Abstract
To date, four species of simian malaria parasites including Plasmodium knowlesi, P. cynomolgi, P. inui and P. fieldi have been incriminated in human infections in Thailand. Although the prevalence of malaria in macaque natural hosts has been investigated, their vectors remain unknown in this country. Herein, we performed a survey of Anopheles mosquitoes during rainy and dry seasons in Narathiwat Province, Southern Thailand. Altogether 367 Anopheles mosquitoes were captured for 40 nights during 18:00 to 06:00 h by using human-landing catches. Based on morphological and molecular identification, species composition comprised An. maculatus (37.06%), An. barbirostris s.l. (31.34%), An. latens (17.71%), An. introlatus (10.08%) and others (3.81%) including An. umbrosus s.l., An. minimus, An. hyrcanus s.l., An. aconitus, An. macarthuri and An. kochi. Analyses of individual mosquitoes by PCR, sequencing and phylogenetic inference of the mitochondrial cytochrome genes of both malaria parasites and mosquitoes have revealed that the salivary gland samples of An. latens harbored P. knowlesi (n = 1), P. inui (n = 2), P. fieldi (n = 1), P. coatneyi (n = 1), P. hylobati (n = 1) and an unnamed Plasmodium species known to infect both long-tailed and pig-tailed macaques (n = 2). The salivary glands of An. introlatus possessed P. cynomolgi (n = 1), P. inui (n = 1), P. hylobati (n = 1) and coexistence of P. knowlesi and P. inui (n = 1). An avian malaria parasite P. juxtanucleare has been identified in the salivary gland sample of An. latens. Three other distinct lineages of Plasmodium with phylogenetic affinity to avian malaria species were detected in An. latens, An. introlatus and An. macarthuri. Interestingly, the salivary gland sample of An. maculatus contained P. caprae, an ungulate malaria parasite known to infect domestic goats. Most infected mosquitoes harbored multiclonal Plasmodium infections. All Plasmodium-infected mosquitoes were captured during the first quarter of the night and predominantly occurred during rainy season. Since simian malaria in humans has a wide geographic distribution in Thailand, further studies in other endemic areas of the country are mandatory for understanding transmission and prevention of zoonotic malaria.
Similar content being viewed by others
Introduction
Plasmodium knowlesi is endemic in Southeast Asia where natural reservoir hosts including long-tailed (Macaca fascicularis) and pig-tailed (M. nemestrina) macaques are ubiquitous1. The distribution of malaria caused by P. knowlesi seems to coincide with the habitats of macaque natural hosts in this region2,3. Despite differential prevalence of human infections with P. knowlesi across these endemic countries, ranging from relatively low to high infection rates among indigenous people, it is considered to be a virulent malaria species implicating in severe and fatal illnesses in patients with high parasitemia4,5. From a global perspective, the number of P. knowlesi-infected individuals may be incomparable to those caused by P. falciparum and P. vivax6. However, this simian malaria species poses an important health problem for people living in high transmission areas as well as travelers to these endemic countries7,8. It is noteworthy that these macaque natural hosts have a wide geographic distribution where humans and macaques interface in several locations in Southeast Asia. Therefore, the zoonotic nature of knowlesi malaria makes it recalcitrant to control by conventional strategies deployed for human malaria. Besides P. knowlesi, other simian malaria species including P. cynomolgi, P. inui, P. fieldi and possible other non-human Plasmodium species are implicated in human infections albeit with relatively lower prevalence8,9,10,11,12,13,14. Therefore, vector control could be an alternative measure for prevention, reduction or elimination of simian malaria transmissible to humans.
Attempts to identify mosquito vectors of P. knowlesi and other simian Plasmodium species circulating in South and Southeast Asia over the past seven decades were entirely based on experimental transmission of primate malaria parasites between laboratory-rear mosquitoes and monkeys1. For P. knowlesi, it has been demonstrated that complete sporogonic development of this parasite was achieved in at least 12 species of Anopheles consisting of An. annularis, An. aztecus, An. stephensi, An. atroparvus, An. balabacensis balabacensis, An. freeborni, An. maculatus, An. vagas, An. introlatus, An. kochi, An. sinensis and An. quadrimaculatus1. However, some of these mosquitoes have not been identified in areas where knowlesi malaria is endemic. After the first discovery of naturally acquired symptomatic human infection of P. knowlesi in 196515, followed by almost three decades later for subsequent identification of this simian malaria in humans in Southeast Asia, a number of Anopheles species belonging to the Leucosphyrus Group have been reported to harbor P. knowlesi sporozoites or its DNA in their salivary glands16. These include An. hackeri17, An. cracens18, An. latens19,20, An. introlatus21, An. balabacensis22,23,24,25,26, An. dirus27,28. Furthermore, P. knowlesi has been identified in other groups of Anopheles mosquitoes including An. collessi, An. roperi and An. donaldi in Malaysian Borneo23,25,29, and An. sundaicus in Andaman and Nicobar Islands30. Meanwhile, several vectors of P. knowlesi may also have potential to transmit other simian malaria parasites in which detailed information about these mosquitoes has been comprehensively reviewed by Vythilingam et al.16.
In Thailand, at least four species of simian Plasmodium species have been incriminated in symptomatic infections in humans which included P. knowlesi, P. cynomolgi, P. inui and P. fieldi8,10,11,12,13. Simian malaria in humans has a wide geographic distribution in Thailand albeit differential prevalence of infections occurred across endemic areas with relatively more cases detected in the southern part of the country where macaque natural hosts are more abundant than other regions10,11,12,13,31,32. To date, it remains unknown whether these simian malaria parasites in Thailand are transmitted by the same mosquito species as those reported from other endemic countries. Since the bionomics of each Anopheles species may differ, it is important to identify natural vectors of these simian malaria parasites in each endemic area. Therefore, we performed a prospective investigation of Anopheles potential vectors of simian malaria in Southern Thailand. Surveys of Anopheles mosquitoes in Narathiwat Province have revealed potential vectors of at least six known species of nonhuman primate malaria parasites as well as four avian and an ungulate Plasmodium species. These findings have provided baseline entomological data for simian malaria in Thailand that are important for prevention and control policy.
Results
Distribution and abundance of Anopheles species
Altogether, 367 female Anopheles mosquitoes were caught during rainy and dry seasons in 2018 and 2019. The total number of Anopheles collected from Sukhirin and Waeng Districts in Narathiwat Province during the raining seasons was 2.7 times more than those captured during the dry seasons (Fig. 1, Table 1). Based on taxonomic keys and molecular analysis of Anopheles33, the predominant species belonged to An. maculatus, An. barbirostris s.l., An. latens and An. introlatus accounting for 96.19% of all Anopheles mosquitoes while those identified as An. umbrosus s.l., An. hyrcanus s.l., An. minimus, An. aconitus and An. kochi were sporadically found, ranging from 1 to 4 specimens for each species (Table 1). The biting rates for An. maculatus, An. barbirostris s.l., An. latens and An. introlatus were 3.400, 2.875, 1.625 and 0.925 mosquito collected/night/collector, respectively. The number of Anopheles mosquitoes captured for each corresponding period of the night between 2018 and 2019 seemed to show a similar trend. The overall number of mosquito collection peaked at 20:00 and 21:00 h whereas remarkably fewer numbers of mosquitoes could be caught after midnight, especially after 2:00 h until daybreak (Fig. 2A). In the mosquito collection sites, the level of temperature peaked during 21:00 and 23:00 h whereas humidity gradually rose from dusk till dawn. The abundance of mosquitoes during the first quarter of the night (18:00 and 21:00 h) seemed to be positively correlated with the environmental temperature (Pearson r = 0.999, p = 0.03) and roughly correlated with the levels of humidity (Pearson r = 0.996, p = 0.05) (Fig. 2A). The number of An. latens and An. introlatus seemed to be early feeders from 18:00 and 19:00 h whereas peak feeding time of An. barbirostris s.l. was between 20:00 and 21:00 h. Meanwhile, An. maculatus was most abundant from 21:00 to 23:00 h. Despite the decline in the number of mosquitoes after midnight, both An. maculatus and An. barbirostris s.l. could be sparsely caught before dawn (Fig. 2B).
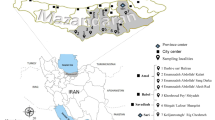
Locations of mosquito collection sites in Waeng (A–C) and Sukhirin (D–F) Districts, Narathiwat Province, Thailand. The images were from Google Earth Pro 7.3.6.9345 (https://www.google.com/intl/en_uk/earth/versions/#earth-pro)65.
Human-biting patterns of Anopheles mosquitoes in Narathiwat Province. (A) Total number of mosquitoes collected per hour in 2018 (blue bars) and 2019 (orange bar) in relation to hourly changes in temperature and humidity shown in corresponding colors. (B) Abundance of Anopheles species/group collected per hour.
Identification of malaria parasites
Of 367 Anopheles mosquitoes analyzed, primary PCR targeting the mitochondrial cytochrome c oxidase subunit 1 (cox1) gene of Plasmodium revealed positive results in 19 samples, accounting for 5.2% of total samples. Most Plasmodium-positive salivary glands (14 of 19 samples, 73.7%) were obtained during the rainy seasons. Species-specific PCR targeting human and simian Plasmodium species could detect 11 mosquitoes bearing simian malarial DNA in their salivary glands including P. inui (n = 5), P. fieldi (n = 2), P. knowlesi (n = 1), P. coatneyi (n = 1), P. cynomolgi (n = 1) and co-existence of P. knowlesi and P. inui (n = 1). DNA sequencing of recombinant plasmid clones from Plasmodium-positive specimens has reaffirmed the species of almost all simian malaria parasites except two P. inui-positive samples (HBT177 and HBT353) whose sequences virtually belonged to P. hylobati (Table 2). The species of Plasmodium in the remaining eight mosquitoes could not be determined by species-specific nested PCR. Sequences from recombinant plasmid clones of these unassigned samples displayed six different Plasmodium species/lineages by phylogenetic analysis. Of these, two mosquitoes (HBT181 and HBT368) harbored Plasmodium species that had phylogenetic affinity to an unnamed simian malaria parasite known to infect long-tailed and pig-tailed macaques in Malaysian Borneo (GenBank accession no. KJ569860) (Fig. 3)34. One sample yielded the cox1 sequence belonging to P. juxtanucleare with 99.79% sequence identity. Four other mosquitoes (HBT253, HBT329, HBT340 and HBT341), each containing 2 to 3 distinct cox1 alleles, seemed to diverge from P. circumflexum and were placed into three distinct phylogenetic clades. The p-distance (d ± S.E.) between the cox1 and its flanking sequences of P. circumflexum and those of the three distinct clades varied from 0.0577 ± 0.0057 to 0.0678 ± 0.0064 which were greater than those between some other known primate Plasmodium species (e.g. P. vivax vs. P. simiovale, d ± S.E. = 0.0139 ± 0.0028, and P. inui vs. P. hylobati d ± S.E. = 0.0237 ± 0.0039 to 0.0307 ± 0.0043). Interestingly, the remaining isolate (HBT314) contained three distinct alleles and displayed 99.71% to 99.85% sequence identity with that of P. caprae (GenBank accession no. LC090215). It is noteworthy that the majority of Plasmodium in the mosquito salivary glands contained clonal variation in the cox1and its flanking sequences except isolates HBT30, HBT368 and HBT179 in which single clones of P. knowlesi, P. sp. (KJ569860) and P. juxtanucleare, respectively, could be obtained.
Maximum likelihood tree of Plasmodium cox1 and its flanking sequences from the salivary glands of Anopheles. GenBank accession nos. of known species are in parentheses. Bootstrap values > 60% are shown along the branches. Scale indicates number of nucleotide substitutions per site. Symbols represent species of infected Anopheles mosquitoes.
Diversity of simian Plasmodium species from mosquitoes, humans and macaques in Thailand
To determine allelic variation in the cox1 and its flanking sequences of simian malaria parasites among different hosts, the sequences obtained from the salivary glands of mosquitoes in this study were compared with the corresponding gene sequences previously reported from macaques and humans in Thailand12,13. Results revealed that the cox1 and its flanking sequences of P. knowlesi, P. cynomolgi and P. fieldi were different among isolates from mosquitoes, macaques and humans. Although allelic variation in this locus occurred among P. inui isolates, the sequences from mosquitoes HBT30 and HBT207 were identical with 11 macaque isolates from Cho-airong, Waeng and Sukhirin Districts in Narathiwat Province and Kabang District in Yala Province collected during 2008–201812,13. Furthermore, these sequences were shared with that of P. inui (AB444114) strain IC (Leucosphyrus)(ATCC 30,195) isolated from Anopheles leucosphyrus from Negri Semilan in Malaysia since 1964 (Fig. 4A)35.
Maximum likelihood tree inferred from the cox1 and its flanking sequences of Plasmodium knowlesi, P. cynomolgi, P. inui and P. fieldi from mosquitoes, macaques and humans in Thailand12,13. GenBank accession nos. are in parentheses. Bootstrap values > 60% are shown along the branches. Scale indicates number of nucleotide substitutions per site. Symbols represent host origins of taxons.
Molecular identification of Anopheles species
The species of all Plasmodium-positive mosquitoes were determined by sequencing of the PCR-amplified 2140 bp region of Anopheles mitochondrial genes encompassing cox1 and cox2. Due to the lack of representative cox2 sequences of some members in the Leucosphyrus Group, species assignment was determined mainly from the cox1 phylogeny (Fig. 5A). Results revealed that An. introlatus carried 4 species of primate malaria parasites including P. knowlesi, P. cynomolgi, P. inui and P. hylobati, and a plausible novel species (HBT341) of an avian malaria parasite related to P. circumflexum. In the salivary glands of An. latens, six species of primate malaria parasites were identified including P. knowlesi, P. inui, P. fieldi, P. hylobati, P. coatneyi and an unnamed macaque malaria parasite Plasmodium sp. (GenBank accession no. KJ569860). Furthermore, P. juxtanucleare and a plausible novel species of avian plasmodia were also found in An. latens (Table 2). It is noteworthy that P. caprae was detected in the salivary glands of An. maculatus in this study. Although the sequence spanning cox1 and cox2 of mosquito HBT329 bearing an unknown Plasmodium sp. related to P. circumflexum could not be assigned from BOLD database36, it has been identified as An. macarthuri from phylogenetic analysis inferred from available reference cox1 sequences of Leucosphyrus mosquitoes spanning the 250-bp fragments (Fig. 6). Likewise, the other unassigned species of mosquito (HBT165) without Plasmodium infection also belonged to An. macarthuri based on phylogenetic analysis (Fig. 6). Meanwhile, the tree constructed from the cox2 locus per se displayed concordant phylogenetic affinity of mosquitoes with that inferred from the cox1 locus (Fig. 5B).
Maximum likelihood tree created from Anopheles cox1 sequences spanning 250 bp. GenBank accession nos. are in parentheses. Bootstrap values > 60% are indicated along the branches. Scale represents number of nucleotide substitutions per site. Plasmodium-infected and -uninfected Anopheles mosquitoes are marked by blue diamond and red spot, respectively.
Feeding time of potential vector species
Both Plasmodium-infected An. latens and An. introlatus were captured during 18:00 and 21.00 h. Likewise, An. macarthuri (HBT329) bearing a plausible novel avian malaria parasite was caught between 20:00 and 21:00 h. Although most An. maculatus in these surveys had maximum feeding time between 21:00 and 23:00 h (Fig. 2B), the infected mosquito was caught during 23:00 and 24:00 h (Table 2).
Discussion
In Thailand, the past couple of decades have seen a dramatic decline in the number of falciparum malaria patients while a relative increase in the proportion of vivax malaria has been envisaged, suggesting that currently applied control measures seem to be less effective against non-falciparum infections10,11,12,13,37,38. Likewise, an increased prevalence of patients infected with P. knowlesi has been observed during the past decade, especially among those who resided in areas where infected domesticated or wild macaques co-existed10,11,12,13,37. In 2022, a total of 10,152 malaria cases were reported in Thailand of which P. vivax, P. falciparum and P. knowlesi were incriminated in 95.3%, 2.5% and 1.8% of infections, respectively37. Importantly, control of zoonotic transmission of simian malaria in humans would require integrative knowledge about the prevalence of simian malaria parasites in humans, macaque natural hosts and Anopheles vectors. Our surveys in Narathiwat Province have revealed a relatively high prevalence of Anopheles mosquitoes carrying malaria parasites in their salivary glands (5.16%, 19 of 368 mosquitoes) in which 13 of these belonged to primate Plasmodium species. This is in stark contrast with the absence of infected mosquitoes in a recent survey in Surat Thani Province, another region in Southern Thailand39. Differential risk of acquiring malaria in different endemic areas could stem from variation in bionomics of mosquito vectors, prevalence of malaria patients and status of reservoir hosts. In this study we deployed both PCR and sequencing to detect primate malarial DNA in the mosquitoes’ salivary gland samples which revealed concordant results in most samples. Discrepancy between PCR and sequencing in samples HBT177 and HBT353 (Table 2) could stem from cross hybridization of the nested PCR primers targeting the cox1 sequence of P. inui and a variant of P. hylobati identified in this study which possessed different sequences of the primer regions from that of P. hylobati strain WAK isolated from a gibbon (Hylobates molock) in Sarawak (GenBank accession no. AB354573)40. However, differentiation between P. inui and P. hylobati could be resolved and correctly assigned by analysis of the cox1 sequences as shown in our previous study13. It is noteworthy that P. hylobati has not been detected among macaques in Thailand13,31,32 while the prevalence of this simian malaria species in gibbons known as its natural hosts awaits further investigation.
The presence of P. knowlesi DNA in the salivary glands of An. latens and An. introlatus in Narathiwat Province has implied that both mosquito members in the Leucosphyrus complex could serve as potential vectors of this simian malaria species in this province where both infected humans and macaques have been previously identified10,11,12,13,31,32. Mosquitoes belonging to the Leucosphyrus Subgroup comprise at least 13 species while seven of these have been detected in Thailand33. The breeding places of these mosquitoes included ground pools, animal footprints, wheel-tracks and small shallow running streams in the forested ecotype with partial or heavily-shaded areas33,41. While An. introlatus seemed to occupy Peninsular Malaysia and Southern Thailand, previous surveys have documented that An. latens was widely distributed across at least 3 countries in Southeast Asia including Kalimantan of Indonesia, Malaysian Borneo, western regions of Peninsular Malaysia and Southern Thailand encompassing Chumporn, Nakhon Si Thammarat, Phang Nga, Satun, Songkhla and Narathiwat Provinces33,41. Consistently, An. latens has been found to vector P. knowlesi in Kapit, Sarawak19. Both An. introlatus and An. hackeri have been incriminated in transmission of this simian malaria species in Selangor, Peninsular Malaysia21,42. Other members in the Leucosphyrus Group as potential vectors of knowlesi malaria include An. dirus in Vietnam27,28, An. cracens in Pahang18 and An. balabacensis in Sabah and Sarawak in Malaysian Borneo and Palawan in The Philippines22,23,24,25,26. It is noteworthy that transmission of knowlesi malaria could be from other groups of mosquitoes including An. donaldi belonging to the Barbirostris Group in Sabah and Sarawak23, An. sundaicus species D in Andaman and Nicobar Islands30,43 and two members in the Umbrosus Group, An. collessi and An. roperi, in Sarawak25,29. The occurrence of diverse species of Anopheles potential vectors of P. knowlesi could enhance transmission of this simian malaria parasite in various ecological niches.
Besides being vectors of P. knowlesi in Thailand, it is plausible that An. latens could be a natural vector of P. inui and P. fieldi. Likewise, An. introlatus seemed to be responsible for transmission of P. cynomolgi and P. inui in the southern part of the country. Our findings have supported previous studies that both An. latens and An. introlatus could be important vectors for simian malaria transmissible to humans19,20,21. Furthermore, An. latens could serve as a vector of P. coatneyi while the presence of P. coatneyi and P. hylobati DNA in the salivary glands of An. introlatus has suggested that they could transmit these simian malarias in the survey areas. In this study, clonal diversity in the cox1 sequences of most primate malaria parasites from infected mosquito salivary glands (Figs. 3, 4) and co-existence of different species of Plasmodium in a single mosquito (An. introlatus sample HBT206, Table 1) could probably stem from multiple feedings of the vectors on different monkeys infected with variant strains or different species of Plasmodium. Alternatively, multiple clones/species of malaria parasites in individual macaques could be prevalent, lending plausibility for the mosquitoes to acquire multiple clones/species of plasmodia upon single feedings. Our previous study has shown considerable clonal diversity of P. knowlesi and other primate malaria parasites in both long-tailed and pig-tailed macaques in Southern Thailand32; thus, the latter scenario could not be excluded. Within mosquitoes, inter-species and intra-species competition could further influence subsequent transmission of malaria parasites to the vertebrate hosts44,45. Meanwhile, multiclonal and mixed species infections with simian malaria in humans occurred less frequently than in mosquitoes and macaques in Thailand12,13,31,32. Intriguingly, differential capability of adaptation to humans might occur among strains/lineages of primate Plasmodium species while genetic/phenotypic diversity of these parasites could contribute to a range of disease severity as observed in knowlesi malaria in humans albeit multiple host factors may also play crucial roles4,5,8,10,11,12,13,46,47.
It has been shown that fluctuation in the levels of asexual parasitemia and gametocytemia occurred in experimental P. knowlesi infections in long-tailed macaques with peak parasitemia between 18:00 and 24:00 h and between 24:00 and 06:00 h, respectively48. The biting period of An. latens in Sarawak during 19:00 and 06:00 h seemed to coincide with the parasitemia of both asexual and sexual stages in peripheral blood of infected hosts; thus, enhancing transmission of simian malaria16,19,23. In field studies, An. latens and An. introlatus had peak biting activity between 19:00 and 20:00 h and 20:00 and 21:00 h, respectively16,20,43. Consistently, in this study simian Plasmodium-infected An. latens and An. introlatus were captured between 18:00 and 21:00 h (Table 2), implying that exposure to mosquito bites in areas where infected macaques coexist in Southern Thailand during the first quarter of the night may pose a high risk for getting zoonotic malaria.
Phylogenetic trees inferred from the Plasmodium cox1 sequences derived from humans, macaques and Anopheles mosquitoes in Thailand have revealed genetic diversity across these hosts (Fig. 4). Despite limited number of available sequences, it is noteworthy that human isolates of P. knowlesi, P. cynomolgi, P. inui and P. fieldi differed from those derived from macaques and Anopheles mosquitoes, implying that the extent of genetic variation in natural populations of these primate malaria parasites could be more extensive. However, it seemed that a certain strain of P. inui derived from An. latens and An. introlatus in this study shared identical cox1 and its flanking sequences across 1504 bp with 11 previously characterized isolates from pig-tailed macaques from Cho-airong, Waeng and Sukhirin Districts in Narathiwat Province obtained in 2008, 2013, 2018 and 202013,32. These isolates also possessed the same sequence as that of strain IC (leucosphyrus) derived from An. leucosphyrus caught from Negri Semilan in Malaysia in 196449. Although identical mitochondrial cox1 sequences may not represent the same entire nuclear genomes, there remains a possibility that certain strains of P. inui could predominate and persist in natural transmission cycle that could probably stem from their adaptive capabilities to survive in diverse species of mosquito vectors and reservoir hosts.
Our sequence analysis of recombinant clones from 8 infected mosquitoes whose species of malaria parasites could not be assigned by PCR has revealed that 2 samples (HBT181 and HBT368) belonged to an unnamed Plasmodium known to infect both pig-tailed and long-tailed macaques in Sabah, northern Borneo (GenBank accession no. KJ569860)34. The placement of these isolates in the phylogenetic tree seemed to be closely related to P. coatneyi while the magnitude of evolutionary distance between the cox1 and its flanking sequences of these malaria species and that of P. coatneyi was comparable or greater than those between other primate Plasmodium species. Therefore, both isolates from An. latens (HBT181 and HBT368) and the previously described isolates (KJ569860) from Sabah34 could belong to a distinct species. It is likely that more novel species of primate malaria parasites remain to be discovered. Meanwhile, Plasmodium species from An. maculatus (HBT314) displayed 99.77% to 99.92% sequence identity with that of P. caprae (GenBank accession no. LC090215). This ungulate malaria parasite has caused infections in domestic goats in Western Thailand and other countries50. It is plausible that An. maculatus could serve as a vector of P. caprae in the southern part of Thailand akin to An. subpictus and An. aconitus that have been recently reported to be implicated in transmission of P. caprae among goat farms in the western provinces of the country51.
Apart from primate malaria, our analysis has identified An. latens as a potential vector of P. juxtanucleare, a predominant avian malaria parasite that infects chicken (Gallus gallus domesticus) and fighting cocks (Burmese red junglefowl, Gallus gallus spadiceus) in Thailand. Meanwhile, this avian malaria parasite has been known to infect other members of the Phasianidae52,53,54 including short-crested flycatcher in Brazil55, turkey, parrot and chugar in Pakistan56, eared-pheasant in Japan57 and black-footed penguins in South Africa58. Besides cosmopolitan in distribution, P. juxtanucleare has been detected among wild passerines in Brazil, suggesting spillover from poultry to free-living birds59. Although P. juxtanucleare is known to be mainly vectored by mosquitoes in the genus Culex (e.g. Cx. vishnui and Cx quinquefasciatus in The Philippines and Cx. saltanensis in Brazil)60,61, identification of An. latens as an additional vector in our study has expanded a range of mosquitoes capable of transmitting this avian malaria species. Herein, we also identified 3 Plasmodium lineages from An. latens and An. introlatus whose cox1 sequences displayed phylogenetic clustering within avian Plasmodium clades. Furthermore, the evolutionary distance (p-distance) between these lineages and a closely related P. circumflexum was comparable to or greater than those between known primate and avian malarial species, suggesting that these newly identified lineages could represent distinct avian Plasmodium species. Taken together, it is plausible that a wide range of mosquito vectors and diverse avian host species could enhance cosmopolitan distribution of avian malaria parasites.
Despite a wealth of knowledge about morphological differences among Anopheles vectors of malaria, molecular identification remains to be a useful method for fine resolution of mosquito species, especially those with minor structural differences. Although our sequence analysis encompassing near complete cox1 and partial cox2 sequences, An. latens and An. introlatus could be identified based on available cox1 reference sequences in the public database. The concordant topologies of phylogenetic trees inferred from either cox1 or cox2 sequences have provided an additional marker for speciation of these mosquito species (Fig. 5).
The midguts and ovaries of the mosquitoes have not been examined in this study. Although the presence of malarial DNA in the midguts may not replicate the results obtained from the mosquito’s salivary gland samples, it can support the integrity of analysis from salivary gland specimens. Likewise, more information on the bionomics of the mosquitoes from examination of the ovary samples has not been available in this study. It is noteworthy that the presence of malarial DNA or sporozoites in the mosquito’s salivary glands does not directly indicate its true vectorial status1. However, consistent identification of specific malarial DNA from the same species of mosquitoes, especially those from different geographic areas, seems to indicate that they could vector a given Plasmodium species16.
To the best of our knowledge, this is the first report on An. latens and An. introlatus as vectors of P. knowlesi in Thailand while both species could potentially transmit other simian and some avian plasmodia. Furthermore, An. maculatus has been identified to vector P. caprae that may parasitize domestic goats in the southern part of the country. Unraveling vectors of malaria and their bionomics could contribute to knowledge about disease transmission in malaria endemic areas that are important for prevention and control policy.
Materials and methods
Study area
Mosquito collections were conducted from three locations in Waeng (A: 5° 48′ 27″ N, 101° 50′ 44″ E; B: 5° 49′ 12″ N, 101° 51′ 20″ N and C: 5° 50′ 5″ N, 101° 51′ 7″ E) and three sites in Sukhirin Districts (D: 5° 48′ 4″ N, 101° 45′ 2″ E; E: 5° 48′ 8″ N, 101° 45′ 13″ N and F: 5° 47′ 40″ N, 101° 42′ 51″ E) in Narathiwat Province during 2018 and 2019 (Fig. 1). These locations were selected because (i) indigenous villagers residing in these areas were diagnosed to be infected with P. knowlesi10,11,12,13, (ii) both wild and domesticated pig-tailed and long-tailed macaques were prevalent and known to be infected with several simian Plasmodium species32, (iii) both districts were situated along forest fringe with natural streams and rivers running through the areas that could be natural breeding places for Anopheles mosquitoes and (iv) rubber plantations were located in the districts where farmers and workers were frequently exposed to mosquito bites during the harvesting process. The collection sites had a tropical monsoon climate and were located near Hala-Bala tropical rain forest, covering an area of approximately 1.3 square kilometers across Yala and Narathiwat Provinces along Thailand-Malaysia border approximately between 5° 37′–6° 14′ N and 101° 8′ E–101° 51′ E. The elevation of Hala-Bala Forest ranged from 50 m to 1.5 km above mean sea level with annual mean temperature of 27.6 °C, annual average rainfall of 2560 mm, and relative humidity between 77 and 80%. The heavy rainy months of the year were from November to December and the relatively dry season from February to April. The number of days with rainfall (≥ 1.0 mm) occurred approximately 200 days per annum.
Collection of mosquitoes
Mosquito collections were carried out every other night for 10 days during each of the four surveys in March and August 2018 and March and November 2019, covering relatively dry and rainy seasons of the study sites. Human-landing catches of mosquitoes were done outdoors. Each collection site comprised two teams, each consisting of two persons: one volunteer served as bait and the other as collector. The first team captured the mosquitoes from 18:00 to 24.00 h and the other from midnight until dawn (06:00 h). The collector captured the mosquitoes upon landing on the human bait by using a plastic collection tube. All captured mosquitoes were kept individually in a 1.5 ml sterile microtube with cap, placed on ice and transferred to 4 °C refrigerator for subsequent morphological identification. All volunteers and field staff were monitored for malaria infection during the field work and thereafter on a weekly basis for 2 months.
Morphological identification of mosquitoes and dissection
Each mosquito was examined under stereomicroscope to exclude those belonging to genera other than Anopheles. All female Anopheles mosquitoes were gently fixed onto plasticine using ultra-thin micro-headless pins. Morphological identification was based on characteristic features of Anopheles genera, groups, complex and species according to illustrated keys to the mosquitoes of Thailand33. After morphological identification, the salivary glands of each female Anopheles were dissected and preserved in absolute ethanol in separate tubes per individual mosquitoes.
Preparation of DNA
Ethanol was allowed to evaporate from mosquito’s salivary glands prior to DNA preparation. Extraction and purification of DNA were performed for each salivary gland sample by using DNeasy Blood and Tissue Kit following the manufacturer’s protocol (Qiagen, Hilden, Germany). DNA was kept at − 40 °C until use.
Molecular detection of Plasmodium species
DNA extracted from salivary glands of each Anopheles mosquito was used as template for species-specific PCR detection targeting the mitochondrial cytochrome c oxidase subunit 1 (cox1) gene of human and simian malaria parasites including P. falciparum, P. vivax, P. malariae, P. ovale, P. knowlesi, P. cynomolgi, P. inui, P. coatneyi and P. fieldi. Amplification reaction mixtures and PCR conditions were the same as those previously described12,13. The primary PCR products of all Plasmodium-positive samples were subcloned into plasmid pGEM-T Easy Vector Systems (Promega, Madison, Wisconsin, USA) with Escherichia coli strain JM109 as a host for transformation. At least five recombinant clones from each positive sample were used as templates for determination of the cox1 sequences.
Molecular detection of Anopheles species
Primers for PCR amplifications of Anopheles mosquitoes’ DNA were derived from the mitochondrial genes encompassing cytochrome c oxidase subunits I and II intervened by the gene encoding tRNA-Leu. The forward and reverse primers were ALMTF0 (5ʹ-ATTTAATCGCGACAATGATTATTTTC-3ʹ) and ALMTR0 (5ʹ-CTATGATTTGCTCCACAAATTTC-3ʹ), respectively. Amplification of the Anopheles DNA was performed in a total volume of 30 μl of the reaction mixture containing template DNA, 300 mM each deoxyribonucleoside triphosphate, 3 μl of 10X LA Taq PCR buffer, 2.5 mM MgCl2, 0.3 μM of each primer and 1.25 units of LA Taq DNA polymerase (Takara, Seta, Japan). Amplification profile consisted of preamplification denaturation at 94 °C for 1 min followed by 35 cycles of 96 °C for 20 s, 55 °C for 30 s and 72 °C for 4 min, and a final extension at 72 °C for 10 min. Thermal cycling of the samples was performed in a GeneAmp 9700 PCR thermal cycler (Applied Biosystems, Foster City, CA). The amplicons were examined by 1% agarose gel electrophoresis and visualized under a UV transilluminator. The PCR product was purified by using QIAquick PCR purification kit (Qiagen, Germany) prior to DNA sequencing. Sequencing primers were used for obtaining overlapping DNA sequences. Sequencing was performed on an ABI3100 Genetic Analyzer using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, USA). Mosquitoes belonging to Leucosphysrus Group were further identified by using multiplex PCR assay for differentiation of An. latens, An. introlatus, An. cracens and An. balabacensis as reported62.
Data analysis
Nucleotide sequences were aligned by using the codon-based option in MUSCLE program with manual adjustment63. For cox1 sequences of malaria parasites, known species or lineage of Plasmodium were used for comparative analysis which included the following GenBank accession nos.: P. falciparum (AJ276845), P. vivax (NC007243), P. malariae (AB354570), P. ovale curtisi (AB354571), P. ovale wallikeri (HQ712053), P. knowlesi (AB444106 and AY598141), P. cynomolgi (AB434919 and AB444126), P. inui (AB444110, AB444111, AB444114, AB444116 and AB444120), P. fieldi (AB354574 and KJ5698631), P. coatneyi (AB354575), P. fragile (AB444135 and AY722799), P. hylobati (AB354573), P. gonderi (AB434918), P. simium (AY800110), P. simiovale (AB434920), P. circumflexum (KY653762), P. juxtanucleare (AB250415), P. relictum (AY733090), P. elongatum (KY653802), P. caprae (LC090215) and P. sp. (KJ569860). The cox1 sequences of human- and macaque-derived P. knowlesi, P. cynomolgi, P. inui and P. fieldi in Thailand from our previous studies were included for comparison (GenBank accession nos. OL437412–OL437468 and OQ436033–OQ436048)12,13. For Anopheles cox1 and cox2 references, both complete and partial sequences of known species were deployed in the phylogenetic analysis: An. introlatus (MW585348), An. nemophilous (DQ897959–DQ897961 and OL742879), An. balabacensis (MH032676 and MH326747), An. cracens (JX219733, An. scanloni (AM180841), An. dirus s.s (JX219731), An. baimaii (AJ877562), An. leucosphyrus s.l. (MN520357), An. latens (OP617572), An. maculatus (KT382822), An. minimus (KT895423), An. culicifacies B (KR732656), An. aconitus (NC039540), An. mirans (DQ897965 and DQ897966), An. sulawesi (DQ890954 and ON908454) and An. macarthuri (DQ897968 and DQ897969). All sites at which the alignment postulated a gap were eliminated in pairwise comparisons of the analysis. For evolutionary distance between sequences, p-distance was determined by dividing the number of nucleotide differences between two sequences by the total number of nucleotides compared and its standard error (d ± S.E.) between sequences was computed by 1000 bootstrap pseudoreplicates. Phylogenetic trees were constructed by using the maximum likelihood method with the best substitution model for the sequence data that yielded the minimum Bayesian Information Criterion (BIC) scores64. Confidence levels of clustering patterns in the phylogenetic tree were assessed by 1000 bootstrap pseudoreplicates.
Ethical approval
This study was reviewed and approved by the Institutional Review Board in Human Research of Faculty of Medicine, Chulalongkorn University (IRB No. 272/61, COA No. 841/2019) and Naresuan University Institutional Review Board of Human Research (IRB No. 0614/62, COA No. 057/2020). Prior to serving as human baits for mosquito collection, written informed consent was obtained from all participants. All procedures were performed in accordance with the relevant guidelines and regulations.
Data availability
The datasets generated during and/or analyses during the current study are available from the corresponding authors upon request.
Code availability
Forty-five sequences of the mitochondrial cox1and its flanking sequences of Plasmodium and 19 sequences of Anopheles mitochondrial genes spanning cox1 and cox2 have been deposited in NCBI GenBank under accession numbers OQ473193–OQ473237 and OQ466059–OQ466077, respectively.
References
Coatney, G. R., Collins, W. E., Warren, M. & Contacos, P. G. The Primate Malarias (Original Book Published 1971) [CD-ROM]. Version 1.0 (Centers for Disease Control and Prevention, 2003).
Singh, B. & Daneshvar, C. Human infections and detection of Plasmodium knowlesi. Clin. Microbiol. Rev. 26, 165–184 (2013).
Shearer, F. M. et al. Estimating geographical variation in the risk of zoonotic Plasmodium knowlesi infection in countries eliminating malaria. PLoS Negl. Trop. Dis. 10, e0004915 (2016).
Cox-Singh, J. et al. Plasmodium knowlesi malaria in humans is widely distributed and potentially life threatening. Clin. Infect. Dis. 46, 165–171 (2008).
Nakaviroj, S., Kobasa, T., Teeranaipong, P., Putaporntip, C. & Jongwutiwes, S. An autochthonous case of severe Plasmodium knowlesi malaria in Thailand. Am. J. Trop. Med. Hyg. 92, 569–572 (2015).
WHO. World Malaria Report 2022 (World Health Organization, 2022).
Müller, M. & Schlagenhauf, P. Plasmodium knowlesi in travellers, update 2014. Int. J. Infect. Dis. 22, 55–64 (2014).
Jongwutiwes, S., Putaporntip, C., Iwasaki, T., Sata, T. & Kanbara, H. Naturally acquired Plasmodium knowlesi malaria in human, Thailand. Emerg. Infect. Dis. 10, 2211–2213 (2004).
Singh, B. et al. A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet 363, 1017–1024 (2004).
Putaporntip, C. et al. Differential prevalence of Plasmodium infections and cryptic Plasmodium knowlesi malaria in humans in Thailand. J. Infect. Dis. 199, 1143–1150 (2009).
Jongwutiwes, S. et al. Plasmodium knowlesi malaria in humans and macaques, Thailand. Emerg. Infect. Dis. 17, 1799–1806 (2011).
Putaporntip, C. et al. Plasmodium cynomolgi co-infections among symptomatic malaria patients, Thailand. Emerg. Infect. Dis. 27, 590–593 (2021).
Putaporntip, C. et al. Cryptic Plasmodium inui and Plasmodium fieldi infections among symptomatic malaria patients in Thailand. Clin. Infect. Dis. 75, 805–812 (2022).
Yap, N. J. et al. Natural human infections with Plasmodium cynomolgi, P. inui, and 4 other simian malaria parasites, Malaysia. Emerg. Infect. Dis. 27, 2187–2191 (2021).
Chin, W., Contacos, P. G., Coatney, G. R. & Kimball, H. R. A naturally acquired quotidian-type malaria in man transferable to monkeys. Science 149, 865 (1965).
Vythilingam, I., Chua, T. H., Liew, J. W. K., Manin, B. O. & Ferguson, H. M. The vectors of Plasmodium knowlesi and other simian malarias Southeast Asia: Challenges in malaria elimination. Adv. Parasitol. 113, 131–189 (2021).
Wharton, R. H. & Eyles, D. E. Anopheles hackeri, a vector of Plasmodium knowlesi in Malaya. Science 134, 279–280 (1961).
Jiram, A. I. et al. Entomologic investigation of Plasmodium knowlesi vectors in Kuala Lipis, Pahang, Malaysia. Malar. J. 11, 213 (2012).
Vythilingam, I. et al. Natural transmission of Plasmodium knowlesi to humans by Anopheles latens in Sarawak, Malaysia. Trans. R. Soc. Trop. Med. Hyg. 100, 1087–1088 (2006).
Tan, C. H., Vythilingam, I., Matusop, A., Chan, S. T. & Singh, B. Bionomics of Anopheles latens in Kapit, Sarawak, Malaysian Borneo in relation to the transmission of zoonotic simian malaria parasite Plasmodium knowlesi. Malar. J. 7, 52 (2008).
Vythilingam, I. et al. Plasmodium knowlesi malaria an emerging public health problem in Hulu Selangor, Selangor, Malaysia (2009–2013): Epidemiologic and entomologic analysis. Parasit. Vectors 7, 436 (2014).
Tsukamoto, M., Miyata, A. & Miyagi, I. Surveys on simian malaria parasites and their vector in Palawan Island, the Philippines. Trop. Med. 20, 39–50 (1978).
Ang, J. X. D. et al. New vectors in northern Sarawak, Malaysian Borneo, for the zoonotic malaria parasite, Plasmodium knowlesi. Parasit. Vectors 13, 472 (2020).
Brown, R. et al. Human exposure to zoonotic malaria vectors in village, farm and forest habitats in Sabah, Malaysian Borneo. PLoS Negl. Trop. Dis. 14, e0008617 (2020).
Hawkes, F. M. et al. Vector compositions change across forested to deforested ecotones in emerging areas of zoonotic malaria transmission in Malaysia. Sci. Rep. 9, 13312 (2019).
Wong, M. L. et al. Seasonal and spatial dynamics of the primary vector of Plasmodium knowlesi within a major transmission focus in Sabah, Malaysia. PLoS Negl. Trop. Dis. 9, e0004135 (2015).
Nakazawa, S. et al. Anopheles dirus co-infection with human and monkey malaria parasites in Vietnam. Int. J. Parasitol. 39, 1533–1537 (2009).
Marchand, R. P. et al. Co-infections of Plasmodium knowlesi, P. falciparum, and P. vivax among humans and Anopheles dirus mosquitoes, Southern Vietnam. Emerg. Infect. Dis. 17, 1232–1239 (2011).
Ang, J. X. D. et al. New vectors that are early feeders for Plasmodium knowlesi and other simian malaria parasites in Sarawak, Malaysian Borneo. Sci. Rep. 11, 7739 (2021).
Vidhya, P. T., Sunish, I. P., Maile, A. & Zahid, A. K. Anopheles sundaicus mosquitoes as vector for Plasmodium knowlesi, Andaman and Nicobar Islands, India. Emerg. Infect. Dis. 25, 817–820 (2019).
Seethamchai, S., Putaporntip, C., Malaivijitnond, S., Cui, L. & Jongwutiwes, S. Malaria and Hepatocystis species in wild macaques, Southern Thailand. Am. J. Trop. Med. Hyg. 78, 646–653 (2008).
Putaporntip, C. et al. Ecology of malaria parasites infecting Southeast Asian macaques: Evidence from cytochrome b sequences. Mol. Ecol. 19, 3466–3476 (2010).
Rattanarithikul, R. et al. Illustrated keys to the mosquitoes of Thailand. IV. Anopheles. Southeast Asian J. Trop. Med. Public Health 37, 1–128 (2006).
Muehlenbein, M. P. et al. Accelerated diversification of nonhuman primate malarias in Southeast Asia: Adaptive radiation or geographic speciation? Mol. Biol. Evol. 32, 422–439 (2015).
Collins, W. E., Contacos, P. G., Guinn, E. G. & Held, J. R. Some observations on the transmission of Plasmodium inui. J. Parasitol. 54, 846–847 (1968).
Ratnasingham, S. & Herbert, P. D. BOLD: The barcode of life data system. Mol. Ecol. Notes 7, 355–364 (2007).
Ministry of Public Health of Thailand, Division of Vector Bourne Diseases. Malaria Online. http://malaria.ddc.moph.go.th (Accessed 29 December 2022).
Cui, L. et al. Multidisciplinary investigations of sustained malaria transmission in the Greater Mekong Subregion. Am. J. Trop. Med. Hyg. 107(Suppl 4), 138–151 (2022).
Wamaket, N. et al. Anopheles bionomics in a malaria endemic area of southern Thailand. Parasit. Vectors 14, 378 (2021).
Collins, W. E., Contacos, P. G., Garnham, P. C., Warren, M. & Skinner, J. C. Plasmodium hylobati: A malaria parasite of the gibbon. J. Parasitol. 58, 123–128 (1972).
Sallum, M. A. M., Peyton, E. L. & Wilkerson, R. C. Six new species of the Anopheles leucosphyrus group, reinterpretation of An. elegans and vector implications. Med. Vet. Entomol. 19, 158–199 (2005).
Wharton, R. H., Eyles, D. E., Warren, M. & Cheong, W. H. Studies to determine the vectors of monkey malaria in Malaya. Ann. Trop. Med. Parasitol. 58, 56–77 (1964).
Manguin, S., Garros, C., Dusfour, I., Harbach, R. E. & Coosemans, M. Bionomics, taxonomy, and distribution of the major malaria vector taxa of Anopheles subgenus Cellia in Southeast Asia: An updated review. Infect. Genet. Evol. 8, 489–503 (2008).
Paul, R. E. L. et al. Interspecific competition during transmission of two sympatric malaria species to the mosquito vector. Proc. R. Soc. Lond. B Biol. Sci. 269, 2551–2557 (2002).
Kamiya, T., Paton, D. G., Catteruccia, F. & Reece, S. E. Targeting malaria parasites inside mosquitoes: Ecoevolutionary consequences. Trends Parasitol. 38, 1031–1040 (2022).
Teeranaipong, P. et al. A functional single-nucleotide polymorphism in the CR1 promoter region contributes to protection against cerebral malaria. J. Infect. Dis. 198, 1880–1891 (2008).
Su, X. Z., Zhang, C. & Joy, D. A. Host-malaria parasite interactions and impacts on mutual evolution. Front. Cell. Infect. Microbiol. 10, 587993 (2020).
Anderios, F., Noorrain, A. & Vythilingam, I. In vivo study of human Plasmodium knowlesi in Macaca fascicularis. Exp. Parasitol. 124, 181–189 (2010).
Collins, W. E., Warren, M., Skinner, J. C. & Alling, D. W. Plasmodium inui: Serologic relationships of Asian isolates. Exp. Parasitol. 27, 507–515 (1970).
Kaewthamasorn, M. et al. Genetic homogeneity of goat malaria parasites in Asia and Africa suggests their expansion with domestic goat host. Sci. Rep. 8, 5827 (2018).
Nguyen, A. H. L. et al. Myzomyia and Pyretophorus series of Anopheles mosquitoes acting as probable vectors of the goat malaria parasite Plasmodium caprae in Thailand. Sci. Rep. 13, 145 (2023).
Tattiyapong, M., Deemagarn, T., Mohkeaw, K., Ngamjiteu, S. & Jiratanh, M. Molecular characterization of Plasmodium juxtanucleare in Burmese red junglefowls (Gallus gallus spadiceus) in Thailand. J. Protozool. Res. 26, 1–10 (2016).
Kissinger, J. C. et al. Molecular phylogenetic analysis of the avian malarial parasite Plasmodium (Novyella) juxtanucleare. J. Parasitol. 88, 769–773 (2002).
Win, S. Y. et al. Detection and molecular identification of Leucocytozoon and Plasmodium species from village chickens in different areas of Myanmar. Acta Trop. 212, 105719 (2020).
Noor, R. et al. Prevalence of parasites in selected captive bird species. Braz. J. Biol. 84, e254251 (2021).
Talat, R. Infection of haematozoan parasites found in birds of NWFP (Pakistan). Pak. J. Biol. Sci. 8, 1–5 (2005).
Murata, K. et al. Plasmodium (Bennettinia) juxtanucleare infection in a captive white eared-pheasant (Crossoptilon crossoptilon) at a Japanese zoo. J. Vet. Med. Sci. 70, 203–205 (2008).
Grim, K. C. et al. Plasmodium juxtanucleare associated with mortality in black-footed penguins (Spheniscus demersus) admitted to a rehabilitation center. J. Zoo Wildl. Med. 34, 250–255 (2003).
Ferreira-Junior, F. C. et al. A new pathogen spillover from domestic to wild animals: Plasmodium juxtanucleare infects free-living passerines in Brazil. Parasitology 145, 1949–1958 (2018).
Lourenco-de-Oliverira, R. & Almiron, W. R. Culex saltanensis Dyar, 1928: Natural vector of Plasmodium juxtanucleare in Rio de Janeiro. Brazil. Mem. Inst. Oswaldo Cruz 86, 87–94 (1991).
Chen, T. H. et al. Avian Plasmodium infection in field-collected mosquitoes during 2012–2013 in Tarlac, Philippines. J. Vector Ecol. 40, 386–392 (2015).
Pramasivan, S. et al. Multiplex PCR assay for the identification of four species of the Anopheles Leucosphyrus sub-group in Malaysia. Insects 13, 195 (2022).
Edgar, R. C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30, 2725–2729 (2013).
Google Earth Pro. Map Data of Thailand. https://www.google.com/intl/en_uk/earth/versions/#earth-pro (2022).
Acknowledgements
The authors are grateful to Siriporn Thongaree for valuable advice and support. The authors wish to thank all staff at Hala-Bala Research Station, Department of National Parks, Wildlife and Plant Conservation for assistance in field studies. S.Y. is a recipient of the Ph.D. scholarship from The Science Achievement Scholarship of Thailand (SAST).
Funding
This study received financial supports from Ratchadapiseksompotch Fund, Faculty of Medicine, Chulalongkorn University (Grant No. RA64/032) to CP and SJ; and The Thailand Research Fund (Grant No. RSA5980054) to C.P.
Author information
Authors and Affiliations
Contributions
C.P., S.S. and S.J. designed the study and funding acquisition. S.Y., C.P., S.K. and S.S. contributed to sample collection. S.Y., C.P., N.K. and S.S. performed the experiments. C.P., S.Y., S.S., W.S. and S.J. performed data analysis. C.P. and S.S. drafted the manuscript. C.P. and S.J. reviewed and finalized the manuscript. All authors approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yanmanee, S., Seethamchai, S., Kuamsab, N. et al. Natural vectors of Plasmodium knowlesi and other primate, avian and ungulate malaria parasites in Narathiwat Province, Southern Thailand. Sci Rep 13, 8875 (2023). https://doi.org/10.1038/s41598-023-36017-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-36017-3
- Springer Nature Limited