Abstract
Hood canarygrass (Phalaris paradoxa L.) is a problematic weed in winter crops of Australia. Experiments were conducted to determine the effects of environmental factors on seed germination of P. paradoxa and wheat (Triticum aestivum L.) and herbicide options for P. paradoxa control. Results revealed that P. paradoxa had higher germination (> 89%) at a temperature range from 15/5 ℃ to 25/15 ℃ [day/night (12 h/12 h] compared with 30/20 ℃ and 35/25 ℃. At a temperature regime of 30/20 ℃, P. paradoxa had 1% germination; however, wheat at this temperature range resulted in 79% germination. Exposure of seeds of P. paradoxa to > 150 ℃ pretreatment (radiant heat for 5 min) resulted in no germination. These results suggest that infestation of P. paradoxa could be reduced by residue burning or by planting wheat crops early in the season when the temperature is relatively high. At a water potential of -0.8 MPa, seed germination of P. paradoxa and wheat was 75 and 96%, respectively. Similarly, at the highest salt concentration (200 mM sodium chloride), seed germination of P. paradoxa and wheat was 73 and 79%, respectively. These observations suggest that like wheat, P. paradoxa is also highly tolerant to water and salt stress conditions, therefore, it could invade the agro-ecosystem under water and salt stress situations. Germination of P. paradoxa was found to be low (10%) on the soil surface, suggesting that no-till systems could inhibit the germination of P. paradoxa. Pre-emergent (PRE) herbicides, namely cinmethylin, pyroxasulfone, and trifluralin, provided 100% control of P. paradoxa; however, in the presence of 2 t ha−1 of residue cover, pyroxasulfone provided better control of P. paradoxa compared with other herbicides. Post-emergent (POST) herbicides clethodim, haloxyfop, and paraquat provided excellent control of P. paradoxa, even if the plant size was large (10-leaf stage). Knowledge generated from this study will help in strengthening the integrated management of P. paradoxa.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Hood canarygrass (Phalaris paradoxa L.) is a major problematic weed in winter season crops of Australia1. It grows very well in alluvial, sandy-clay, or clay texture soils with high soil moisture conditions2. It has been observed that the high infestation of P. paradoxa in wheat (Triticum aestivum L.) paddocks of Australia could cause a 40% reduction in yield3. These observations suggest that P. paradoxa is a very competitive weed in wheat crops. In South Asia, it was observed that heavy infestation of P. minor (close relative species of P. paradoxa) could cause an 80% reduction in wheat yield4. It has been estimated that one plant of P. paradoxa can produce up to 21,000 seeds per plant in fallow conditions5 (www.crop.bayer.com.au). High seed production and seed shattering tendency of P. paradoxa could lead to reinfestation of this weed in paddocks. Under field conditions, P. paradoxa produced 3100 seeds m-2 in wheat and 50 seeds m-2 in barley (Hordeum vulgare L.)6.
Phalaris paradoxa is very similar to P. minor in appearance; however, P. minor has one sterile floret per spikelet whereas, P. paradoxa has two sterile florets per spikelet7. Both P. minor and P. paradoxa originated from the Mediterranean1. Phalaris paradoxa has become a serious weed in Australia due to distinct attributes such as its high seed production, innate dormancy, periodicity in emergence patterns, and the adoption of no-till agriculture6. As a result, this weed becomes the third most difficult grass weed after Avena fatua L. and Avena ludoviciana (wild oats) in the northern cropping regions of subtropical Australia. In Australia, P. paradoxa has evolved multiple herbicide resistance across two sites of action [“Fop” (aryloxyphenoxy propionates) and ALS group of herbicides]; therefore, there are reports of poor control of P. paradoxa with the use of these herbicides6,8.
Knowledge of the seed biology of weeds is essential for developing effective weed management strategies to control and delay the onset of herbicide resistance9. Seed germination behavior is an important factor for the establishment of weeds in cropping regions and understanding the weed seed ecology could play an important role in the decision-making process for weed control by formulating effective strategies10. Weed seed persistence in the soil is highly influenced by the germination and dormancy behaviors of the weeds under different environmental conditions11.
Despite the significant yield losses caused by P. paradoxa in Australian wheat production systems, knowledge regarding the seed germination ecology of P. paradoxa is limited. Weed seed persistence in a cropping region is always dependent on critical events of seed germination under different environmental conditions2. Environmental parameters, such as temperature, light, and water stress conditions, regulate seed germination12,13,14. As P. paradoxa is a significant weed in wheat crops, a better understanding of the germination and emergence behavior of P. paradoxa and wheat in response to environmental conditions (temperature, light, radiant heat, salinity, water stress, and burial depth) would be useful in developing effective management strategies for P. paradoxa in wheat.
Knowledge of optimum temperature conditions for weed seed germination could help in altering the sowing time of crops for reduced infestation of weeds15. Similarly, knowledge of the light requirement of seeds could help in managing weeds by using different tillage systems. It has been estimated that a 60% area of the Australian cropping region has sodic soils16. The accumulation of alkaline salts may increase the pH of many soils to as high as 9.0 17. The germination response of P. paradoxa and wheat seeds in relation to these factors (e.g., salinity) may determine weed invasiveness, the extent of crop-weed competition, and the pattern of weed spread18. From an ecological point of view, therefore, it is important to evaluate the effect of these environmental conditions on the germination and emergence of P. paradoxa and wheat.
There may be differing germination responses for P. paradoxa and wheat in relation to temperature regimes18. For instance, germination in the closely related species P. minor was highest (96%) at a temperature regime of 25/15 ℃ (day/night) and nil at 35/25 ℃19. However, such information is very limited for P. paradoxa under Australian conditions.
It has been observed that dormancy in P. paradoxa seeds could be overcome by placing imbibed seeds in the dark at 16 ℃ for either 7 or 14 d, followed by exposure to red or white light for a 15-min period2. In another study on a closely related species, P. minor, it was observed that germination was reduced in dark environments compared with light conditions 20. Considering these differences in germination responses of Phalaris species under different light and temperature conditions, it is pertinent to evaluate the germination behavior of P. paradoxa and wheat in response to different temperature and light conditions. Therefore, it is possible that the temperature and light response of P. paradoxa may be similar or different from that of wheat. Seed burial depth has an important role in inducing depth-mediated dormancy, especially in small-sized weed seeds21. Therefore, a better understanding of the burial depth factor that could affect the germination of P. paradoxa under no-till systems is needed. Such understanding will help in developing effective cultural management practices for this problematic weed.
There are reports of poor control of P. paradoxa with ACCase inhibiting herbicides in Australia. A similar type of situation was also observed in central and southern Italy, and Mexico22. Therefore, there is a need to evaluate alternative pre-emergent (PRE) and post-emergent (POST) herbicides for the control of P. paradoxa. The efficacy of PRE herbicides may vary under different residue covers in paddocks due to the adsorption of herbicides23. The efficacy of POST herbicides may vary with the size of weed plants in the paddock. Therefore, it is pertinent to evaluate PRE and POST herbicides under these situations for better control of P. paradoxa.
The primary objective of this study was to evaluate the effects of temperature, light quantity, radiant heat, salt stress, water stress, and burial depth on seed germination between both P. paradoxa and wheat. A secondary objective was to evaluate the performance of different PRE and POST herbicides on P. paradoxa control.
Material and methods
Seed collection and multiplication
For experimental studies, seeds of P. paradoxa were initially collected from approximately 200 plants grown at Millmerran, Queensland, Australia (27.82625°S, 151.3513°E) in January 2018. For collecting seeds, permission was accorded from the consultants through phone calls and personal meetings. The spikes of P. paradoxa were collected at the maturity stage of plants. Seeds were then threshed from spikes by gentle rubbing on hands at the Weed Science Laboratory (27.5552°S, 152.343°E), Gatton, Australia. After cleaning, seeds were stored in paper bags at room temperature (25 °C). Seeds were multiplied in a screenhouse in 2019 by growing plants in pots using potting mix (Centenary Landscaping, Brisbane, Queensland, Australia). For the multiplication of seeds, plants of P. paradoxa were grown in May 2019, and seeds were collected in October 2019. Fresh seeds from the matured plants grown in 2019 were used for experimental studies. For studies on wheat, seeds of a commercial variety Spitfire were used. Each study was conducted two times. All local, national or international guidelines and legislation were adhered for the use of plants in this study.
Effect of temperature
The study was conducted in a factorial, completely randomized design with three replications. The first factor was plant species (wheat and P. paradoxa) and the second factor was temperature regime [15/5, 20/10, 25/15, 30/20, and 35/25 °C (day/night temperatures 12 h/12 h)]. Treatment-wise, 25 seeds per treatment were sown in Petri dishes lined with double-layer WhatmanTM filter paper that was moistened with 5 mL of tap water. A micropipette (BOECO Germany) was used to add 5 mL water. After sowing, Petri dishes were put in sealable plastic bags. The bags were sealed and transferred to incubators (Labec Laboratory Pvt. LTD, Australia) at the respective temperature regime. Germination data were taken 21 d after sowing. Seeds were considered germinated when at least a 2 mm radical was observed.
Effect of light intensity
The study was conducted in a factorial, completely randomized design with three replications. The first factor was two plant species: wheat (Triticum aestivum L.) and P. paradoxa; and the second factor was five light intensity levels (0, 30, 50, 70, and 100%). Seeds were sown in Petri dishes in a similar way as in the temperature experiment and were incubated at 20/10 °C (day/night 12 h/12 h temperature). For zero light intensity, Petri dishes were covered with double aluminum foils. For 30, 50, and 70% light intensity, Petri dishes were covered with meshes of different intensities. The light intensity was measured using a Quantum meter (MQ-200 Apogee Instruments, USA). Germination data were taken 21 d after sowing.
Effect of radiant heat
To examine the effect of radiant heat on seed germination, 600 seeds of P. paradoxa were divided into six paper bags, each containing 100 seeds. The paper bags were labeled as per respective preheat treatments (25, 50, 100, 150, 200, and 250 °C). The hotter temperatures of the heat treatment were selected to simulate temperatures during the burning of crop straws in a field (Cook 1939). Seeds stored at room temperature were used for the 25 °C treatment and other treatments were conducted in an oven at specific temperatures for 5 min. After heat treatment, seeds were sown in Petri dishes in a similar way as in the temperature experiment and incubated at 20/10 °C. Germination data were taken 21 d after sowing.
Effect of osmotic stress
The study was conducted in a factorial, completely randomized design with three replications. The first factor was plant species (wheat and P. paradoxa) and the second factor was water stress levels (0, −0.1, −0.2, −0.4, −0.8, and −1.6 MPa). Seeds were sown in Petri dishes in a similar way to the temperature experiment, but filter papers were moistened with 5 mL of different concentrations of osmotic potentials, which were prepared using polyethylene glycol (PEG 8000) (Sigma-Aldrich Co., St. Louis, MO). Seeds were incubated at 20/10 °C. Germination data were taken 21 d after sowing.
Effect of salinity
The study was conducted in a factorial, completely randomized design with three replications. The first factor was plant species (wheat and P. paradoxa) and the second factor was salinity levels (0, 25, 50, 100, 150, and 200-mM sodium chloride, NaCl). Seeds were sown in Petri dishes in a similar way as in the temperature experiment, but filter papers were moistened with 5 ml of different concentrations of NaCl. Seeds were incubated at 20/10 °C. Germination data were taken 21 d after sowing.
Effect of burial depth
For the burial depth experiment, field soil (after sieving through a 0.3 cm mesh) was used. The soil used was clay loam in texture, with organic matter comprising 2.7%, and a pH of 7.2. The experiment was conducted in pots having a diameter of 12 cm. Pots were filled with the sieved soil leaving the corresponding depth to the top of the pot as 0, 0.5, 1, 2, 4, and 8 cm. Forty seeds of P. paradoxa were sown in each pot and covered with the same soil to achieve the desired depth. The experiment was conducted in a completely randomized design and each treatment was replicated thrice. The pots were regularly irrigated using a sprinkler system to avoid any moisture stress. Seedling emergence data were collected at 28 d after sowing. Seedlings were considered emerged when the coleoptiles were seen above the soil surface.
Evaluation of PRE-herbicides
The study was conducted in a factorial, completely randomized design with three replications. The first factor was plant species (wheat and P. paradoxa) and the second factor was herbicide treatment (pendimethalin 594 g ai ha−1, triallate 800 g ai ha−1, trifluralin 384 g ai ha−1, atrazine 585 g ai ha−1, pyraxosulfone 100 g ai ha−1, s-metolachlor 240 g ai ha−1, isoxaflutole 75 g ai ha−1, dimethenamid-P 720 g ai ha−1, imazethapyr 70 g ai ha−1, prosulfocarb + s-metolachlor 2300 g ai ha−1, bixlozone 500 g ai ha−1, and cinmethylin 375 g ai ha−1). The study was conducted in 12 cm diameter pots filled with potting mix. Twenty seeds were sown in each pot. After sowing, seeds were covered with a thin layer (0.5 cm) of potting mix. Spraying was done using a research track sprayer equipped with Teejet XR 110,015 flat fan nozzles calibrated to an output spray volume of 108 L ha-1. Plant survival was recorded 28 d after sowing.
Efficacy of PRE-herbicides in relation to crop residue cover
The study was conducted in a factorial, completely randomized design with three replications. The first factor was crop residue level (0, 2, and 6 t ha−1) and the second factor was PRE herbicide (pyraxosulfone 100 g ai ha−1, cinmethylin 375 g ai ha−1, triallate 800 g ai ha−1, pendimethalin 594 g ai ha−1, isoxaflutole 75 g ai ha−1, imazethapyr 70 g ai ha−1). Pots (12 cm diameter) were filled with potting mix and 20 seeds were sown in each pot. After sowing, seeds were covered with chopped (about 2 cm in length) wheat residue according to treatment. The residue was used after oven drying the wheat straw in an oven at 70 C for 72 h. Herbicide spray was done using a research track sprayer equipped with Teejet XR 110,015 flat fan nozzles calibrated to an output spray volume of 108 L ha−1. Data on plant survival was recorded 28 d after sowing.
Efficacy of POST herbicides in relation to plant size
The study was conducted in a factorial, completely randomized design with three replications. The first factor was the size of the plant (4-leaf and 10-leaf stages) and the second factor was herbicide treatment (clodinafop 20 g ai ha−1, propaquizafop 30 g ai ha−1, clethodim 120 g ai ha−1, butroxydim 45 g ai ha−1, pinoxaden 20 g ai ha−1, haloxyfop 78 g ai ha−1, imazamox + imazapyr 36 g ai ha−1, glyphosate 741 g ae ha−1, glufosinate 750 g ai ha−1 and paraquat 600 g ai ha−1). For this study, pots (12 cm diameter) were filled with potting mix and 10 seeds were sown in each pot. After emergence, only 6 plants per pot were maintained. Plants were watered regularly to avoid any water stress. For larger plants (10-leaf stage), sowing was done two weeks earlier than the 4-leaf stage plants, so that herbicide spray could be done on the same day. Herbicide spray was done using a research track sprayer equipped withTeejet XR 110,015 flat fan nozzles calibrated to an output spray volume of 108 L ha−1. Data on plant survival was recorded 28 days after sowing.
Statistical analyses
Each experiment was repeated once over time. Each dataset represents the average of the two runs, as there was no time by treatment interaction as determined by analysis of variance (ANOVA). Except for the PRE herbicide x residue experiment, the homogeneity of variance was not improved by transformation, and residuals were normally distributed; thus, ANOVA was performed on nontransformed germination percentage values (using GENSTAT 16th Edition; VSN International, Hemel Hempstead, UK). Survival data in the PRE herbicide x residue experiment were subjected to square-root transformation (√x + 0.5) before analysis. ANOVA was used to identify any significant treatment and interaction effects (P ≤ 0.05). Where treatments were significant (P ≤ 0.05), means were separated using Fisher’s protected LSD test.
Results and discussion
Effects of light intensity and temperature
The germination of P. paradoxa (91 to 95%) and wheat (93 to 97%) was not affected by light intensity (data not shown). Our results conform to previous studies which revealed that light intensity had little role in influencing P. paradoxa germination24.
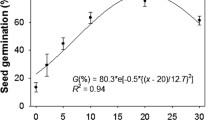
The germination of wheat and P. paradoxa was influenced by temperature regimes (Fig. 1). At temperature regimes of 15/5 °C and 20/10 °C, germination of wheat and P. paradoxa did not vary. Seed germination in wheat remained similar at temperatures ranging between 15/5 °C to 30/20 °C. However, in P. paradoxa, germination was reduced at higher temperature regimes (35/25 C) compared with lower temperature regimes (15/5 °C to 25/15 °C). At the highest temperature regime (35/25 °C), the germination of wheat was 79%, while, at this temperature regime, the germination of P. paradoxa was only 1%. This suggests that wheat can germinate at high-temperature ranges, while, germination of P. paradoxa may be reduced at high temperatures (35/25 °C). These results implied that at the time of planting wheat in Australia if the air temperature is low, the chances of emergence of P. paradoxa are very high. This suggests that efforts should be made towards early control of P. paradoxa in wheat if the air temperature in the winter season falls early. These results also suggest that early planting of wheat could reduce the emergence of P. paradoxa as the prevailing temperature conditions are relatively high in early planting (e.g., end of April). In the Indo-Gangetic Plains, better control of P. minor was observed in the early planting of wheat (high-temperature conditions) due to less emergence of P. minor25.
Previous studies have also revealed that germination of P. paradoxa was highest at 10 °C and then failed to germinate at 30 °C 24,26, however, these studies were conducted at constant temperatures and the germination response of P. paradoxa was not studied in comparison with wheat in those studies.
Effect of radiant heat
The germination of P. paradoxa seeds that were stored at room temperature (25 °C) was 97%, which reduced to 88% after exposure to the 100 °C pretreatment for 5 min and became nil at 150 °C (Fig. 2). About 88% of P. paradoxa at 100 °C suggests that it can tolerate heat stress for short periods.
Germination was nil at 150 °C and above, suggesting that burning could help in managing P. paradoxa, particularly in a no-till field where seeds are on the soil surface or at shallow depths. Exposure of seeds to fire could inhibit germination by desiccating the seed coat or by damaging the embryo27,28,29.
Burning of residue in the fields could kill weed seeds and other pests in the topsoil layer30. Windrow burning proved to be an effective tool for killing weed seeds in paddocks31. However, the crop residue burning may cause environmental destruction by killing microbes and polluting the air. Also, it reduces the amount of soil organic matter due to the high heat, causing soil degradation. Therefore, these aspects should also be considered while formulating weed management strategies through crop residue burning. Burning may also release the dormancy of other weed seeds present in the subsoil and thus may increase infestation; therefore, this technique should be used cautiously32,33.
Effect of osmotic stress
Germination of P. paradoxa was highest (95%) in the control treatment and germination reduced to 75% at an osmotic potential of −0.8 MPa, and became nil at −1.6 MPa (Fig. 3). However, in wheat, germination did not reduce with an increase in water potential and it was 94% in the control treatment.
At a very high concentration of PEG, the metabolic activity of P. paradoxa might be reduced due to water stress. Seed germination is affected when seeds are not able to get critical moisture threshold levels for imbibitions34,35. These results indicate that high water stress may inhibit the seed germination of P. paradoxa. However, under no water stress or mild water stress conditions, P. paradoxa may infest the wheat crop.
Contrary to these results, previous studies reported that germination of P. paradoxa was reduced by 90% at an osmotic potential of −0.25 MPa25. Good germination of wheat at high osmotic potential indicates that the wheat variety used in this study may have water stress tolerance traits for germination. It was observed that wheat could germinate well (75%) at a high-water stress level (−1.6 MPa)36. This suggests that it is possible to menace P. paradoxa by growing stress-tolerant varieties of wheat and manipulating irrigation. In a previous study, less infestation of P. paradoxa was observed in drip-irrigated wheat crops due to optimal soil moisture conditions for the crop37.
Effect of salt stress
Germination of P. paradoxa was highest (93%) in the control treatment, and at a NaCl of 150 mM, germination was reduced to 76% (Fig. 4). Similarly, in wheat, germination was highest (94%) in the control treatment and at a salt concentration of 150 and 200 mM, germination was reduced to 84 and 79%, respectively. These results suggest that at a high salt concentration, P. paradoxa may infest the wheat crop owing to its ability to germinate under high salt concentrations.
Contrary to this, in Iran, it was observed that germination of P. paradoxa was reduced by 70% at a NaCl of 160 mM24. Most of the Australian soils are saline; therefore, it is quite possible that P. paradoxa in Australia might have developed traits for salt tolerance38. The variable response of populations of P. paradoxa to salt concentrations in Iran and Australia might be due to genetic differences between the P. paradoxa populations38. These observations suggest that P. paradoxa could invade the agroecosystem under the saline conditions of Australia.
Effect of seed burial depth on emergence
Germination of P. paradoxa was very low (10%) on the soil surface, and seedling emergence was highest (74%) at a soil burial depth of 0.5 cm (Fig. 5). Seedling emergence was similar when seeds were buried in the soil at a depth ranging from 0.5 to 4 cm. Seedling emergence was 32% at a burial depth of 8 cm.
The results from this experiment suggest that a no-till production system may inhibit the germination of P. paradoxa. This study also suggests that deep tillage (> 4 cm) could reduce the emergence of P. paradoxa to some extent; therefore, inversion tillage could be a weed management strategy if the seedbank is in the shallow layer of the soil. It has been reported that the emergence of small-seeded weeds is reduced from deeper burial depths, as the soil-gas exchange is limited 21. However, it is important to know the seed longevity of this weed in different soil and environmental conditions when considering tillage operations39.
Likewise, previous studies also reported that seed germination of P. paradoxa was lowest on the soil surface and no seedlings emerged from a soil depth of 10-cm2,40. Contrary to this in Iran, germination of P. paradoxa was found to be > 65% on the soil surface 24.
Evaluation of PRE-herbicides
Results revealed that cinmethylin, pyroxasulfone, and trifluralin provided 100% control of P. paradoxa. Atrazine, bixlozone, imazethapyr, isoxaflutole, prosulfocarb + s-metolachlor, and s-metolachlor were not found to be effective against P. paradoxa (Table 1). Pendimethalin and triallate controlled P. paradoxa by 80 and 42%, respectively, compared with the nontreated control.
In wheat, all tested herbicides performed similarly for plant survival except dimethenamid-P and prosulfocarb + s-metolachlor, which caused wheat mortality by 41 and 16%, respectively, compared with the nontreated control. These results suggest that pyroxasulfone, pendimethalin, and trifluralin can be successfully used for the management of P. paradoxa in wheat. Alternative use of these herbicides in wheat crops could provide sustainable weed control of P. paradoxa. In previous studies conducted in Australia, herbicides namely cinmethylin, pyroxasulfone, and trifluralin were found safe for wheat and provided excellent grass weed control41.
Efficacy of PRE-herbicides in relation to crop residue cover
Cinmethylin, pendimethalin, and pyroxasulfone were proven to be very effective against P. paradoxa under no residue cover conditions (Table 2). However, at the residue cover of 6 t ha-1 (high output systems), the efficacy of these herbicides decreased and these three herbicides failed to provide effective control of P. paradoxa. At the residue cover of 2 t ha-1 (low output systems), the efficacy of pyroxasulfone in controlling P. paradoxa was not affected; however, cinmethylin and pendimethalin at the residue load of 2 t ha-1 did not control P. paradoxa. These results suggest that in a residue-retained, no-till system, pyroxasulfone could provide better control of P. paradoxa compared with cinmethylin and pendimethalin.
The crop residue binds some herbicides, which results in a reduced dose to target weeds and provides poor weed control42. A crop residue cover of 1 t ha-1 may prevent 50% of the herbicide from reaching the target weed seeds in the soil and thus provide poor weed control43.
Efficacy of POST herbicides in relation to plant size
When plants were sprayed at the 4-leaf stage, the herbicides clodinafop and propaquizafop were not effective against P. paradoxa compared with the other tested herbicides (Table 3). The efficacy of clethodim, glyphosate, haloxyfop, and paraquat in controlling P. paradoxa was not decreased even when plants were sprayed at the 10-leaf stage. In previous studies, poor control of P. paradoxa was observed with ACCase-inhibiting herbicides44,45. These results also suggest that under noncropped or fallow situations, early and late cohorts of P. paradoxa can be controlled successfully by delaying applications of clethodim, paraquat, haloxyfop, and glyphosate.
Germination of P. paradoxa at 25/15 °C (day/night) was lower compared with 20/10 °C. This suggests that early sowing of wheat (relatively high-temperature conditions) could reduce the emergence of P. paradoxa in fields. Phalaris paradoxa did not germinate after exposure to radiant heat of 150 °C (for 5 min), which suggests that burning may be a useful tool for managing P. paradoxa, particularly when seeds are on the soil surface or at the shallow surface. A high level of tolerance of P. paradoxa to water and salt stress was observed. These observations suggest that this weed can dominate under saline and water stress conditions in Australia. Low germination of P. paradoxa was observed on the soil surface, suggesting that a no-till system could provide better control of P. paradoxa. PRE herbicides cinmethylin, pyroxasulfone, pendimethalin, and trifluralin were effective for control of P. paradoxa in wheat; however, under a conservation tillage system, pyroxasulfone provided better control of P. paradoxa compared with other herbicides. Haloxyfop and clethodim were the most effective herbicides among the ACCase-inhibiting herbicides. Under noncropped or fallow land situations, larger plants of P. paradoxa can be successfully controlled with the application of clethodim, glyphosate, and paraquat.
Data availability
All relevant data are within the paper and its Supporting Information files.
References
Baldini, R. M. Revision of the genus Phalaris L. (Gramineae). Webbia 49, 65–329 (1995).
Taylor, I. N., Walker, S. R. & Adkins, S. W. Burial depth and cultivation influence emergence and persistence of Phalaris paradoxa seed in an Australian sub-tropical environment. Weed Res. 45, 33–40 (2005).
Dellow, J. J. & Milne, B. R. Control of Phalaris paradoxain wheat. Australas. Weeds 31, 22–23 (1986).
Singh, S., Kirkwood, R. C. & Marshall, G. Biology and control of Phalaris minor Retz. (little seed canary grass) in wheat. Crop Prot. 18, 1–16 (1999).
Wilson, B. J. Effect of time of seedling emergence on seed production and time to flowering of eight weeds. In Proceedings of the Sixth Australian Weeds Conference, Volume 1, City of Gold Coast, Queensland, Australia, 13–18 September 1981 1981 (pp. 35–38). Queensland Weed Society.
Walker, S. R., Robinson, G. R. & Medd, R. W. Management of Avena ludoviciana and Phalaris paradoxa with barley and less herbicide in subtropical Australia. Aust. J. Exp. Agric. 41, 1179–1185 (2001).
Matus-C’adiz, M. & Hucl, P. Morphological variation within and among five annual Phalaris species. Can. J. Plant Sci. 82, 85–88 (2002).
Storrie, A.& Edwards, J. Herbicide resistance in northern New South Wales. Agnote DPI/211. New South Wales Department of Agriculture. 1998.
Mahajan, G. & Chauhan, B. S. Weed biology—A required foundation for effective weed management. Weeds J. Asian-Pacific Weed Sci. Soc. 2, 30–34 (2020).
Chauhan, B. S. & Johnson, D. E. The role of seed ecology in improving weed management strategies in the tropics. Adv. Agron. 105, 221–262 (2010).
Baskin, J. M. & Baskin, C. C. Temperature requirements for after ripening in seeds of nine winter annuals. Weed Res. 26, 375–380 (1986).
Taylorson, R. B. Environmental and chemical manipulation of weed seed dormancy. Weed Sci. 3, 135–154 (1987).
Chachalis, D. & Reddy, K. N. Factors affecting Campsis radicans seed germination and seedling emergence. Weed Sci. 48, 212–216 (2000).
Koger, C. H., Reddy, K. N. & Poston, D. H. Factors affecting seed germination, seedling emergence, and survival of texasweed (Caperonia palustris). Weed Sci. 52, 989–995 (2004).
Xu, G. F., Zhang, F. D., Li, T. L., Shen, S. C. & Zhang, Y. H. Influence of environmental factors on seed germination of Phalaris paradoxa and Phalaris minor. Acta Botanical Boreali-Occidentalia Sinica 31, 1458–1465 (2011).
Rengasamy, P. Soil processes affecting crop production in salt–affected soils. Funct. Plant Biol. 37, 613–620 (2010).
de Caritat, P., Cooper, M. & Wilford, J. The pH of Australian soils: field results from a national survey. Soil Res. 49, 173–182 (2011).
Ahmadi, A., Hosseinin, M. & Zeidali, E. Study of ecological characteristics of canary grass Phalaris minor. Tech. J. Eng. Appl. Sci. 3, 1835–1840 (2013).
Ahmadi, A. The effect of temperature, salinity and soil burial on canary grass (Phalaris minor) germination. Jordan J. Agric. Sci. 405, 1–10 (2016).
Alshallash, K. S. Germination of weed species (Avena fatua, Bromus catharticus, Chenopodium album and Phalaris minor) with implications for their dispersal and control. Annals Agric. Sci. 63, 91–97 (2018).
Benvenuti, S., Macchia, M. & Miele, S. Quantitative analysis of emergence of seedlings from buried weed seeds with increasing soil depth. Weed Sci. 49, 528–535 (2001).
Gasparetto, M. A. Dinamica di emergenza delle infestanti del frumento duro. Informatore Agrario 59(12), 77–82 (2003).
Chauhan, B. S. & Mahajan, G. Role of integrated weed management strategies in sustaining conservation agriculture system. Curr. Sci. 103, 135–136 (2012).
Rezvani, M., Nadimi, S., Zaefarian, F. & Chauhan, B. S. Environmental factors affecting seed germination and seedling emergence of three Phalaris species. Crop Protection 148, 105743 (2021).
Mahajan, G. & Brar, L. S. Integrated management of Phalaris minor in wheat. Indian J. Weed Sci. 33, 9–13 (2001).
Jimenez Hidalgo, M.J., Saavedra, M. & Garcia-Torres, L. Germination of Phalaris species as affected by temperature and light. In Proceedings of the 1993 Congress of the Spanish Weed Science Society, Lugo, Spain, 1–3 December 1993. (pp. 98–102). Sociedad Española de Malherbología (Spanish Weed Science Society) (1993)
Baskin, C. C. & Baskin, J. M. Seeds: Ecology, biogeography, and evolution of dormancy and germination (Elsevier Academic Press, 2014).
Jeffery, D. J., Holmes, P. M. & Rebelo, A. G. Effects of dry heat on seed germination in selected indigenous and alien legume species in South Africa. S. Afr. J. Bot. 54, 28–34 (1988).
Van de Venter, H. A. & Esterhuizen, A. D. The effect of factors associated with fire on seed germination of Erica sessiliflora and E. hebecalyx (Ericaceae). South African J. Botany 54, 301–304 (1988).
Virto, I., Imaz, M. J., Enrique, A., Hoogmoed, W. & Bescansa, P. Burning crop residues under no-till in semi-arid land, Northern Spain—effects on soil organic matter, aggregation, and earthworm populations. Soil Res. 45, 414–421 (2007).
Walsh, M. & Newman, P. Burning narrow windrows for weed seed destruction. Field Crop Res 104, 24–30 (2007).
Ali, H. H., Tanveer, A. & Nadeem, M. A. Evaluation of some seed dormancy breaking methods on germination of Rhynchosia capitata (Roth DC). Pakistan J. Weed Sci. Res. 18, 423–432 (2012).
Moreira, B. & Pausas, J. G. Tanned or burned: The role of fire in shaping physical seed dormancy. PLoS ONE 5, e51523 (2012).
Bittencourt, H. V., Bonome, L. T., Trezzi, M. M., Vidal, R. A. & Lana, M. A. Seed germination ecology of Eragrostis plana, an invasive weed of South American pasture lands. S. Afr. J. Bot. 109, 246–252 (2017).
Fenner, M. & Thompson, K. Seeds (Cambridge University Press, 2005).
Wuest, S. B. & Lutcher, L. K. Soil water potential requirement for germination of winter wheat. Soil Sci. Soc. Am. J. 77, 279–283 (2013).
Nadeem, M. A., Tanveer, A., Ali, A., Ayub, M. & Tahir, M. Effect of weed-control practice and irrigation levels on weeds and yield of wheat Triticum aestivum. Indian J. Agron. 52, 60–63 (2007).
Taylor, I. N., Peters, N. C., Adkins, S. W. & Walker, S. R. Germination response of Phalaris paradoxa L. seed to different light qualities. Weed Research 44, 254–64 (2004).
Mahajan, G. & Chauhan, B. S. Seed longevity and seedling emergence behavior of Avena fatua and Avena ludoviciana in response to burial depth in eastern Australia. Weed Sci. 69, 362–371 (2021).
Taylor, I., Walker, S., Adkins, S., Bullen, K. & Peters, N. Dynamics of paradoxa grass (Phalaris paradoxa L.) soil seedbank. In12th Australian Weeds Conference, Papers and Proceedings, Hobart, Tasmania, Australia, 12–16 September 1999: Weed management into the 21st century: do we know where we're going? 1999 (pp. 71–74). University of Tasmania (1999).
Boutsalis, P., Gill, G. S. & Preston, C. Control of rigid ryegrass in Australian wheat production with pyroxasulfone. Weed Technol. 28, 332–339 (2014).
Chauhan, B. S. & Abugho, S. B. Interaction of rice residue and PRE herbicides on emergence and biomass of four weed species. Weed Technol. 26, 627–632 (2017).
Banks, P. A. & Robinson, E. L. Soil reception and activity of acetochlor, alachlor, and metolachlor as affected by wheat (Triticum aestivum) straw and irrigation. Weed Sci. 34, 607–611 (1986).
Welsh, R.D. Propargyl-a post-emergent herbicide for selective control of wild oats and canary grasses in wheat, triticale and durum. In Proceedings of the forty-seventh New Zealand plant protection conference, Waitangi, New Zealand, 9–11 August 1994. (pp. 22–26). New Zealand Plant Protection Society (1994).
Cruz-Hipolito, H., Domínguez-Valenzuela, J. A., Osuna, M. D. & De Prado, R. Resistance mechanism to acetyl coenzyme A carboxylase inhibiting herbicides in Phalaris paradoxa collected in Mexican wheat fields. Plant Soil 355, 121–130 (2012).
Acknowledgements
This research did not receive any specific funding.
Author information
Authors and Affiliations
Contributions
V.K. ran the experiment and recorded the data. G.M. and B.S.C. designed the study. V.K. and G.M. wrote the initial draft; B.S.C. edited the manuscript. All authors read and approved the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kibasa, V., Mahajan, G. & Chauhan, B.S. Seed germination ecology of hood canarygrass (Phalaris paradoxa L.) and herbicide options for its control. Sci Rep 12, 15241 (2022). https://doi.org/10.1038/s41598-022-19418-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19418-8
- Springer Nature Limited