Abstract
Post-stroke arrhythmias represent a risk factor for complications and worse prognosis after cerebrovascular events. The aims of the study were to detect the rate of atrial fibrillation (AF) and other cardiac arrhythmias after acute ischemic stroke, by using a 7-day Holter ECG which has proved to be superior to the standard 24-h recording, and to evaluate the possible association between brain lesions and arrhythmias. One hundred and twenty patients with cryptogenic ischemic stroke underwent clinical and neuroimaging assessment and were monitored with a 7-day Holter ECG. Analysis of the rhythm recorded over 7 days was compared to analysis limited at the first 24 h of monitoring. 7-day Holter ECG detected AF in 4% of patients, supraventricular extrasystole (SVEB) in 94%, ventricular extrasystole (VEB) in 88%, short supraventricular runs (SVRs) in 54%, supraventricular tachycardia in 20%, and bradycardia in 6%. Compared to the first 24 h of monitoring, 7-Holter ECG showed a significant higher detection for all arrhythmias (AF p = 0.02; bradycardia p = 0.03; tachycardia p = 0.0001; SVEB p = 0.0002; VEB p = 0.0001; SVRs p = 0.0001). Patients with SVRs and bradycardia were older (p = 0.0001; p = 0.035) and had higher CHA2DS2VASc scores (p = 0.004; p = 0.026) respectively, in the comparison with patients without these two arrhythmias. An association was found between SVEB and parietal (p = 0.013) and temporal (p = 0.013) lobe lesions, whereas VEB correlated with insular involvement (p = 0.002). 7-day Holter ECG monitoring proved to be superior as compared to 24-h recording for the detection of all arrhythmias, some of which (SVEB and VEB) were associated with specific brain areas involvement. Therefore, 7-day Holter ECG should be required as an effective first-line approach to improve both diagnosis and therapeutic management after stroke.
Similar content being viewed by others
Introduction
Cardiac complications, such as arrhythmias, congestive heart failure, and myocardial injuries, are fairly frequent after acute stroke1,2. New-onset arrhythmias, including supraventricular tachycardia, supraventricular extrasystole (SVEB), ventricular extrasystole (VEB), bradycardia, atrial fibrillation (AF) and QT interval prolongation, tend to occur often during the first few hours after stroke3. Although preexisting cardiac pathologies may worsen clinical outcomes4, arrhythmias are common in stroke patients even in the absence of previous heart diseases, suggesting also a role for central nervous system (CNS) in these abnormalities2,5.
However, rhythmic disturbances may also lead to brain ischemia. AF represents a major cause of approximately 30% of strokes6 and, because of its possible paroxysmal pattern, it could be underdiagnosed7.
A strict interaction between cardiovascular and neurological systems is well known8. As cardioembolism is a common cause of stroke, acute cerebral ischemia itself seems to induce an imbalance of central autonomic control, leading then to cardiac dysfunctions. The sympathetic and parasympathetic imbalance after stroke is documented by impairment of heart rate and blood pressure regulation9 and by an increased catecholamine release10, which mediates beta-adrenergic effects on the myocardium. Moreover, the significant increase of cardiac enzymes following acute stroke enhances the evidence of an interplay between stroke and cardiac muscle disturbances11.
Previous studies12,13 suggested also that the brain cortex exerts a control of the autonomic system, therefore specific cerebral areas involved in the ischemic damage might contribute to arrhythmogenesis.
As a result, current recommendations suggest a prolonged cardiac monitoring after stroke in order to detect arrhythmias14. Despite recent studies15 reported higher sensitivity of continuous electrocardiographic (ECG) recording, using either external/implantable loop recorders16 or long-term Holter ECG17, as compared to 24-h Holter monitoring, those methods are still not widely used. Currently, among all these methods, implantable loop recording has shown the highest sensitivity in arrhythmias detection, especially AF18,19,20. An early and effective identification of cardiac disturbances is required for treatment management to prevent morbidity and mortality after acute stroke.
The aim of the present study was to detect the rate of both AF and other cardiac arrhythmias after stroke. A further sub-analysis was also carried out to confirm the advantage of using 7-day Holter ECG, which has been suggested to be more accurate than 24-h recordings for arrhythmias detection.
A second aim was to evaluate a possible correlation between cerebral ischemic areas and specific arrhythmia occurrence to support the hypothesis of a role for CNS in arrhythmogenesis.
Methods
One hundred and twenty patients with cryptogenic stroke21, referred to the Neurology Clinic of “SS Annunziata” Hospital of Chieti from July 2018 to March 2020 for ischemic stroke, were included in the study. Exclusion criteria were a previous diagnosis of AF (or at the admission with a standard ECG exam), known etiology of stroke, and ongoing anticoagulation therapy.
The study was performed according to the declaration of Helsinki and its later amendments, and it was approved by the ethical committee of the Department of Neuroscience, Imaging and Clinical Sciences of Chieti. A written consent for research purposes was obtained from all patients.
Clinical and diagnostic assessment
Each patient underwent a complete neurological examination and the “National Institutes of Health Stroke Scale" (NIHSS)22 was estimated both at admission and discharge.
At admission, a thorough medical history was collected, including cardiovascular risk factors such as hypertension, diabetes, smoking, heart failure, and previous coronary disease or transient ischemic attacks/strokes. A CHA2DS2VASc score was calculated. Blood test with lipid profile was performed for each patient, whereas thrombophilia screening was done only in those younger than 60 years.
All patients underwent carotid doppler ultrasonography to evaluate the presence of extracranial atherosclerosis (< 50% if in the same vascular territory of the infarct or ≥ 50% if in a different territory from the infarct), and brain Computed Tomography. Sixty-two patients also underwent a brain Magnetic Resonance Imaging to better characterize ischemic lesions.
Considering patient history, only eight subjects performed a Transcranial Doppler Sonography to investigate the presence of patent foramen ovale.
Sixty-two patients underwent a Transthoracic Echocardiography (TTE) and left atrial volume index (LAVI) and ejection fraction were estimated. LAVI, calculated by dividing left atrial volume by body surface area, was classified as follows: normal (16–28 ml/m2), mildly abnormal (29–33 ml/m2), moderately abnormal (34–39 ml/m2), and severely abnormal (≥ 40 ml/m2). The other fifty-eight patients performed a TTE in a different Center after hospital discharge. These exams were not considered in the study because of missing data (e.g., LAVI measurements).
Within one week after the admission, all patients were monitored with a 7-day continuous Holter ECG recording.
7-day Holter ECG monitoring
7-day Holter ECG (AthenaDiax, Medtronic)23, a continuous ECG recording device, is a small and non-invasive body sensor composed of two devices, an ECG recorder (ARES) and an ECG patch (ZEUS 7D). ARES records heart rhythm, detecting episodes of AF (of at least 30 s duration), bradycardia (< 50 beats for minute, bpm), supraventricular tachycardia (> 109 bpm), SVEB, short supraventricular runs (SVRs, ≥ 120 bpm, 3 beats = 1 run), VEB, and cardiac pauses (> 3000 ms, ms). ZEUS 7D is a single-use product located on the patient’s chest. After recording, ARES is placed into the reader (IRIS), which is connected to a PC via USB cable. Data were analyzed using ThemisLight Software (v2.015; https://www.athenadiax.de/index.php?option=com_content&view=article&id=54:themis-italienisch&catid=13&lang=it&Itemid=264)23. All data were manually inspected and validated by an expert cardiologist.
Statistical analysis
Categorical variables were reported as absolute numbers and percentages. Distributed continuous variables were reported as mean ± standard deviation (SD). Categorical variables were compared using ANOVA, continuous variables using the Mann–Whitney U test. Correlations between variables were assessed by means of the Spearman’s correlation coefficient.
All tests were performed using SPSS Statistic (v26) and the level of significance was set at 0.05.
Results
Study population
Demographic characteristics of the study sample are described in Table 1. The most frequent risk factors for stroke were hypercholesterolemia (62% of patients), hypertension (60%), diabetes (35%), and carotid stenosis (20%), including either < 50% of stenosis in the same vascular territory as the infarct, or ≥ 50% if in a different vascular territory.
At the admission, systemic fibrinolytic therapy was performed in 23 patients (19%).
Arrhythmic events
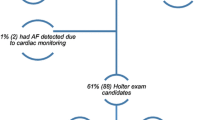
All patients experienced arrhythmias after stroke. Among them, AF was detected in 5 (4%) patients, and it was asymptomatic in all cases. As regards as other rhythmic disturbances, bradycardia was observed in 7 (6%) individuals, supraventricular tachycardia in 25 (20%), SVEB in 116 (94%), SVRs in 67 (54%), and VEB in 108 (88%) (Fig. 1).
The patients with AF (AF group) underwent thrombolysis more frequently than patients without AF detection (no-AF group) (60 vs. 17%; p = 0.018). No correlation was found between NIHSS scores and AF (p = 0.26) and between AF and either SVRs (p = 0.27) or bradycardia (p = 0.52).
A sub-analysis compared all types of cardiac arrythmia detected at the first 24 h of 7-day Holter ECG to those reported during the whole 7-day ECG recording. 7-day Holter ECG monitoring proved to be superior as compared to 24-h recording for the detection of all arrhythmias (statistics is reported in Table 2).
None of the arrhythmic events, considered as dependent variable using repeated measures of ANOVA, showed any association with a prior history of hypertension, diabetes, coronary artery disease, heart failure, smoking, patent foramen ovale, or previous stroke.
In addition, patients with SVRs were older (77.3 ± 13.3 vs. 66.5 ± 12.6 years; p = 0.0001) and had higher CHA2DS2VASc scores (5.3 ± 1.4 vs. 4.0 ± 1.5; p = 0.004) compared to subjects without SVRs (Fig. 2a,b). CHA2DS2VASc score (6.0 ± 0.8 vs. 4.8 ± 1.5; p = 0.026) and age (83.1 ± 9.2 vs. 69.8 ± 14.0 years; p = 0.035) were also higher in patients with bradycardia (Fig. 2c,d). No significant results were found for the presence of SVEB, VEB, supraventricular tachycardia, and AF in relation to age and CHAD2VASC2 scores.
The Box Plots represent the comparisons among different cardiac arrythmias (SVRs, bradycardia, and AF), detected by 7-day Holter ECG, with age, CHA2DS2VASc score, and LAVI. Data are presented as mean ± standard deviation (SD). (a) Patients with SVRs were older (77.3 ± 13.3) compared to those without SVRs (66.5 ± 12.6), p = 0.0001; (b) patients with SVRs showed a higher CHA2DS2VASc score (5.3 ± 1.4) in comparison with the group without SVRs (4.0 ± 1.5), p = 0.004; (c) patients with bradycardia were older (83.1 ± 9.2) compared with those without bradycardia (69.8 ± 14.0), p = 0.035; (d) CHA2DS2VASc score was higher in patients with bradycardia (6.0 ± 0.8) in comparison with individuals without bradycardia (4.8 ± 1.5), p = 0.026; (e) A higher LAVI was detected in AF group compared to no-AF group (p = 0.002); 1 = normal LAVI (16–28 ml/m2), 2 = mildly abnormal (29–33 ml/m2), 3 = moderately abnormal (34–39 ml/m2), and 4 = severely abnormal (≥ 40 ml/m2). SVRs short supraventricular runs, CHA2DS2VASc score assessing risk of atrial fibrillation, AF atrial fibrillation, LAVI left atrial volume index.
No significant difference was found in the detection rate of any type of arrhythmia, considering the time of 7-day Holter ECG placement (measured in days) after stroke.
Transthoracic echocardiography
A higher LAVI was detected in AF group compared to no-AF group (p = 0.002) (Fig. 2e). No difference in ejection fraction was documented between the two groups.
Site of ischemic lesion
The cerebral ischemic lesions were divided into the following categories: side (right or left), insular involvement (yes or not), size (lacunar or no-lacunar stroke), and site (brainstem, cerebellum, basal ganglia, insula, frontal cortex, parietal cortex, temporal cortex, occipital cortex, thalamus, corona radiata, and extensive lesion, when more than a region was involved).
Using repeated measures of ANOVA, each arrhythmic event was considered as a dependent variable compared to the site of ischemic lesion. A significant correlation was found among SVEB onset and parietal (25 vs. 2%; p = 0.013) and temporal (25 vs. 2%; p = 0.013) cortical lesions. Conversely, no associations were found between other arrhythmic events and the remaining sites of ischemic infarction. In addition, VEB occurrence correlated with insular involvement (64 vs. 33%; p = 0.002). No association was found for lacunar stroke and side of lesion compared to each cardiac arrhythmia.
Discussion
The present study shows the rate of arrhythmias occurring after stroke, suggesting the importance of a strict monitoring after cerebrovascular events to prevent possible worse outcomes due to cardiological accidents. Our findings confirm also the higher sensitivity of 7-day Holter ECG, in the detection of all arrhythmias considered, compared to 24-h ECG recording24. As expected, the rate of arrhythmia detection increases with prolonged heart rhythm monitoring24,25 (see Table 2). Even if all our cases of AF were asymptomatic and characterized by a paroxysmal pattern, AF was identified in 4% of our patients using the 7-day Holter ECG. No AF episodes were found with 24-h ECG recording. Hence, our finding seems to be in accordance with previous literature, which showed that 7-day Holter ECG provides a higher AF detection rate, approximately up to 10–15% of cases14,26 as compared to nearly 2% in 24 h recording24. Therefore, the use of 7-day Holter ECG can represent a first-line approach in arrhythmia detection after stroke because of its low costs, effectiveness, and non-invasiveness27. A prolonged cardiac monitoring, such as 7-day Holter ECG, should be placed after stroke to detect rhythm disturbances for both therapeutical management and prognostic evaluation. For the well-known interaction between cerebrovascular events and cardiac disturbances1,2, this device should be considered in all individuals with diagnosis of acute stroke, and especially in those with a cryptogenic or embolic origin.
Newly diagnosed arrythmias are commonly observed after stroke, even if it is currently unclear whether they should be considered as cardiogenic or neurogenic events28.
As regards to the cardiogenic hypothesis, although the presence of AF is a well-established cause of stroke, other coexisting cardiological factors (e.g., endothelial dysfunction, fibrosis, and atrial abnormalities) seem to be associated with rhythm disturbances and, consequentially, thromboembolism29. Of interest in our study, LAVI was higher in AF group compared to no-AF group, confirming the previous assumption of the strict association between LAVI and AF30. As previously reported20,31, a higher LAVI is considered a well-known risk factor for AF and, therefore, it might be a worthwhile clinical predictor for identifying individuals with a major risk of AF and thromboembolic complications, optimizing both therapeutic management and outcomes30,31. Considering other cardiac disturbances, episodes of SVRs were found to correlate with age and CHA2DS2VASc score, in accordance with previous findings32,33. A similar association with age and CHA2DS2VASc score was also observed for bradycardia, which may be an expression of sinus node dysfunctions, such as in the Brady-Tachy Syndrome. These conditions are commonly associated with AF, suggesting the presence of a cardiac rhythm disturbance, which represents a risk factor for embolic ischemic strokes34,35. However, in our results, the lack of correlation between bradycardia and AF and between AF and either CHAD2VASC2 score or age might be explained by the small sample size of subject with AF (only 4% of the whole sample).
Hence, sinus and atrio-ventricular nodal diseases, including Brady-Tachy Syndrome, SVRs, and AF, might represent the result of either an underlying myocardial pathology (shown also by atrial abnormalities, such as a higher LAVI) or a central autonomic imbalance due to stroke29. Moreover, the copresence of cardiovascular risk factors, calculated for instance by CHA2DS2VASc score, may worsen the incidence of both cerebrovascular and cardiac accidents2,32.
As concerns the assumption of a CNS involvement in arrhythmogenesis, the association between cerebral infarction and cardiac events has been largely debated12,36. A pivotal role in autonomic regulation is represented by different cerebral structures, which may act after their damage in atrial remodelling and arrhythmia generation28. Because of its autonomic and limbic connections, converging evidences point toward the role of insula in VEB occurrence13,37,38,39, data confirmed in our results. As well as insula involvement, also temporal and parietal lobes have been related to the onset of severe cardiac arrhythmias12,13. Our findings showed a significant correlation among SVEB and temporal and parietal regions. A previous study showed as atrial premature beats were connected to parieto-temporal area in up to 20% of cases40. However, it remains unclear whether the effects of parietal infarction are directly due to a central autonomic dysfunction or it is necessary to consider parietal lobe only as a bystander region, which shares with the insula the same vascular supply13. Certainly, lesions of parietal cortex have been shown to be an independent predictor of both long-term cardiac morbidity and mortality3,12,13.
Limitations of our study are related to the sample size, which is not wide enough for generalisable considerations. Indeed, the low statistical power of our cohort did not allow to identify a strict association between all arrhythmias and cerebral infarctions and to support the hypothesis of a CNS role for arrhythmogenesis. Moreover, it might be useful to compare the rates of arrhythmias after stroke with those of a population of healthy controls to better evaluate the higher incidence of heart rhythm disturbances after stroke. Additional investigations are needed to confirm our findings.
Conclusions:
Cardiac arrhythmias represent an increased risk factor for complications after stroke. Prolonged cardiac monitoring is a well-recognized strategy to identify individuals to submit to further cardiological examinations and therapies. For its technical characteristics (e.g., non-invasiveness and effectiveness) and its low costs, 7-day Holter ECG should be considered as a first-line approach. Therefore, 7-day Holter ECG should be placed in the first hours after stroke and in all subjects with cryptogenic stroke in order to put in place timely therapeutical strategies.
Further investigations are needed to deeply understand the correlation between cerebral ischemic lesions and the occurrence of arrhythmic events.
Data availability
The data are deposited in the SS Annunziata Hospital of Chieti repository.
References
Samuels, M. A. The brain-heart connection. Circulation 116, 77–84 (2007).
Chen, Z. et al. Brain-heart interaction: Cardiac complications after stroke. Circ. Res. 121, 451–468 (2017).
Ruthirago, D., Julayanont, P., Tantrachoti, P., Kim, J. & Nugent, K. Cardiac arrhythmias and abnormal electrocardiograms after acute stroke. Am. J. Med. Sci. 351, 112–118 (2016).
Wolf, P. A., Abbott, R. D. & Kannel, W. B. Atrial fibrillation as an independent risk factor for stroke: The framingham study. Stroke 22, 983–988 (1991).
Wang, Y. et al. Newly detected atrial fibrillation after acute stroke: A narrative review of causes and implications. Cardiol. 144, 112–121 (2019).
Asplund, K. High prevalence of atrial fibrillation among patients with ischemic stroke. Stroke 45, 2599–2605 (2014).
Chou, P. S. et al. Delayed diagnosis of atrial fibrillation after first-ever stroke increases recurrent stroke risk: a 5-year nationwide follow-up study. Intern. Med. J. 48, 661–667 (2018).
Battaglini, D. et al. Brain-heart interaction after acute ischemic stroke. Crit. Care 24, 163 (2020).
Naver, H. K., Blomstrand, C. & Wallin, B. G. Reduced heart rate variability after right-sided stroke. Stroke 27, 247–251 (1996).
Myers, M. G., Norris, J. W., Hachinski, V. C. & Sole, M. J. Plasma norepinephrine in stroke. Stroke 12, 200 (1981).
Oppenheimer, S. Cerebrogenic cardiac arrhythmias: Cortical lateralization and clinical significance Stephen Oppenheimer. Clin. Auton. Res. 16, 6–11 (2006).
Seifert, F. et al. Neuroanatomical correlates of severe cardiac arrhythmias in acute ischemic stroke. J. Neurol. 262, 1182–1190 (2015).
Rincon, F. et al. Stroke location and association with fatal cardiac outcomes: Northern manhattan study (NOMAS). Stroke 39, 2425–2431 (2008).
Stahrenberg, R. et al. Enhanced detection of paroxysmal atrial fibrillation by early and prolonged continuous holter monitoring in patients with cerebral ischemia presenting in sinus rhythm. Stroke 41, 2884–2888 (2010).
Higgins, P. et al. Noninvasive cardiac event monitoring to detect atrial fibrillation after ischemic stroke: A randomized, controlled trial. Stroke 44, 2525–2531 (2013).
Gladstone, D. J. et al. Atrial fibrillation in patients with cryptogenic stroke. N. Engl. J. Med. 370, 2467–2477 (2014).
Wachter, R. & Freedman, B. The role of atrial fibrillation in patients with an embolic stroke of unknown source (ESUS). Thromb. Haemost. 117, 6–8 (2017).
Sanna, T. et al. Cryptogenic stroke and underlying atrial fibrillation. N. Engl. J. Med. 370, 2478–2486 (2014).
Ziegler, P. D. et al. Long-term detection of atrial fibrillation with insertable cardiac monitors in a real-world cryptogenic stroke population. Int. J. Cardiol. 244, 175–179 (2017).
De Angelis, M. V. et al. Cryptogenic stroke and atrial fibrillation in a real-world population: The role of insertable cardiac monitors. Sci. Rep. 10, 1–6 (2020).
Adams, H. P. J. et al. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24, 35–41 (1993).
DeGraba, T. J., Hallenbeck, J. M., Pettigrew, K. D., Dutka, A. J. & Kelly, B. J. Progression in acute stroke: Value of the initial NIH stroke scale score on patient stratification in future trials. Stroke 30, 1208–1212 (1999).
Medtronic. AthenaDiax. https://www.athenadiax.de/index.php?lang=it.
Jabaudon, D., Sztajzel, J., Sievert, K., Landis, T. & Sztajzel, R. Usefulness of ambulatory 7-day ECG monitoring for the detection of atrial fibrillation and flutter after acute stroke and transient ischemic attack. Stroke 35, 1647–1651 (2004).
Liao, J., Khalid, Z., Scallan, C., Morillo, C. & O’Donnell, M. Noninvasive cardiac monitoring for detecting paroxysmal atrial fibrillation or flutter after acute ischemic stroke: A systematic review. Stroke 38, 2935–2940 (2007).
Ritter, M. A. et al. Occult atrial fibrillation in cryptogenic stroke: Detection by 7-day electrocardiogram versus implantable cardiac monitors. Stroke 44, 1449–1452 (2013).
Chew, D. S., Rennert-May, E., Spackman, E., Mark, D. B. & Exner, D. V. Cost-effectiveness of extended electrocardiogram monitoring for atrial fibrillation after stroke: A systematic review. Stroke 51, 2244–2248 (2020).
Cerasuolo, J. O., Cipriano, L. E. & Sposato, L. A. The complexity of atrial fibrillation newly diagnosed after ischemic stroke and transient ischemic attack: Advances and uncertainties. Curr. Opin. Neurol. 30, 28–37 (2017).
Kamel, H., Okin, P. M., Elkind, M. S. V. & Iadecola, C. Atrial fibrillation and mechanisms of stroke: Time for a new model. Stroke 47, 895–900 (2016).
Tsang, T. S. M. et al. Left atrial volume: Important risk marker of incident atrial fibrillation in 1655 older men and women. Mayo Clin. Proc. 76, 467–475 (2001).
Goldberger, J. J. et al. Evaluating the Atrial Myopathy Underlying Atrial Fibrillation: Identifying the Arrhythmogenic and Thrombogenic Substrate. Circulation 132, 278–291 (2015).
Suzuki, S. et al. Usefulness of frequent supraventricular extrasystoles and a high CHADS 2 score to predict first-time appearance of atrial fibrillation. Am. J. Cardiol. 111, 1602–1607 (2013).
Locati, E. T., Bagliani, G., Ciconte, G. & Pappone, C. Sex and Cardiac Electrophysiology (Elsevier Inc., 2020).
Bodin, A. et al. P5675Ischemic stroke in patient with sinus node disease in comparison to atrial fibrillation, bradycardia-tachycardia syndrome and other cardiac conditions: A nationwide cohort-study. Eur. Heart J. 40, 5675 (2019).
Capucci, A. et al. Monitored atrial fibrillation duration predicts arterial embolic events in patients suffering from bradycardia and atrial fibrillation implanted with antitachycardia pacemakers. J. Am. Coll. Cardiol. 46, 1913–1920 (2005).
Hilz, M. J. & Schwab, S. Stroke-induced sudden-autonomic death: Areas of fatality beyond the insula: Editorial. Stroke 39, 2421–2422 (2008).
Oppenheimer, S. M., Gelb, A., Girvin, J. P. & Hachinski, V. C. Cardiovascular effects of human insular cortex stimulation. Neurology 42, 1727–1732 (1992).
Oppenheimer, S. M., Kedem, G. & Martin, W. M. Left-insular cortex lesions perturb cardiac autonomic tone in humans. Clin. Auton. Res. 6, 131–140 (1996).
Di Stefano, V. et al. Clinical presentation of strokes confined to the insula: a systematic review of literature. Neurol. Sci. 42, 1697–1704 (2021).
Yamour, B. J., Sridharan, M. R., Rice, J. F. & Flowers, N. C. Electrocardiographic changes in cerebrovascular hemorrhage. Am. Heart J. 99, 294–300 (1980).
Author information
Authors and Affiliations
Contributions
C.C.: collection of data; conceptualization; writing; tables and figure. V.D.S.: writing; tables and figure; statistical analysis. M.R.: collection of data; tables and figure. F.D.: collection of data. M.D.P.: collection of data. N.F.: review and editing. M.O.: review and editing. L.B.: supervision; writing—review and editing. M.F.: collection of data; data analysis; review and editing. M.V.D.: conceptualization; design of study; collection of data; supervision; writing—review and editing. All the authors provided final approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carrarini, C., Di Stefano, V., Russo, M. et al. ECG monitoring of post-stroke occurring arrhythmias: an observational study using 7-day Holter ECG. Sci Rep 12, 228 (2022). https://doi.org/10.1038/s41598-021-04285-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-04285-6
- Springer Nature Limited
This article is cited by
-
Arrhythmia detection by the graph convolution network and a proposed structure for communication between cardiac leads
BMC Medical Research Methodology (2024)
-
Heartbeat detector from ECG and PPG signals based on wavelet transform and upper envelopes
Physical and Engineering Sciences in Medicine (2023)