Abstract
Osteoporosis (OP) is characterized by diminished bone mass and deteriorating bone structure that increases the chance of fractures in the spine, hips, and wrists. In this paper, a novel data processing method of artificial intelligence (AI) is used for evaluating, predicting, and classifying OP risk factors in clinical data of men and women separately. Additionally, artificial intelligence was used to suggest the most appropriate sports programs for treatment. Data was obtained from dual-energy x-ray absorption scanning center of Ayatollah Kashani, Milad, and Khatam al-Anbia hospitals in Tehran, Iran. The subjects included 1224 men and women. Models were developed using decision tree, random forest (RF), k-nearest neighbor, support vector machine, gradient boosting (GB), Extra trees, Ada Boost (AB), and artificial neural network multilayer perceptron analysis to predict osteoporosis and to recommend sports programs. Data was divided into training (80%) and test dataset (20%). The results were obtained on a 20% test dataset. Area under receiver operating characteristic curve (AUROC) was used to compare the performance of the models. To predict healthy individuals, osteopenia and osteoporosis, the FR algorithm with AUROC 0.91 performed best in men and the GB algorithm with AUROC 0.95 performed best in women compared to other classification algorithms. Prediction of RF algorithm in women and men with AUROC 0.96 and 0.99, respectively, showed the highest performance in diagnosing the type of exercise for healthy individuals and those with osteopenia and OP. Eight AI algorithms were developed and compared to accurately predict osteoporosis risk factors and classify individuals into three categories: healthy, osteopenia, and OP. In addition, the AI algorithms were developed to recommend the most appropriate sports programs as part of treatment. Applying the AI algorithms in a clinical setting could help primary care providers classify patients with osteoporosis and improve treatment by recommending appropriate exercise programs.
Similar content being viewed by others
Introduction
Osteoporosis (OP) is a common metabolic systemic bone disease characterized by increased bone fragility, low bone mass, and a high risk of fractures leading to falls and decreased bone mineral density (BMD)1. OP is considered a great public health problem and the most common metabolic bone disease as it causes more than 8.9 million fractures per year, resulting in one fracture every three seconds and affecting more than 200 million people worldwide2. To increase physicians' awareness of asymptomatic osteoporosis and to identify at-risk patients, it is crucial to understand the risk factors and appropriately diagnose the disease3. Several factors such as gender, age, body mass index (BMI), height, low body weight, adequate levels of physical activity, poor nutritional status, family history, calcium, and vitamin D intake, back pain, and other endocrine and cardiometabolic factors are associated with osteoporosis and very important in diagnosing it during lifetime4,5. Although the most widely used clinical tool for measuring BMD and assessing bone strength is laboratory dual-energy X-ray absorption (DXA), the availability of DXA is very limited6 and does not indicate bone quality7. Therefore, appropriate methods for screening, diagnosis and monitoring of these patients are needed8. Many researchers have also aimed to develop predictive models using risk factors for the screening of osteoporosis9,10.
In recent years, other than traditional modeling, classification algorithms have gained popularity because of their ability to detect more complex relationships between input and output features and flexible modeling11,12. Classification algorithms, using large volumes of data, make new information and relationships embedded in large and complex datasets visible through inferring and learning new patterns and relationships13. At present, the machine learning approach is not sufficient to predict osteoporosis with a larger data set in men and women and requires further study. Hence, the first goal of the present study was to determine osteoporosis risk factors in clinical data comprising of physical characteristics, personal and medical history, and laboratory tests in men and women. By using classification algorithms in clinical practice as a screening tool, both physicians and patients would be more aware of osteoporosis risk factors and take more preventive measures in the early stages of the disease to avoid adverse outcomes.
Drug treatments, fall precautions, and lifestyle changes suggested to patients with osteoporosis have led to a 21–66 percent reduction in fracture risk14. Despite the availability of effective anabolic and anti-absorption drugs, osteoporosis and related fractures remain an unsolvable problem, forcing health organizations to recently launch a “Call to Action” to address the crisis in osteoporosis treatment15. Exaggerated concerns regarding the side effects of some medications have resulted in the use of exercise to prevent osteoporosis16.
Physical activity (PA) is recommended as a safe and low-cost non-pharmacological intervention strategy to change bone risk factors and maintain musculoskeletal health17. It has been shown that the mechanical load resulting from PA increases muscle mass, creates mechanical stress on the skeleton, and increases osteoblast activity18. Various physical activities are effective in preventing and treating OP and AI is effective in data classification and quick access to results19,20. Due to the beneficial effects of PA in the prevention of osteoporosis, physician and patient accessibility to appropriate and effective sports activities is essential21. The second goal of this study was to develop artificial intelligence to propose appropriate exercise protocols for OP patients' improvement.
Materials and methods
Ethics
The present study was a prospective cohort design approved by the Research Ethics Committee of Allameh Tabataba’i University with ID: IR.ATU.REC.1399.038. Subjects provided written informed consent to participate in the study and all research was conducted according to the relevant instructions. In order to protect the privacy of these clinical data, all ethical principles of patients’ rights have been heed and participants’ names were not mentioned.
Study participants
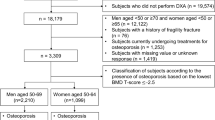
A total of 1224 patients of Ayatollah Kashani, Milad, and Khatam al-Anbia hospitals (Tehran, Iran) in the period 2019–2021 were included in the study. The inclusion criteria comprised of the age range of 35–85 years, with clinical risk factors related to osteoporosis. The sample size using G-Power software with effect size of 0.15, test power of 0.89 and error value of 0.05 was 368 participants. For greater assurance in this study, the sample size was increased to 1224 participants. Patients were separated into women (n = 754) and men groups (n = 470).
Outcome T-score measurements
A dual-energy X-ray absorptiometry (QDR 4500; Hologic, Bedford, MA, USA) was used to measure T-score and BMD in the femoral neck, lumbar spine (L1–L4) and total femur. The most important aspect of DXA interpretation is estimating a patient’s risk of developing an osteoporosis-related fracture. The DXA information includes the BMD, T-score, and Z-score. A T-score indicates the number of standard deviations below or above the BMD average, expressed in grams per square centimeter. This BMD value is compared to the BMD of a population of young adults of the same gender, and several standard deviations (SD) near each of these values. The difference between the mass of the population of young adults of the same gender and the current bone mass of the patient examined is called T-score22. According to World Health Organization (WHO), healthy or normal is diagnosed when the T-score is greater than − 1.0 SD, osteopenia is diagnosed when the T-score is between − 1.5 and − 2.5, and osteoporosis is diagnosed when the T-score is more than − 2.5 SD23.
Feature selection and assessment of covariates
The present study was a prospective cohort design. In this study, by reviewing similar articles and consulting with a specialist physician, and distributing and collecting relevant questionnaires from the subjects, the most significant clinical factors of osteoporosis were prepared. In addition, in a review of relevant books and articles, clinical factors related to osteoporosis were extracted24. Finally, 19 input features for women and 17 input features for men (excluding menopause and menopausal age) were selected as the input of the algorithms.
The input features applied to the algorithms included: age, height, weight, body mass index (BMI), curvature of the spine, family history of osteoporosis, Vitamin D intake, physical activity, back pain, bone fracture, parathyroid disease, history of smoking, and serum levels of calcium, phosphorus, vitamin D, and alkaline phosphatase (ALP). 0 and 1 were used for questions with yes and no answers (0 meaning no, 1 meaning yes). A questionnaire was used to diagnose parathyroid disease. A family history of osteoporosis was considered when at least one first-degree relative was diagnosed with osteoporosis. A physical activity questionnaire was used to assess the level of subject physical activity. Smoking was classified as "never" and "now". For women, menopausal status and menopausal age were also considered. For analysis, the data set in Excel format was transferred to Python version 3.10 (Python Software Foundation, Wilmington, DE, USA).
Subjects had to meet the following criteria: to be aged between 35 and 85 years, male/female gender, BMI of 18 to 40, T-score of 2 to − 3, absence of metabolic disease, rheumatoid arthritis and bone cancer, neither taking corticosteroids nor exercising regularly in the past year. Subjects also had to demonstrate they had no secondary illness, history of surgery or injury, or any physical problems. Table 1 shows the description of the dataset features.
Data analysis to predict Healthy people, osteopenia, osteoporosis and proposing sports protocols
Candidate algorithms in this study included DT, RF, KNN, SVM, GB, ET, AB and ANN. Due to differences in input characteristics, baseline characteristics, and the prevalence of positive predictions, the models were trained separately in men and women Experimental data sets were applied to different models to obtain predictive probabilities of osteopenia, osteoporosis, and disease probability or absence in each model, and according to age and health status in both groups of men and women using the AI methods and to provide suitable sports protocol. For this purpose, all sports therapy protocols collected in scientific articles and classified based on various effective parameters (such as age, gender and health status). Finally, to predict and prescribe the type of exercise, appropriate algorithms were trained.
The model was trained by predicting three groups, which included healthy, osteopenia, and osteoporosis. During the training process, the goal was to predict the multivariate model (1, 2, 3). In the testing process, the prediction results were marked “1” for the healthy group, “2 and 3” for the osteopenia and osteoporosis groups, respectively. For classifying sports, several binary variables were presented (1, 2, 3, 4, 5, 6, 7, 8, 9) shown separately for men and women in Tables 4 and 5.
The separate datasets for men and women were divided into training and testing datasets with an 80:20 split. During each training section of models, used a 20% test dataset to experiment the performance of the models. The data was analyzed, and the algorithm with best performance identified. This resulted in 376 men in training and 94 men in testing datasets, and 603 women in training and 151 women in testing datasets. Figure 1 illustrates data processing in the present study.
DT is the denotative representation of a decision-making process. DT in AI is used to arrive at conclusions based on the data available from decisions made in the past25. The random forest (RF) is comprised of a large number of individual decision trees that act as a collection. Each single tree in the random forest publishes a class prediction, which uses a randomly drawn subsample and a random subset of the available features for each splitting decision25. The KNN algorithm uses 'feature similarity' to predict the values of any new data points. This means that the new point is assigned a value based on how closely it resembles the points in the training set26. SVM falls into the category of supervised learning which is useful for solving classification problems. This method creates the best decision boundary for dividing the next n space into different classes to place the new data points in the appropriate class category. SVM always selects strong vectors to create hyperplanes. These extremes are called backup vectors. The dimensions of the hyperplane are determined by the number of data set features27. GB combines the predictions from multiple decision trees to generate the final predictions. The ET algorithm operates by creating a large number of unpruned decision trees from the training dataset. Predictions are made by averaging the prediction of the decision trees in the case of classification. AB functions by putting more weight on difficult to classify instances and less on those already handled well28. A multilayer perceptron (MLP) is a fully connected class of feedforward artificial neural network (ANN). The term MLP is used ambiguously, sometimes loosely to mean any feedforward ANN, sometimes strictly to refer to networks composed of multiple layers of perceptrons. MLP is a feedforward artificial neural network that generates a set of outputs from a set of inputs. An MLP is characterized by several layers of input nodes connected as a directed graph between the input and output layers29. The process and the final selected hyperparameters for each model are presented in Table 2.
Various combinations of the number of layers (1 or 40 hidden layers), learning rate (from 0.01 to 0.0001) were investigated in the ANN model for hyperparameter tuning. For the DT model, the depth and leaf parameters and SVM, the kernel type (linear, polynomial, or radial basis function), regularization parameter C (from 2–2 to 29) and for the RF model, the max depth (from 3 to 13), random state (from 3 to 17), were examined; for the KNN model, the number of neighbors (from 1 to 10) were tested. All models were created with a balanced class weight. Randomization for the split of validation dataset and the training process was repeated numerous times for each hyperparameter set. The area under the receiver operating characteristic curve (AUROC) was estimated for the test dataset in each training process, and the mean values of AUROC were compared. The Area Under the Curve (AUC) is a measure of the capability of a classifier to distinguish between classes and is utilized as a summary of the ROC curve. The greater the AUC, the better the performance of the model at discerning positive and negative classes. Other values were calculated in the performance of the algorithms for the data set in each training process including accuracy, precision, sensitivity, specificity, and F-score. Accuracy is an evaluation criteria of classification models and is a fraction of the predictions that the model has made accurately (Eq. 1). Precision is a metric calculating the percentage of correct predictions for the positive class. Maximizing precision will minimize the false-positive errors, whereas maximizing recall will minimize the false-negative errors (Eq. 2). Sensitivity (also known as recall) estimates the percentage of correct predictions for the positive class out of all possible positive predictions made (Eq. 3). Specificity calculates the proportion of true negatives correctly identified by the model (Eq. 4). The F-measure, also named the F-score, is used to verify the accuracy of a model in a data set. F-score is widely used in evaluation of information retrieval systems such as search engines and many types of machine learning models. In addition, the F-measure is a configurable single-score metric for evaluating a binary classification model based on the predictions made for the positive class (Eq. 5). The F-measure is estimated using sensitivity and precision30. The following criteria were calculated to evaluate the performance of the predicted models.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Allameh Tabataba’i University with research ID Code: IR.ATU.REC.1399.038. Subjects provided written informed consent to participate in this study and all research was conducted according to the relevant instructions.
Results
1224 subjects participated in the study (men: n = 470 and women: n = 754). The results of DXA showed that 72 men and 149 women had osteoporosis, 233 men and 427 women had osteopenia, and 165 men and 178 women were healthy. The demographic information is summarized in Table 3.
Step 1 At this stage, 8 classification algorithms (DT, RF, KNN, SVM, GB, ET, AB, ANN) were used to predict and divide people into three categories: healthy, osteopenia and osteoporosis.
19 features for women and 17 features for men (excluding menopause and menopausal age) were selected as input characteristics of these algorithms with three objectives of healthy (T-score > − 1.0), osteopenia (T-score between − 1.5 and − 2.5) and osteoporosis (T-score ≤ − 2.5).
According to the results of Table 3 machine learning models achieved 73–85% accuracy, 71–90% precision, 64–76% sensitivity and 81–89% specificity in classifying men participants into the three categories of healthy, osteopenia, and osteoporosis. To predict healthy, osteopenia, and osteoporosis in women, machine learning models achieved an accuracy of 75–93%, a precision of 74–93%, a sensitivity of 75–91% and a specificity of 82–95%. In addition, the AUROC between machine learning models were compared. The RF algorithm showed the highest performance in the men group with AUROC 0.91, and in the women's group, the GB algorithm with AUROC 0.95 performed best. The ROC curves of different models of machine learning in men and women are presented in Figs. 2 and 3 (Table 4).
Step 2 In the second stage, sports protocols were proposed to healthy individuals and those with osteopenia and osteoporosis using 8 classification algorithms and 9 labels to predict exercise protocols for men and women separately.
Table 5 shows the recommended exercises for men in the three categories of health status. Numbers 1, 2, and 3 denote healthy men’s (T-score > − 1.0) age ranges of 35–50, 51–65 and 66–85, respectively; numbers 4, 5 and 6 correspond to the same age ranges of 35–50, 51–65 and 66–85 in men with osteopenia (T-score between − 1.5 and − 2.5); and finally the last category of men with osteoporosis (T-score ≤ − 2.5) is represented by numbers 7, 8 and 9 with respective age ranges of 35–50, 51–65 and 66–85.
Table 6 indicates the recommended exercises for women in the three categories of health status. Numbers 1, 2, and 3 denote healthy women's (T-score > − 1.0) age ranges of 35–50, 51–65 and 66–85, respectively; numbers 4, 5 and 6 correspond to the same age ranges of 35–50, 51–65 and 66–85 in women with osteopenia (T-score between − 1.5 and − 2.5); and finally the last category of women with osteoporosis (T-score ≤ − 2.5) is represented by numbers 7, 8 and 9 with respective age ranges of 35–50, 51–65 and 66–85.
In Table 7, the results of the men and women data classification algorithms for the proposed sports protocols are compared.
Based on the findings in Table 7, predicting exercise appropriate for healthy male individuals and those with osteopenia and osteoporosis, machine learning models achieved 56–84% accuracy, 45–78% precision, 50–69% sensitivity and 87–97% specificity. To predicting exercise appropriate for healthy female individuals and those with osteopenia and osteoporosis, machine learning models achieved an accuracy of 53–98%, a precision of 45–96%, a sensitivity of 37–97% and a specificity of 89–99%. The AB algorithm had the best performance in men and women (AUROC of 0.96, and 0.99, respectively). The ROC curves of different models of machine learning in men and women are presented in Figs. 4 and 5.
Discussion
In the present study, various AI algorithms were used to predict and classify subjects into three categories of healthy, osteopenia and OP as well as to recommend appropriate sports programs. Accuracy was used to evaluate the prediction performance of algorithms.
In the first stage, The RF algorithm showed the highest performance in the men group with AUROC 0.91, and in the women's group, the GB algorithm with AUROC 0.95 performed best to identify healthy individuals, osteopenia and OP. Numerous studies have been previously carried out on diagnosing osteoporosis using AI.
A study published in 2016 by Yu et al.31 with a study sample of 119 hospitalized patients (49 men and 79 women) with average age of 65 years to diagnose osteoporosis using AI demonstrated that 55 patients had osteoporosis while 64 patients did not. In another study which targeted 1,792 postmenopausal women published in 2020 by Shim et al.32 compared seven machine learning models; the best performance was shown by the RF model with AUROC of 0.763. Ilio et al.33 used a set of 589 records extracted from the Greek population to measure bone density. In this study, three and five diagnostic factors were considered to predict the risk of osteoporosis, and finally, using multilayer perceptron classification, individuals were classified into three categories of normal, osteopenia and osteoporosis. In 2021, Wang et al.34 studied 1559 Chinese women over the age of 20 to develop an ANN model using age and weight as input for prediction of osteoporosis, achieving an AUROC of 0.78. A study in 2021 by Yang et al.3 entitled Development of Machine Learning Models for Prediction of Osteoporosis from Clinical Health Examination Data used a sample population of 3053 Taiwanese men and 2929 women; in this research, the best AUROC of 0.843 and 0.811 in men and in women, respectively was achieved with the RF algorithm for predicting osteoporosis. The following input characteristics were used: medical history of diabetes and hypertension, history of smoking and alcohol consumption liver function, thyroid function, lipid profile, blood protein content, electrolytes, hematological profile, renal function, and for women, history of obstetrics and gynecology were also included. Among male patients, secondary causes such as alcohol abuse, steroid therapy, and other metabolic disorders account for up to 65% of cases of osteoporosis3. In contrast, the prevalence of secondary osteoporosis is much lower in women than in men35. In women, estrogen deficiency after menopause and osteoporosis is one of the main causes of osteoporosis36. In the present study, for men and women, due to differences in cause and baseline characteristics, prediction algorithms were trained separately, obtaining acceptable results. Feature selection was based on accessibility and known relevance to bone health. In comparison with previous research, this had the benefits of predicting osteopenia and osteoporosis with a larger sample size selection (1224 patients) and the inclusion of more input features (17 features in men and 19 features in women) from different aspects. Reducing the number of variables (features) leads to poorer performance and the inclusion of more features contributes to better performance of AI models3. Input features were selected based on known association with bone health and easy availability.
Proposing appropriate and separate sport programs for healthy individuals and those with osteopenia and osteoporosis, the AB algorithm had the best performance in men and women (AUROC of 0.96, and 0.99, respectively).
To the best of our knowledge, the present research study is the first to use AI methods to recommend sports programs to individuals with osteoporosis and osteopenia. Age, and health status (healthy, osteopenia, and osteoporosis) were used as input to the algorithm, and appropriate exercise programs for improvement and treatment were selected based on effective and meaningful protocols mentioned in ISI authoritative articles. This research had the capability of recommending reliable exercise programs with different intensities and amounts based on patients' health status.
In the current study, accuracy was used to observe the performance of algorithms and categorize individuals into healthy, osteopenia, and osteoporosis categories, and accordingly propose appropriate exercise protocols. The great strengths of the study included the identification of a large number of risk factors directly associated with an increased risk of fracture. However, the present study has limitations such as removing inaccessible indicators of osteoporosis risk factors and not taking into consideration the alcohol consumption index because all recorded responses were negative. In addition, data from only three hospitals were included in this study and as such might not represent the entire population.
Conclusion
Osteoporosis as a silent disease affects individuals worldwide and causes long-term loss of movement, serious injuries, severe pain, and even premature death. Using AI to predict risk groups can help the economy and reduce the burden on health systems. The results of our study showed that AI models with high accuracy and using several clinical and physiological indicators had acceptable performance in classifying individuals at risk for osteoporosis. Our study also demonstrated that AI algorithms have acceptable performance for accessing appropriate sport protocols, and by incorporating these algorithms into clinical practice, both physicians and patients can easily reap the benefits.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- OP:
-
Osteoporosis
- AI:
-
Artificial intelligence
- DXA:
-
Dual-energy x-ray absorption
- BMD:
-
Bone mineral density
- PA:
-
Physical activity
- AUROC:
-
Area under the receiver operating characteristic curve
- DT:
-
Decision tree
- RF:
-
Random forest
- KNN:
-
K-nearest neighbor
- SVM:
-
Support vector machine
- GB:
-
Gradient boosting
- ET:
-
Extra trees
- AB:
-
Ada Boost
- ANN:
-
Artificial neural networks
- MLP:
-
Multilayer perceptron
References
Sirufo, M. M., De Pietro, F., Bassino, E. M., Ginaldi, L. & De Martinis, M. Osteoporosis in skin diseases. Int. J. Mol. Sci. 21(13), 4749 (2020).
Hussein, R. S. & Wahdan, M. M. Knowledge about symptoms and risk factors of osteoporosis among adult women in Cairo, Egypt. Egypt. J. Commun. Med. 39(2) (2021).
Ou Yang, W.-Y., Lai, C.-C., Tsou, M.-T. & Hwang, L.-C. Development of machine learning models for prediction of osteoporosis from clinical health examination data. Int. J. Environ. Res. Public Health 18(14), 7635 (2021).
Bijelic, R., Milicevic, S. & Balaban, J. Risk factors for osteoporosis in postmenopausal women. Med. Arch. 71(1), 25 (2017).
Aspray, T. J. & Hill, T. R. Osteoporosis and the ageing skeleton. Biochem. Cell Biol. Ageing Part II Clin. Sci. 91, 453–476 (2019).
Looker, A. C., Melton, L. J. III., Harris, T. B., Borrud, L. G. & Shepherd, J. A. Prevalence and trends in low femur bone density among older US adults: NHANES 2005–2006 compared with NHANES III. J. Bone Miner. Res. 25(1), 64–71 (2010).
McLaughlin, E. C. Cause or Consequence? (University of Waterloo, 2021).
Abdalbaryb, K. A. M. & Kakanib, Y. A. E. Bone quality in CKD patients: Current concepts and future directions-Part I. Kidney Dis. 7, 268–277 (2021).
Forgetta, V. et al. Development of a polygenic risk score to improve screening for fracture risk: A genetic risk prediction study. PLoS Med. 17(7), e1003152 (2020).
Zhang, B. et al. Deep learning of lumbar spine X-ray for osteopenia and osteoporosis screening: A multicenter retrospective cohort study. Bone 140, 115561 (2020).
Nam, K. H. et al. Machine learning model to predict osteoporotic spine with hounsfield units on lumbar computed tomography. J. Korean Neurosurg. Soc. 62(4), 442 (2019).
Benke, K. & Benke, G. Artificial intelligence and big data in public health. Int. J. Environ. Res. Public Health 15(12), 2796 (2018).
Vatansever, S. et al. Artificial intelligence and machine learning-aided drug discovery in central nervous system diseases: State-of-the-arts and future directions. Med. Res. Rev. 41(3), 1427–1473 (2021).
Yusuf, A. A. et al. Real-world effectiveness of osteoporosis therapies for fracture reduction in post-menopausal women. Arch. Osteoporos. 13(1), 1–10 (2018).
DegliEsposti, L. et al. Use of antiosteoporotic drugs and calcium/vitamin D in patients with fragility fractures: Impact on re-fracture and mortality risk. Endocrine 64(2), 367–377 (2019).
Waltman, N. et al. Bone-loading exercises versus risedronate for the prevention of osteoporosis in postmenopausal women with low bone mass: A randomized controlled trial. Osteoporos. Int. 33(2), 475–486 (2022).
Marini, S. Effects of Adapted Physical Activity on Quality of Life, Physical Fitness and Fear of Falling in People with Osteoporosis (2021).
Negri, S., Samuel, T. J. & Lee, S. The potential role of exercise training and mechanical loading on bone-associated skeletal nerves. J. Bone Metab. 28(4), 267 (2021).
Senderovich, H. & Kosmopoulos, A. An insight into the effect of exercises on the prevention of osteoporosis and associated fractures in high-risk individuals. Rambam Maimonides Med. J. 9(1), e0005 (2018).
Pinheiro, M. B. et al. Evidence on physical activity and osteoporosis prevention for people aged 65+ years: A systematic review to inform the WHO guidelines on physical activity and sedentary behaviour. Int. J. Behav. Nutr. Phys. Act. 17(1), 1–53 (2020).
Tong, X. et al. The effect of exercise on the prevention of osteoporosis and bone angiogenesis. BioMed Res. Int. https://doi.org/10.1155/2019/8171897 (2019).
Aldieri, A. et al. Osteoporotic hip fracture prediction: Is T-score-based criterion enough? A hip structural analysis-based model. J. Biomech. Eng. 10(1115/1), 4040586 (2018).
Prevention WSGo, Osteoporosis Mo, Organization WH. Prevention and Management of Osteoporosis: Report of a WHO Scientific Group (World Health Organization, 2003).
Miyagi, M. et al. Factors associated with pain-related disorders and gait disturbance scores from the Japanese orthopedic association back pain evaluation questionnaire and Oswestry Disability Index in patients with osteoporosis. Arch. Osteoporos. 17(1), 1–8 (2022).
Islam, M. Z., Liu, J., Li, J., Liu, L. & Kang, W. (eds.) A semantics aware random forest for text classification. In Proceedings of the 28th ACM International Conference on Information and Knowledge Management (2019).
Cui, L., Zhang, Y., Zhang, R. & Liu, Q. H. A modified efficient KNN method for antenna optimization and design. IEEE Trans. Antennas Propag. 68(10), 6858–6866 (2020).
Bisgin, H. et al. Comparing SVM and ANN based machine learning methods for species identification of food contaminating beetles. Sci. Rep. 8(1), 1–12 (2018).
Tanha, J., Abdi, Y., Samadi, N., Razzaghi, N. & Asadpour, M. Boosting methods for multi-class imbalanced data classification: An experimental review. J. Big Data 7(1), 1–47 (2020).
Kruthi, C., Tejashwini, H., Poojitha, G., Shreelakshmi, H. & Shobha Chandra, K. Detection of Autism Spectrum Disorder Using Machine Learning (2021).
Chakraborty, P., Nawar, F. & Chowdhury, H. A. Sentiment analysis of Bengali facebook data using classical and deep learning approaches. In Innovation in Electrical Power Engineering, Communication, and Computing Technology 209–218 (Springer, 2022).
Yu, X., Ye, C. & Xiang, L. Application of artificial neural network in the diagnostic system of osteoporosis. Neurocomputing 214, 376–381 (2016).
Shim, J.-G. et al. Application of machine learning approaches for osteoporosis risk prediction in postmenopausal women. Arch. Osteoporos. 15(1), 1–9 (2020).
Iliou, T., Anagnostopoulos, C.-N., Stephanakis, I. M. & Anastassopoulos, G. A novel data preprocessing method for boosting neural network performance: A case study in osteoporosis prediction. Inf. Sci. 380, 92–100 (2017).
Wang, Y. et al. Prediction model for the risk of osteoporosis incorporating factors of disease history and living habits in physical examination of population in Chongqing, Southwest China: based on artificial neural network. BMC Public Health 21(1), 1–10 (2021).
Rinonapoli, G. et al. Osteoporosis in men: A review of an underestimated bone condition. Int. J. Mol. Sci. 22(4), 2105 (2021).
Hemmati, E. et al. Prevalence of primary osteoporosis and low bone mass in postmenopausal women and related risk factors. J. Educ. Health Promot. 10, 204 (2021).
Acknowledgements
The authors thank the participants of the study. In addition, we thank Ayatollah Kashani, Milad, and Khatam Al-Anbia hospitals for granting access to the subjects and data support.
Funding
This research was a part of the MSc dissertation of the first author at Department of Exercise Physiology of Allameh Tabataba’i University. This research received no external funding.
Author information
Authors and Affiliations
Contributions
Intellectual concept and design of the research, B.T. and L.F.; Acquisition of data, L.F.; Analysis of data, H.F. and L.F.; Interpretation of data, all authors; Original writing L.F.; Critical editing of written text, B.T. and R.E.; Approval of the definitive version of the manuscript, B.T. and R.E.; Accountable for legal and ethical aspects of the work, B.T. and L.F.; Accountable for all other aspects of the work, B.T. and L.F.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fasihi, L., Tartibian, B., Eslami, R. et al. Artificial intelligence used to diagnose osteoporosis from risk factors in clinical data and proposing sports protocols. Sci Rep 12, 18330 (2022). https://doi.org/10.1038/s41598-022-23184-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-23184-y
- Springer Nature Limited