Abstract
Rheumatoid arthritis (RA) patients often exhibit finger/wrist joint symptoms and reduced grip strength. This study aimed to validate grip strength as a measure of frailty in RA patients. Subjects were 424 female RA patients (mean age ± standard deviation, 66.8 ± 14.5 years). Frailty was defined as a score of ≥ 8 points on the Kihon Checklist (KCL). Finger/wrist joint symptoms were defined based on tender or swollen joints. Associations between frailty and grip strength were determined using receiver operating characteristic (ROC) curve analysis and multivariable logistic regression analysis. There were 179 subjects with frailty (42.2%). Multivariable logistic regression analysis revealed that frailty was significantly associated with grip strength independently of finger/wrist joint symptoms. In ROC curves, cut-off scores of grip strength for frailty in subjects without and with finger/wrist joint symptoms were 17 kg (sensitivity, 62.1%; specificity, 69.0%) and 14 kg (sensitivity, 63.2%; specificity, 73.0%), respectively. The results of the present study suggest that grip strength in female RA patients is associated with frailty, with a cut-off score of 17 kg (equivalent to Cardiovascular Health Study criteria, < 18 kg) when RA patients have no finger/wrist joint symptoms. However, when RA patients have finger/wrist joint symptoms, it may be considered to reduce the cut-off score of grip strength.
Similar content being viewed by others
Introduction
In a rapidly advancing aging society, a general goal for reducing nursing costs and improving quality of life is to maintain and improve physical function1,2. Frailty is a condition that leads to an increased need for long-term care and defined as the intermediate stages of health and disability with age-related declines in physical and cognitive function3. Various negative health outcomes, including COVID-19-related mortality are significantly associated with frailty4,5. Frailty can reduce healthy life expectancy, however, it can also be recovered with proper interventions since it is clinically considered as the condition of predisability3.
Rheumatoid arthritis (RA) is a chronic systemic inflammatory autoimmune disease which can lead to joint destruction and physical dysfunction due to synovial inflammation, as well as physical frailty6. The proportion of RA patients with frailty is estimated to be higher than that of the general population7, and the aging of RA patients is progressing8,9. As RA treatment has advanced significantly with the introduction of methotrexate, biologics, and Janus kinases inhibitors, the survival rate of RA patients has also improved in tandem10. In an aging society, especially in one like that of Japan, the extension of health life expectancy is important also for RA patients.
Grip strength reflects upper limb function as well as whole body physical function11. It has been reported that a decline in grip strength was associated with mortality, and monitoring grip strength may improve the identification of women at greatest risk of death12. Decreased function and muscle weakness have also been evaluated by grip strength in cancer patients13. Reduced grip strength is one of the Cardiovascular Health Study (CHS) criteria for diagnosing frailty3,14. A major benefit of grip strength is that it is easy to measure in clinical settings.
Findings from previous studies have highlighted the importance of detecting frailty early and preventing it in order to promote good health outcomes. However, RA patients often exhibit finger/wrist joint symptoms and reduced grip strength. Accordingly, this study aimed to validate the use of grip strength as a measure of frailty in RA patients, taking into consideration disease activity, especially finger/wrist joint symptoms.
Materials and methods
Subjects
This study included the RA patients who consecutively visited Japanese Red Cross Nagoya Daiichi Hospital, Japan Community Health Care Organization Kani Tono Hospital, and Yokkaichi Municipal Hospital between June and August 2021. All patients fulfilled the 2010 American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) classification criteria15. This study targeted only female RA patients. Cut-off scores of CHS criteria for males and females differ (i.e., < 18 kg for females and < 28 kg for males)3,14. Since this study aimed to confirm the validity of the cut-off score of grip strength, we first targeted females given the small number of male patients in the source population. Comorbidities were defined as diseases that were currently or previously treated by other medical specialists and included diabetes mellitus, hypertension, osteoporosis, interstitial pneumonia, and ever or current malignancy.
This retrospective study was approved by the Ethics Committees of Nagoya University School of Medicine (2017-0271), Japanese Red Cross Nagoya Daiichi Hospital (2020-451), Japan Community Health Care Organization Kani Tono Hospital (20110901), and Yokkaichi Municipal Hospital (2017-29). We disclosed information pertaining to the study at the cooperating facilities according to the procedure stipulated by the respective Ethics Committees. Informed consent was obtained from all subjects. The study was conducted in accordance with the World Medical Association of Helsinki ethical principles for medical research involving human subjects. Patients’ individual information was anonymized.
Frailty
Frailty categories were defined based on Kihon Checklist (KCL) scores (≥ 8 points corresponds to frailty; 4–7 points corresponds to pre-frailty; 0–3 points corresponds to normal16), which is a widely used instrument developed by the Ministry of Health, Labour and Welfare in Japan to identify older people at risk of requiring care/support17. KCL consists of 25 self-reporting yes/no questions in total, including seven domains: activities of daily living, physical strength, nutrition, oral function, isolation, cognitive function, and depressive mood16,17. KCL was reported to be significantly correlated with Fried’s Cardiovascular Health Study criteria, and its validity as a screening tool to assess frailty has been demonstrated3,16.
Grip strength
Grip strength was measured with the elbow fully extended in the standing position, using Smedley spring handgrip dynamometers (TTM Smedley Dynamo Meter; Tsutsumi, Tokyo, Japan). Measurements were carried out twice with the right hand and the left hand respectively, and the higher value was used for analysis, according to Asian Working Group for Sarcopenia18.
Finger/wrist joint symptoms
First, we defined the dominant side as the higher value of grip strength. Then, finger/wrist joint symptoms were defined as tender and/or swollen joints on the dominant side. Tender and swollen joints referred to articular and periarticular manifestations including joint tenderness and swelling to palpation respectively19. Tender and swollen 28-joint count meant the number of tender and swollen joints out of 28 joints (knee, shoulder, elbow, wrist, metacarpophalangeal, proximal interphalangeal joints)20, and were incorporated into Disease Activity Score 28- C-reactive protein (DAS28-CRP): DAS28-CRP = 0.56*√(tender 28-joint count) + 0.28*√(swollen 28-joint count) + 0.014*(subject’s global assessment of disease activity visual analog scale) + 0.36*ln(CRP + 1) + 0.9621. DAS28-CRP was categorized as follows: clinical remission (DAS28-CRP < 2.3); low disease activity (LDA; 2.3 ≤ DAS28-CRP < 2.7); moderate disease activity (MDA; 2.7 ≤ DAS28-CRP ≤ 4.1); and high disease activity (HDA; DAS28-CRP > 4.1)22,23.
Health Assessment Questionnaire-Disability Index (HAQ-DI)
HAQ-DI is 20 self-reporting questions consisting of eight categories (Dressing and grooming, Arising, Eating, Walking, Hygiene, Reaching, Gripping, and Other activities), and widely used as an index of physical function in RA patients24. HAQ-DI is calculated as the average of the highest score of questions in each category, and response coding scores of questions are as follows: “without any difficulty” = 0, “with some difficulty” = 1, “with much difficulty” = 2, and “unable to do” = 324. HAQ-DI ≤ 0.5 is defined as functional remission25.
Statistical analysis
Continuous variables are expressed as mean and standard deviation (SD) and were analyzed using the unpaired t-test. Ordinal variables and categorical variables are expressed as percentages and were analyzed using Fisher’s exact test. For the purpose of clarifying the associations between HAQ-DI and grip strength, and DAS28-CRP and finger/wrist joint symptoms, linear regression analyses adjusted for age and duration of disease were performed. Multivariable logistic regression analyses were performed to confirm the independent impact of related variables (e.g., grip strength) on frailty. Receiver operating characteristic (ROC) curves were generated to assess the associations between frailty, clinical remission (DAS28-CRP < 2.3)23, functional remission (HAQ-DI ≤ 0.5)25, and grip strength with or without finger/wrist joint symptoms. The point closest to the upper left corner was identified as the best cut-off point. Univariate analysis of variance was performed, and estimated marginal means of grip strength were calculated with age, duration of disease, and BMI as covariates, and stratifying by frailty and finger/wrist joint symptoms.
Statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan; http://www.jichi.ac.jp/saitama-sct/SaitamaHP.files/statmed.html), a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria)26, and SPSS version 28.0.0 software (IBM Corp., Armonk, NY, USA). P < 0.05 was considered statistically significant.
Results
A total of 630 RA patients consecutively visited our hospitals between June and August 2021. Among these patients, data on clinical characteristics, including scores for grip strength, KCL, and DAS28-CRP were available for 591. Among these 591 patients, 424 were female. Subject characteristics are summarized in Table 1. 179 (42.2%) of 424 subjects had frailty. Mean age, DAS28-CRP, HAQ-DI, grip strength, and the proportions of those with finger/wrist joint symptoms, diabetes mellitus, hypertension, osteoporosis, and ever or current malignancy were all significantly different between those with and without frailty.
Table 2 shows significant regression coefficients of finger/wrist joint symptoms and grip strength for DAS28-CRP and HAQ-DI respectively. In ROC curves, cut-off scores of DAS28-CRP for finger/wrist joint symptoms were 2.2 (area under curve (AUC), 0.881; sensitivity, 82.7%; specificity, 76.9%), which corresponded to remission of disease activity23. Thus, we found that finger/wrist joint symptoms were significantly associated with DAS28-CRP, and grip strength was significantly associated with HAQ-DI.
Table 3 shows ORs for frailty based on multivariable logistic regression analyses. Since this study aimed to validate grip strength instead of HAQ-DI as a measure of frailty, we decided not to include HAQ-DI in the same model as grip strength. DAS28-CRP was entered as a variable in Model 1, whereas finger/wrist joint symptoms was entered as a variable in Model 2 (with comorbidities) and Model 3 (without comorbidities). Frailty was significantly associated with age, body mass index (BMI), DAS28-CRP (Model 1), finger/wrist joint symptoms (Model 3), and grip strength (Model 1/Model 2/Model 3).
In the ROC curve analysis for frailty in subjects without finger/wrist joint symptoms, the best cut-off score of grip strength corresponding to frailty was 17 kg (sensitivity, 62.1%; specificity, 69.0%) (Fig. 1a). In subjects with finger/wrist joint symptoms, the best cut-off score of grip strength corresponding to frailty was 14 kg (sensitivity, 63.2%; specificity, 73.0%) (Fig. 1b).
ROC curves for frailty and grip strength (a) without finger/wrist joint symptoms, and (b) with finger/wrist joint symptoms. Area under curve and Cut-offs were (a) 0.697 (95% CI: 0.633–0.761) and 17 kg (sensitivity, 62.1%; specificity, 69.0%), and (b) 0.726 (95% CI: 0.645–0.808) and 14 kg (sensitivity, 63.2%; specificity, 73.0%).
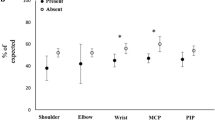
Figure 2 shows estimated marginal means of grip strength adjusted for age, duration of disease, and BMI as covariates. Mean grip strength values were 19.8 kg (95% CI: 18.9–20.7) in subjects with no frailty and no finger/wrist joint symptoms, 16.2 kg (95% CI: 15.0–17.3) in subjects with frailty and no finger/wrist joint symptoms, 17.0 kg (95% CI: 15.6–18.4) in subjects with no frailty and finger/wrist joint symptoms, and 13.4 (95% CI: 12.0–14.7) in subjects with frailty and finger/wrist joint symptoms.
Estimated marginal means of grip strength with age, duration of disease, and BMI as covariates. In order from left to right, subjects with no frailty and no finger/wrist joint symptoms (n = 171, 40.3%), with frailty and no finger/wrist joint symptoms (n = 103, 24.3%), with no frailty and finger/wrist joint symptoms (n = 74, 17.5%), and with frailty and finger/wrist joint symptoms (n = 76, 17.9%).
In ROC curve analysis, cut-off scores of grip strength for clinical remission (DAS28-CRP < 2.3)23/functional remission (HAQ-DI ≤ 0.5)25 in subjects without finger/wrist joint symptoms and with finger/wrist joint symptoms were 16 kg (sensitivity, 66.1%; specificity, 68.7%/sensitivity, 80.0%; specificity, 75.5%) and 14 kg (sensitivity, 51.7%; specificity, 76.5%/sensitivity, 69.6%; specificity, 75.3%), respectively, which were similar to cut-off scores of grip strength corresponding to frailty. When subjects were dichotomized according to CHS grip strength criteria to diagnose frailty (i.e., < 18 kg for females)3,14, the proportion of those with frailty was 58.5/26.7% (grip strength < 18 kg / ≥ 18 kg), the proportion of those with finger/wrist joint symptoms was 43.5/27.6%, and mean grip strength (SD) was 11.2 (4.5)/23.2 (4.4) kg, which were all significantly different between those with grip strength < 18 kg vs those with grip strength ≥ 18 kg.
Discussion
The present study, to our knowledge, is the first attempt to investigate the association between grip strength and frailty in RA patients, with a specific focus on finger/wrist joint symptoms (i.e., tender and swollen joints) and disease activity (DAS28-CRP). Frailty was significantly associated with grip strength independently of age, comorbidities, and disease activity, which were previously reported to be factors related to frailty27, as well as finger/wrist joint symptoms. While the cut-off score of grip strength corresponding to frailty in RA patients without finger/wrist joint symptoms was 17 kg, which was similar to one of the CHS criteria for diagnosing frailty (i.e., < 18 kg for females)3,14, the cut-off score of grip strength in RA patients with finger/wrist joint symptoms was 14 kg, which was lower than that in those without finger/wrist joint symptoms. Furthermore, the upper limit of 95% CI for mean grip strength in subjects with frailty and no finger/wrist joint symptoms was 17.3 kg, and that for those with frailty and finger/wrist joint symptoms was 14.7 kg, indicating that RA patients with no finger/wrist joint symptoms having a grip strength less than 18 kg or RA patients with finger/wrist joint symptoms having a grip strength less than 15 kg are frailty. These findings suggest that grip strength in RA patients reflects frailty regardless of finger/wrist joint symptoms and disease activity. The cut-off score of grip strength corresponding to frailty is < 18 kg (equivalent to CHS criteria) when RA patients have no finger/wrist joint symptoms. However, when RA patients have finger/wrist joint symptoms, it may be considered to reduce the cut-off score of grip strength.
Finger/wrist joint symptoms were significantly associated with DAS28-CRP. In daily clinical practice, compared to the calculations of DAS28-CRP, information on finger/wrist joint symptoms can be obtained simply. We also found that grip strength was significantly associated with HAQ-DI. Based on the results of this study, we propose a screening method for evaluating frailty that uses grip strength in combination with finger/wrist joint symptoms. Actually, it is better to calculate DAS28-CRP and HAQ-DI to evaluate the condition of RA patients in detail. However, according to one report, grip strength of RA patients in remission of disease activity was almost equivalent to that of the healthy population28. Moreover, grip strength can be measured more easily than lower limb muscle strength and done in a shorter time than answering the questions of KCL, and finger/wrist joint symptoms can be identified immediately. Accordingly, it will be important to clarify which RA patients can be evaluated for frailty using grip strength, and when necessary, to lower the grip strength cut-off score when the patients have finger/wrist joint symptoms. The significance of our present findings is that it reveals the need to reduce the cut-off score of grip strength to predict frailty in RA patients with finger/wrist joint symptoms.
Cut-off scores of grip strength corresponding to clinical remission (DAS28-CRP < 2.3) and functional remission (HAQ-DI ≤ 0.5) were 16 kg (without finger/wrist joint symptoms) and 14 kg (with finger/wrist joint symptoms), respectively, which were similar to the cut-off score of grip strength corresponding to frailty. It was reported that frailty had significant association with disease activity27 and HAQ29. Notably, maintaining grip strength from the perspective of preventing frailty may lead to also aiming for clinical and functional remission, which can be the underlying goal of care in RA patients.
Factors that may influence grip strength in RA patients include age30 and disease activity31. In this study, even when adjusting for these factors, grip strength was significantly associated with frailty. Furthermore, reduced grip strength may also result from sarcopenia in RA patients32. While higher grip strength is reportedly associated with lower levels of inflammation, leading to lower mortality33, hand involvement in early inflammatory arthritis has been shown to be a strong predictor of a poor long-term disease outcome34. These results suggest that regular follow-up of grip strength and finger/wrist joint symptoms in RA patients may allow for the determination that disease activity is changing (worsening) and the prevention of frailty and the negative health outcomes. Adequate exercise can reduce pain in RA patients35 and improve grip strength36. From the perspective of preventing frailty, exercise may have a synergistic effect when performed in combination with RA drug therapy.
This study has several limitations. First, radiographic evaluations were not performed. However, we believe our findings are valid because tender and swollen joints could lead to reduced grip strength regardless of radiographic changes. Second, this study targeted only Japanese female RA patients. It will be important to conduct a study with a larger male RA patient population to confirm the cut-off score for males. Third, we did not obtain information regarding the history of upper limb surgery, which can affect grip strength37. Finally, physical factors of lower limb function such as walking time and psychosocial factors such as depression, anxiety, and social support, which are related to frailty38, were not evaluated in detail. Since some RA patients do not have frailty despite having poor grip strength, evaluating frailty based only on upper limb function is insufficient. Nonetheless, upper limb function measurements are simple to perform and may serve as a screening index for evaluating frailty in daily clinical practice.
In conclusion, we investigated the association between grip strength and frailty in RA patients. Frailty was significantly associated with grip strength, independently of age, disease activity, and finger/wrist joint symptoms. Measuring grip strength and checking finger/wrist joint symptoms offer a useful and simple way to assess frailty in daily clinical practice. Our findings serve as a foundation for the development of methods to detect and screen for frailty, as well as interventions.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Kontis, V. et al. Future life expectancy in 35 industrialised countries: projections with a Bayesian model ensemble. Lancet (London, England) 389, 1323–1335. https://doi.org/10.1016/s0140-6736(16)32381-9 (2017).
Freedman, V. A. et al. Resolving inconsistencies in trends in old-age disability: Report from a technical working group. Demography 41, 417–441. https://doi.org/10.1353/dem.2004.0022 (2004).
Fried, L. P. et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 56, M146-156. https://doi.org/10.1093/gerona/56.3.m146 (2001).
Hewitt, J. et al. The effect of frailty on survival in patients with COVID-19 (COPE): A multicentre, European, observational cohort study. Lancet Public Health 5, e444–e451. https://doi.org/10.1016/s2468-2667(20)30146-8 (2020).
Vermeiren, S. et al. Frailty and the prediction of negative health outcomes: A meta-analysis. J. Am. Med. Dir. Assoc. 17(1163), e1161-1163.e1117. https://doi.org/10.1016/j.jamda.2016.09.010 (2016).
Sobue, Y. et al. Relationship between locomotive syndrome and frailty in rheumatoid arthritis patients by locomotive syndrome stage. Mod. Rheumatol. 32, 546–553. https://doi.org/10.1093/mr/roab024 (2021).
Tada, M., Yamada, Y., Mandai, K. & Hidaka, N. Correlation between frailty and disease activity in patients with rheumatoid arthritis: Data from the CHIKARA study. Geriatr. Gerontol. Int. 19, 1220–1225. https://doi.org/10.1111/ggi.13795 (2019).
Kojima, M. et al. Epidemiological characteristics of rheumatoid arthritis in Japan: Prevalence estimates using a nationwide population-based questionnaire survey. Mod. Rheumatol. 30, 941–947. https://doi.org/10.1080/14397595.2019.1682776 (2020).
Kato, E. et al. The age at onset of rheumatoid arthritis is increasing in Japan: A nationwide database study. Int. J. Rheum. Dis. 20, 839–845. https://doi.org/10.1111/1756-185x.12998 (2017).
Zhang, Y. et al. Improved survival in rheumatoid arthritis: A general population-based cohort study. Ann. Rheum. Dis. 76, 408–413. https://doi.org/10.1136/annrheumdis-2015-209058 (2017).
Bohannon, R. W., Magasi, S. R., Bubela, D. J., Wang, Y. C. & Gershon, R. C. Grip and knee extension muscle strength reflect a common construct among adults. Muscle Nerve 46, 555–558. https://doi.org/10.1002/mus.23350 (2012).
Xue, Q. L., Beamer, B. A., Chaves, P. H., Guralnik, J. M. & Fried, L. P. Heterogeneity in rate of decline in grip, hip, and knee strength and the risk of all-cause mortality: The Women’s Health and Aging Study II. J. Am. Geriatr. Soc. 58, 2076–2084. https://doi.org/10.1111/j.1532-5415.2010.03154.x (2010).
Norman, K. et al. Determinants of hand grip strength, knee extension strength and functional status in cancer patients. Clin. Nutr. 29, 586–591. https://doi.org/10.1016/j.clnu.2010.02.007 (2010).
Satake, S. & Arai, H. The revised Japanese version of the Cardiovascular Health Study criteria (revised J-CHS criteria). Geriatr. Gerontol. Int. 20, 992–993. https://doi.org/10.1111/ggi.14005 (2020).
Aletaha, D. et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 62, 2569–2581. https://doi.org/10.1002/art.27584 (2010).
Satake, S. et al. Validity of the Kihon Checklist for assessing frailty status. Geriatr. Gerontol. Int. 16, 709–715. https://doi.org/10.1111/ggi.12543 (2016).
Arai, H. & Satake, S. English translation of the Kihon Checklist. Geriatr. Gerontol. Int. 15, 518–519. https://doi.org/10.1111/ggi.12397 (2015).
Chen, L. K. et al. Asian Working Group for Sarcopenia: 2019 Consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 21, 300-307.e302. https://doi.org/10.1016/j.jamda.2019.12.012 (2020).
Grassi, W., De Angelis, R., Lamanna, G. & Cervini, C. The clinical features of rheumatoid arthritis. Eur. J. Radiol. 27(Suppl 1), S18-24. https://doi.org/10.1016/s0720-048x(98)00038-2 (1998).
Fuchs, H. A. & Pincus, T. Reduced joint counts in controlled clinical trials in rheumatoid arthritis. Arthritis Rheum. 37, 470–475. https://doi.org/10.1002/art.1780370406 (1994).
Wells, G. et al. Validation of the 28-joint Disease Activity Score (DAS28) and European League Against Rheumatism response criteria based on C-reactive protein against disease progression in patients with rheumatoid arthritis, and comparison with the DAS28 based on erythrocyte sedimentation rate. Ann. Rheum. Dis. 68, 954–960. https://doi.org/10.1136/ard.2007.084459 (2009).
Prevoo, M. L. et al. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 38, 44–48 (1995).
Inoue, E., Yamanaka, H., Hara, M., Tomatsu, T. & Kamatani, N. Comparison of Disease Activity Score (DAS)28-erythrocyte sedimentation rate and DAS28-C-reactive protein threshold values. Ann. Rheum. Dis. 66, 407–409. https://doi.org/10.1136/ard.2006.054205 (2007).
Pincus, T., Summey, J. A., Soraci, S. A. Jr., Wallston, K. A. & Hummon, N. P. Assessment of patient satisfaction in activities of daily living using a modified Stanford Health Assessment Questionnaire. Arthritis Rheum. 26, 1346–1353 (1983).
Wells, G. A. et al. Minimal disease activity for rheumatoid arthritis: A preliminary definition. J. Rheumatol. 32, 2016–2024 (2005).
Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant 48, 452–458. https://doi.org/10.1038/bmt.2012.244 (2013).
Salaffi, F., Di Carlo, M., Farah, S., Di Donato, E. & Carotti, M. Prevalence of frailty and its associated factors in patients with rheumatoid arthritis: A cross-sectional analysis. Clin. Rheumatol. 38, 1823–1830. https://doi.org/10.1007/s10067-019-04486-5 (2019).
Sheehy, C., Gaffney, K. & Mukhtyar, C. Standardized grip strength as an outcome measure in early rheumatoid arthritis. Scand. J. Rheumatol. 42, 289–293. https://doi.org/10.3109/03009742.2012.747624 (2013).
Andrews, J. S. et al. The impact of frailty on changes in physical function and disease activity among adults with rheumatoid arthritis. ACR Open Rheumatol. 1, 366–372. https://doi.org/10.1002/acr2.11051 (2019).
Vianna, L. C., Oliveira, R. B. & Araújo, C. G. Age-related decline in handgrip strength differs according to gender. J. Strength Cond. Res. 21, 1310–1314. https://doi.org/10.1519/r-23156.1 (2007).
Palamar, D. et al. Disease activity, handgrip strengths, and hand dexterity in patients with rheumatoid arthritis. Clin. Rheumatol. 36, 2201–2208. https://doi.org/10.1007/s10067-017-3756-9 (2017).
Salaffi, F., Di Matteo, A., Farah, S. & Di Carlo, M. Inflammaging and frailty in immune-mediated rheumatic diseases: How to address and score the issue. Clin. Rev. Allergy Immunol. https://doi.org/10.1007/s12016-022-08943-z (2022).
Smith, L., Yang, L. & Hamer, M. Handgrip strength, inflammatory markers, and mortality. Scand. J. Med. Sci. Sports 29, 1190–1196. https://doi.org/10.1111/sms.13433 (2019).
Schumacher, H. R. Jr., Habre, W., Meador, R. & Hsia, E. C. Predictive factors in early arthritis: Long-term follow-up. Semin. Arthritis Rheum. 33, 264–272. https://doi.org/10.1053/s0049-0172(03)00130-6 (2004).
Sobue, Y. et al. Does exercise therapy improve patient-reported outcomes in rheumatoid arthritis?—A systematic review and meta-analysis for the update of the 2020 JCR guidelines for the management of rheumatoid arthritis. Mod. Rheumatol. 32, 96–104. https://doi.org/10.1080/14397595.2021.1886653 (2021).
Cima, S. R., Barone, A., Porto, J. M. & de Abreu, D. C. Strengthening exercises to improve hand strength and functionality in rheumatoid arthritis with hand deformities: A randomized, controlled trial. Rheumatol. Int. 33, 725–732. https://doi.org/10.1007/s00296-012-2447-8 (2013).
Ishikawa, H. et al. Overall benefits provided by orthopedic surgical intervention in patients with rheumatoid arthritis. Mod. Rheumatol. 29, 335–343. https://doi.org/10.1080/14397595.2018.1457468 (2019).
Feng, Z. et al. Risk factors and protective factors associated with incident or increase of frailty among community-dwelling older adults: A systematic review of longitudinal studies. PLoS ONE 12, e0178383. https://doi.org/10.1371/journal.pone.0178383 (2017).
Acknowledgements
This research was supported by AMED under Grant Number JP21ek0410086. We thank Ms. Sachiko Kato, Ms. Emi Yokota, Ms. Ritsuko Otake, and Ms. Takako Sashikata for their assistance with information collection.
Author information
Authors and Affiliations
Contributions
Y.S. and T.K. contributed to the conception and design of the study, acquisition of data, analysis and interpretation of data, and drafting/revising the manuscript critically for important intellectual content. M.S., Y.O., H.K., N.O., K.F., H.I., H.I., M.K., S.A., K.T., K.K., M.M., and D.K. contributed to the conception and design of the study, acquisition of data, and revising the manuscript critically for important intellectual content. S.I. contributed to the conception and design of the study, analysis and interpretation of data, and revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sobue, Y., Suzuki, M., Ohashi, Y. et al. Validation of grip strength as a measure of frailty in rheumatoid arthritis. Sci Rep 12, 21090 (2022). https://doi.org/10.1038/s41598-022-21533-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-21533-5
- Springer Nature Limited
This article is cited by
-
Association of relative hand grip strength with myocardial infarction and angina pectoris in the Korean population: a large-scale cross-sectional study
BMC Public Health (2024)
-
Persistence on subcutaneous tocilizumab as monotherapy or in combination with synthetic disease-modifying anti-rheumatic drugs in rheumatoid arthritis patients in Greece (EMBRACE study): a multicenter, post-marketing, non-interventional, observational trial
Clinical Rheumatology (2024)