Abstract
Evidence for an association between the amount of particulate matter (PM) in the atmosphere and vitamin D status of pregnant women is limited. We aimed to examine the independent association between PM and maternal levels of serum 25-hydroxyvitamin D (25OHD) during the second trimester and to explore possible modifications to the association by meteorological factors. 27,768 pregnant women presenting for prenatal examination who were tested for serum 25OHD concentration during the second trimester between January 1, 2016, and December 31, 2020, were included in this retrospective analysis. Exposure to PM was evaluated based on daily average PM with an aerodynamic diameter of ≤ 2.5 μm (PM2.5) and PM with an aerodynamic diameter of ≤ 10 μm (PM10). Corresponding meteorological data for daily average atmospheric temperature, atmospheric pressure, relative humidity, sunshine duration, and wind speed were collected. The maximum cumulative effects of PM2.5 occurred at lag 45 days, and the maximum cumulative effects of PM10 occurred at lag 60 days. In crude models, 45-day moving daily average PM2.5 concentrations were negatively associated with 25OHD levels (β, − 0.20; 95% CI − 0.21 to − 0.19), as were 60-day moving daily average PM10 concentrations (β, − 0.14; 95% CI − 0.15 to − 0.14). After adjusting for temporal and meteorological factors, the effect values were drastically reduced (adjusted β of PM2.5, − 0.032; 95% CI − 0.046 to − 0.018; adjusted β of PM10, − 0.039; 95% CI − 0.049 to − 0.028). Our study showed there was a small, independent, negative association between PM in the atmosphere and maternal serum 25OHD levels during the second trimester of pregnancy after adjusting for temporal and/or meteorological factors, which indicates that PM may have a limited influence on maternal serum 25OHD levels. Besides taking vitamin D supplements, pregnant women should keep participating in outdoor activities while taking PM protection measures to improve their vitamin D levels when PM levels are high in winter and spring.
Similar content being viewed by others
Introduction
The key fat-soluble nutrient vitamin D has multiple functions, and its deficiency is thought to be a risk factor in skeletal health and various non-skeletal conditions, such as schizophrenia, skin disorders, certain types of cancer, type 2 diabetes, and infections1. For pregnant women, sufficient vitamin D stores need to be maintained to provide for both themselves and their fetus2. Research continues to provide evidence of an association between maternal vitamin D deficiency (VDD) and a higher risk of numerous undesirable pregnancy outcomes, such as a low body weight, respiratory tract infections, and neurocognitive developmental problems in newborns and preeclampsia and high blood pressure in mothers3,4,5,6,7,8. Therefore, VDD is a key clinical issue for pregnant women and deserves research attention. Throughout the world at present, VDD during pregnancy is still a frequent occurrence. In one study of pregnant women in Finland, 77.4% were found to suffer from VDD (25-hydroxyvitamin D [25OHD] < 50 nmol/L) during their first trimester9. While in, the USA, Ginde et al.10 reported 46%, 32%, and 18% of women with VDD (25OHD < 50 nmol/L) in the first, second, and third trimesters, respectively, and there were comparable observations among British11, Indian12, and Chinese13 women.
For the most part, factors determining the vitamin D status during pregnancy do not differ from those important for the general population, such as the number of melanocytes in the skin, the amount of exposure of the skin to the sun, the amount of adipose tissue in the body, geographical latitude, dietary factors, and intake of supplementary vitamins14,15,16,17,18. An indoor lifestyle can also increase the risk of developing a VDD19. Moreover, the amount of PM in the atmosphere has been associated with circulating levels of 25OHD in humans20,21,22,23, including pregnant women24.
It is not yet clear what mechanisms coordinate the links between maternal vitamin D and air pollution levels. Because vitamin D is a relatively scarce resource in food, humans generally rely on the vitamin D produced by cells in the skin on exposure to natural solar ultraviolet (UV) B radiation25,26. Some scholars have hypothesized that the reduction in the amount of solar UVB radiation reaching ground level by particulate matter (PM) is the main mechanism linking PM to circulating 25OHD levels in humans23,24. In an investigation into the relationship between certain pollutants and ground-level UVB intensity, PM with an aerodynamic diameter of ≤ 10 μm (PM10) was shown to be a significant negative predictor of solar UVB radiation, but the impact of PM10 was very small27. In addition, vitamin D status correlated with temporal and meteorological factors. During their second trimester, the 25OHD levels of women in eastern China demonstrated variation with season and air temperature28. Another study reported 25OHD levels were positively correlated with the prior month’s temperature29. Thus, when exploring the independent association between PM and vitamin D status, meteorological factors may need to be taken into account.
Thus, our study objective was to examine the independent association between PM and maternal serum 25OHD levels during the second trimester in pregnant women and to explore if meteorological factors have an impact on this association.
Methods
Study population
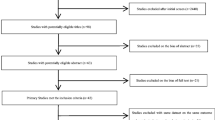
We designed a single-hospital-based cross-sectional retrospective observational study to test our hypotheses. All prenatal examination data were collected from the hospital medical database in Affiliated Kunshan Hospital of Jiangsu University located in Kunshan, eastern China, at 31.2° N latitude and approximately 30 km from Shanghai. This is the largest hospital that provides prenatal care in all the districts of Kunshan. Consecutively, 35,476 pregnant women at the 15–20th week gestation (during the second trimester) who visited our institution for prenatal examination and were tested for serum 25OHD concentrations between January 1, 2016, and December 31, 2020, were included in this study. We excluded 7708 women for the following reasons: those (1) with a chronic metabolic disease with consequences for the metabolism of vitamin D (n = 5389); (2) with a high-risk pregnancy (n = 1087), or (3) who had lived in Kunshan for less than 1 year (n = 1232). Finally, 27,768 pregnant women were included in this analysis (Fig. S1).
The study protocol, submitted for review by the ethical committee at the Affiliated Kunshan Hospital of Jiangsu University (approval No. 2020-03-046-K01), was approved, and it complied with the Declaration of Helsinki. Patient information was initially documented for hospital’s quality improvement purposes. The blood samples used for measuring 25OHD were taken as part of prenatal examinations. The requirement for informed consent was waived because of the anonymous and observational design of this investigation. Data analyzers were blinded to the identity of the patients.
Measurement of vitamin D
In humans, the most abundant form of vitamin D in the blood is 25OHD, and serum levels are used to reliably estimate a patient’s vitamin D status. All pregnant women in this study were in their 15–20th week of gestation (during the second trimester of pregnancy) and were asked to fast before blood samples were taken. Serum 25OHD concentrations were measured immediately using an automated electrochemiluminescence immunoassay on a Roche Cobas 8000/e602 analyzer (Roche Diagnostics, Mannheim, Germany). There is no medical consensus regarding the status cut-off values of 25OHD concentrations. However, the Institute of Medicine (National Academy of Sciences, Washington, DC, USA) and the National Osteoporosis Society (Bath, England) concur that, for bone health, a serum 25OHD level (2.5 nmol/L 25OHD = 1 ng/mL 25OHD) of < 30 nmol/L (12 ng/mL) is deficient, 30–50 nmol/L (12–20 ng/mL) is insufficient for certain individuals, while > 50 nmol/L (20 ng/mL) is sufficient for most people30,31. The time of blood collection was included in the analysis. Seasons were defined as: Spring, March–May; Summer, June–August; Autumn, September–November; Winter, December–February.
PM exposure and meteorological data assessment
We received data for the daily average concentration of PM with an aerodynamic diameter of ≤ 2.5 μm (PM2.5; μg/m3) and PM10 (μg/m3) from the Meteorological Bureau of Kunshan City. Meteorological data (daily average atmospheric temperature [°C], atmospheric pressure [hPa], relative humidity [%], sunshine duration [hour], and wind speed [m/s]) were obtained from the Ecology and Environment Bureau of Kunshan City. The distances between the three environmental (No. 2 Middle School monitoring site, Zhenchuan Middle School monitoring site, and Kunshan Deng-yun College monitoring site) and one meteorological (National Meteorological Observing Station) monitoring site and the hospital are within 11 km. Kunshan City covers an area of 927.7 square kilometers; this distance is shorter than that used for sensitivity analysis in the study by Di et al. in 201732.
Because the serum 25OHD half-life is about 15 days33, the cumulative effects of PM2.5 and PM10 on maternal serum 25OHD levels during the second trimester of pregnancy were approximated using the moving-average lag structures (blood collection day [day 0] was not included) as follows: 0–3 days (3-day moving daily average PM concentration), 0–7 days (7-day moving daily average PM concentration), 0–15 days (15-day moving daily average PM concentration), 0–30 days (30-day moving daily average PM concentration), 0–45 days (45-day moving daily average PM concentration), 0–60 days (60-day moving daily average PM concentration), 0–75 days (75-day moving daily average PM concentration), and 0–90 days (90-day moving daily average PM concentration). When adjusting for the influence of meteorological factors, the corresponding moving average lag structures for the meteorological factors were calculated.
Statistics
The summary statistics for the characteristics of pregnant women were expressed as frequencies (proportions) for categorical variables and as the means (standard deviation [SD]) or medians (Q1–Q3) for continuous variables. We also conducted univariate logistic regression analysis to evaluate the association between the characteristics of pregnant women and maternal serum 25OHD concentrations during the second trimester. The daily average meteorological variables and daily average PM2.5, PM10, and 25OHD concentrations were screened for correlations by Pearson’s.
Using generalized estimating equations (GEE), we evaluated the relationships between the cumulative effects of PM on maternal serum 25OHD levels. The cumulative effects of PM were divided into four levels on the basis of quartiles. After adjusting for year and age at blood collection (adjust I) or year, season, and age at blood collection (adjust II), β-values (95% confidence interval [CI]) of the maternal serum 25OHD levels were calculated based on group trends. As part of the sensitivity analysis, we also calculated the β-values (95% CI) for the 25OHD levels when using the concentrations of PM2.5 and PM10 as continuous variables.
Controlling for the influence of meteorological covariances by applying multivariate linear regression analysis allowed us to evaluate independent associations between the maximum cumulative effects of PM and maternal serum 25OHD. We calculated the results of the unadjusted or minimally adjusted analysis and those from fully adjusted analysis. First, collinearity diagnosis of covariances was performed using variance inflation factor (VIF) analysis (the variable average atmospheric temperature was first removed due to VIF > 10). Then, a judgement on whether to adjust covariances was made using the following principles: Criteria 1, the covariate is added to the basic model or removed from the full model and the matched odds ratio (OR) is changed by at least 10%; Criteria 2: Criteria 1 or a covariate P-value of < 0.1 in the univariate model34. As part of the sensitivity analysis, we transformed 25OHD quantitative variables into dichotomous qualitative variables (1 = vitamin D deficiency and inadequacy [< 20 ng/mL]; 0 = adequacy [≥ 20 ng/mL]), then the OR and 95% CI for maternal vitamin D deficiency and inadequacy (< 20 ng/mL) associated with a 10 μg/m3 increase in PM2.5 or PM10 was determined.
Non-linear relationships were additionally identified via a generalized additive model (GAM), and on finding a non-linear correlation, the threshold effect in terms of the smoothing curve was calculated using a two-piecewise linear regression model. When a clear ratio was apparent in the smoothing curve, the recursive method was applied to automatically calculate the turning point at which to use the maximum likelihood model35. In addition, to test the robustness and potential variation in the different subgroups, we repeated the subgroup analyses while stratifying by season, age, and meteorological factors. The age threshold was derived from the turning point calculated by the GAM followed by an inspection of the modification and interaction of the subgroups with the likelihood ratio test.
All statistical analyses were performed using the Empower Stats (www.empowerstats.com, X&Y solutions, Inc., Boston, MA, USA) and R software version 3.6.3 (http://www.r-project.org). A P-value < 0.05 was set as the significance threshold.
Ethics approval
This study complies with the Declaration of Helsinki and has been approved by the Ethics Committee of the First People’s Hospital of Kunshan (no. 2020-03-046-K01).
Results
Table 1 displays the data on the pregnant women and their vitamin D statuses. This analysis included 27,768 individuals with a mean age of 28.86 (SD, 4.27) years. The mean (SD) and median (Q1–Q3) values of maternal serum 25OHD concentrations during the second trimester of pregnancy were 17.7 (7.9) ng/mL and 16.0 (12.0–22.0) ng/mL, respectively. Vitamin D deficiency, inadequacy, and adequacy were present in 23.5%, 41.3%, and 35.2% of women, respectively. Univariate logistic regression analysis revealed that maternal 25OHD concentration was positively associated with maternal age and showed seasonal variation, with the peak in September and the nadir in February.
Pearson’s correlation analysis was conducted to compare serum 25OHD concentration, meteorological variables, and air pollutant exposure. Figure S2 presents that, except PM2.5 vs. sunshine duration (P-value = 0.82), all correlations among the variables were statistically significant (P-value < 0.001). There was a strong positive correlation between the daily average PM2.5 concentration and PM10 concentration, and the Pearson coefficient was 0.92. There was also a strong negative correlation between daily average atmospheric temperature and atmospheric pressure, and the Pearson coefficient was − 0.89. There was a moderate negative correlation between daily sunshine duration and relative humidity, and the correlation coefficient was − 0.68.
Figure 1 shows periodic changes in the above indicators in terms of monthly average values over time. It can be seen that the variation in the average temperature of the last month was most consistent with the periodic variation of monthly average serum 25OHD concentrations. Similar periodic changes were seen for sunshine duration and relative humidity. However, monthly average PM2.5 and PM10 concentrations and atmospheric pressure showed periodic changes that were diametrically opposite to those of the monthly average serum 25OHD concentration.
Temporal trends in monthly average serum 25OHD concentration vs. monthly average PM2.5 concentration (A), PM10 concentration (B), atmospheric pressure (C), temperature (D), relative humidity (E), sunshine duration (F), and wind speed (G) from January 2016 to December 2020. 25OHD 25-hydroxy vitamin D, PM2.5 particulate matter with an aerodynamic diameter of ≤ 2.5 μm, PM10 particulate matter with an aerodynamic diameter of ≤ 10 μm.
Figure 2 and Table S1 show the cumulative effects of PM2.5 and PM10 on maternal serum 25OHD levels during the second trimester of pregnancy. After we adjusted for year, season, and age at blood collection, the maximum cumulative effects occurred at lag 0–45 days of PM2.5 and lag 0–60 days of PM10. When PM concentrations were used as continues variables, similar results were observed, and these are provided in Table S2.
Association among exposure to PM2.5 (A) or PM10 (B) and maternal serum 25OHD levels during pregnancy. The cumulative effects of PM were divided into four levels based on quartiles. After adjusting for year and age at blood collection (adjust I) or year, season, and age at blood collection (adjust II), the β-values (95% CI) of maternal serum 25OHD levels during pregnancy were calculated based on the group trend. The dashed vertical line represents lag days corresponding to the maximum cumulative effects. 25OHD 25-hydroxy vitamin D, PM2.5 particulate matter with an aerodynamic diameter of ≤ 2.5 μm, PM10 particulate matter with an aerodynamic diameter of ≤ 10 μm, CI confidence interval.
The independent associations between the maximum cumulative effects of PM and serum 25OHD levels were further investigated, and Table 2 shows the results adjusted for different covariances. In the crude models, the effect sizes of PM2.5 (β, − 0.20; 95% CI − 0.21 to − 0.19; P-value < 0.00001) and PM10 (β, − 0.14; 95% CI − 0.15 to − 0.14; P-value < 0.00001) were relatively high. After adjustments for year, season, and age at blood collection (Model 1), the effect sizes of PM2.5 (β, − 0.11; 95% CI − 0.13 to − 0.10; P-value < 0.00001) and PM10 (β, − 0.090; 95% CI − 0.099 to − 0.081; P-value < 0.00001) were reduced. On the basis of the Model 1 adjustment, the effect values of PM2.5 (β, − 0.042; 95% CI − 0.055 to − 0.028; P-value < 0.00001) and PM10 (β, − 0.039; 95% CI − 0.049 to − 0.030; P-value < 0.00001) were further reduced after adding atmospheric pressure adjustment (Model 2). After further adjustment for sunshine duration, the effect values of Model 3 were similar to those of Model 2. In the fully adjusted Model 4 (adjusted for year, age, season, daily average atmospheric pressure, sunshine duration, relative humidity, and wind speed), there was a negative relationship between the 45-day moving daily average PM2.5 concentration and the women’s 25OHD levels (β, − 0.032; 95% CI − 0.046 to − 0.018; P-value < 0.00001), and the 60-day moving daily average PM10 concentration was negatively associated with 25OHD levels (β, − 0.039; 95% CI − 0.049 to − 0.028; P-value < 0.00001). As part of the sensitivity analysis, the crude and adjusted OR for PM exposure’s association with maternal vitamin D deficiency and inadequacy (< 20 ng/mL) were determined, which are given in Table S3. We observed greater odds of maternal vitamin D deficiency and inadequacy (< 20 ng/mL) with higher PM levels. In the fully adjusted Model 4, an increase in the PM2.5 45-day moving daily average by 10 μg/m3 was associated with a 12.4% (OR 1.12; 95% CI 1.07 to 1.18, P-value < 0.00001) increase in the odds for maternal vitamin D deficiency and inadequacy (< 20 ng/mL). In the fully adjusted Model 4, a 10 μg/m3 increase in the PM10 60-day moving daily average was associated with a 11.3% (OR 1.11; 95% CI 1.07 to 1.15, P-value < 0.00001) increase in the odds of maternal vitamin D deficiency and inadequacy (< 20 ng/mL).
In the subgroup analyses stratified by season, we further investigated the role of season on the association between the maximum cumulative effects of PM and serum 25OHD levels. For PM2.5 (Table 3), both linear and nonlinear effect values were higher in summer and autumn and lower in winter and spring (P-values for interaction < 0.001). Figure 3A shows the different nonlinear associations between the 45-day moving daily average PM2.5 concentration and maternal serum 25OHD levels stratified by season. In addition, we calculated, using the two-piecewise linear regression model, the turning point of the adjusted smoothed curve. Specifically, the difference between the two slopes was at its maximum in autumn, and the turning point (45-day moving daily average PM2.5 concentration) was 31.11 μg/m3. For PM10 (Table 4), linear regression analysis showed a significant interaction for season (interaction P-value < 0.001), but the non-linear model showed this interaction was not significant (interaction P-value = 0.38). Figure 3B shows the different nonlinear associations between the 60-day moving daily average PM10 concentration and maternal serum 25OHD levels stratified by season. It is worth mentioning that, in autumn, the relationship between PM10 and 25OHD levels was nonlinear, and the turning point was 38.10 μg/m3. Specifically, when the 60-day moving daily average PM10 concentration ranged from 32.32 to 38.10 μg/m3, a stronger negative relationship was found between PM10 and serum 25OHD levels (β, − 1.12; 95% CI − 1.51 to − 0.73; P-value < 0.0001; number of pregnant women, 878).
Adjusted smoothed curves for 45-day moving daily average PM2.5 concentration (A), 60-day moving daily average PM10 concentration (B), and maternal serum 25OHD levels during pregnancy stratified by seasons. Thresholds were nonlinear associations between PM and 25OHD, as evidenced in generalized additive models. Adjustment factors included year, age, corresponding-day moving daily average atmospheric pressure, sunshine duration, relative humidity, and wind speed. 25OHD 25-hydroxy vitamin D, PM2.5 particulate matter with an aerodynamic diameter of ≤ 2.5 μm, PM10 particulate matter with an aerodynamic diameter of ≤ 10 μm.
The threshold effect analysis used to examine the associations between maternal age and serum 25OHD levels during the second trimester of pregnancy revealed a stronger positive relationship between age and serum 25OHD levels when the women were 15 to 25 years of age and a weaker positive relationship when they were 25 to 47. The results of four different adjusted models were robust (Fig. S3, Table S4). We then categorized the pregnant women using a threshold of 25 years according to the results of the threshold effect analysis and further investigated the modification effect of age on the association between the maximum cumulative effects of PM and serum 25OHD levels. Table S5 shows that the interaction between age and PM2.5 was not significant (linear interaction P-value = 0.08 and non-linear interaction P-value = 0.06), while Table S6 shows the interaction between age and PM10 had a marginally significant effect on serum 25OHD levels (linear P-value for interaction = 0.045 and non-linear P-value for interaction = 0.04).
In the subgroup analyses stratified by meteorological factors, Tables 5 and 6 showed the associations between PM and serum 25OHD levels during the second trimester of pregnancy were generally modified by meteorological factors, although wind speed had no modification effect on PM2.5. Of particular interest, there was a positive association between PM concentration and 25OHD levels under low relative humidity. The result of this stratification was the opposite to the final conclusion.
Discussion
Based on our knowledge of the literature, this epidemiological study is the first to focus on the independent relationship between PM and maternal serum 25OHD levels during the second trimester of pregnancy after adjusting for meteorological factors. We found vitamin D deficiency, inadequacy, and adequacy in 23.5%, 41.3%, and 35.2% of pregnant women during the second trimester of pregnancy, respectively. We found that the maximum cumulative effects of PM2.5 occurred at lag 45 days and the maximum cumulative effects of PM10 occurred at lag 60 days. However, the effect values were drastically reduced after adjusting for temporal and/or meteorological factors. The results indicated that PM has a limited influence on maternal serum 25OHD levels.
The list of studies that have linked VDD to complications of pregnancy continues to grow: vitamin D status has been associated with gestational diabetes36,37,38,39, aeroallergen sensitization40, and markers of regulatory immunity41. A meta-analysis of eight studies found a significant association between VDD and the risk of pre-eclampsia, which was more evident in studies that defined VDD as 25OHD < 50 nmol/L and those from the USA42. In addition, a meta-analysis of 24 observational studies confirmed the association between VDD (< 50 nmol/L) and an increased risk of preterm birth (OR 1.58; 95% CI 1.08 to 2.31)43. With respect to birthweight, a meta-analysis of three observational studies found a weak positive association between maternal vitamin D status and birthweight after adjusting for potential confounders44. In addition, recent reviews suggested that appropriate levels of vitamin D during pregnancy are associated with less morbidity during pregnancy45,46.
Zhao et al. reported an association between prenatal exposure to higher PM2.5 and PM10 levels and a decrease in circulating 25OHD concentrations in women in the third trimester and the entire pregnancy24. They reported a 10 μg/m3 increase in PM2.5 and PM10 exposure during the entire pregnancy was associated with a 4.62% (95% CI 26.31% to 22.93%) and 5.06% (95% CI 26.50% to 23.62%) decrease in 25OHD levels, respectively24. In contrast to the analysis they conducted, we first investigated the time window for the largest cumulative effect of PM at the individual level and then adjusted for temporal and meteorological factors. Although both effect sizes of PM2.5 and PM10 in the fully adjusted models were small (β-value of PM2.5 = − 0.032; β-value of PM10 = − 0.039, respectively), they were highly significant (P-value < 0.00001). The β-values showed two large drops, one after adjusting for season and the other after adjusting for atmospheric pressure. Due to the collinearity of atmospheric pressure and temperature, it can be inferred that season and atmospheric pressure/temperature may have been important confounders in the regression model. Thus, in some correlation analyses of PM and vitamin D levels, the independent role of PM may have been overestimated when there was no adjustment for meteorological factors. On the other hand, there is now evidence that 25OHD accumulates in skeletal muscle cells, which provide a functional store during the winter months47,48. The mechanism is mediated by the muscle cell uptake of circulating vitamin-D-binding protein (DBP) through a megalin–cubilin membrane transport process47. If ways to optimize the efficiency of the muscle conservation mechanism for 25OHD could be found, e.g., a pharmacological agent or some exercise regime, perhaps that optimization would ensure that vitamin D status is also optimized by a process that has evolved to adapt to seasonal changes in vitamin D supply48. Therefore, we also proposed, when PM levels are high in winter and spring (when the population’s vitamin D levels are also low), pregnant women, besides taking vitamin D supplements, should keep participating in outdoor activities while taking PM protection measures, e.g., wearing a facemask or face covering and avoiding high-PM areas such as alongside high-volume traffic roads, to increase their vitamin D levels.
Indeed, in areas with distinct seasons, 25OHD concentrations in the population fluctuate over time29,49,50,51,52. Many studies showed season to be the primary factor affecting serum 25OHD levels15,29,53,54,55. Periodic changes in the seasons affect periodic changes in meteorological factors, such as temperature, humidity, and sunshine duration. Hence, unlike other studies that only adjusted for season, our study precisely adjusted for meteorological factor exposure at the individual level.
On the other hand, meteorological factors are also related to periodic changes in PM concentrations. Weather and climate are the most influential forces affecting the chemistry and atmospheric residence time of PM56. In a study in China, the geographical and temporal variations in PM levels and coinciding meteorological conditions were analyzed for 366 cities, and peak PM concentrations occurred in winter in most regions, and there were negative correlations between PM levels and precipitation, relative humidity, air temperature, and wind speed but a positively correlation was found between PM levels and surface pressure57. Guan et al.58 reported the spatiotemporal variability of PM in three cities in western China and the influence of meteorological factors on PM. Crawford et al.59, who analyzed the impact of meteorology on particulate source types in Lucas Heights, Australia, reported that temperature significantly affected PM2.5 levels. An Indian study reported that PM displayed substantial seasonal variations and a strong negative association with temperature, with considerable dependency levels56.
Over 90% of the vitamin D in the body is synthesized in human skin after the exposer of precursor 7-dehydrocholesterol to UVB (290–315 nm) radiation from the sun60. The strength of the UVB during sun exposure, and therefore the amount of vitamin D synthesized, is affected by solar zenith angle (SZA)—the angle between the local vertical and the sun’s position. Hence, the most intense solar radiation occurs when the SZA is small, i.e., which at lower altitudes, is around 11:00–15:00 h in the summer, at which time the synthesis of vitamin D by the skin is most active61. At latitudes > 50°, human skin participates in very little vitamin D synthesis during winter and spring for all skin types and ethnicities62. In addition, the amount of vitamin D produced is dependent on local weather conditions, such as the percentage cloud cover, which filters UVB radiation, and has an impact in all seasons and hours of the day50. Thus, because PM can absorb and diffuse solar irradiation, some scholars have hypothesized that PM can indirectly reduce vitamin D formation by reducing UVB exposure23,24. However, other scholars have argued that there is significant spatiotemporal variation in the morphology, chemical makeup, density of PM, and it is difficult to determine its effect on UV radiation63. More recently, PM10 was found to be a significant negative predictor of solar UVB radiation, but the effect of PM10 was miniscule27. This conclusion can be used to further deduce that PM10 has an effect on vitamin D levels, but the effect is small, which is consist with the results of our study. Combined with the above views, we propose a hypothesis (see Fig. S4) that PM and meteorological factors indirectly influence the cyclical changes of vitamin D in pregnant women by impacting the level of personal UVB exposure. For example, in winter, when the temperature is lower and there are fewer hours of sunshine, the PM concentration is higher, but pregnant women spend less time outdoors and wear more clothes, which lead to lower solar UVB radiation exposure, reducing the synthesis of vitamin D.
In the subgroup analysis, with the exception of the low relative humidity, a negative effect between PM and 25OHD was evident in all subgroups considered and after careful adjustments. In the interaction analysis, season and most meteorological factors interacted with the association between PM and 25OHD. However, the mechanisms of their interactions were unclear and need to be further investigated in the future.
There were some limitations regarding our study. First, our results were obtained from a Chinese population of pregnant women during the second trimester and cannot be extrapolated to other populations. Second, the study was an analytical retrospective study and hence provides limited evidence that PM exposure and vitamin D outcomes were related, and the difference between cause and effect is uncertain. Third, demographic information was lacking (education level, vitamin D/calcium supplement use, outdoor activities, use of sun protection, and BMI, etc.) for the individuals whose test results were used. However, valuable insights can be gleaned from the study, as it involved the retrospective analysis of a large dataset from a prenatal examination population. We used precise adjustments at the individual level to reveal whether PM levels are independently associated with 25OHD concentration. In the future, a more informative vitamin D database will be established, and vitamin D supplementation will be studied. Fourth, solar UVB dose information was not studied; thus, this important indicator could be included in the future studies.
Conclusions
Our study showed there was a small, independent, negative association between PM in the atmosphere and maternal serum 25OHD levels during the second trimester of pregnancy after adjusting for temporal and/or meteorological factors, which indicates that PM may have a limited influence on maternal serum 25OHD levels. Besides taking vitamin D supplements, pregnant women should keep participating in outdoor activities while taking PM protection measures to improve their vitamin D levels when PM levels are high in winter and spring.
Data availability
All data generated or analysed during this study are included in this published article and its Supplementary Information files.
References
Holick, M. F. Vitamin D: Evolutionary, physiological and health perspectives. Curr. Drug Targets 12(1), 4–18. https://doi.org/10.2174/138945011793591635 (2011).
Hatun, S. et al. Vitamin D deficiency in early infancy. J. Nutr. 135(2), 279–282. https://doi.org/10.1093/jn/135.2.279 (2005).
Achkar, M. et al. Vitamin D status in early pregnancy and risk of preeclampsia. Am. J. Obstet. Gynecol. 212(4), 511–517. https://doi.org/10.1016/j.ajog.2014.11.009 (2015).
Arnold, D. L. et al. Early pregnancy maternal vitamin D concentrations and risk of gestational diabetes mellitus. Paediatr. Perinat. Epidemiol. 29(3), 200–210. https://doi.org/10.1111/ppe.12182 (2015).
Wang, H., Xiao, Y., Zhang, L. & Gao, Q. Maternal early pregnancy vitamin D status in relation to low birth weight and small-for-gestational-age offspring. J. Steroid Biochem. Mol. Biol. 175, 146–150. https://doi.org/10.1016/j.jsbmb.2017.09.010 (2018).
Murthi, P. et al. Maternal 25-hydroxyvitamin D is inversely correlated with foetal serotonin. Clin. Endocrinol. (Oxf.) 86(3), 401–409. https://doi.org/10.1111/cen.13281 (2017).
Meems, L. M. et al. Parental vitamin D deficiency during pregnancy is associated with increased blood pressure in offspring via Panx1 hypermethylation. Am. J. Physiol. Heart Circ. Physiol. 311(6), H1459–H1469. https://doi.org/10.1152/ajpheart.00141.2016 (2016).
Weinert, L. S. & Silveiro, S. P. Maternal-fetal impact of vitamin D deficiency: A critical review. Matern. Child Health J. 19(1), 94–101. https://doi.org/10.1007/s10995-014-1499-7 (2015).
Viljakainen, H. T. et al. Maternal vitamin D status determines bone variables in the newborn. J. Clin. Endocrinol. Metab. 95(4), 1749–1757. https://doi.org/10.1210/jc.2009-1391 (2010).
Ginde, A. A., Sullivan, A. F., Mansbach, J. M. & Camargo, C. A. Jr. Vitamin D insufficiency in pregnant and nonpregnant women of childbearing age in the United States. Am. J. Obstet. Gynecol. 202(5), 436–438. https://doi.org/10.1016/j.ajog.2009.11.036 (2010).
McAree, T. et al. Vitamin D deficiency in pregnancy—Still a public health issue. Matern. Child Nutr. 9(1), 23–30. https://doi.org/10.1111/mcn.12014 (2013).
Vijayendra Chary, A. et al. Vitamin D deficiency in pregnant women impairs regulatory T cell function. J. Steroid Biochem. Mol. Biol. 147, 48–55. https://doi.org/10.1016/j.jsbmb.2014.11.020 (2015).
Song, S. J. et al. The high prevalence of vitamin D deficiency and its related maternal factors in pregnant women in Beijing. PLoS ONE 8(12), e85081. https://doi.org/10.1371/journal.pone.0085081 (2013).
Bodnar, L. M., Catov, J. M., Roberts, J. M. & Simhan, H. N. Prepregnancy obesity predicts poor vitamin D status in mothers and their neonates. J. Nutr. 137(11), 2437–2442. https://doi.org/10.1093/jn/137.11.2437 (2007).
Bodnar, L. M. et al. High prevalence of vitamin D insufficiency in black and white pregnant women residing in the northern United States and their neonates. J. Nutr. 137(2), 447–452. https://doi.org/10.1093/jn/137.2.447 (2007).
Brembeck, P., Winkvist, A. & Olausson, H. Determinants of vitamin D status in pregnant fair-skinned women in Sweden. Br. J. Nutr. 110(5), 856–864. https://doi.org/10.1017/s0007114512005855 (2013).
Jensen, C. B. et al. Sources and determinants of vitamin D intake in Danish pregnant women. Nutrients 4(4), 259–272. https://doi.org/10.3390/nu4040259 (2012).
Richard, A., Rohrmann, S. & Quack Lötscher, K. C. Prevalence of vitamin D deficiency and its associations with skin color in pregnant women in the first trimester in a sample from Switzerland. Nutrients 9(3), 9030260. https://doi.org/10.3390/nu9030260 (2017).
Sowah, D., Fan, X., Dennett, L., Hagtvedt, R. & Straube, S. Vitamin D levels and deficiency with different occupations: A systematic review. BMC Public Health 17(1), 519. https://doi.org/10.1186/s12889-017-4436-z (2017).
Agarwal, K. S. et al. The impact of atmospheric pollution on vitamin D status of infants and toddlers in Delhi, India. Arch. Dis. Child 87(2), 111–113. https://doi.org/10.1136/adc.87.2.111 (2002).
Kelishadi, R. et al. Independent association between air pollutants and vitamin D deficiency in young children in Isfahan. Iran. Paediatr. Int. Child Health 34(1), 50–55. https://doi.org/10.1179/2046905513y.0000000080 (2014).
Feizabad, E. et al. Impact of air pollution on vitamin D deficiency and bone health in adolescents. Arch. Osteoporos. 12(1), 34. https://doi.org/10.1007/s11657-017-0323-6 (2017).
Yang, C., Li, D., Tian, Y. & Wang, P. Ambient air pollutions are associated with vitamin D status. Int. J. Environ. Res. Public Health 18(13), 6887. https://doi.org/10.3390/ijerph18136887 (2021).
Zhao, Y. et al. Particulate air pollution exposure and plasma vitamin D levels in pregnant women: A longitudinal cohort study. J. Clin. Endocrinol. Metab. 104(8), 3320–3326. https://doi.org/10.1210/jc.2018-02713 (2019).
Glerup, H. et al. Commonly recommended daily intake of vitamin D is not sufficient if sunlight exposure is limited. J. Intern. Med. 247(2), 260–268. https://doi.org/10.1046/j.1365-2796.2000.00595.x (2000).
Chen, T. C. et al. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 460(2), 213–217. https://doi.org/10.1016/j.abb.2006.12.017 (2007).
Rahman, A. & Elmi, A. Air pollutants are negatively associated with vitamin D-synthesizing UVB radiation intensity on the ground. Sci. Rep. 11(1), 21480. https://doi.org/10.1038/s41598-021-00980-6 (2021).
Xiao, J. P. et al. Low maternal vitamin D status during the second trimester of pregnancy: A cross-sectional study in Wuxi, China. PLoS ONE 10(2), e0117748. https://doi.org/10.1371/journal.pone.0117748 (2015).
Ding, F., Nie, X., Li, X., He, Y. & Li, G. Data mining: Biological and temporal factors associated with blood parathyroid hormone, vitamin D, and calcium concentrations in the Southwestern Chinese population. Clin. Biochem. 90, 50–57. https://doi.org/10.1016/j.clinbiochem.2021.01.014 (2021).
Aspray, T. J. et al. National Osteoporosis Society vitamin D guideline summary. Age Ageing 43(5), 592–595. https://doi.org/10.1093/ageing/afu093 (2014).
Institute of Medicine Committee to Review Dietary Reference Intakes for Vitamin D, Calcium The National Academies Collection: Reports funded by National Institutes of Health. In Dietary Reference Intakes for Calcium and Vitamin D (eds Ross, A. C. et al.) (National Academies Press, 2011).
Di, Q. et al. Air pollution and mortality in the medicare population. N. Engl. J. Med. 376(26), 2513–2522. https://doi.org/10.1056/NEJMoa1702747 (2017).
Jones, K. S. et al. Predictors of 25(OH)D half-life and plasma 25(OH)D concentration in the Gambia and the UK. Osteoporos. Int. 26(3), 1137–1146. https://doi.org/10.1007/s00198-014-2905-0 (2015).
Kernan, W. N. et al. Phenylpropanolamine and the risk of hemorrhagic stroke. N. Engl. J. Med. 343(25), 1826–1832. https://doi.org/10.1056/nejm200012213432501 (2000).
Liu, S. et al. The effects of intraoperative cryoprecipitate transfusion on acute renal failure following orthotropic liver transplantation. Hepatol. Int. 7(3), 901–909. https://doi.org/10.1007/s12072-013-9457-9 (2013).
Zhang, Q. et al. Effect of various doses of vitamin D supplementation on pregnant women with gestational diabetes mellitus: A randomized controlled trial. Exp. Ther. Med. 12(3), 1889–1895. https://doi.org/10.3892/etm.2016.3515 (2016).
Sablok, A. et al. Supplementation of vitamin D in pregnancy and its correlation with feto-maternal outcome. Clin. Endocrinol. (Oxf.) 83(4), 536–541. https://doi.org/10.1111/cen.12751 (2015).
Mojibian, M., Soheilykhah, S., Fallah Zadeh, M. A. & Jannati Moghadam, M. The effects of vitamin D supplementation on maternal and neonatal outcome: A randomized clinical trial. Iran. J. Reprod. Med. 13(11), 687–696 (2015).
Zhang, C. et al. Maternal plasma 25-hydroxyvitamin D concentrations and the risk for gestational diabetes mellitus. PLoS ONE 3(11), e3753. https://doi.org/10.1371/journal.pone.0003753 (2008).
Grant, C. C. et al. Vitamin D supplementation during pregnancy and infancy reduces aeroallergen sensitization: A randomized controlled trial. Allergy 71(9), 1325–1334. https://doi.org/10.1111/all.12909 (2016).
Zerofsky, M. et al. Effects of early vitamin D deficiency rickets on bone and dental health, growth and immunity. Matern. Child Nutr. 12(4), 898–907. https://doi.org/10.1111/mcn.12187 (2016).
Tabesh, M., Salehi-Abargouei, A., Tabesh, M. & Esmaillzadeh, A. Maternal vitamin D status and risk of pre-eclampsia: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 98(8), 3165–3173. https://doi.org/10.1210/jc.2013-1257 (2013).
Wei, S. Q., Qi, H. P., Luo, Z. C. & Fraser, W. D. Maternal vitamin D status and adverse pregnancy outcomes: A systematic review and meta-analysis. J. Matern. Fetal Neonatal Med. 26(9), 889–899. https://doi.org/10.3109/14767058.2013.765849 (2013).
Harvey, N. C. et al. Vitamin D supplementation in pregnancy: A systematic review. Health Technol. Assess. 18(45), 1–190. https://doi.org/10.3310/hta18450 (2014).
Suárez-Varela, M. M. et al. Vitamin D-related risk factors for maternal morbidity during pregnancy: A systematic review. Nutrients 14(15), 3166. https://doi.org/10.3390/nu14153166 (2022).
Wagner, C. L. & Hollis, B. W. The implications of vitamin D status during pregnancy on mother and her developing child. Front. Endocrinol. (Lausanne) 9, 500. https://doi.org/10.3389/fendo.2018.00500 (2018).
Mason, R. S., Rybchyn, M. S., Abboud, M., Brennan-Speranza, T. C. & Fraser, D. R. The role of skeletal muscle in maintaining vitamin D status in winter. Curr. Dev. Nutr. 3(10), 087. https://doi.org/10.1093/cdn/nzz087 (2019).
Rybchyn, M. S. et al. Skeletal muscle and the maintenance of vitamin D status. Nutrients 12, 11. https://doi.org/10.3390/nu12113270 (2020).
Dovnik, A. et al. Determinants of maternal vitamin D concentrations in Slovenia: A prospective observational study. Wien Klin. Wochenschr. 129(1–2), 21–28. https://doi.org/10.1007/s00508-016-1142-2 (2017).
Saraff, V. & Shaw, N. Sunshine and vitamin D. Arch. Dis. Child 101(2), 190–192. https://doi.org/10.1136/archdischild-2014-307214 (2016).
Hyppönen, E. & Power, C. Hypovitaminosis D in British adults at age 45 y: Nationwide cohort study of dietary and lifestyle predictors. Am. J. Clin. Nutr. 85(3), 860–868. https://doi.org/10.1093/ajcn/85.3.860 (2007).
Kroll, M. H. et al. Temporal relationship between vitamin D status and parathyroid hormone in the United States. PLoS ONE 10(3), e0118108. https://doi.org/10.1371/journal.pone.0118108 (2015).
Yuan, Y. et al. Association of maternal serum 25-hydroxyvitamin D concentrations in second trimester with delivery mode in A Chinese population. Int. J. Med. Sci. 14(10), 1008–1014. https://doi.org/10.7150/ijms.20337 (2017).
Krieger, J. P. et al. Prevalence and determinants of vitamin D deficiency in the third trimester of pregnancy: A multicentre study in Switzerland. Br. J. Nutr. 119(3), 299–309. https://doi.org/10.1017/s0007114517003634 (2018).
Li, H. et al. Prevalence of vitamin D deficiency in the pregnant women: An observational study in Shanghai, China. Arch. Public Health 78, 31. https://doi.org/10.1186/s13690-020-00414-1 (2020).
Murari, V., Kumar, M., Mhawish, A., Barman, S. C. & Banerjee, T. Airborne particulate in Varanasi over middle Indo-Gangetic Plain: Variation in particulate types and meteorological influences. Environ. Monit. Assess. 189(4), 157. https://doi.org/10.1007/s10661-017-5859-9 (2017).
Li, X. et al. Particulate matter pollution in Chinese cities: Areal-temporal variations and their relationships with meteorological conditions (2015–2017). Environ. Pollut. 246, 11–18. https://doi.org/10.1016/j.envpol.2018.11.103 (2019).
Guan, Q. et al. Spatio-temporal variability of particulate matter in the key part of Gansu Province, Western China. Environ. Pollut. 230, 189–198. https://doi.org/10.1016/j.envpol.2017.06.045 (2017).
Chambers, S., Williams, A., Crawford, J., Stelcer, E. & Dyer, L. Impact of meteorology on fine aerosols at Lucas Heights, Australia. Atmos. Environ. 145, 135 (2016).
Diehl, J. W. & Chiu, M. W. Effects of ambient sunlight and photoprotection on vitamin D status. Dermatol. Ther. 23(1), 48–60. https://doi.org/10.1111/j.1529-8019.2009.01290.x (2010).
Webb, A. R. Who, what, where and when-influences on cutaneous vitamin D synthesis. Prog. Biophys. Mol. Biol. 92(1), 17–25. https://doi.org/10.1016/j.pbiomolbio.2006.02.004 (2006).
Webb, A. R., Kline, L. & Holick, M. F. Influence of season and latitude on the cutaneous synthesis of vitamin D3: Exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J. Clin. Endocrinol. Metab. 67(2), 373–378. https://doi.org/10.1210/jcem-67-2-373 (1988).
Zegarska, B. et al. Air pollution, UV irradiation and skin carcinogenesis: What we know, where we stand and what is likely to happen in the future? Postepy Dermatol. Alergol. 34(1), 6–14. https://doi.org/10.5114/ada.2017.65616 (2017).
Funding
The study was supported by National Natural Science Foundation of China (CN) (82172441), Elderly Health Research Project of Jiangsu Province (CN) (LKZ2022020), Suzhou Collaborative Innovation Research Project of Medical and Industrial Integration (CN) (SLJ2022023), Clinical Medical Science and Technology Development Fund of Jiangsu University (CN) (JLY2021048) and Suzhou Key Clinical Diagnosis and Treatment Technology Project (CN) (LCZX202024).
Author information
Authors and Affiliations
Contributions
K.L. contributed to the study conception and design. Y.G., Y.X., X.W., L.C. and S.Y. contributed to the acquisition of data. K.L. and Y.G. contributed to the analysis and interpretation of data. C.L. drafted the manuscript, R.L. and K.L. revised it. All authors critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, C., Gong, Yq., Xia, Yy. et al. Particulate matter may have a limited influence on maternal vitamin D levels. Sci Rep 12, 16807 (2022). https://doi.org/10.1038/s41598-022-21383-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-21383-1
- Springer Nature Limited