Abstract
The huntsman spiders’ genus Eusparassus are apex arthropod predators in desert ecosystems of the Afrotropical and Palearctic ecoregions. The Eusparassus dufouri and E. walckenaeri clades are two distinct taxonomic, phylogenetic, and geographic units concerning morphology, molecular phylogeny, and spatial data; but little is known about their ecological niche. We applied the maximum-entropy approach and modelled ecologic niches of these two phylogenetically closely related clades. Ecological niches of the two clades were compared using identity and background tests and two different metrics, the Schooner’s D and Warren’s I. We also predicted the impacts of climate change on the distribution of the two clades. The results of the identity test showed that the ecological niches of the two clades were different in geographic space but were similar in environmental space. While results of the background test revealed that the ecological niches of the two clades were similar in geographic and environmental space. This indicated that “niche conservatism” had an important role over the evolutionary time of allopatric diversification. However, the normalized difference vegetation index vs. topographic heterogeneity had influenced the niches of the dufouri and walckenaeri clades, respectively. The analyses recovered that the two clades’ climatically suitable habitats will increase under future climate (the year 2070). However, since the two clades are characterized by the narrow range of environmental optimum and the accordingly high limits of tolerance, they are vulnerable to climate change.
Similar content being viewed by others
Introduction
Spiders, order Araneae, are the main arthropod predators in the terrestrial ecosystems1. Spiders with nearly 50,000 species in 130 families are one of the most diverse groups of metazoan2. These venomous arachnids are proposed to be used for pest control in agroecosystems3. The spider venom and silk are subject of composition studies4 and application in pharmaceutical5, and technology of biomaterial6, respectively.
The Huntsman spiders, family Sparassidae, are large ambush predators partitioned in various habitats from humid rain forests to arid sand deserts. The family currently comprises 1290 described species in 89 genera2. The Stone huntsman spiders, genus Eusparassus Simon, 1903, composed of the largest predatory spiders (leg-span up to 14 cm) inhabiting xeric and subxeric regions of Africa, Mediterranean Europe via the Middle East to Central Asia7,8. Like other spiders, Eusparssus species are generalist predators but they mainly prey on insects and are able to subdue small vertebrates, indicating them as potential apex arthropod predators in the desert ecosystems8,9. Eusparassus represents one of the successful taxa from evolutionary perspective since it exists from at least 50 million years ago in the early Tertiary according to evidence of the amber fossil in Europe10. The genus currently comprises 30 species, majority of them are classified into six morphologic species-groups: the Eusparassus doriae, dufouri, jaegeri, vestigator, and walckenaeri8 while molecular phylogeny recovered and confirmed them as phylogenetically defined clades (exc. vestigator; no DNA data) based on four molecular markers11. The dufouri clade members are distributed in NW Africa and the Iberian Peninsula neighbouring the walckenaeri clade members which occur in Eastern Mediterranean, NE Africa, and the Arabian Peninsula. These two clades recovered as closely related taxa according to phylogenetic inferences11. The Eusparassus members and the entire family Sparassidae have never been the subject of any ecological niche modelling studies to date and our knowledge about factors that shaped their evolutionary history is scarce.
Every kind of living form has a range of specific environmental characteristics within which can have positive population growth; this range is called an ecological niche12. There are two kinds of ecological niches, the fundamental niche which is solely influenced by abiotic, and the realized one which is the result of interaction with biotic factors13. Species Distribution Modelling (SDM) methodologically uses a variety of statistical methods concerning large-scale climatic and topographic variables to estimate the ecological niche of taxa in relation to underlying environmental gradients14. These models are important tools in ecology, evolution and biogeography studies15,16,17,18. They have been used in studying niche evolution, niche dynamic, and niche comparisons in different taxonomic groups above and below species level15,16,19,20,21. Spiders have rarely been the subject of ecological niche modelling studies22,23. The previous SDM studies on spiders were geographically restricted to Europe and America devoting mainly to medically important biting spiders24,25,26,27, potentially model taxa to study biogeography and conservation biology28,29,30, and potentially endangered spiders31,32,33.
Here we applied the independent lines of evidence-based studies including morphology7,8 and molecular phylogeny11 integrated to geographical and environmental data to investigate the distribution modelling of two spider clades, E. dufouri clade (western Mediterranean Europe and Africa) and E. walckenaeri clade (eastern Mediterranean and partly in the Middle East and NE Africa), to test whether these two allopatric clades are different based on their ecological niches.
Results
Models performance
All niche models developed in this study performed well based on AUC metric (the dufouri clade’s ecological niche model: AUC = 0.976 ± 0.011, the walckenaeri clade’s ecological niche model: AUC = 0.982 ± 0.009, the dufouri clade’s climate niche model: AUC = 0.922 ± 0.016, and the walckenaeri clade’s climate niche model: AUC = 0.947 ± 0.021).
Ecological niche model
The model predicted that the environmental condition of the present distribution range of the dufouri clade is suitable for the walckenaeri clade members and vice versa (Figs. 1, 2). The potential distribution range of the walckenaeri clade was estimated much more widely extending into those of the dufouri clade in NW Africa and Iberia. On the other hand, the environmental condition in the western Mediterranean (Iberian and NW Africa), where the dufouri clade is distributed, is potentially appropriate for the walckenaeri clade (Fig. 2). Moreover, the eastern Mediterranean (Greece and Turkey) had suitable environmental conditions for the dufouri clade as well (Fig. 1).
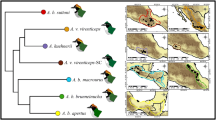
Distribution model of the dufouri clade. The distribution model was created using Maxent software 3.4.1 (https://biodiversityinformatics.amnh.org/open_source/maxent/) and mapped in QGIS 3.22.3 (https://www.qgis.org). The spider was photographed by Majid Moradmand.
Distribution model of the walckenaeri clade. The distribution model was created using Maxent software 3.4.1 (http://biodiversityinformatics.amnh.org/open_source/maxent/) and mapped in QGIS 3.22.3 (https://www.qgis.org). The spider was photographed by Majid Moradmand.
Ecological niche across geographical and environmental spaces
Comparing the ecological niche of the dufouri and walckenaeri clades using two identity tests, the Schoener’s D and Warren’s I showed that their niches are different only in geographical space but are similar in environmental space (Fig. 3). Results of two background tests, the Schoener’s D and Warren’s I showed that ecological niches of the dufouri and walckenaeri clades are similar in both geographical and environmental spaces (Fig. 4).
Results of identity test for the two clades (dufouri and walckenaeri) across the geographical and environmental spaces. Panels show histograms of 100 simulations of identity tests using two different metrics; the Schoener’s D and Warren’s I. Dotted lines indicate observed values. The figure is created using ENMTools 1.0.4 (https://github.com/danlwarren/ENMTools).
Results of background test for the two clades (dufouri and walckenaeri) across the geographical and environmental spaces. Panels show histograms of 100 simulations of background tests using two different metrics; the Schoener’s D and Warren’s I. Dotted lines indicate observed values. The figure is created using ENMTools 1.0.4 (https://github.com/danlwarren/ENMTools).
Environmental variables and response curves
Results showed that the annual mean temperature (Bio1) with 44.9% contribution followed by the Normalized Difference Vegetation Index (NDVI) with 15.4% contribution were the most influential variables in shaping the ecological niche of the dufouri clade (Table 1). On the other side, for the walckenaeri clade, the Topographic Heterogeneity (TH) with 34% contribution and the annual mean temperature with 24.9% contribution were found to be the most important determinants of the ecological niche. Comparing the two clades, revealed that on one hand, the NDVI had much more influence on the dufouri clade (15.4%) than on the walckenaeri clade (3.6%); on the other hand, the TH had a greater impact on the walckenaeri clade (34%) than on the dufouri clade (7.9%). The response curves of the dufouri and the walckenaeri clades which were created using the environmental variables were similar and mostly unimodal (Fig. S1).
Climate change modelling
Results of predicting the impacts of climate change on the distribution of the two clades showed that climatically suitable areas will increase in the future (Fig. 5). Based on RCP 8.5 until 2070, climatically suitable habitats for the dufouri clade will increase between 16% (CCSM4) to 29% (MIROC5). Climatically suitable habitats for the walckenaeri clade will increase by 14% based on CCSM4 and while based on MIROC5 will remain stable under climate change (only 0.07% increase is expected). The response curves of the dufouri and the walckenaeri clades which were created using the bioclimatic variables were very similar and all unimodal (Fig. S2).
Climate suitability maps for the dufouri clade (left) and walckenaeri clade (right) for current (A) and future climate (2070), under CCSM4 RCP 8.5 (B) and MIROC5 RCP 8.5 (C), representatives of each clade, habitus from frontal view (D). The suitability maps were created using Maxent software 3.4.1 (http://biodiversityinformatics.amnh.org/open_source/maxent/) and mapped in QGIS 3.22.3 (https://www.qgis.org). The spiders were photographed by Majid Moradmand.
Discussion
Despite being the most diverse group of organisms, but the terrestrial arthropods have rarely been used in the species distribution modelling studies compared to other animal phyla27,34, since the scarcity of available distribution data and fewer taxonomic studies23. Here, we estimated the potential distribution of the two clades of the spider genus Eusparassus in the Western Palearctic using ecological niche modelling. The two Eusparassus clades: dufouri and walckenaeri, are diagnosed by morphology, and distributed allopatrically in different geographic regions8, indeed molecular phylogeny recovered them as distinct monophyletic clades11. The result of the current study revealed that the environmental factors are not drivers of their distribution patterns and no significant niche differentiation was observed between these two clades.
The majority of the SDMs are built at the species level. But it is recommended that the taxonomic resolution below or above species level should be considered in distribution modelling21,35,36. Recently, applying other evolutionary taxonomic units to estimate spatial modelling has been developing but under different terms. For instance, the lineage-level based distribution models or LDM37 used intraspecific lineages of various species (e.g.16,21,38), and spatial distribution modelling at the Clade-level, applied data of inter-and intraspecific sampling for spatial modelling (e.g.39).
Most studies showed that climatic variables are the main driver of organism distribution and diversification40. The distributions pattern of spider communities is mainly influenced by the environmental variables (e.g.41,42), however, niche conservatism was observed in closely related spider taxa26. Studies on the subterranean spiders showed partial niche overlap between close relatives33. Investigation on spider diversification during evolutionary time with conservative climatic niche but allopatric distribution pattern is also reported in the spider family Ctenidae30. Ecologically similar but geographically different taxa are found in different groups of organisms too39 including insects40,44,45 and vertebrates46,47.
The diversification in two allopatric Eusparassus clades without divergence in the ecological niche is reported here. Both Eusparassus clades: dufouri and walckenaeri are recovered highly monophyletic and closely related lineage to each other and partitioned into two distinct geographic regions. There is no evidence of sympatric distribution of the species of these two clades except occasional overlap in their neighbouring borders8. Our result revealed that the geography and niche conservatism are the main drivers of diversification of these clades.
The responses of the dufouri and walckenaeri clades to environmental variables were generally similar. However, the impacts of environmental variables on the clades seem to be different between the two clades. Importantly, the impacts of NDVI vs. TH on the clades are correspondence with the field observation of the clade members in their natural habitats. Some species of the dufouri clade were collected from vegetation and trees showing foliage and arboreal habitat preferences of this group (see references7,8). In contrast, the walckenaeri clade members are well known as inhabitants of stony deserts where they mainly build their silken retreats under stones8,9. These pieces of evidence explain why NDVI and TH have significant impact differences in the dufouri and walckenaeri clades, respectively.
The topology of the respond curves to environmental and bioclimatic variables showed some levels of the narrow range in the optimum and accordingly high limits of tolerance. Any change in the optimal environmental condition can lead to a decrease in taxa performance and reach the zone of intolerance48. These data indicate that the clades are environmental specialists and sensitive to change in the corresponding environmental variables. We found out that the two clades’ climatically suitable habitats will increase under future climate (the year 2070) but because the two clades are characterized with a narrow range in the environmental optimum, we believe that they are vulnerable to climate change. The environmentally specialist spiders have poorly developed abilities of dispersal49. While aerial dispersal or ballooning is an efficient mechanism in spiders1, there is no evidence of this behaviour in Sparassidae, indicating their limited capability of dispersal compared to ballooning competitors. The lack of dispersal performance would cost the clade members by limiting them from reaching a suitable habitat. Consequently, the ongoing climate change would negatively affect the clades, and probably eliminate them from their habitats producing them as “climate change losers”. Since these spiders are the apex predators in their environment, thus planning conservation strategies are highly recommended.
Methods
Distribution data
Species distribution data of the dufouri and walckenaeri clades (three species per each, total N = 6 species) reconstructed after molecular phylogenetics11 were applied for georeferencing. The locality data of these species occurrences were obtained from different sources, partially by direct sampling in the field using GPS but mainly from museum collection labels and publications7,8 and GBIF source50. For the collection labels without coordination (containing just the exact geographical names), the online global gazetteers version 2.2 (http://www.fallingrain.com/world) was applied to assign longitude and latitude (georeferencing) to each locality. All forms of geographic coordinates were converted into decimal degrees. The data were used to build up an occurrence database of the studied taxa (Fig. 6). We carefully checked the clades data to clean the duplicate records. Because the environmental data were available at 5 km resolution, therefore we thinned the dataset to ensure that observations were at least 5 km apart. Overall, 133 distribution records were used in ecological niche modelling of walckenaeri clade and 111 occurrence records for dufouri clade.
Distribution records of the dufouri clade and walckenaeri clade on a topographic overview of the study area. Elevation map was obtained from the Shuttle Radar Topography Mission (SRTM) elevation model (https://srtm.csi.cgiar.org/) and mapped in QGIS 3.22.3 (https://www.qgis.org). The spiders were photographed by Majid Moradmand.
Environmental predictors
In this study, to develop an ecological niche model for each clade we used a combination of variables reflecting climate, topography, and plant productivity (Table 2). Climate variables (annual temperature (Bio1) and annual precipitation (Bio12)) and Solar Radiation Index at ~ 5 km spatial resolution were downloaded from WorldClim51. A digital elevation model(from the Shuttle Radar Topography Mission (SRTM) elevation model)52 of the study area was used to generate two topography variables; elevation and topographic heterogeneity (TH) in the raster package53. Then, the normalized difference vegetation index (NDVI) was used as an indicator of plant productivity, which may reflect resource availability. To avoid multicollinearity problems among variable predictors54, a variance inflection factor (VIF)55 analysis was run using the ‘usdm’ package56. We found low collinearity among the environmental variables (annual temperature = 6.977, annual precipitation = 2.554, elevation = 3.323, topographic heterogeneity = 1.636 and NDVI = 2.637).
Ecological niche modelling and niche comparison
There are various statistical methods for SDM, but one of the most frequently applied methods is the maximum-entropy approach implemented in Maxent57. The efficacy of Maxent is proved to be useful in estimating the suitable environmental distribution range of the organisms such as spiders (e.g.,24,28,29,58). In this study, we used the maximum entropy approach59 to develop the ecological niche of the two phylogenetically clades (dufouri and walckenaeri) across their distribution range. For our models, maximum iterations were set to 500, convergence threshold was 0.0001, and 10,000 background locations were chosen from the entire study area (Fig. 5). Maxent output format was set Cloglog which gives an estimate between 0 and 1 of probability of presence. We applied the cross-validation method in Maxent and distribution points were randomly split into 10 folds containing an equal number of occurrences, and training models were created by eliminating each fold in turn60. Models performance was assessed using the Area Under the Curve (AUC) metric of the Receiving Operator Characteristic (ROC) curve43,59,61.
Ecological niches of the two clades were compared using identity and background tests and two different metrics, the Schoener’s D62 and Warren’s I63. Further, to understand whether niche similarity is smaller or larger than expected with respect to the clades’ geographical distribution we performed a background test using the Schoener’s D62 and Warren’s I63. We compared the two clades' ecological niches in geographic and environmental space separately64.
Climate change assessment
We used the maximum entropy approach to predict the impacts of climate change on the two phylogenetically clades (dufouri and walckenaeri) across their distribution range. Two general circulation models of CCSM4 and MIROC5 for 2070 (average for 2061–2080) under the representative concentration pathways (RCP 8.5) were used. To predict the future distribution of the two clades the following nine bioclimatic variables were used: mean diurnal range (Bio2), temperature seasonality (Bio4), mean temperature of wettest quarter (Bio8), mean temperature of driest quarter (Bio9), precipitation of wettest month (Bio13), precipitation of driest month (Bio14), precipitation seasonality (Bio15), precipitation of warmest quarter (Bio18) and precipitation of coldest quarter for current (Bio19) and future climatic conditions. Bioclimatic variables for current and future climatic conditions were downloaded from Worldclim (https://www.worldclim.org/). In Maxent models, maximum iterations were set to 500, convergence threshold was 0.0001, and 10,000 background locations were chosen. To estimate the area of suitable habitats of each clade under current and future climatic conditions, we first converted continuous model outputs to presence/absence maps using the 10th percentile training presence threshold35,65,66. Then calculated area of suitable habitats under current and future climatic conditions using Raster package in R environment v. 3.4.3. Like ecological niche models for climate niche models, we applied the cross-validation method in Maxent and distribution points were randomly split into 10 folds containing an equal number of occurrences, and training models were created by eliminating each fold in turn60. Models' performance was assessed using the Area Under the Curve (AUC) metric of the Receiving Operator Characteristic (ROC) curve43,59,61.
Data availability
The datasets generated and analysed during the current study are available from described sources in the manuscript. Further information can be obtained from the corresponding author on reasonable request.
References
Foelix, R. F. Biology of Spiders (Oxford University Press, 2011).
World Spider Catalog. World Spider Catalog, Version 23.0. Natural History Museum Bern, online at http://wsc.nmbe.ch (2022).
Nyffeler, M. & Sunderland, K. D. Composition, abundance and pest control potential of spider communities in agroecosystems: A comparison of European and US studies. Agric. Ecosyst. Environ. 95, 579–612 (2003).
Oldrati, V. et al. Peptidomic and transcriptomic profiling of four distinct spider venoms. PLoS ONE 12, e0172966 (2017).
Herzig, V. et al. Animal toxins—Nature’s evolutionary-refined toolkit for basic research and drug discovery. Biochem. Pharmacol. 181, 114096 (2020).
Vollrath, F. & Knight, D. P. Liquid crystalline spinning of spider silk. Nature 410, 541–548 (2001).
Moradmand, M. & Jäger, P. Taxonomic revision of the huntsman spider genus Eusparassus Simon, 1903 (Araneae: Sparassidae) in Eurasia. J. Nat. Hist. 46, 2439–2496 (2012).
Moradmand, M. The stone huntsman spider genus Eusparassus (Araneae: Sparassidae): Systematics and zoogeography with revision of the African and Arabian species. Zootaxa 3675, 1–108 (2013).
Levy, G. The family of huntsman spiders in Israel with annotations on species of the Middle East (Araneae: Sparassidae). J. Zool. 217, 127–176 (1989).
Dunlop, J. A. et al. Computed tomography recovers data from historical amber: An example from huntsman spiders. Naturwissenschaften 98, 519–527 (2011).
Moradmand, M., Schönhofer, A. L. & Jäger, P. Molecular phylogeny of the spider family Sparassidae with focus on the genus Eusparassus and notes on the RTA-clade and ‘Laterigradae’. Mol. Phylogenet. Evol. 74, 48–65 (2014).
Hutchinson, G. E. Cold spring harbor symposium on quantitative biology. Concl. Remarks 22, 415–427 (1957).
Pearman, P. B., Guisan, A., Broennimann, O. & Randin, C. F. Niche dynamics in space and time. Trends Ecol. Evol. 23, 149–158 (2008).
Wake, D. B., Hadly, E. A. & Ackerlya, D. D. Biogeography, changing climates, and niche evolution. Proc. Natl. Acad. Sci. U. S. A. 106, 19631–19636 (2009).
Smith, A. B., Godsoe, W., Rodríguez-Sánchez, F., Wang, H. H. & Warren, D. Niche estimation above and below the species level. Trends Ecol. Evol. 34, 260–273 (2019).
Peñalver-Alcázar, M., Jiménez-Valverde, A. & Aragón, P. Niche differentiation between deeply divergent phylogenetic lineages of an endemic newt: implications for Species Distribution Models. Zoology 144, 125852 (2021).
Di Pasquale, G. et al. Coastal Pine-Oak Glacial Refugia in the mediterranean basin: A biogeographic approach based on charcoal analysis and spatial modelling. Forests 11, 673 (2020).
Du, Z., He, Y., Wang, H., Wang, C. & Duan, Y. Potential geographical distribution and habitat shift of the genus Ammopiptanthus in China under current and future climate change based on the MaxEnt model. J. Arid Environ. 184, 104328 (2021).
Kafash, A. et al. The Gray Toad-headed Agama, Phrynocephalus scutellatus, on the Iranian Plateau: The degree of niche overlap depends on the phylogenetic distance. Zool. Middle East 64, 47–54 (2018).
Namyatova, A. A. Climatic niche comparison between closely related trans-Palearctic species of the genus Orthocephalus (Insecta: Heteroptera: Miridae: Orthotylinae). PeerJ 8, e10517 (2020).
Zhang, Z. et al. Lineage-level distribution models lead to more realistic climate change predictions for a threatened crayfish. Divers. Distrib. 27, 684–695 (2021).
Mammola, S. & Leroy, B. Applying species distribution models to caves and other subterranean habitats. Ecography (Cop.) 41, 1194–1208 (2018).
Mammola, S. et al. Challenges and opportunities of species distribution modelling of terrestrial arthropod predators. Divers. Distrib. 00, 1–19 (2021).
Saupe, E. E., Papes, M., Selden, P. A. & Vetter, R. S. Tracking a medically important spider: Climate change, ecological niche modeling, and the brown recluse (Loxosceles reclusa). PLoS ONE 6, 2 (2011).
Planas, E., Saupe, E. E., Lima-Ribeiro, M. S., Peterson, A. T. & Ribera, C. Ecological niche and phylogeography elucidate complex biogeographic patterns in Loxosceles rufescens (Araneae, Sicariidae) in the Mediterranean Basin. BMC Evol. Biol. https://doi.org/10.1186/s12862-014-0195-y (2014).
Taucare-Ríos, A., Nentwig, W., Bizama, G. & Bustamante, R. O. Matching global and regional distribution models of the recluse spider Loxosceles rufescens: to what extent do these reflect niche conservatism?. Med. Vet. Entomol. 32, 490–496 (2018).
Wang, Y., Casajus, N., Buddle, C., Berteaux, D. & Larrivée, M. Predicting the distribution of poorly-documented species, Northern black widow (Latrodectus variolus) and Black purse-web spider (Sphodros Niger), using museum specimens and citizen science data. PLoS ONE 13, e0201094 (2018).
Jiménez-Valverde, A., Decae, A. E. & Arnedo, M. A. Environmental suitability of new reported localities of the funnelweb spider Macrothele calpeiana: An assessment using potential distribution modelling with presence-only techniques. J. Biogeogr. 38, 1213–1223 (2011).
Monsimet, J., Devineau, O., Pétillon, J. & Lafage, D. Explicit integration of dispersal-related metrics improves predictions of SDM in predatory arthropods. Sci. Rep. https://doi.org/10.1038/s41598-020-73262-2 (2020).
Salgado-Roa, F. C., Gamez, A., Sanchez-Herrera, M., Pardo-Diaz, C. & Salazar, C. Divergence promoted by the northern Andes in the giant fishing spider Ancylometes bogotensis (Araneae: Ctenidae). Biol. J. Linn. Soc. 132, 495–508 (2021).
Mammola, S., Goodacre, S. L. & Isaia, M. Climate change may drive cave spiders to extinction. Ecography (Cop.) 41, 233–243 (2018).
Ferretti, N. E., Soresi, D. S., González, A. & Arnedo, M. An integrative approach unveils speciation within the threatened spider Calathotarsus simoni (Araneae: Mygalomorphae: Migidae). Syst. Biodivers. 17, 439–457 (2019).
Pavlek, M. & Mammola, S. Niche-based processes explaining the distributions of closely related subterranean spiders. J. Biogeogr. 48, 118–133 (2021).
Bosso, L. et al. Nature protection areas of Europe are insufficient to preserve the threatened beetle Rosalia alpina (Coleoptera: Cerambycidae): evidence from species distribution models and conservation gap analysis. Ecol. Entomol. 43, 192–203 (2018).
Kafash, A. et al. Climate change produces winners and losers: Differential responses of amphibians in mountain forests of the Near East. Glob. Ecol. Conserv. 16, e00471 (2018).
Vásquez-Aguilar, A. A., Ornelas, J. F., Rodríguez-Gómez, F. & Cristina MacSwiney, G. Modeling future potential distribution of buff-bellied hummingbird (Amazilia yucatanensis) under climate change: species vs subspecies. Trop. Conserv. Sci. 25, 2 (2021).
Rosauer, D. F., Catullo, R. A., VanDerWal, J., Moussalli, A. & Moritz, C. Lineage range estimation method reveals fine-scale endemism linked to pleistocene stability in Australian rainforest herpetofauna. PLoS ONE 10, e0126274 (2015).
Eyres, A., Eronen, J. T., Hagen, O., Böhning-Gaese, K. & Fritz, S. A. Climatic effects on niche evolution in a passerine bird clade depend on paleoclimate reconstruction method. Evolution 75, 1046–1060 (2021).
Loyola, R. D., Lemes, P., Brum, F. T., Provete, D. B. & Duarte, L. D. S. Clade-specific consequences of climate change to amphibians in Atlantic Forest protected areas. Ecography (Cop.) 37, 65–72 (2014).
Muñoz, M. M. & Bodensteiner, B. L. Janzen’s hypothesis meets the bogert effect: Connecting climate variation, thermoregulatory behavior, and rates of physiological evolution. Integr. Org. Biol. 1, 1–12 (2019).
Entling, W., Schmidt, M. H., Bacher, S., Brandl, R. & Nentwig, W. Niche properties of Central European spiders: Shading, moisture and the evolution of the habitat niche. Glob. Ecol. Biogeogr. 16, 440–448 (2007).
Lafage, D., Maugenest, S., Bouzillé, J. B. & Pétillon, J. Disentangling the influence of local and landscape factors on alpha and beta diversities: opposite response of plants and ground-dwelling arthropods in wet meadows. Ecol. Res. 30, 1025–1035 (2015).
Peterson, A. T., Soberón, J. & Sánchez-Cordero, V. Conservatism of ecological niches in evolutionary time. Science 285, 1265–1267 (1999).
Wellenreuther, M., Larson, K. W. & Svensson, E. I. Climatic niche divergence or conservatism? Environmental niches and range limits in ecologically similar damselflies. Ecology 93, 1353–1366 (2012).
Nosil, P. & Sandoval, C. P. Ecological niche dimensionality and the evolutionary diversification of stick insects. PLoS ONE 3, e1907 (2008).
McCormack, J. E., Zellmer, A. J. & Knowles, L. L. Does niche divergence accompany allopatric divergence in Aphelocoma jays as predicted under ecological speciation?: Insights from tests with niche models. Evolution 64, 1231–1244 (2010).
Goudarzi, F., Hemami, M. R., Malekian, M. & Fakheran-Esfahani, S. Ecological Characterization of the breeding habitat of Luristan newt (Neurergus kaiseri) at local scale. J. Nat. Environ. 72, 113–127 (2019).
Chase, J. M. & Leibold, M. Ecological Niches: Linking Classical and Contemporary Approaches (University of Chicago Press, 2003).
Bonte, D., Vandenbroecke, N., Lens, L. & Maelfait, J. P. Low propensity for aerial dispersal in specialist spiders from fragmented landscapes. Proc. R. Soc. B Biol. Sci. 270, 1601–1607 (2003).
GBIF.org. GBIF Occurrence Download. https://doi.org/10.15468/dl.2tc2ja (2021) doi:https://doi.org/10.15468/dl.2tc2ja.
Fick, S. E. & Hijmans, R. J. WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 37, 4302–4315 (2017).
Jarvis, A., Reuter, H. I., Nelson, A. & Guevara, E. Hole-Filled SRTM for the Globe Version 4. Available from the CGIAR-CSI SRTM 90m Database. (2008) doi:https ://srtm.csi.cgiar .org.
Hijmans, R. J. raster: Geographic Data Analysis and Modeling. R package version 3, 3–7 (2020).
Guisan, A., Thuiller, W. & Zimmermann, N. E. Habitat suitability and distribution models: With applications in R. (2017). doi:10.1017/ 9781139028271.
Quinn, G. P. & Keough, M. J. Experimental Design and Data Analysis for Biologists (Cambridge University Press, 2002).
Naimi, B. Uncertainty Analysis for Species Distribution Models. R package version (2015).
Phillips, S. J., Dudík, M. & Schapire, R. E. Maxent software for modeling species niches and distributions (Version 3.4.1). Available from url: http://biodiversityinformatics.amnh.org/open_source/maxent/. Accessed on 2022–2–12.
Nǎpǎruş, M. & Kuntner, M. A GIS model predicting potential distributions of a lineage: a test case on hermit spiders (Nephilidae: Nephilengys). PLoS ONE 7, e30047 (2012).
Phillips, S. J., Anderson, R. P. & Schapire, R. E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 190, 231–259 (2006).
Merow, C., Smith, M. J. & Silander, J. A. A practical guide to MaxEnt for modeling species’ distributions: What it does, and why inputs and settings matter. Ecography (Cop.) 36, 1058–1069 (2013).
Swets, J. A. Measuring the accuracy of diagnostic systems. Science 240, 1285–1293 (1988).
Schoener, T. W. The anolis lizards of Bimini: Resource partitioning in a complex fauna. Ecology 49, 704–726 (1968).
Warren, D. L., Glor, R. E. & Turelli, M. Environmental niche equivalency versus conservatism: Quantitative approaches to niche evolution. Evolution 62, 2868–2883 (2008).
Warren, D. L. et al. ENMTools 1.0: an R package for comparative ecological biogeography. Ecography 44, 504–511 (2021).
Liu, C., Berry, P. M., Dawson, T. P. & Pearson, R. G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography (Cop.) 28, 385–393 (2005).
Vale, C. G., Tarroso, P. & Brito, J. C. Predicting species distribution at range margins: Testing the effects of study area extent, resolution and threshold selection in the Sahara-Sahel transition zone. Divers. Distrib. 20, 20–33 (2014).
Acknowledgements
The first author is thankful to GRADE (Goethe Graduate Academy, Goethe University, Frankfurt am Main, Germany). Maryam Rajabi (Frankfurt am Main, Isfahan) assisted in georeferencing the data, Roohollah Abbasi (Manitoba, Isfahan) edited the early draft, and Anooshe Kafash (Isfahan) helped in developing ecological niche models, for which we are very thankful.
Author information
Authors and Affiliations
Contributions
M.M. (core idea, georeferencing, interpretation of data, writing the main text); M.Y. (data analyses, interpretation of data, writing, preparation of figures). The authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moradmand, M., Yousefi, M. Ecological niche modelling and climate change in two species groups of huntsman spider genus Eusparassus in the Western Palearctic. Sci Rep 12, 4138 (2022). https://doi.org/10.1038/s41598-022-08145-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-08145-9
- Springer Nature Limited
This article is cited by
-
Global habitat suitability modeling reveals insufficient habitat protection for mangrove crabs
Scientific Reports (2022)