Abstract
Median nerve cross-sectional area (CSA) was used for screening and diagnosis of neuropathy, but few studies have suggested reference range. Hence, this systematic review was performed to evaluate a normative values of median nerve CSA at various landmarks of upper limb based on ultrasonography. PubMed and Web of science were used to search relevant articles from 2000 to 2020. Forty-one eligible articles (2504 nerves) were included to access median nerve CSA at different landmarks (mid-arm, elbow, mid-forearm, carpal tunnel (CT) inlet and CT outlet). Data was also stratified based on age, sex, ethnicity, geographical location, and method of measurement. Random effects model was used to calculate pooled weighted mean (95% confidence interval (CI), [upper bound, lower bound]) at mid-arm, elbow, mid-forearm, CT inlet and outlet which found to be 8.81 mm2, CI [8.10, 9.52]; 8.57 mm2 [8.00, 9.14]; 7.07 mm2 [6.41, 7.73]; 8.74 mm2 [8.45, 9.03] and 9.02 mm2 [8.08, 9.95] respectively. Median nerve CSA varies with age, geographical location, and sex at all landmarks. A low (I2 < 25%) to considerable heterogeneity (I2 > 75%) was observed, indicating the variation among the included studies. These findings show that median nerve CSA is varying not only along its course but also in other sub-variables.
Similar content being viewed by others
Introduction
In the long course of the median nerve through the upper limb, there are multiple potential sites for lesions or injuries to occur. These lesions could be secondary to local aetiologies like trauma and compressive masses, or polyneuropathies such as diabetes mellitus, demyelinating diseases and rheumatoid arthritis causing nerve entrapment1,2. To evaluate the morphological changes of the median nerve in peripheral neuropathy, ultrasound technology has become a popular means3. Ultrasonography allowed nerve conduction studies to be supplemented via measurements of the nerve cross-sectional area (CSA).
Ultrasonography studies have commonly compared median nerve CSA between healthy controls and patients with peripheral neuropathy, thus establishing that CSA tend to increase in carpal tunnel syndrome and diabetic neuropathy4,5, whereas it is decreased in neurodegenerative conditions such as amyotrophic lateral sclerosis6. While these studies have stated the CSA cut-off for diagnosing their conditions, limited studies have suggested normative ranges of median nerve CSA which could potentially be applied for screening and diagnosis of other conditions. Therefore, this systematic review was aimed to report the normal median nerve CSA along its course at predetermined anatomical landmarks based on the reported ultrasound studies. This study also analysed the median nerve CSA in subgroup variables such as age, sex, ethnicity, geographical location, and method of CSA measurement. A common reference would be helpful for clinicians in improving screening and diagnostic methods for neuropathy of the median nerve.
Materials and methods
Study design, search strategy and selection process
This is a non-interventional systematic review of published articles from online databases to evaluate the range of median nerve CSA in the upper limb using ultrasonography. Article selection process was conducted and documented with the Preferred Reporting Items for Systematic reviews (PRISMA) and Meta-Analyses guidelines7. Using broader search terms, authors systematically searched articles from electronic databases—PubMed and Web of Science until 31 December 2020. The keywords used for the search strategy were “Median nerve”, “reference values” or “normative range”, “ultrasound imaging” and/or “ultrasonography”.
The studies were selected and included as per following eligibility criteria.
-
1.
Peer-reviewed studies, written and published in the English language were included. Non-English articles, case reports, conference abstracts, editorial letters, expert opinion, articles without primary datas or incomplete results were excluded8. Given the development of ultrasound technology over the last 20 years3, empirical studies published from the year 2000 onwards were considered.
-
2.
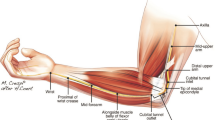
Articles were screened and included for the preliminary review that encompassed the primary variable of median nerve CSA, measured using ultrasonography in healthy volunteers, including control populations, at minimally one of the five proxy landmarks: the mid-arm, elbow (cubital fossa), mid-forearm, carpal tunnel (CT) inlet (level of the pisiform), and CT outlet (level of the hook of the hamate) (Fig. 1). These landmarks were the most common sites for measuring median nerve CSA.
-
3.
Studies were excluded if the mean CSA was not reported, not obtained from main trunk of median nerve with the proxy landmarks not listed or undistinguished, and/or if data was obtained from pregnant women, nursing mothers or patients with history of neuropathy or associated risk factors. The list of risk factors associated with neuropathy is reported in the Supplementary Table 1.
-
4.
Study subjects were assessed with minimal age of 19 years because epiphyseal closure in the longer bones of upper limb is expected to happen by this age9.
-
5.
Duplicated records of articles were excluded, remaining records were screened by titles and abstracts according to the eligibility criteria described above. Subsequently, articles included from screening were assessed in full text for review and the reasons for exclusion were noted. The flowchart for study identification and evaluation is shown in the Fig. 2.
Data extraction
Datas from the included articles were manually extracted and reviewed as per PRISMA guidelines. Sample size, mean, standard deviation and range of median nerve CSA for each of the anatomical landmark were recorded in Microsoft excel 2010 spreadsheet (Microsoft Corp., Redmond, WA) for the analysis. Secondary variables such as age, sex, geographical location, ethnicity, and method of measurement, were also noted for further stratification of the CSA. Some studies did not report data based on ethnicity, hence meta-analysis was reported as “unspecified ethnicity” and calculations were performed based on geographical location. The methods of measuring median nerve CSA and the boundary defining the median nerve was also noted, where this information was explicitly stated in the included studies. Information on positioning of upper limb, the number of operators for ultrasonic measurements was also recorded to supplement the discussion of results.
Statistical analysis
Statistical analyses were performed using Open Meta software using R console (CEBM, Brown University)10. The data from the included articles were noted as continuous variables and the analysis was performed using mean, standard deviation and reported sample size (number of median nerves). The overall mean CSA was calculated for the articles that reported the mean value of CSA for subpopulations (for example male and female), using this equation—Overall mean CSA = [sum of (N × mean CSA)] ÷ sum of N, where N represents the number of upper limbs/median nerves measured11. Likewise, for the studies where the standard deviations were not provided, standard deviation was calculated manually using interquartile range, median and/or P values12,13.
The frequency of variations from the included studies were computed from weighted mean estimates using DerSimonian–Lard method14,15. A continuous random-effects model was used for the meta-analysis. The measures for all included studies were calculated at 95% confidence intervals (CI), showing the upper and lower boundary limits. Statistical heterogeneity of the pooled weighted mean was quantified using chi square \(x\)2 (represented by Cochran’s Q and P value) and I2 range, interpreted using Cochrane Handbook16. The I2 statistic measured the degree of inconsistency among the studies, indicated by the percentage of total variation effects. The thresholds for I2 were indicated as low or might not important (I2 < 25%), moderate (I2 = 30–60%), substantial (I2 = 50–90%) and a considerable heterogenicity (I2 = 75–100%)17. In addition, a Chi square test with Cochran’s Q, P < 0.10 was also considered for heterogenecity17.
In addition, appropriate subgroup analysis was performed to make comparisons and to identify the sources of heterogeneity. The variability was investigated based on sex, geographical location, ethnicity, and method of measurement with the datas stratified for each anatomical landmark in upper limb measuring median nerve CSA. Majority of the studies did not report median nerve CSA based on age groups, instead the overall mean age was available. Hence, for the purpose of analysis, we classified the age groups as young (19–40 years), middle age (41–65) and elderly (> 65) based on the mean age reported from the eligible studies. There were no clear scientific guidelines on the classification of age into different groups in the context of the evaluation of the CSA in human peripheral nerves such as median nerve. The age classification reported in this study is consistent in a clinicopathological study involving the gastric adenocarcinoma patients from different age groups18. The I2 statistic, weighted mean and standard error was calculated for each subgroup(s) at 95% confidence intervals based on Cochrane guidelines17.
Quality and risk of bias assessment
The quality of the included studies was evaluated using Anatomical Quality Assessment (AQUA) tool19. This reporting is important as the poorly reported studies with lack of information may decrease the reliability, thus increase the potential risk of bias19. Studies were probed individually and judged by assessing five domains—objectives and characteristics of the study/subjects, design of the research study, methodology, description of anatomical structures and reporting of results. AQUA assessment guidelines define “low” as low risk of bias when the reported information is sufficient to reproduce. Studies that reported ambiguous and missing baseline information were considered to lack proper quality, hence they were ranked as “High” and “Unclear” risk of bias. Investigators #1 and #2 rated the included studies independently and all the disagreements were resolved through detailed discussion among the investigators.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Informed consent is not relevant in this case as this is a non-interventional literature review of published articles from online databases.
Results
The initial literature search screened and potentially identified a total of 311 articles (158 records in PubMed and 153 in Web of Science), searched up to 31st December 2020. Subsequently, 61 duplicates were removed. The remaining 250 articles were scrutinized based on titles and abstracts. 130 papers were excluded further as their abstracts lacks the information on the median nerve and ultrasonography. From this, 120 potentially eligible articles were identified and 78 were excluded further (studies neither does not have full text nor failed to meet inclusion criteria), resulting in extraction of 42 studies meeting the inclusion criteria. Among the 42 studies, one study20 was excluded for the meta-analysis due to missing descriptive statistics information and hence 41 studies, reporting 2504 median nerves in 1614 healthy individuals were used for final systematic review. The number of articles that presented the mean median nerve CSA were seven6,21,22,23,24,25,31, seven4,22,23,26,27,28,29, fourteen4,6,22,24,25,26,27,29,30,31,32,33,34,35, twenty-seven5,21,22,23,26,27,36,37,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,58 and ten 5,21,26,27,36,38,50,52,54,57 based on mid-arm, elbow, mid-forearm, CT inlet and CT outlet, respectively. The study identification process and PRISMA flowchart outlining the systematic article review is provided in Fig. 2.
Assessment of risk of bias of individual studies
The risk was evaluated as “low” for majority of the included studies in all five domains. The summary of assessment of risk of bias is shown in Fig. 3 and detailed evaluation of individual studies is given in Supplementary Table 2. A few studies were rated as high risk of bias in domain one (objectives and study characteristics) and domain three (characteristics of study methodology).
Median CSA by anatomical landmarks
The pooled weighted mean and 95% CI [upper bound, lower bound] of the median nerve CSA at the mid-arm, elbow, mid-forearm, CT inlet and outlets were 8.81 mm2, CI [8.10, 9.52]; 8.57 mm2, CI [8.00, 9.14]; 7.07 mm2, CI [6.41, 7.73]; 8.74 mm2, CI [8.45, 9.03] and 9.02 mm2, CI [8.08, 9.95], respectively. This indicates that the median nerve CSA is variable at different points in its course, such that it decreases from the mid-arm to mid-forearm and then increases distally at the CT inlet and outlet. A significant Cochran Q statistic I2 > 90% for each landmark showed the presence of considerable heterogeneity variation among the included studies (Table 1). A summary of weights of each individual study, categorised based on respective anatomical landmarks is given in Supplementary Table 3.
Median nerve CSA by age
A total of thirty-four studies reported the overall mean age of the participants which ranged from 21 to 82. The median nerve CSA for age groups at different landmarks were summarised in Table 2. At mid-arm and CT outlet, middle age group CSA appeared to be higher compared to other groups. In younger adults, CSA at mid-forearm was higher compared to other groups. Interestingly, the CSA at elbow and CT inlet were similar in young and middle age groups. A considerable heterogeneity (I2 > 90% with P value < 0.001) was observed in most of the studies.
Median CSA by sex
Seven studies23,29,32,37,48,50,52 involving 534 and 614 median nerves were reported for males and females based on different landmarks (Table 3). The weighted mean median nerve CSA of males showed generally larger than females at all the landmarks (> 1 mm2) except for CT inlet and mid-forearm where CSA values were similar (< 0.5 mm2).
Median nerve CSA by geographical location and ethnicity
Forty-one studies reported median nerve CSA from different geographical locations. Majority of the studies (n = 29) did not report the sub-population ethnicity and hence the meta-analysis was performed based on geographical location reported in the studies (Table 4). At mid-arm, the mean median nerve CSA was smaller for Europe (7.00 mm2) compared to Oceania (9.28 mm2) and Asia (9.06 mm2) studies. At elbow, the mean median nerve CSA was found higher for Europe (9.20 mm2) compared to Asia (8.27 mm2). At mid-forearm, the mean median nerve CSA appears to be larger in Americas (9.38 mm2) followed by Europe (7.15 mm2), Asia (6.24 mm2) and Oceania (5.91 mm2). Interestingly, the mean median nerve CSA for CT inlet seems to be similar for middle east (8.77 mm2), Oceania (8.71 mm2), Europe (8.90 mm2) and Asia (8.68 mm2) studies. However, the mean median nerve CSA at CT outlet is larger for Asia (9.20 mm2) compared to the middle East (8.18 mm2) reports. These results indicate that there is a considerable variation of median nerve CSA among the different geographical locations for different landmarks of the upper limb except CT inlet (< 0.25mm2).
Regarding ethnicity, only twelve4,21,23,26,32,34,36,38,39,41,46,55 articles clearly reported the ethnicity of the sub-populations. Of this, seven articles reported for Chinese21,26,36,38,39,46,55, two for Caucasian34,41, two for Indians4,32 and one article presented for Japanese23. At mid-arm, the pooled weighted mean CSA of Chinese (9.57 mm2) is more than the Japanese (8.20 mm2). The mean CSA of median nerve appears to be smallest at elbow in Indian population (6.90 mm2), compared to the Japanese (9.10 mm2) and Chinese (8.65 mm2). Likewise at mid-forearm, the mean CSA of median nerve is higher in Caucasian (8.20 mm2) compared to the Indians (5.64 mm2) and Chinese (5.70 mm2) subjects. At CT inlet, the Caucasian (8.50 mm2), Japanese (8.50 mm2) and Chinese (8.12 mm2) CSA values were similar. The median nerve mean CSA at CT outlet (8.97 mm2) was available for Chinese subjects only. These findings represent the differences in median nerve CSA along its course of upper limb for different ethnicities (Table 4).
Median nerve CSA based on method of measurement
Thirty-one articles reported measurement of CSA by the method of direct tracing method, out of which 26 articles5,6,21,22,23,24,25,27,28,29,30,32,36,37,38,39,40,41,42,43,44,45,46,47 traced CSA without the hyperechogenic rim of the epineurium of the median nerve. The echogenic rim of the epineurium is reported but did not provide any image of ultrasound trace for interpretation34. Seven articles measured CSA using the elliptical selection (CSA = diameter × width × π), out of which one article53 included the epineurium. Two articles49,53 inferred to have excluded the epineurium, while studies from Bayrak et al., (2007)54 and Lu et al., (2015)55 did not provide any ultrasound images of median nerve CSA for inference54,55. Two studies35,36 used unique methods of measurement (freehand lasso tool on the Adobe Photoshop and automatically using integrated software) but failed to mention if the epineurium was included or not. One article31 reported the measurement of direct tracing within the hyperechoic epineurial rim and continuous trace tool within the outer epineurial rim. In summary, direct tracing was most reported method that used to measure the CSA and this method yielded higher CSA value at CT outlet (9.24 mm2), followed by mid-arm (9.14mm2), CT inlet (8.96 mm2), elbow (8.57 mm2) and mid-forearm (6.73 mm2) compared to the other methods. Likewise, other methods such as Freehand lasso adobe photoshop produced abnormally higher CSA value in mid-forearm (11.75 mm2). In conclusion, considerable heterogeneity (I2 > 90%) was observed for this variable at all landmarks, except at CT outlet reported a moderate heterogeneity (I2 = 43.12%) when measured using elliptical selection method (Table 5).
Discussion
The current study systematically reviewed the existing literature to demonstrate the normative median nerve CSA along its course in the upper limb at various anatomical landmarks. The median nerve is one of the most clinically important and commonly afflicted nerves in the upper limb. Meta-analysis findings from this study generated weighted pooled results of median nerve CSA at different points as well as based on sub-group analysis. This evidence-based observation of normative median nerve CSA would be helpful to the clinicians, neuroradiologists, ultra-sonographers in clinical screening and diagnosis of the normal and pathological median nerve based on the ultrasonography studies.
Analysis of CSA by landmark
Interestingly, the median nerve CSA was much higher at the mid- forearm35, CT inlet48 and CT outlet38 compared to the other studies for the same landmarks. The discrepancy in mean CSA at the mid-forearm is likely related to the deviation of methodology in measuring CSA35. While all other studies have used the ultrasound machine to either trace the echogenic boundary of the median nerve or input values for the ellipsoid formula, this study35 utilised computer software Adobe Photoshop to trace the median nerve. However, the detailed information on how the median nerve boundary defined was not available. On the other hand, the median nerve CSA at the CT inlet reported by Kaymak et al.48 can be further investigated, as the study reported the widest range of CSA (5.90–20.7 mm2) among the rest of the studies included in this review. The authors included the epineurium in its CSA measurement but were unable to elucidate the contributing factors behind this variation. Lastly, the study from Tsai et al.38 did not report the results of the median nerve conduction in the control population, thus investigators were unable to determine whether the control population included individuals with subclinical median nerve neuropathy.
Despite these anomalies, the overall measurement of median nerve CSA was reported to decrease in size, followed by an increase distally down the upper limb. The decrease of median nerve CSA in the proximal upper limb could be explained by the branching of the median nerve at the elbow as it provides an innervation to the forearm muscles, such as the pronator teres, flexor carpi radialis, flexor digitorum sublimis and palmaris longus59. The weighted pool estimates of median nerve CSA tends to be the smallest at the mid-forearm (7.07 mm2). This can be explained by the branching of the anterior interosseous nerve at the forearm which would further reduce the median nerve CSA in the mid-forearm before increasing again prior to reaching the carpal tunnel, as evidenced in cadaveric studies60,61. The distal increase of CSA could be possibly explained by the redistribution of nerve fibres into more fascicular bundles62. However, a cadaveric study by Perumal and Stringer63 showed no significant difference in the number of fascicles in the median nerve around the carpal tunnel area and the redistribution of fascicles affected the antero-posterior and transverse diameters, but not due to the CSA. Yet, the authors of the same study acknowledged the limitations such as the smaller sample size, age, and gender distribution of the samples63.
Analysis of CSA by age
There is no uniform age classification to investigate the median nerve CSA changes in the normal subjects. This is a limitation to understand the progress of the median nerve CSA from young and elderly. However, we classified the age and analysed the changes in CSA based on the overall mean age reported in the studies. Except for elbow and CT inlet, the CSA was found to be varied for different age groups at different landmarks. This observation may be due to limited number of studies with sample size (Table 2) and different methods of CSA measurements. For example, at mid-forearm, Li et al. (2015) reported higher CSA in young age group 13.6 mm2 which is considerably higher compared to other studies for the similar age group. This could be due to use of adobe photoshop to trace the median nerve boundary instead of tracing method in the ultrasound machine. Another reason for varied CSA for different age groups at different landmarks might be due to accumulation of inter-fascicular adipose, connective tissue, redistribution of fascicles and compartmentalisation in the median especially at CT region64,65. One article34 reported mean age based on body mass index, highlighting that the weight considered as major parameter for predicting median nerve CSA. Chen et al. (2011)21 compared median nerve CSA at the mid-arm, CT inlet and CT outlet among different age groups and reported no statistical difference between the middle-aged and elderly groups. Hence, considering the limited number of articles that reported age range, further research is recommended to support both the trend in CSA at these landmarks and their causative mechanisms.
Analysis of CSA by geographical location and ethnicity
Sub-group analysis based on geographical location showed a considerable variation of median nerve CSA among the different geographical locations for different landmarks of the upper limb except CT inlet. This indicates the CSA values reported in one part of the world may not be applicable to other geographical locations. So, it may be worth to have their own data of median CSA representing the country specific/ ethnicity. This sub-group analysis also suffers from the small number of available studies that explicitly reported median nerve CSA based on the geographical location and ethnicity. Therefore, future studies would be needed for determining the relationships of median nerve CSA across different ethnic groups.
Analysis of CSA by sex
Comparison of median nerve CSA by sex indicated that males generally have larger CSA than females. A proposed reason could be males usually having greater weight and body mass index (BMI)66, which contribute to correlate with peripheral nerve CSA67. However, with limited number of eight articles reporting median nerve CSA by sex, more research has to be conducted in future to substantiate the finding from this review. Notably, Kaymak et al.48 presented a larger mean CSA of 11.5 mm2 at the CT inlet in females, which is larger than the mean CSA noted in both the male23,37,50 and female populations23,37,50,52. The reason for Kaymak et al. study48 deviating from the trend discussed above is uncertain, although it could be related to the reduced reliability of this study as discussed above. If data from this study was omitted, the mean CSA at the CT inlet in females would range from 7.94 to 8.83 mm2 as reported in these three articles37,50,53.
Differences in method of measuring CSA and definition of CSA boundary
There was lack of standardised protocol in measuring median nerve CSA across all articles. This included the various upper limb positions that were adopted for the measurement in healthy volunteers, involving wrist hyperextension reported by Kaymak et al.48, and Li et al.35 who performed CSA measurements using computer Freehand lasso adobe photoshop software instead using ultrasound machine. Although there was variation in the method of measuring median nerve CSA, both direct tracing and ellipsoid formula (CSA = diameter × width × π) measurements yielded similar results68 and thus should not affect reliability of the findings in this review. On the other hand, although there was disparity in whether the epineurium was included within the median nerve CSA measurement, the significance of epineurium thickness in the comparison of median nerve CSA has hardly been discussed in current literature69. Furthermore, with ultrasonography being operator-dependant, only six articles27,30,35,38,42,52 reported using more than one operator and seventeen5,6,20,24,25,30,39,40,42,43,45,46,49,50,52,53,57 articles blinded their operators to reduce bias in interpreting measurements.
Limitations of this review
Overall, this review analysed the mean median nerve CSA at different anatomical landmarks in the upper limbs of healthy individuals. However, it was limited by the presence of considerable heterogenicity among the included studies. This could be due to the slightly different anatomical variations among the healthy individuals and calculated across different geographical locations. Despite this observation, the source of heterogeneity was not identified even when subgroup analysis was performed based on age, sex, geographical location, and method of measurement. Although a very high heterogeneity was observed in most subgroups with an I2 range greater than 80%, we also found low to moderate variations (I2 = 0–50%). Results from the latter could be due to the inclusion of the studies from the same research group and limited number of studies.
Furthermore, the ranges of mean median nerve CSA calculated in this study were not categorized based on right and left hands, as other studies reported no significant bilateral differences regardless of hand dominance48,70. The median nerve CSA was also not stratified by BMI as this review aimed to report the range of normative median nerve CSA values applicable to asymptomatic individuals regardless of BMI. Authors believed that this information may be useful in establishing a standardised reference range for the screening and diagnosis of median nerve neuropathy or determining the threshold for treatment like surgical repairs of median nerve injuries. As different studies used different scanning protocols and measurement methods (direct tracing, freehand adobe photoshop and elliptical tracing) to obtain the ultrasound images, this may create bias in interpreting the measurements. Future studies may include establishing standardised protocols and methods to ensure data accuracy and repeatability when measuring median nerve CSA in various upper limb positions.
In addition to a study with limited sample size as low as seven6, there were paucity of articles reporting the detailed ethnicities and age range for subgroup analysis, which may over- or understate the CSA range.
Lastly, a publication bias might have occurred as authors restricted the literature search to English language. This might have limited the complete inclusion of all existing ultrasonography studies.
Future research
Investigations could explore whether differences in median nerve CSA with or without epineurium of the median nerve can be considered, although this difference may be statistically relevant and important. Beyond the range of mean CSA, inclusion of other methods for reviewing normal median nerve CSA cut-offs between articles could potentially explored in future research such as comparing the ratio between wrist-to-forearm median nerve CSA or wrist circumference to median nerve CSA. Moreover, given that the CSA of the median nerve that comprised of area formed by fascicular bundles and non-fascicular area, ultrasound measurements and comparison of the ratio between these areas can be studied further in order to potentially establish the causal mechanisms behind the trends noted in this review. There are no clear scientific suggestions on how to categorize age groups when measuring CSA in human peripheral nerves such as the median nerve. This work might encourage future research on determining the median nerve CSA in the age categories suggested.
Conclusion
This study reported the pooled weighted mean of median nerve CSA along its length (in normal subjects) as well as across geographical locations, age, sex, and method of measurements based on the ultrasonography studies. Our findings on normative CSA values may be considered as a resource for clinicians and surgeons as a cut-off point for investigating preclinical screening and diagnosing the median nerve neuropathy. However, further studies are recommended to obtain normative median nerve CSA values based on BMI across the various ethnic populations and different age groups using standardised protocols.
Data availability
The datasets used in this systematic study were taken from the published articles extracted from online databases such as PubMed and Web of Science.
References
Barnes, C. & Currey, H. Carpal tunnel syndrome in rheumatoid arthritis. A clinical and electrodiagnostic survey. Ann. Rheum. Dis. 26, 226 (1967).
Cranford, C. S., Ho, J. Y., Kalainov, D. M. & Hartigan, B. J. Carpal tunnel syndrome. J. Am. Acad. Orthop. Surg. 15, 537–548 (2007).
Simon, N. G., Talbott, J., Chin, C. T. & Kliot, M. Peripheral nerve imaging. Handb. Clin. Neurol. 136, 811–826 (2016).
Singh, Y., Dixit, R., Singh, S., Garg, S. & Chowdhury, N. High resolution ultrasonography of peripheral nerves in diabetic peripheral neuropathy. Neurol. India 67, 71 (2019).
Kwon, B. C., Jung, K.-I. & Baek, G. H. Comparison of sonography and electrodiagnostic testing in the diagnosis of carpal tunnel syndrome. J. Hand Surg. 33, 65–71 (2008).
Pelosi, L. et al. Peripheral nerve ultrasound in cerebellar ataxia neuropathy vestibular areflexia syndrome (CANVAS). Muscle Nerve 56, 160–162 (2017).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 6, e1000097 (2009).
Henry, B. M., Tomaszewski, K. A. & Walocha, J. A. Methods of evidence-based anatomy: A guide to conducting systematic reviews and meta-analysis of anatomical studies. Ann. Anat. Anat. Anz. 205, 16–21 (2016).
Cardoso, H. F. Age estimation of adolescent and young adult male and female skeletons II, epiphyseal union at the upper limb and scapular girdle in a modern Portuguese skeletal sample. Am. J. Phys. Anthropol. Off. Publ. Am. Assoc. Phys. Anthropol. 137, 97–105 (2008).
Wallace, B. C. et al. Closing the gap between methodologists and end-users: R as a computational back end. J. Stat. Softw. 49, 1–15 (2012).
Higgins, J. P. T., Deeks, J. J. Chapter 7: Selecting studies and collecting data. In Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011) (eds. Higgins, J. P. T. & Green, S.) (The Cochrane Collaboration, 2011). www.handbook.cochrane.org.
Wan, X., Wang, W., Liu, J. & Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 14, 1–13 (2014).
Sharma, D., Ulaganathan, S. P., Sharma, V., Piplani, S. & Niraj, R. R. K. Deep Meta Tool: GUI tool to obtain mean and standard deviation (SD) from median and interquartile range (IQR) (2021).
Hedges, L. V. & Olkin, I. Statistical Methods for Meta-Analysis (Academic Press, Cambridge, 2014).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 7, 177–188 (1986).
Cumpston, M., et al. Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 10, ED000142 (2019).
Higgins, J. P. green S. Cochrane Handb. Syst. Rev. Interv. Version 5, 3 (2011).
Lai, J. F., et al. Clinicopathologic characteristics and prognosis for young gastric adenocarcinoma patients after curative resection. Ann. Surg. Oncol. 15, 1464–1469 (2008).
Tomaszewski, K. A. et al. Development of the Anatomical Quality Assurance (AQUA) checklist: Guidelines for reporting original anatomical studies. Clin. Anat. 30, 14–20 (2017).
Marciniak, C. et al. High-resolution median nerve sonographic measurements: Correlations with median nerve conduction studies in healthy adults. J. Ultrasound Med. 32, 2091–2098 (2013).
Chen, J., Wu, S. & Ren, J. Ultrasonographic measurement of median nerve cross-sectional area reference values in a healthy Han population from Guiyang, China*. Neural Regen. Res. 6, 1883–1887 (2011).
Won, S. J., Kim, B., Park, K. S., Yoon, J. S. & Choi, H. Reference values for nerve ultrasonography in the upper extremity. Muscle Nerve 47, 864–871 (2013).
Sugimoto, T. et al. Ultrasonographic reference sizes of the median and ulnar nerves and the cervical nerve roots in healthy Japanese adults. Ultrasound Med. Biol. 39, 1560–1570 (2013).
Pelosi, L. et al. Peripheral nerves are pathologically small in cerebellar ataxia neuropathy vestibular areflexia syndrome: A controlled ultrasound study. Eur. J. Neurol. 25, 659–665 (2018).
Mulroy, E. et al. Peripheral nerve ultrasound in Friedreich ataxia. Muscle Nerve 57, 852–856 (2018).
Niu, J. et al. Cross-sectional area reference values for sonography of nerves in the upper extremities. Muscle Nerve 61, 338–346 (2020).
Jang, J. H., Cho, C. S., Yang, K.-S., Seok, H. Y. & Kim, B.-J. Pattern analysis of nerve enlargement using ultrasonography in chronic inflammatory demyelinating polyneuropathy. Clin. Neurophysiol. 125, 1893–1899 (2014).
Grimm, A. et al. A look inside the nerve–morphology of nerve fascicles in healthy controls and patients with polyneuropathy. Clin. Neurophysiol. 128, 2521–2526 (2017).
Grimm, A., Axer, H., Heiling, B. & Winter, N. Nerve ultrasound normal values–readjustment of the ultrasound pattern sum score UPSS. Clin. Neurophysiol. 129, 1403–1409 (2018).
Grimm, A., Décard, B. F. & Axer, H. Ultrasonography of the peripheral nervous system in the early stage of Guillain–Barré syndrome. J. Peripher. Nerv. Syst. 19, 234–241 (2014).
van Rosmalen, M., Lieba-Samal, D., Pillen, S. & van Alfen, N. Ultrasound of peripheral nerves in neuralgic amyotrophy. Muscle Nerve 59, 55–59 (2019).
Bathala, L., Kumar, P., Kumar, K., Shaik, A. B. & Visser, L. H. Normal values of median nerve cross-sectional area obtained by ultrasound along its course in the arm with electrophysiological correlations, in 100 Asian subjects. Muscle Nerve 49, 284–286 (2014).
Lothet, E. H., Bishop, T. J., Walker, F. O. & Cartwright, M. S. Ultrasound-derived nerve cross-sectional area in extremes of height and weight. J. Neuroimaging 29, 406–409 (2019).
Cartwright, M. S., Mayans, D. R., Gillson, N. A., Griffin, L. P. & Walker, F. O. Nerve cross-sectional area in extremes of age. Muscle Nerve 47, 890–893 (2013).
Li, X., Li, J. W., Ho, A.M.-H. & Karmakar, M. K. Age-related differences in the quantitative echo texture of the median nerve. J. Ultrasound Med. 34, 797–804 (2015).
Wang, L.-Y. et al. Best diagnostic criterion in high-resolution ultrasonography for carpal tunnel syndrome. Chang Gung Med. J. 31, 469–476 (2008).
Kerasnoudis, A., Pitarokoili, K., Behrendt, V., Gold, R. & Yoon, M.-S. Cross sectional area reference values for sonography of peripheral nerves and brachial plexus. Clin. Neurophysiol. 124, 1881–1888 (2013).
Tsai, N. W. et al. The diagnostic value of ultrasonography in carpal tunnel syndrome: A comparison between diabetic and non-diabetic patients. BMC Neurol. 13, 1–8 (2013).
Horng, Y. S. et al. Ultrasonographic median nerve changes under tendon gliding exercise in patients with carpal tunnel syndrome and healthy controls. J. Hand Ther. 27, 317–324 (2014).
Kim, M.-K., Jeon, H.-J., Park, S.-H., Park, D.-S. & Nam, H.-S. Value of ultrasonography in the diagnosis of carpal tunnel syndrome: Correlation with electrophysiological abnormalities and clinical severity. J. Korean Neurosurg. Soc. 55, 78 (2014).
Böhm, J., Scheidl, E., Bereczki, D., Schelle, T. & Arányi, Z. High-resolution ultrasonography of peripheral nerves: Measurements on 14 nerve segments in 56 healthy subjects and reliability assessments. Ultraschall Med. Eur. J. Ultrasound 35, 459–467 (2014).
Kerasnoudis, A., Pitarokoili, K., Behrendt, V., Gold, R. & Yoon, M. Correlation of nerve ultrasound, electrophysiological and clinical findings in chronic inflammatory demyelinating polyneuropathy. J. Neuroimaging 25, 207–216 (2015).
Borire, A. A., Hughes, A. R., Lueck, C. J., Colebatch, J. G. & Krishnan, A. V. Sonographic differences in carpal tunnel syndrome with normal and abnormal nerve conduction studies. J. Clin. Neurosci. 34, 77–80 (2016).
Arslan, H. et al. The efficiency of acoustic radiation force impulse (ARFI) elastography in the diagnosis and staging of carpal tunnel syndrome. J. Med. Ultrason. 45, 453–459 (2018).
Atan, T. & Günendi, Z. Diagnostic utility of the sonographic median to ulnar nerve cross-sectional area ratio in carpal tunnel syndrome. Turk. J. Med. Sci. 48, 110–116 (2018).
Chang, Y. W. et al. Ratio and difference of the cross-sectional area of median nerve to ulnar nerve in diagnosing carpal tunnel syndrome: A case control study. BMC Med. Imaging 19, 1–9 (2019).
De Kleermaeker, F. G., Meulstee, J. & Verhagen, W. I. The controversy of the normal values of ultrasonography in carpal tunnel syndrome: Diagnostic accuracy of wrist-dependent CSA revisited. Neurol. Sci. 40, 1041–1047 (2019).
Kaymak, B. et al. A comparison of the benefits of sonography and electrophysiologic measurements as predictors of symptom severity and functional status in patients with carpal tunnel syndrome. Arch. Phys. Med. Rehabil. 89, 743–748 (2008).
Rahmani, M. et al. The ultrasonographic correlates of carpal tunnel syndrome in patients with normal electrodiagnostic tests. Radiol. Med. (Torino) 116, 489–496 (2011).
Kim, H. S., Joo, S. H., Han, Z.-A. & Kim, Y. W. The nerve/tunnel index: A new diagnostic standard for carpal tunnel syndrome using sonography: A pilot study. J. Ultrasound Med. 31, 23–29 (2012).
Cingoz, M. et al. Evaluation of median nerve by shear wave elastography and diffusion tensor imaging in carpal tunnel syndrome. Eur. J. Radiol. 101, 59–64 (2018).
Keles, I., Kendi, A. T. K., Aydn, G., Zög, S. G. & Orkun, S. Diagnostic precision of ultrasonography in patients with carpal tunnel syndrome. Am. J. Phys. Med. Rehabil. 84, 443–450 (2005).
Ažman, D. et al. Median nerve imaging using high-resolution ultrasound in healthy subjects. Acta Clin. Croat. 48, 265–268 (2009).
Bayrak, I. K., Bayrak, A. O., Tilki, H. E., Nural, M. S. & Sunter, T. Ultrasonography in carpal tunnel syndrome: Comparison with electrophysiological stage and motor unit number estimate. Muscle Nerve Off. J. Am. Assoc. Electrodiagn. Med. 35, 344–348 (2007).
Lu, Y., Meng, Z., Pan, X., Qin, L. & Wang, G. Value of high-frequency ultrasound in diagnosing carpal tunnel syndrome. Int. J. Clin. Exp. Med. 8, 22418 (2015).
Kele, H., Verheggen, R., Bittermann, H.-J. & Reimers, C. D. The potential value of ultrasonography in the evaluation of carpal tunnel syndrome. Neurology 61, 389–391 (2003).
Kuo, M.-H., Leong, C.-P., Cheng, Y.-F. & Chang, H.-W. Static wrist position associated with least median nerve compression: Sonographic evaluation. Am. J. Phys. Med. Rehabil. 80, 256–260 (2001).
Wilkinson, M., Grimmer, K. & Massy-Westropp, N. Ultrasound of the carpal tunnel and median nerve: A reproducibility study. J. Diagn. Med. Sonogr. 17, 323–328 (2001).
Murphy, K. A. & Morrisonponce, D. Anatomy, Shoulder and Upper Limb, Median Nerve. in StatPearls (StatPearls Publishing, 2022). [Updated 2021 May 27]
Sunderland, S. & Smith, J. W. Nerves and nerve injuries. Plast. Reconstr. Surg. 44, 601 (1969).
Brill, N. A. & Tyler, D. J. Quantification of human upper extremity nerves and fascicular anatomy. Muscle Nerve 56, 463–471 (2017).
Reina, M. A., Sala-Blanch, X., Fabiola, M., Arriazu, R. & Prats-Galino, A. Connective tissues of peripheral nerves. In Textbook of Regional Anesthesia and Acute Pain Management 2nd edn. (McGraw-Hill Med., N. Y., 2015).
Perumal, V. & Stringer, M. D. The intrinsic arterial vascularity and morphology of the median nerve within the carpal tunnel: A microscopic study. Anat. Sci. Int. 89, 28–33 (2014).
Werner, R. A., Albers, J. W., Franzblau, A. & Armstrong, T. J. The relationship between body mass index and the diagnosis of carpal tunnel syndrome. Muscle Nerve 17, 632–636 (1994).
Sladjana, U. Z., Ivan, J. D. & Bratislav, S. D. Microanatomical structure of the human sciatic nerve. Surg. Radiol. Anat. 30, 619–626 (2008).
Flegal, K. Body mass index of healthy men compared with healthy women in the United States. Int. J. Obes. 30, 374–379 (2006).
Bedewi, M. A., et al.. Estimation of ultrasound reference values for the upper limb peripheral nerves in adults: A cross-sectional study. Medicine (Baltimore) 96, e9306 (2017).
Duncan, I., Sullivan, P. & Lomas, F. Sonography in the diagnosis of carpal tunnel syndrome. AJR Am. J. Roentgenol. 173, 681–684 (1999).
Goedee, H. et al. High resolution sonography in the evaluation of the peripheral nervous system in polyneuropathy—a review of the literature. Eur. J. Neurol. 20, 1342–1351 (2013).
Klauser, A. S. et al. Carpal tunnel syndrome assessment with US: Value of additional cross-sectional area measurements of the median nerve in patients versus healthy volunteers. Radiology 250, 171–177 (2009).
Author information
Authors and Affiliations
Contributions
J.T.A.N and R.C equally involved in literature search, data collection, data analysis and writing of manuscript. A.P. contributed to data analysis and assisted with the writing of manuscript. S.R.M conceptualized and developed this project, assisted with data analysis, writing of manuscript, and supervised R.C and J.T.A.N. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ng, A.J.T., Chandrasekaran, R., Prakash, A. et al. A systematic review: normative reference values of the median nerve cross-sectional area using ultrasonography in healthy individuals. Sci Rep 12, 9217 (2022). https://doi.org/10.1038/s41598-022-13058-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-13058-8
- Springer Nature Limited