeled step lengths using a gamma distribution and turning angles using a Von Mises distribution53. We defined initial parameter values for 3 states: a stationary movement state with small step lengths (gamma distribution with a mean of 27 m and standard deviation of 27 m) and uniform turning angles (Von Mises distribution with a mean of π and a concentration of 0.1), a restricted movement state with small/moderate step lengths (gamma distribution with a mean of 150 m and standard deviation of 150 m) and uniform turning angles (Von Mises distribution with a mean of 2.5 and a concentration of 0.5), and a travelling movement state with large step lengths (gamma distribution with a mean of 400 m and a standard deviation of 1000 m) and directed turning angles (Von Mises distribution with a mean of 0.001 and a concentration of 0.99). We used the Viterbi algorithm to assign each step to the most likely behavioral state based on results of the HMM54. We conducted our analysis in R (v.4.1.0; R Core Team, 2022) using package momentuHMM55.
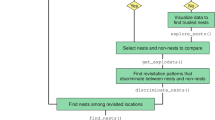
Conceptual framework for evaluating resource selection as a response to behavioral dependent recursive movements. This approach relies on standardizing movement data (1) that are used within a hidden Markov model to classify behavioral states (blue: mobile, orange: restrictive; 2). We then used each behavioral state to identify recursive movements based on a diameter appropriate to each behavioral state (star represents the GPS location of interest; 3) and created 2 raster layers representing revisit and residence time (4). We identified used versus available points based on step length and turn angle (5) and extracted environmental and recursive movement covariates (6). Finally, are data was fitted within a step selection function (7) to quantify in resource selection as broods aged.
Recursive analysis
Following Bracis et al.44, we calculated the revisit rate (hereafter, revisitation) as the sum of visits to previously visited locations as follows. We first assigned a unique identification to each female GPS location for the duration of known brooding. To quantify behavioral-specific revisitation, we buffered GPS locations using the mean step-length from our HMMs for restricted and mobile movements (90-m radius and 250-m radius) to identify the area likely used by each brood each hour (Fig. 1). We considered revisits to be GPS locations that fell within a spatial buffer for any previous day for the entire period the brood was monitored. Additionally, we calculated residence time, defined as the total elapsed time between successive GPS locations within the circular buffer for all visits during the observed observation period for each brood (i.e., up to 28 days), and return time, defined as the amount of time (days)between visits using the R package recurse44. For the recursion analyses, we evaluated brooding females independently of one another.
To incorporate the effect of recursive movements on resource selection, we used a modified Brownian bridge approach, the Biased Random Bridge kernel utilization56,57. We produced a 30-m by 30-m raster to represent behavioral-specific recursive movements for each individual (Fig. 1). We buffered GPS locations identified from our HMMs as restricted or mobile, using either the 90-m or 250-m radius as noted above. We created a 2-dimensional utilization distribution of each individual’s trajectory that represented the relative number of revisits made to each location58. We also assessed the time that a female spent in each area, which provided a biologically relevant measure of intensity of use. Specifically, we used Biased Random Bridge kernel utilization distributions based on residence time to evaluate intensity of use. We converted each behavioral-specific recursion map to a continuous value between 0 and 100, where 0 identified areas that were most strongly associated with the recursive behavior, and values around 100 identified areas not associated with recursive behaviors. We created recursive movement maps using the R package adehabitatHR59.
Environmental covariates
We examined resource selection in relation to a set of environmental covariates relevant to ecology of brooding female wild turkeys. We obtained year-specific, 30-m resolution spatial data on landcover from the Cropland Data Layer (Cropscape) provided by the National Agricultural Statistics Service (National Agricultural Statistics Service 2015). We recoded and combined landcover in R to create 7 unique landcover types (water, pine forest, hardwood forest, mixed pine-hardwood forest, open treeless areas, shrub/scrub, and infrastructure). We then calculated the Euclidean distance in ArcMap 10.8 (Esri, Redlands, CA, USA) to get the distance a GPS location was located from each landcover type. We used 30‐m resolution imagery from United States Geological Survey (USGS) Landsat–8 Operational Land Imager to compute a normalized difference vegetation index (NDVI) in ArcMap 10.8 (Esri, Redlands, CA, USA) as an index of vegetation density60. Measurements of NDVI allowed us to understand sparseness of vegetation, which has been shown to influence maneuverability, concealment, and foraging ability for poults36,41. We used the USGS National Transportation Dataset (https://nationalmap.gov/transport.html) and information provided by the Department of Defense to obtain secondary road layers.
Habitat and model selection
Before analysis, we scaled and centered all variables, so that we could compare effect sizes of variables within individuals on each respective study sites61. We tested for correlation among all continuous predictor variables using Pearson’s correlation and none were highly correlated (correlation coefficient > 0.7). We used a step selection function (SSF;62,63) to assess resource selection, where available habitat associated with a given female location was conditional on where the individual occurred at the time of the previous GPS location. We considered a used point as the GPS location of a female, whereas available points were 100 locations that were theoretically available for selection by that female during the hour the GPS location was recorded. We generated available locations using the amt package in R64. To assess temporal resource selection and recursive movement behaviors, we separated data into 2 periods based on whether females with broods were ground (day 1–13) or tree roosting (day 14–28). For each behavioral state (restricted or mobile), we then parameterized 6 models separately. Within each model, we included the logarithm of step length and the cosine of the turning angle as covariates to account for the underlying movement process65. Furthermore, we parameterized a landcover model, which included only landcover and secondary roads. We parameterized 2 models, one of which was based on number of revisits only, and the other based on only residence time. Finally, we parameterized 2 models that contained all covariates and which interacted with either number of revisits or residence time (composite model; Table 1).
We used mixed conditional Poisson regression models with stratum-specific intercepts to estimate resource selection63. To account for variability among individuals within our models, we included random slope for each covariate for each unique individual66. We did not include random slopes for each interaction of landcover and recursive movement as models failed to converge due to quasi-complete separation. We fitted the SSF using the Poisson formulation where the stratum-specific random intercept variance was fixed to a large value to avoid shrinkage, following Muff et al.63. We used the R package glmmTMB to conduct the step selection analysis67.
We used second-order Akaike's Information Criteria (AICc) to assess the amount of support for the different candidate models68,69. We calculated ΔAICc values between the AICc value for candidate model i and the lowest-ranked AIC value. We also calculated Akaike's weights (wi) for each model. We then calculated parameter estimates and their standard errors for all covariates in models within 2 ΔAICc units of the lowest-ranked AIC value. To assess how well our SSF models explained the data, we used area under the receiver-operating characteristic curves (AUC) calculated with the pROC package70. An AUC value of 0.5 indicated the model provided estimates of no better than random predictions but values greater than 0.7 indicated a better model fit with more accurate predictions.
Ethical approval
All methods were performed in accordance with the relevant guidelines including turkey capture, handling, and marking procedures which were approved by the Institutional Animal Care and Use Committee at the University of Georgia (Protocol #A2014 06008Y1A0, A343701, A2016 04-001-R1, A2019 01-025-R2, and A2020 06-018-R1) and the Louisiana State University Agricultural Center (Protocol #A2014-013, A2015-07 and A2018-13).
Results
We captured and radio‐marked 663 female wild turkeys during 2014–2021. We monitored 692 nest attempts, 147 (21.2%) of which successfully hatched. We censored data from 10 broods that were presumably lost during or within hours of hatching, as we were not able to visually document poult presence via brood surveys. We censored an additional 26 broods due to GPS failure. Hence, we used 111 broods in our analyses, which we visually monitored until brood failure or 28 days after hatch. Of these 111 broods, 36 (32%) survived to 28 days post‐hatch. After removing roosting locations, we used 33,819 GPS locations to use for subsequent analyses.
Behavioral classifications
From our HMM, the step length distribution had an estimated mean of 13.3 m (95% CI = 13.2–13.4 m) and standard deviation of 10.1 m (95% CI = 10.0–10.2 m) for the stationary state, an estimated mean of 92.2 m (95% CI = 90.7–93.8 m) and standard deviation of 74.1 m (95% CI = 72.7–75.5 m) for the restricted state, and an estimated mean of 249.1 m (95% CI = 245.7–252.4 m) and standard deviation of 175.9 m (95% CI = 175.0–177.2 m) for the mobile state (Fig. 2). The turning angle distribution had an estimated mean of 3.1 (95% CI = 3.1–3.2) and concentration parameter of 0.7 (95% CI = 0.7–0.8) for the stationary state, a mean of 2.4 (95% CI = 0.7–3.1) and concentration parameter of 0.01 (95% CI = − 0.01 to 0.02) for the restricted state, and a mean of − 0.1 (95% CI = − 0.04 to 0.01) and concentration parameter of 0.5 (95% CI = 0.5–0.6) for the mobile state (Fig. 2). For subsequent analyses, we combined the stationary and restricted states as they were both associated with relatively shorter distances moved and sharper turn angles characteristic of foraging bouts. From the HMM, we considered 84.5% of movements restricted whereas 15.5% were mobile.
Step length and turning angle distributions for movements of eastern wild turkey (Meleagris gallopavo silvestris) broods across the southeastern United States during 2014–2021. The black curves depict distributions based on parameter values estimated with a Hidden Markov Model, which are overlaid on histograms of the raw data.
Recursive movements and behavioral dependent resource selection
On average, broods revisited previous locations 9.2 times (SD = 8.1, range = 0–58 visits; Fig. 3). Mean residence time was 43.2 h (SD = 47.3 h, range = 0.1–564.0 h; Fig. 4), whereas the return time averaged 1.4 days (SD = 2.2, range = 0–25.9 days; Fig. 5).
Boxplots of daily revisitation events by each behavioral state (mobile and restricted) and density plots showing the distribution of the data for eastern wild turkey (Meleagris gallopavo silvestris) broods across the southeastern United States during 2014–2021. The dashed black line in the density plot is the mean of the data.
Boxplots of daily residence time by each behavioral state (mobile and restricted) and density plots showing the distribution of the data for eastern wild turkey (Meleagris gallopavo silvestris) broods across the southeastern United States during 2014–2021. The dashed black line in the density plot is the mean of the data.
The composite models that included both recursive movements and landcover performed better than those based exclusively on landcover or recursive movements (Table 2). Within each model broods revisited and exhibited increased residence time regardless of landcover (Figs. 6, 7). Ground roosting broods in a restricted state spent less time in mixed pine-hardwoods (β = 0.04, 95% CI = 0.00–0.07) and more time at locations with greater vegetation density (β = 0.08, 95% CI = 0.04–0.1). Ground roosting broods selected locations closer to secondary roads (β = − 0.33, 95% CI = − 0.59 to − 0.07) but did not revisit or spend time in these areas. Ground roosting broods selected areas farther from pine forest (β = 0.1, 95% CI = 0.04–0.2).
Coefficient plot depicting habitat selection of eastern wild turkey (Meleagris gallopavo silvestris) broods across the southeastern United States during 2014–2021 while in a restricted behavioral state. The left plot refers to revisit composite model while the right corresponds to the residence composite model. The whiskers depict 95% confidence intervals around mean estimates.
Coefficient plot depicting habitat selection of eastern wild turkey (Meleagris gallopavo silvestris) broods across the southeastern United States during 2014–2021 while in a mobile behavioral state. The left plot refers to revisit composite model while the right corresponds to the residence composite model. The whiskers depict 95% confidence intervals around mean estimates.
When in a mobile state, ground roosting broods in a mobile state revisited and spent more time at locations closer to secondary roads (β = − 0.2, 95% CI = − 0.4 to − 0.1), and mixed pine-hardwoods (β = − 0.1, 95% CI = − 0.2 to − 0.02), but farther from hardwoods (β = 0.1, 95% CI = 0.02–0.21; Fig. 6). Tree roosting broods revisited areas closer to secondary roads (β = − 0.1, 95% CI = − 0.2 to − 0.03), and shrub/scrub (β = − 0.05, 95% CI = − 0.1 to − 0.01), and with greater vegetation density (β = 0.09, 95% CI = 0.05–0.1) while in a restricted state. Likewise, tree roosting broods in a restricted state spent less time near mixed pine-hardwoods (β = 0.05, 95% CI = 0.005–0.1), but more time in areas with greater vegetation density (β = 0.05, 95% CI = 0.02–0.09). Tree roosting broods in a restricted state selected areas closer to hardwoods (β = − 0.1, 95% CI = − 0.3 to − 0.003) and shrub/scrub (β = − 0.1, 95% CI = − 0.2 to − 0.03), regardless of revisitation and residence time.
When in a mobile state, tree roosting broods revisited areas with greater vegetation density (β = 0.1, 95% CI = 0.007–0.2) while in a mobile state. Likewise, tree roosting broods spent more time in areas closer to pine forest (β = − 0.1, 95% CI = − 0.2 to − 0.04). Tree roosting broods in a mobile state selected areas closer to shrub/scrub (β = − 0.2, 95% CI = − 0.4 to − 0.07), regardless of revisitation and residence time.
Discussion
We found that resource selection of wild turkey broods varied depending on behavioral state and recursive movements. Further, we noted that resource selection differed for broods that roosted on the ground (1–14 days old) versus those that roosted off the ground (15–28 days old), although broods exhibited consistent recursive movements regardless of their age. Our approach allowed us to integrate movement behaviors into an improved understanding of resource selection9,58,71. Our SSF indicated that incorporating recursive movements with landcover improved model fit relative to standard SSF approaches, which typically only consider habitat variables and disregard movement behaviors that could influence selection72,73. Incorporating behavioral states that have potential to influence animal movements and decision-making can increase our ability to understand species movement ecology74.
Our behavioral analysis identified a restricted state characterized by shorter step lengths and less concentrated turning angles, and a mobile state characterized by longer step lengths and turning angles concentrated around zero, which was not surprising given similar findings in contemporary literature22,75. We observed that broods spent most of their time in a restricted rather than mobile state, consistent with Chamberlain et al.35. Restricted movements are often characterized as area-restricted search, loafing, or foraging behaviors76,77, whereas mobile movements are those such as walking, which represent directional movements away from a patch or along travel corridors28,30. We offer that ignoring behavioral states and how they influence movement could lead to misinterpretation of resource selection models. For instance, mobile movements occur at a much larger spatial scale than restricted movements as individuals are covering more area, and within our analysis we defined availability by step lengths of each state, making selection more representative of the behavior22,62. Overall, our findings indicate that recursive movements occur in each behavioral state, and resource selection differs by behavioral state whether individuals were ground or tree roosting.
Recursive movements are common across wildlife species9. We found recursive movements to be important on both a behavioral and temporal scale during brooding. Brooding females are faced with the challenge of finding quality foraging opportunities near vegetative cover that provides concealment78,79. Our results support the idea that wild turkey broods increased residence time at locations and develop behaviors that reflect area-restricted searching35,43,80. Area-restricted foraging presumably allows broods to limit movements and space use, which may positively influence foraging success and reduce predation risk81,82,83. Our results also indicate that recursive movements occur while broods are in a mobile state, suggesting broods were moving through familiar areas. Recursive movements to areas previously visited increases environmental predictability for individuals, which may increase fitness as individuals familiarize themselves with resources on the landscape84. Overall, our results suggest that broods were returning to and spending more time in specific locations, presumably to areas which increase maneuverability and foraging opportunities.
When animals are moving from one resource patch to the next, they may exhibit differential resource selection85,86. We observed differences in resource selection between broods that were ground versus tree roosting when they were in a mobile state. When in a mobile state, ground roosting broods were more likely to be closer to secondary roads and mixed pine-hardwoods, while avoiding areas closer to hardwoods. As broods aged and began to roost in trees, they selected for pine forest, shrub/scrub, and areas with increasing vegetation density. Traveling and feeding rates of brooding galliforms are impacted by the ability of individuals to maneuver through ground cover36,87. Thus, behavioral strategies in relation to vegetation composition is critical to brooding individuals and will depend on morphological development32,88. The differences we observed in resource selection as broods aged provide evidence that selection becomes more plastic as broods age.
In precocial birds, resource requirements and degree of resource specialization may vary due to body size that can influence mobility, thermoregulation, foraging, and responses to predation risk89,90. Our findings demonstrate that accounting for underlying behavior and temporal scales as broods age may change assessments of resource selection. For many species, broods are inextricably linked to early successional vegetation communities that offer quality foraging opportunities with reduced predation risk88,91,92,93. We observed that broods in a restricted behavioral state (i.e., foraging, loafing) selected secondary roads and areas with increased vegetation density during the first 14 days of life when they roosted on the ground. Conversely, after broods reached 14 days of age and began roosting in trees, they selected areas closer to shrub/scrub and hardwood landcover types, and areas with increased vegetation density when they were in a restricted behavioral state. Hence, broods exhibited rapid changes in behavioral plasticity as they aged, which would contribute to the temporal changes in resource selection we observed, and parallel similar observations in contemporary literature on wild turkey broods35,39. Increasing behavioral plasticity as broods age has been reported in other gallinaceous species, in that as broods age their diet breadth88,94,95 and mobility87 change, allowing them to exploit more profitable patches within their ranges.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Pyke, G. H. Optimal foraging theory: A critical review. Annu. Rev. Ecol. Syst. 15, 523–575 (1984).
Owen-Smith, N., Fryxell, J. & Merrill, E. Foraging theory upscaled: The behavioural ecology of herbivore movement. Philos. Trans. R. Soc. B Biol. Sci. 365(1550), 2267–2278 (2010).
Makino, T. T. & Sakai, S. Findings on spatial foraging patterns of bumblebees (Bombus ignitus) from a bee-tracking experiment in a net cage. Behav. Ecol. Sociobiol. 56(5), 155–163 (2004).
Van Moorter, B. et al. Memory keeps you at home: A mechanistic model for home range emergence. Oikos 118(5), 641–652 (2009).
Van Moorter, B., Rolandsen, C. M., Basille, M. & Gaillard, J. M. Movement is the glue connecting home ranges and habitat selection. J. Anim. Ecol. 85(1), 21–31 (2016).
Börger, L., Dalziel, B. D. & Fryxell, J. M. Are there general mechanisms of animal home range behaviour? A review and prospects for future research. Ecol. Lett. 11(6), 637–650 (2008).
Piper, W. H. Making habitat selection more “familiar”: A review. Behav. Ecol. Sociobiol. 65(7), 1329–1351 (2011).
Merkle, J., Fortin, D. & Morales, J. M. A memory-based foraging tactic reveals an adaptive mechanism for restricted space use. Ecol. Lett. 17(8), 924–931 (2014).
Berger-Tal, O. & Bar-David, S. Recursive movement patterns: Review and synthesis across species. Ecosphere 6(9), 1–12 (2015).
Ohashi, K. & Thomson, J. D. Efficient harvesting of renewing resources. Behav. Ecol. 16(3), 592–605 (2005).
Berger-Tal, O. & Avgar, T. The glass is half-full: Overestimating the quality of a novel environment is advantageous. PLoS ONE 7(4), e34578 (2012).
Riotte-Lambert, L., Benhamou, S. & Chamaillé-Jammes, S. Periodicity analysis of movement recursions. J. Theor. Biol. 317, 238–243 (2013).
Bar-David, S. et al. Methods for assessing movement path recursion with application to African buffalo in South Africa. Ecology 90(9), 2467–2479 (2009).
English, M. et al. Foraging site recursion by forest elephants Elephas maximus borneensis. Curr. Zool. 60(4), 551–559 (2014).
Bista, D. et al. Space use, interaction and recursion in a solitary specialized herbivore: A red panda case study. Endanger. Species Res. 47, 131–143 (2022).
Boyce, M. S. & McDonald, L. L. Relating populations to habitats using resource selection functions. Trends Ecol. Evol. 14(7), 268–272 (1999).
Boyce, M. S., Vernier, P. R., Nielsen, S. E. & Schmiegelow, F. K. Evaluating resource selection functions. Ecol. Model. 157(2–3), 281–300 (2002).
Manly, B., McDonald, L., Thomas, D., McDonald, T. & Erickson, W. Resource Selection by Animals: Statistical Analysis and Design for Field Studies 2nd edn. (Kluwer Academic Publishers, 2002).
Buderman, F. E., Hooten, M. B., Alldredge, M. W., Hanks, E. M. & Ivan, J. S. Time-varying predatory behavior is primary predictor of fine-scale movement of wildland-urban cougars. Mov. Ecol. 6(1), 1–16 (2018).
McKeown, B., Walton, Z. & Willebrand, T. Does recursive use of resource locations shape a home range? Exploring the red fox’s cognitive map. Wildl. Biol. 2020(1), 1–9 (2020).
Togunov, R. R., Derocher, A. E. & Lunn, N. J. Windscapes and olfactory foraging in a large carnivore. Sci. Rep. 7(1), 1–10 (2017).
Picardi, S. et al. Behavioural state-dependent habitat selection and implications for animal translocations. J. Appl. Ecol. 59(2), 624–635 (2022).
Whittington, J., Hebblewhite, M., Baron, R. W., Ford, A. T. & Paczkowski, J. Towns and trails drive carnivore movement behaviour, resource selection, and connectivity. Mov. Ecol. 10(1), 1–18 (2022).
Creel, S. & Christianson, D. Relationships between direct predation and risk effects. Trends Ecol. Evol. 23(4), 194–201 (2008).
Buchholz, R. et al. Behavioural research priorities for the study of animal response to climate change. Anim. Behav. 150, 127–137 (2019).
Collier, B.A. & Chamberlain, M.J. Redirecting research for wild turkeys using global positioning system transmitters. In National Wild Turkey Symposium Vol. 10, 81–92.
Wilson, R. R., Gilbert-Norton, L. & Gese, E. M. Beyond use versus availability: Behaviour-explicit resource selection. Wildl. Biol. 18(4), 424–430 (2012).
Ferreira, E. M. et al. Assessing behaviour states of a forest carnivore in a road-dominated landscape using Hidden Markov Models. Nat. Conserv. 47, 155–175 (2022).
Roever, C. L., Van Aarde, R. J. & Leggett, K. Functional connectivity within conservation networks: Delineating corridors for African elephants. Biol. Conserv. 157, 128–135 (2013).
Abrahms, B. et al. Lessons from integrating behaviour and resource selection: Activity-specific responses of African wild dogs to roads. Anim. Conserv. 19(3), 247–255 (2016).
Abrahms, B. et al. Does wildlife resource selection accurately inform corridor conservation?. J. Appl. Ecol. 54(2), 412–422 (2017).
Healy, W. M. Behavior. In The Wild Turkey: Biology and Management (ed. Dickson, J. G.) 46–65 (Stackpole Books, 1992).
Hubbard, M. W., Garner, D. L. & Klaas, E. E. Wild turkey poult survival in southcentral Iowa. J. Wildl. Manag. 63(1), 199–203 (1999).
Spears, B. L. et al. Habitat use and survival of preflight wild turkey broods. J. Wildl. Manag. 71(1), 69–81 (2007).
Chamberlain, M. J., Cohen, B. S., Bakner, N. W. & Collier, B. A. Behavior and movement of wild turkey broods. J. Wildl. Manag. 84(6), 1139–1152 (2020).
Healy, W. M. Turkey poult feeding activity, invertebrate abundance, and vegetation structure. J. Wildl. Manag. 49(2), 466–472 (1985).
Lafon, N. W., Norman, G. W., Jeffreys, J. C., Steffen, D. E. & Fell, R. D. Forest clearings management: Insects and vegetation for wild turkey broods. Proc. Southeastern Assoc. Fish Wildl. Agencies 55, 547–559 (2001).
Backs, S.E., & Bledsoe, L.W. Invertebrate abundance and vegetative structure in forest openings. In National Wild Turkey Symposium, Vol. 10, 51–63 (2011).
Nelson, S. D. et al. Fine-scale resource selection and behavioral tradeoffs of eastern wild turkey broods. J. Wildl. Manag. 86(5), e22222 (2022).
Barwick, L. H., Austin, D. H. & Williams, L. Roosting of young turkey broods during summer in Florida. Proc. Southeastern Assoc. Fish Wildl. Agencies 25, 231–243 (1970).
Williams, L., Austin, D., Peoples, T. & Phillips, R. Observations on movement, behavior, and development of turkey broods. Wild turkey management: current problems and programs. (University of Missouri Press, 1973).
Pelham, P. H. & Dickson, J. G. Physical characteristics. In The Wild Turkey: Biology and Management (ed. Dickson, J. G.) 32–45 (Stackpole Books, 1992).
Bakner, N. W., Cohen, B. S., Collier, B. A. & Chamberlain, M. J. Recursive movements of eastern wild turkey broods in the southeastern United States. Wildl. Soc. Bull. 46(2), e1274 (2022).
Bracis, C., Bildstein, K. L. & Mueller, T. Revisitation analysis uncovers spatio-temporal patterns in animal movement data. Ecography 41(11), 1801–1811 (2018).
Guthrie, J. D. et al. Evaluation of a global positioning system backpack transmitter for wild turkey research. J. Wildl. Manag. 75(3), 539–547 (2011).
Cohen, B. S., Prebyl, T. J., Collier, B. A. & Chamberlain, M. J. Home range estimator method and GPS sampling schedule affect habitat selection inferences for wild turkeys. Wildl. Soc. Bull. 42(1), 150–159 (2018).
Conley, M., Oetgen, J., Barrow, J., Chamberlain, M., Skow, K., & Collier, B. Habitat selection, incubation, and incubation recess ranges of nesting female Rio Grande wild turkeys in Texas. In National Wild Turkey Symposium, Vol. 11, 117–126 (2015).
Bakner, N. W., Schofield, L. R., Cedotal, C., Chamberlain, M. J. & Collier, B. A. Incubation recess behaviors influence nest survival of wild turkeys. Ecol. Evol. 9(24), 14053–14065 (2019).
Conley, M. D., Yeldell, N. A., Chamberlain, M. J. & Collier, B. A. Do movement behaviors identify reproductive habitat sampling for wild turkeys?. Ecol. Evol. 6(19), 7103–7112 (2016).
Yeldell, N. A., Cohen, B. S., Little, A. R., Collier, B. A. & Chamberlain, M. J. Nest site selection and nest survival of eastern wild turkeys in a pyric landscape. J. Wildl. Manag. 81(6), 1073–1083 (2017).
Yeldell, N. A., Cohen, B. S., Prebyl, T. J., Collier, B. A. & Chamberlain, M. J. Prescribed fire influences habitat selection of female eastern wild turkeys. J. Wildl. Manag. 81(7), 1287–1297 (2017).
Hillestad, H. O. & Speake, D. W. Activities of wild turkey hens and poults as influenced by habitat. Proc. Southeastern Assoc. Game Fish Comm. 24, 244–251 (1970).
Langrock, R. et al. Flexible and practical modeling of animal telemetry data: Hidden Markov models and extensions. Ecology 93(11), 2336–2342 (2012).
Zucchini, W., MacDonald, I. L. & Langrock, R. Hidden Markov Models for Time Series: An Introduction Using R 2nd edn. (CRC Press, 2017).
McClintock, B. T. & Michelot, T. momentuHMM: R package for generalized hidden Markov models of animal movement. Methods Ecol. Evol. 9(6), 1518–1530 (2018).
Benhamou, S. Dynamic approach to space and habitat use based on biased random bridges. PLoS ONE 6(1), e14592 (2011).
Benhamou, S. & Riotte-Lambert, L. Beyond the utilization distribution: Identifying home range areas that are intensively exploited or repeatedly visited. Ecol. Model. 227, 112–116 (2012).
Oliveira-Santos, L. G. R., Forester, J. D., Piovezan, U., Tomas, W. M. & Fernandez, F. A. S. Incorporating animal spatial memory in step selection functions. J. Anim. Ecol. 85(2), 516–524 (2016).
Calenge, C. The package “adehabitat” for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 197(3–4), 516–519 (2006).
Wakefield, C. T. et al. Hunting activity effects on roost selection by male wild turkeys. J. Wildl. Manag. 84(3), 458–467 (2020).
Schielzeth, H. Simple means to improve the interpretability of regression coefficients. Methods Ecol. Evol. 1(2), 103–113 (2010).
Avgar, T., Potts, J. R., Lewis, M. A. & Boyce, M. S. Integrated step selection analysis: Bridging the gap between resource selection and animal movement. Methods Ecol. Evol. 7(5), 619–630 (2016).
Muff, S., Signer, J. & Fieberg, J. Accounting for individual-specific variation in habitat-selection studies: Efficient estimation of mixed-effects models using Bayesian or frequentist computation. J. Anim. Ecol. 89(1), 80–92 (2020).
Signer, J., Fieberg, J. & Avgar, T. Animal movement tools (amt): R package for managing tracking data and conducting habitat selection analyses. Ecol. Evol. 9(2), 880–890 (2019).
Forester, J. D., Im, H. K. & Rathouz, P. J. Accounting for animal movement in estimation of resource selection functions: Sampling and data analysis. Ecology 90(12), 3554–3565 (2009).
Duchesne, T., Fortin, D. & Courbin, N. Mixed conditional logistic regression for habitat selection studies. J. Anim. Ecol. 79(3), 548–555 (2010).
Magnusson, A. et al. glmmTMB: Generalized linear mixed models using template model builder. R package version 0.1.3. Retrieved from https://github.com/glmmTMB (2017).
Akaike, H. Information theory and an extension of the maximum likelihood principle. Proc. Int. Symp. Inf. Theory 2, 267–281 (1973).
Anderson, D. R. & Burnham, K. P. Avoiding pitfalls when using information-theoretic methods. J. Wildl. Manag. 66(3), 912–918 (2002).
Zipkin, E. F., Grant, E. H. C. & Fagan, W. F. Evaluating the predictive abilities of community occupancy models using AUC while accounting for imperfect detection. Ecol. Appl. 22(7), 1962–1972 (2012).
Fagan, W. F. et al. Spatial memory and animal movement. Ecol. Lett. 16(10), 1316–1329 (2013).
Fortin, D. et al. Wolves influence elk movements: Behavior shapes a trophic cascade in Yellowstone National Park. Ecology 86(5), 1320–1330 (2005).
Merkle, J. A. et al. Spatial memory shapes migration and its benefits: Evidence from a large herbivore. Ecol. Lett. 22(11), 1797–1805 (2019).
Nathan, R. et al. A movement ecology paradigm for unifying organismal movement research. Proc. Natl. Acad. Sci. 105(49), 19052–19059 (2008).
Gonnerman, M. et al. Dynamic winter weather moderates movement and resource selection of wild turkeys at high-latitude range limits. Ecol. Appl. https://doi.org/10.1002/eap.2734 (2022).
Bennison, A. et al. Search and foraging behaviors from movement data: A comparison of methods. Ecol. Evol. 8(1), 13–24 (2018).
Zhang, J. et al. GPS telemetry for small seabirds: Using hidden Markov models to infer foraging behaviour of Common Diving Petrels (Pelecanoides urinatrix urinatrix). Emu Austral Ornithol. 119(2), 126–137 (2019).
Williams, L. E. Flight attainment in wild turkeys. J. Wildl. Manag. 38(1), 151–152 (1974).
Rumble, M. A. & Anderson, S. H. Feeding ecology of Merriam’s turkeys (Meleagris gallopavo merriami) in the Black Hills, South Dakota. Am. Midland Nat. 136(1), 157–171 (1996).
Byrne, M. E., Guthrie, J. D., Hardin, J., Collier, B. A. & Chamberlain, M. J. Evaluating wild turkey movement ecology: An example using first-passage time analysis. Wildl. Soc. Bull. 38(2), 407–413 (2014).
Erikstad, K. E. & Spidsø, T. K. The influence of weather on food intake, insect prey selection, and feeding behaviour in willow grouse chicks in northern Norway. Ornis Scand. 13(3), 176–182 (1982).
Park, K. J., Robertson, P. A., Campbell, S. T. & Foster, R. The role of invertebrates in the diet, growth, and survival of red grouse (Lagopuslagopus scoticus) chicks. J. Zool. 254(2), 137–145 (2001).
Mainguy, J., Gauthier, G., Giroux, J. F. & Bêty, J. Gosling growth and survival in relation to brood movements in greater snow geese (Chencaerulescens atlantica). Auk 123(4), 1077–1089 (2006).
Riotte-Lambert, L. & Matthiopoulos, J. Environmental predictability as a cause and consequence of animal movement. Trends Ecol. Evol. 35(2), 163–174 (2020).
Haas, C. A. Dispersal and use of corridors by birds in wooded patches on an agricultural landscape. Conserv. Biol. 9(4), 845–854 (1995).
Rosenberg, D. K., Noon, B. R. & Meslow, E. C. Biological corridors: Form, function, and efficacy. BioScience 47(10), 677–687 (1997).
Giroux, W., Blanchette, P., Bourgeois, J. C. & Cabana, G. Ruffed grouse brood habitat use in mixed softwood-hardwood nordic-temperate forests, Quebec, Canada. J. Wildl. Manag. 71(1), 87–95 (2007).
Jin-Ming, Z., Yun, F., Ying-Qiang, L. & Yue-Hua, S. Brood habitat selection of Chinese grouse (Tetrastes sewerzowi) at Lianhuashan, Gansu, China. Wilson J. Ornithol. 127(2), 310–318 (2015).
Brown, J. H., Gillooly, J. F., Allen, A. P., Savage, V. M. & West, G. B. Toward a metabolic theory of ecology. Ecology 85(7), 1771–1789 (2004).
Preisser, E. L. & Orrock, J. L. The allometry of fear: Interspecific relationships between body size and response to predation risk. Ecosphere 3(9), 1–27 (2012).
Novoa, C., Aebischer, N. J. & Landry, P. Upland habitat use by Pyrenean grey partridges Perdix perdix hispaniensis during the breeding season. Wildl. Biol. 8(2), 99–108 (2002).
Tirpak, J. M., Giuliano, W. M. & Miller, C. A. Ruffed grouse brood habitat selection at multiple scales in Pennsylvania: Implications for survival. Can. J. Zool. 86(4), 329–337 (2008).
Signorell, N. et al. Concealment from predators drives foraging habitat selection in brood-rearing Alpine black grouse Tetrao tetrix hens: Habitat management implications. Wildl. Biol. 16(3), 249–257 (2010).
Wegge, P. & Kastdalen, L. Habitat and diet of young grouse broods: Resource partitioning between Capercaillie (Tetrao urogallus) and Black Grouse (Tetrao tetrix) in boreal forests. J. Ornithol. 149(2), 237–244 (2008).
Goddard, A. D., Dawson, R. D. & Gillingham, M. P. Habitat selection by nesting and brood-rearing sharp-tailed grouse. Can. J. Zool. 87(4), 326–336 (2009).
Acknowledgements
We thank A. P. Gerrits, A. K. Lohr, E. Ulrey, N. A. Yeldell, L. Schofield, C. J. Wakefield, P. H. Wightman, S. D. Nelson, S. Watkins, P. Goodman, C. Argabright, and J. W. Wood for their efforts collecting field data. We also thank M. T. Kohl for providing constructive comments on earlier drafts of this manuscript.
Funding
Funding was provided by the Georgia Department of Natural Resources—Wildlife Resources Division, the Louisiana Department of Wildlife and Fisheries, the Louisiana State University Agricultural Center, the South Carolina Department of Natural Resources, the United States Forest Service, and the Warnell School of Forestry and Natural Resources at the University of Georgia. This material is partially based on work supported by the National Institute of Food and Agriculture and United States Department of Agriculture under McIntire Stennis project (7001494).
Author information
Authors and Affiliations
Contributions
N.B.: writing-original draft (lead); conceptualization (supporting); data curation (lead); formal analysis (lead). B.C.: conceptualization (equal); writing-review and editing (supporting); funding acquisition (equal). M.C.: conceptualization (supporting); writing-review and editing (supporting); funding acquisition (equal); project administration (lead). All authors consent for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bakner, N.W., Collier, B.A. & Chamberlain, M.J. Behavioral-dependent recursive movements and implications for resource selection. Sci Rep 13, 16632 (2023). https://doi.org/10.1038/s41598-023-43907-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-43907-z
- Springer Nature Limited