Abstract
The ClpP protease is found in all kingdoms of life, from bacteria to humans. In general, this protease forms a homo-oligomeric complex composed of 14 identical subunits, which associates with its cognate ATPase in a symmetrical manner. Here we show that, in contrast to this general architecture, the Clp protease from Mycobacterium smegmatis (Msm) forms an asymmetric hetero-oligomeric complex ClpP1P2, which only associates with its cognate ATPase through the ClpP2 ring. Our structural and functional characterisation of this complex demonstrates that asymmetric docking of the ATPase component is controlled by both the composition of the ClpP1 hydrophobic pocket (Hp) and the presence of a unique C-terminal extension in ClpP1 that guards this Hp. Our structural analysis of MsmClpP1 also revealed openings in the side-walls of the inactive tetradecamer, which may represent sites for product egress.
Similar content being viewed by others
Introduction
Bacterial Clp proteases are generally formed by two components, a single peptidase component (ClpP) which associates with one or more members of the AAA+ (ATPases associated with a variety of cellular activities) superfamily (e.g. ClpA, ClpX or ClpC)1,2,3,4,5,6. In Escherichia coli, ClpP is expressed as a proenzyme and the N-terminal propeptide is autocatalytically removed7. The active complex is a barrel-shaped oligomer composed of two heptameric rings stacked back-to-back8. The catalytic residues (Ser-His-Asp), of this complex, are encapsulated within the barrel-shaped proteolytic chamber and access to the chamber is restricted to a narrow entry portal at either end of the complex. This design hinders entry of correctly folded proteins into the catalytic chamber, and as such prevents the indiscriminate turnover of cytosolic proteins. Substrate recognition and unfolding is mediated by the ATPase component of these machines, and ATPase docking to ClpP also couples substrate delivery to peptidase activation by triggering “gate-opening” of the peptidase entry portal9. ATPase docking to ClpP is mediated by two types of contacts – static and dynamic10,11. The primary contact is mediated by a static interaction between a highly conserved loop (commonly referred to as the IGF/L loop) located on the proximal face of the ATPase component, which docks into a hydrophobic pocket (Hp) on ClpP. The Hp is located at the periphery of the interface and is composed of several highly conserved aromatic and hydrophobic residues which are critical for interaction with the ATPase component12. The second dynamic contact, which involves the N-terminal loops of ClpP and the pore-2 loops of the ATPase, is axial in nature and regulated by the nucleotide state of the ATPase component13. The Hp is also the site of binding of a novel class of antibiotic that dysregulates ClpP function. These compounds (e.g. acyldepsipeptides (ADEPs)) not only inhibit regulated protein turnover by ClpP by blocking ATPase docking to ClpP, but they also facilitate unregulated access of cytosolic proteins into the catalytic chamber of ClpP thereby triggering their uncontrolled turnover14,15,16,17.
In contrast to E. coli several bacterial species contain two or more ClpP homologs, which form a diverse array of oligomeric complexes18,19,20,21,22,23,24. Interestingly, in Mycobacterium tuberculosis (Mtb), despite both proteins (i.e. ClpP1 and ClpP2), containing catalytic residues and a propeptide, neither protein alone is processed nor are they proteolytically active21. In a landmark study, Goldberg and colleagues identified Benzyloxycarbonyl-Leu-Leucinal (here termed z-LL) as a potent activator of the Mtb hetero-oligomeric ClpP1P2 complex, which led the group to propose that z-LL facilitated assembly of an “active” hetero-oligomer21. Structural analysis later confirmed that the active Mtb Clp protease complex was asymmetric in nature, composed of a single heptameric ring of each protein and that the activator docked into the substrate binding pocket as a substrate agonist24. Finally, the asymmetric nature of the ClpP1P2 complex from Mtb was also shown to extend to its peptidase specificity and its interaction with its cognate ATPase components25,26.
In this study we have examined the activation and assembly of the ClpP1P2 complex from Mycobacterium smegmatis (Msm). Similar to the ClpP1P2 complex from Mtb, we show that MsmClpP1P2 forms an asymmetric complex. The asymmetric nature of this complex is not limited to the composition of the tetradecamer but also extends to the peptidase activity of the complex (including propeptide processing), and interaction with its cognate ATPase components. In this case, our biochemical and structural analysis revealed that asymmetric docking of the ATPase components (to MsmClpP1P2) is controlled by two elements within MsmClpP1. Firstly, by a C-terminal extension in MsmClpP1 that obstructs access of the ATPase component to the Hp and secondly by residues that line the Hp. Together with our analysis of the EcClpP Hp, we have identified Y88 (I104 in EcClpP) as a key feature of ATPase docking specificity. In addition, our structure of MsmClpP1 reveals small openings in the side wall of the ClpP1 tetradecamer, which may represent exit portals for the egress of cleaved polypeptides.
Results and Discussion
Processing of the MsmClpP2 propeptide by the active site residues of MsmClpP1 does not require an activator
Given that correct processing of ClpP propeptides is crucial for ClpP peptidase activity27, we examined the processing of both MsmClpP components. Initially, in order to determine if both MsmClpP subunits were processed, we co-expressed untagged ClpP1 together with His10 tagged ClpP2 (ClpP2H10) in E. coli and co-purified the active MsmClpP1P2 complex. Although recovery of the active hetero-oligomeric complex was poor, only one component (MsmClpP2) was processed, and this processing only occurred when the other component (ClpP1) was present (Fig. S1). To determine the location of the processing site we cloned Msm clpP1 and clpP2, expressed them in E. coli and purified the individual components to analyse propeptide cleavage in vitro. Consistent with the co-expression experiment, the processing of MsmClpP2, required MsmClpP1 (Fig. 1). Interestingly, although these data are similar to the processing of the MtbClpP1P2 complex, in which both ClpP subunits are processed by a hetero-oligomeric complex21,28 processing of MsmClpP2 occurred in the absence of any additional components (Fig. 1b). In contrast to the processing of MsmClpP2, processing of both MtbClpP subunits required either the artificial activator z-LL21 (also see Fig. S2) or a cognate ATPase component25. To determine the site of processing, we performed Edman degradation of ClpP1 and processed ClpP2 (pClpP2) from the in vitro processing assay. Similar to EcClpP and MtbClpP2 (processed between Ala12 and Arg13), MsmClpP2 was processed between residues Ala16 and Arg17. However, in contrast to MtbClpP1, which was processed between Met7 and Arg821,28, no processing of MsmClpP1 was observed.
Propeptide processing of MsmClpP2 occurs via the active site residues of MsmClpP1 in the absence of the activator (z-LL). (a) Cartoon representation of MsmClpP1 and MsmClpP2 indicating the position of the propeptide (grey) and the catalytic Ser residues (residue 95 for MsmClpP1 and 114 for MsmClpP2). The in vitro processing of (b) wild type MsmClpP2 into processed (p) MsmClpP2 (pClpP2) or (c) inactive MsmClpP2 (ClpP2in) into processed ClpP2in (pClpP2in), was monitored in the absence of activator (z-LL). (d) Processing of MsmClpP2 (in the absence of z-LL), failed to occur in the presence of the proteolytically inactive ClpP1 mutant (ClpP1in).
Next we asked, how does processing of MsmClpP2 occur? Initially, to address this question, we generated active site mutants of MsmClpP1 (ClpP1in) and MsmClpP2 (ClpP2in), in which the active site Ser (Ser95 and Ser114, respectively) were replaced with Ala. Similar to the processing of Mtb ClpP228, mutation of the active site Ser in MsmClpP2 (MsmClpP2in) did not affect processing (Fig. 1c), while in contrast processing was completely abolished by mutation of the active site Ser in MsmClpP1 (Fig. 1d). Collectively, these data demonstrate that processing of MsmClpP2 is not autocatalytic, rather it appears to occur in trans via the catalytic triad of MsmClpP1. An alternative interpretation of these data is that the catalytic triad of MsmClpP2 was not active. Therefore, to ensure that the catalytic triad of wild type MsmClpP2 was active, we examined the turnover of different model substrates (from short peptides to a folded protein), by various mixed (wild type and mutant) ClpP1P2 complexes (Fig. S3). These data demonstrated that although the catalytic triad of ClpP2 was active it was not essential for the turnover of all substrates. For example, although the active site of ClpP2 was dispensable for the turnover of short peptide substrates (Fig. S3a,b, compare lanes 2 and 6) it was necessary for efficient EcClpX-mediated turnover of the model protein substrate, GFP-EcSsrA (Fig. S3c, blue triangles). Interestingly, although mutation of ClpP1 completely inhibited the turnover of peptide substrates it had little effect on the rate of native protein turnover (Fig. S3c, red squares). In contrast mutation of ClpP2 slowed the turnover of GFP-EcSsrA (Fig. S3c, blue triangles). Collectively, these data suggest that both ClpP1 and ClpP2 exhibit unique substrate specificities and that entry of a protein substrate (via EcClpX) into the proteolytic chamber may be unidirectional.
Next, in order to gain a better understanding of how in trans processing might occur we examined propeptide processing of various heterologous component combinations (Fig. 2). Initially we monitored the ability of MtbClpP1 to facilitate the processing of MsmClpP2 in the absence or presence of z-LL (Fig. 2a). Interestingly, consistent with the processing of MsmClpP2 by MsmClpP1 (Fig. 1b), z-LL was not required for the MtbClpP1-mediated processing of MsmClpP2, despite an absolute requirement of z-LL for all MtbClpP1 activities (Fig. S2). Furthermore, the processing of MtbClpP1 was not required for processing of MsmClpP2 to occur. Hence these data suggest that the initial processing event (of ClpP2) may occur by a mechanism that is distinct from the downstream processing of ClpP1. We then monitored the ability of MsmClpP1 to facilitate the processing of MtbClpP2 (Fig. 2b). Surprisingly, despite the fact that z-LL was not required for the MsmClpP1-mediated processing of MsmClpP2, it (z-LL) was essential for the processing of MtbClpP2. This suggests that z-LL is required to trigger a conformational change in MtbClpP2 that mediates its processing by MsmClpP1.
Propeptide processing of mixed Mtb/MsmClpP1P2 complexes in the presence or absence of z-LL. (a) In vitro processing of MtbClpP1MsmP2 in the presence (lanes 1–5) or absence of z-LL (lane 6–10). (b) In vitro processing of MsmClpP1MtbP2 in the presence (lanes 1–5) or absence of z-LL (lane 6–10). Following processing proteins were separated by SDS-PAGE and visualised by staining with CBB.
MsmClpP2 is processed via a transient complex
Although propeptide processing of homo-oligomeric ClpP complexes such as EcClpP and Homo sapiens ClpP (HsClpP) has long been described as autocatalytic7, the mechanism by which this step occurs is poorly defined and currently it remains unclear if processing occurs before (or as a result of) assembly of the tetradecamer, or indeed if processing occurs via the cis or trans ring7,27,29. Therefore, we examined the composition of the active “processing” complex. For homo-oligomeric ClpP complexes, it is plausible that processing is mediated by the cis ring and the processing site is determined via a molecular “ruler” mechanism. For hetero-oligomeric complexes such as ClpP1P224, the distance between the processing site (within the propeptide) and the active site within the trans ring (~47 Å) is estimated to be much greater than the distance between the propeptide and the active site in the cis ring (~25 Å). Hence, to determine if processing of ClpP2 is mediated by formation of the ClpP1P2 complex, we generated single point mutations in both MsmClpP proteins to disrupt the ring-ring interface. This mutation was based on a critical Arg residue that stabilises the ring-ring interface of Staphylococcus aureus ClpP (SaClpP)30. Specifically, we replaced the Arg-finger residue in MsmClpP1 and MsmClpP2 (Arg168 and Arg189, respectively) with Ala. Initially, to ensure the overall structure of each mutant protein was not compromised, we compared the oligomeric state of each protein in the absence of z-LL using analytical ultracentrifugation (AUC). Importantly, both wild type and mutant MsmClpP1 and MsmClpP2 each formed heptamers (Fig. S4, red circles). Next, to monitor the effect of these mutations on MsmClpP1P2 complex formation we performed a series of pull-down experiments, in which wild type or mutant MsmClpP1H10 was immobilised to Ni-NTA-agarose beads. Importantly, mutation of a single component was sufficient to significantly reduce its interaction with the other component (Fig. 3), while mutation of both components almost completely abolished the interaction of the two components (Fig. 3b, lane 6). These data demonstrate that, similar to SaClpP, the Arg finger plays a crucial role in stabilising the MsmClpP1P2 complex. Consistent with this loss of the ClpP1P2 tetradecamer, the turnover of a model peptide (Fig. 3c) or protein (Fig. 3d) substrate, by each of the different mutant protein complexes, was completely abolished. Surprisingly, and in contrast to the peptidase activity of each mutant protein complex, all three complexes retained the ability to process ClpP2, with only a modest change to the rate of processing (Fig. 3e). One explanation for these data is that the substrate (i.e. the propeptide) is, in this case, tethered to the peptidase and hence any residual interaction between the two rings may be sufficient to facilitate rapid cleavage of the propeptide. An alternative explanation for these data is that processing of ClpP2 is not mediated by the classic ClpP1P2 tetradecamer, but rather by an interaction that does not require inter-ring contacts mediated by the Arg fingers. Consistent with this idea, propeptide processing of MsmClpP2 (by MsmClpP1) is independent of z-LL activity (Fig. 1), while in contrast the activator is essential for all other proteolytic activities of the MsmClpP1P2 complex.
In contrast to peptide and protein degradation, propeptide processing of MsmClpP2 does not require stable interaction between MsmClpP1 and MsmClpP2. (a) Cartoon representation of MsmClpP1-H10 and MsmClpP2 indicating the position of the conserved Arg finger in ClpP1 (R168) and ClpP2 (R189). (b) Co-immunoprecipitation (co-IP) of wild type (lanes 1–3) or mutant ClpP1-H10 (lanes 5–7) in the presence of either wild type (lanes 3 and 7) or mutant pClpP2 (lane. 2 and 6). The total amount of wild type or mutant pClpP2 (lane 9 and 10, respectively) added to the co-IP is indicated. (c–d) The turnover of AAF-amc peptide (c) or rate of GFP-EcssrA degradation was monitored by fluorescence using wild type or mutant ClpP1P2 complexes (e) comparison of propeptide processing by the wild type ClpP1P2 complex (left panel) and the ClpP1R168AP2R189A complex (right panel).
Mutation of the hydrophobic pocket modulates the peptidase activity of the MsmClpP1pP2 complexes
Next, in order to study the interaction of the MsmClpP1P2 complex with its cognate ATPase components (ClpX and ClpC1), we generated specific point mutations within the hydrophobic pockets of MsmClpP1 and MsmClpP2. Initially, we targeted two residues in the Hp (the first was a highly conserved tyrosine residue found in all ClpP sequences, Y60 in ClpP1 and Y79 in ClpP2, the second hydrophobic residue was less conserved across ClpP sequences, Y110 in ClpP1 and L129 in ClpP2). Each of the hydrophobic residues (described above) was replaced with alanine, to generate a series of single point mutants (P1Y60A, P1Y110A, P2Y79A or P2L129A) and one double mutant of MsmClpP2 in which both hydrophobic residues (Y79 and L129) were replaced with alanine, here referred to as P2dbl. To ensure that mutation of the Hp of MsmClpP1 and MsmClpP2, did not alter the overall structure of each protein, we examined the oligomeric state of each Hp mutant using AUC and compared them to the wild type proteins (Fig. S4). Importantly, with the exception of P1Y60A, the oligomeric state (as determined by AUC) of each mutant was largely unaffected (Fig. S4). A similar trend for ClpP1 mutants was also observed using Native-PAGE (Fig. S4), although in this case, again with the exception of P1Y60A the gel appeared to stabilize the 14-mer. Stabilisation of the 14-mer by Native-PAGE has also been observed for human ClpP29. Surprisingly, neither the 7-mer nor the 14-mer complexes of ClpP2 were observed in Native-PAGE.
Next, we compared the peptidase activity of the wild type MsmClpP1P2 complex with the various MsmClpP1 and MsmClpP2 mutant proteins (Fig. S5). Interestingly, despite the changes to the oligomeric state of P1Y60A only a small change in the rate of AAF-amc turnover (by P1Y60AP2) was observed (Fig. S5a), while in contrast, the rate of LY-amc turnover by P1Y60AP2 was unexpectedly increased by ~4-fold (Fig. S5b). Collectively these data appear to suggest that the replacement of aromatic residues (e.g. Y60A) within the Hp of ClpP1 may affect the conformation of the substrate binding pocket (S1) directly, which in the presence of the peptide agonist z-LL, could modulate substrate affinity, specificity and/or cleavage. Consistently, ClpP1 is directly responsible for the turnover of both peptide substrates, as replacement of the active site Ser of MsmClpP1 with Ala (P1in) abolished the turnover of both substrates (Fig. S3). In contrast to P1Y60A the relative peptidase activity of P1Y110A (by the P1Y110AP2 complex) was essentially unchanged for both substrates (Fig. S5a,b). Importantly, given that P1Y110A did not exhibit any substrate-dependent defects in peptidase activity, we limited our subsequent analysis to the ClpP1Y110AP2 complex. Next, we examined the effect of the different Hp mutations in MsmClpP2. Surprisingly, the peptidase rate for both substrates was significantly reduced (by up to 80%) for both single point mutants (Y79A and L129A) of MsmClpP2 (P2Y79A and P2L129A) (Fig. S5c,d). Intriguingly, the rate of peptide degradation by the double mutant (P2dbl) was unchanged for both substrates (Fig. S5c,d, compare columns 1 and 4). Currently it remains unclear why the peptidase activity of the two single mutants in complex with wild type ClpP1 is reduced. However, given that the double mutant exhibited a similar peptidase activity to the wild type complex, all further analysis was limited to the ClpP1P2dbl complex.
MsmClpP1P2 forms asymmetric complexes with its cognate ATPase
To determine the mode of ATPase docking to the MsmClpP1P2 complex, we examined the degradation of several ATPase-dependent substrates in the presence of wild type or mutant complexes of MsmClpP1P2. Initially, we examined the ATPase-dependent turnover of GFP-EcSsrA by Hp mutants of MsmClpP1 (in the presence of wild type ClpP2) using the non-cognate ATPase, EcΔNClpX. Consistent with recent findings31, EcΔNClpX was able to mediate the turnover of GFP-EcSsrA by MsmClpP1P2, either in the absence or presence of the peptide activator, z-LL. Significantly, despite differences in peptidase activity of the various ClpP1 Hp mutants (Fig. S5) the EcΔNClpX -mediated turnover of GFP-EcSsrA by each mutant protein complex was equivalent to the wild type complex (Fig. S6). Next, we examined the EcΔNClpX-mediated turnover of the same substrate by ClpP1P2 complexes bearing Hp mutations in ClpP2 (Fig. S6). Importantly, mutation of either or both Hp residues on ClpP2 abolished the ATPase-mediated turnover of GFP-EcSsrA (Fig. S6). Collectively, these data suggest that the ATPase component docks exclusively to ClpP2. To confirm the asymmetric nature of the MsmClpP1P2 complex we monitored the turnover of two additional model substrate that are mediated by Mycobacterial ATPase components (i.e. GFP-MtbSsrA for ClpX and fluorescently-labelled model unfolded protein, FITC-casein for ClpC1). In this case, given that the “ClpP docking loop” of each ATPase component (ClpX and ClpC1) is conserved across Msm and Mtb (Fig. S7), the turnover of each substrate by the various Hp mutant complexes was examined in the presence of either MtbClpX or MtbClpC1, respectively. Consistent with the asymmetric binding observed for EcΔNClpX, the MtbClpX-dependent turnover of GFP-MtbSsrA was unaffected by mutation of the Hp in MsmClpP1 (Fig. 4 lane 5). In contrast, the equivalent Hp mutation in MsmClpP2 completely abolished substrate turnover (Fig. 4, lane 6). Similarly, the MtbClpC1-dependent turnover of FITC-casein was unaffected by mutation of the Hp in MsmClpP1 (Fig. 4, lane 8), while the equivalent Hp mutation in MsmClpP2 effectively abolished the turnover of FITC-casein (Fig. 4, lane 9). Collectively these data indicate that, mutation of the Hp in MsmClpP2 is sufficient to completely abolish the turnover of substrates by all ATPases tested. Indeed, both the cognate ATPases (MtbClpX and MtbClpC1) and the heterologous ATPase (EcClpX) bind specifically to MsmClpP2 and not to MsmClpP1 forming single-headed, asymmetric complexes (ClpP1P2X and ClpP1P2C1). These data are consistent with the findings by Weber-Ban and colleagues who showed that the MtbClpP1P2 complex only binds to its partner unfoldase components through ClpP225.
MsmClpP1P2 form an obligate single-headed complex with its cognate ATPase components. (a) The EcΔNClpX-mediated degradation of GFP-EcSsrA is mediated by docking to MsmClpP2. Although mutation of the Hp residue (Y110) to Ala did not affect the ATPase-mediated delivery of GFP-EcSsrA (red squares), mutation of the Hp residues (Y79 and Y129) abolished turnover (blue triangles). (b) The rate of either GFP-EcSsrA degradation by EcΔNClpX (white bars), GFP-MtbSsrA degradation by MtbClpX (grey bars) or FITC-casein degradation by MtbClpC1 (black bars) was determined from three independent experiments (n = 3) using either wild type ClpP1P2 (lanes 1, 4 and 7), ClpP1Y110AP2 (lanes 2, 5 and 8) or ClpP1P2dbl (lanes 3, 6 and 9). Error bars represent SEM.
Next, to better understand the molecular basis of this asymmetric specificity we compared the Hp residues of several ClpP homologs. From this analysis we noticed that in contrast to most ClpP sequences, MsmClpP1 contained an additional aromatic residue (Y88) within the Hp and speculated that this aromatic residue inhibited ATPase docking (Fig. S8). To test this hypothesis, we first replaced the equivalent residue in EcClpP (I104) with tyrosine to generate EcClpPI104Y and tested the ability of this mutant protein to interact with its cognate ATPase components (ClpA and ClpX). Consistent with the idea that Y88 is a crucial inhibitory element within the Hp, EcClpPI104Y prevented the ClpA-mediated degradation of GFP-EcSsrA (Fig. 5a). However, the same mutation had no effect on the ClpX-mediated turnover of SsrA-tagged GFP (Fig. 5b). Interestingly, although the introduction of tyrosine at residue 104 (in EcClpP) was sufficient to inhibit ClpA docking, EcClpP (bearing Ile at residue 104) was unable to functionally interact with either MtbClpC1 (Fig. 5c, open red triangles) or MtbClpX (Fig. 5d, open red triangles). In contrast to ClpA docking, replacement of Ile104 with Tyr in EcClpP (EcClpPI104Y), did not affect EcClpX-docking. However, this mutation was sufficient to recover wild type-like activity with MtbClpX. Collectively these data indicate that tyrosine (at residue 104/88) although inhibitory to ClpA and ClpC1 docking is permissive to ClpX docking. This in turn suggests that residue 104/88 may act as a “sensor” of ATPase docking and not simply a key inhibitory element within MsmClpP1, that obstructs ATPase docking.
Single point mutations in the Hp of EcClpP inhibit ClpA-mediated substrate-turnover but facilitate MtbClpX-mediated substrate-turnover. The degradation of GFP-EcSsrA mediated by (a) EcClpA and (b) EcΔNClpX was monitored by fluorescence in the presence of wild type EcClpP (black symbols) or EcClpPI104Y (blue symbols). (c) The degradation of FITC-casein was monitored by fluorescence in the presence of either EcClpAP (filled circles) or MtbClpC1 (triangles) together with MsmClpP1P2 (filled triangles), EcClpP (open red triangles) or EcClpPI104Y (open blue triangles). (d) The turnover of GFP-EcSsrA was monitored by fluorescence in the absence of any addition (green circles) or in the presence of either EcΔNClpXP (filled black triangles) or MtbClpX together with either MsmClpP1P2 (filled red circles), EcClpP (open red triangles) or EcClpPI104Y (open blue triangles). Degradation rates were determined from three independent experiments (n = 3). Error bars represent SEM.
The C-terminal extension (CTE) of MsmClpP1 obstructs ATPase docking and substrate delivery. (a) Ribbon representation of the oligomeric tetradecamer of MsmClpP1 in top view (left panel) and side-view (right panel). Individual subunits are indicated in green, blue and orange. (b) Close up view of the catalytic triad of MsmClpP1 (slate blue) in comparison to EcClpP (light pink, PDB code 3MT6) demonstrating that the His120 is dislocated ~5.8 Å from the catalytic Ser residue (Ser 95) (c) Surface representation of MsmClpP1 tetradecamer highlighting side-wall openings located between the rings, adjacent to the catalytic triad. (d) Three tyrosine residues (Tyr60, Tyr88 and Tyr110) line the Hp and interact with the CTE (pink). (e) The EcΔNClpX-mediated degradation of GFP-EcSsrA was monitored by fluorescence in the presence of various ClpP1P2 complexes. Although the turnover of GFP-EcSsrA by wild type ClpP1P2 (open black circles) was not affected by deletion of the CTE (open blue squares) it was blocked by mutation of the Hp in ClpP2 (filled black circles). Importantly, degradation via the ClpP1P2dbl was recovered when the CTE was removed from ClpP1 (filled blue squares). Degradation assays were determined from three independent experiments (n = 3). Error bars represent SEM.
Structural basis for docking asymmetry of MsmClpP1P2
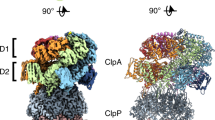
To better understand the molecular features that define ATPase docking we crystallised MsmClpP1 (Fig. 6). The structure of MsmClpP1 was determined to 2.0 Å resolution and refined to an Rfree value of 21.5% (Table 1). Similar to other ClpP structures, MsmClpP1 was composed of two heptameric rings stacked back-to-back (the asymmetric unit consisted of one heptamer, the biological tetradecamer is formed from crystallographically related heptamers). The tetradecamer forms a compact barrel-shaped oligomer with approximate dimensions 86 × 102 Å (Fig. 6a). The overall fold of the MsmClpP1 protomer is similar to most ClpP structures; structural superposition of MsmClpP1 with MtbClpP1 (2CBY)32, MtbClpP1P2 (4U0G)24 and EcClpP (3MT6)17 resulted in overall r.m.s.d. values of 0.8 Å (1183 Cα aligned), 2.3 Å (1024 Cα aligned) and 2.6 Å (1071 Cα aligned), respectively. However, there are a number of notable differences between these structures. Firstly, the structure of MsmClpP1, similar to MtbClpP132, is in an inactive conformation, as His120 is dislocated from the catalytic triad (i.e. the distance between Ser95 and His120 is 5.8 Å (Fig. 6b, right panel) (6.55 Å for the equivalent residues MtbClpP1). In contrast, in the ClpP1 component of the MtbClpP1P2 complex (4U0G) and in EcClpP, these residues are only 2.9 Å apart (Fig. 6b, left panel). Another key difference between these structures relates to the formation of the tetradecamer, via the handle domains. In EcClpP and MtbClpP1P2 complex, the two heptameric rings associate via the formation of an antiparallel β-sheet in the handle domains, whereby each β-sheet is composed of strands from opposing monomers across the ring-ring interface. Compared to these active structures, both MsmClpP1 and MtbClpP1, have shorter handle domains, which lack a structured β-strand region. As a result, association of the heptameric rings in MsmClpP1 (and MtbClpP1) is mediated largely by a trans association of a short section of the α-helix in the handle domain creating several openings in the side-walls of the ClpP1 tetradecamer (Figs. 6c and S9). Although handle flexibility has been observed in a selection of inactive ClpP structures, the size and precise location of pores resulting from this flexibility has been difficult to assess due to the large number of unmodeled residues in these structures. Significantly, the breaches in the side wall of MsmClpP1 are located adjacent to the catalytic residues of the protease and as such provide a direct path between the active site of the protease and the external solution. Hence as has been proposed by Kay and Houry33, these openings could represent an exit portal for the egress of peptides. Therefore, we postulate that this structure of MsmClpP1 represents a post “substrate-cleavage” snapshot of the protease. Following peptide cleavage, this protease acquires an inactive conformation where the catalytic His residue adopts a distorted configuration, which initiates a conformational change in the adjacent handle domain whereby the β-strand that typically forms an antiparallel β-sheet with the equivalent β-strand in the opposite monomer, becomes disordered. As a result of this disorder in the β-strand, several interactions which stabilise the tetradecamer are lost. This results in a shorter interface between opposing subunits in the tetradecamer and opening of “large pores” along the surface of the protein which could allow release of the cleaved peptides. Smaller solvent exposed channels have also been observed in the side walls of the MtbClpP1P2 complex (Fig. S9), however these openings were only located between ClpP1 subunits, above the equatorial interface, ~15 Å from the catalytic triad24.
Another distinctive feature of MsmClpP1, which is absent in many ClpP homologues including MtbClpP124,32, is a short C-terminal extension (CTE) ~12 residues long, which docks into its own Hp (Fig. 6d). Interestingly, the CTE interacts with residues within the Hp of ClpP in a similar manner to ADEP16,17,24. Specifically, the CTE interacts with three tyrosine residues (Y60, Y88 and Y110) within the Hp (Fig. 6d). Hence, we speculated that, similar to ADEPs, the CTE of MsmClpP1 may restrict ATPase docking. To test this idea, we deleted the CTE of MsmClpP1 and monitored the ability of various wild type and mutant MsmClpP1P2 complexes to interact with different ATPase components (Figs. 6e and S10). First, we examined the EcClpX-mediated turnover of GFP-EcSsrA by MsmClpP1ΔCTE in the presence of wild type MsmClpP2. Importantly, this complex retained wild type-like activity against each ATPase/substrate combination tested, demonstrating that removal of the CTE does not alter ClpP1P2 activity (Fig. 6e, open symbols). Next, in order to switch ATPase-docking specificity (from MsmClpP2 to MsmClpP1), we repeated the above experiments in the presence of MsmClpP2dbl (which is unable to dock to any of the ATPase components tested). Remarkably, deletion of the CTE was sufficient to facilitate the EcClpX-mediated turnover of GFP-EcSsrA (Fig. 6e, filled squares). These data demonstrate that the CTE of MsmClpP1 plays a crucial inhibitory role in ATPase docking. Next, we monitored the docking of the cognate ATPase components (MtbClpX and MtbClpC1) to the ClpP1ΔCTEP2dbl complex. Surprisingly, neither MtbClpX nor MtbClpC1 were able to mediate the turnover of either GFP-MtbSsrA (Fig. S10b) or FITC-casein (Fig. S10c) respectively. Taken together, these data suggest that although the CTE obstructs ATPase docking (of EcClpX), removal of this feature alone is not sufficient to permit docking to the physiological relevant ATPase components (MtbClpX/MtbClpC1). Hence, both the presence of the CTE (that occludes the Hp of ClpP1) and the altered specificity of the Hp may have important implications for the development of novel ADEP-like antibiotics that dysregulate Clp proteases.
Interestingly, several ClpP homologs contain a C-terminal extension (including human ClpP which contains a 28-residue extension). To date however, the role of these CTEs has remained unclear. In contrast to our structure of MsmClpP1, the CTE of HsClpP extends away from the heptameric ring27. Despite this “snapshot” placing the CTE of human ClpP away from the ATPase-interface we speculate that this region (in various ClpP homologues) could play an important role in regulating ClpP function in vivo. One possibility is that the CTE (or in the case of MsmClpP1 - the atypical Hp) may have co-evolved with an alternate Clp-protease activator to further diversify the function of the peptidase. Consistent with this idea, novel (ATP-independent) Clp protease activators have recently been identified in plants34. Likewise, the Mycobacterial proteasome has also been shown to function with a variety of activators (both ATP-dependent and ATP-independent)3,35,36,37,38. Therefore, we speculate that novel ClpP activators may exist in a variety of species which contain ClpP homologs with either an extended C-terminus or an atypical Hp.
In conclusion, our findings clearly demonstrate that, similar to MtbClpP1P2, the hetero-oligomeric Clp-protease complex in Msm is highly regulated. Although processing of MsmClpP2 (and hence “activation” of the MsmClpP1P2 complex) can proceed in the absence of an activator, the in vitro peptidase activity of MsmClpP1P2 (for peptide and protein turnover) requires either a cognate ATPase component or a chemical activator (i.e. z-LL). Significantly, both cognate ATPase components dock to only one face of the peptidase. This asymmetry provides direct competition for ATPase-docking to the peptidase, and as a result (dependent on the abundance of each component), likely controls the delivery of specific substrates to this peptidase and hence their turnover in a tightly regulated fashion. The specific docking of both ATPase components to a single face of this machine may also provide an opportunity for further diversification of the peptidase, through docking of additional specific activators to the vacant platform on the ClpP1P2 complex.
Material and Methods
Cloning
Msm clpP1 (MSMEG_4673) and clpP2 (MSMEG_4672) were amplified from M. smegmatis mc2155 genomic DNA (kindly provided by Prof. R. Manganelli) with specific primers (see Table S1), using Phusion DNA polymerase (New England Biolabs). Mtb clpP1 (Rv2460c) and clpP2 (Rv2461c) were amplified from Mtb genomic DNA (kindly provided by Ms. M. Globan, VIDRL). The amplified DNA was digested with the appropriate restriction enzymes and ligated into similarly digested plasmids. Fragments coding for unprocessed ClpP1 and ClpP2 were cloned into pET10C (and pET10N)39 to generate either a C- or N-terminal His10-tagged fusion protein, while fragments coding for processed forms of ClpP1 and ClpP2 (i.e. lacking propeptide) were cloned into pHUE40 to generate N-terminal His6-Ub fusion proteins. Mtb clpX cloned into pET15(b) was a kind gift from Dr. P. Genevaux (Université de Toulouse, France), and Mtb clpC1 cloned into pET30(a) was a kind gift from Dr. D. Vasudevan (Institute of Life Sciences, Bhubaneswar, India)41. The SsrA-tag from Mtb/Msm (AADSNQRDYALAA) was cloned into pDD17342 by annealing specific primers (see Table S1) to generate a C-terminal GFP fusion (GFP-MtbSsrA). All clones (Table S2) were verified by nucleotide sequencing.
Protein expression and purification
His6-tagged EcClpX and EcClpP were expressed in E. coli and purified as described previously43. MtbClpC1 and MtbClpX were both expressed as C-terminal His6-tagged fusion proteins. MtbClpC1-His6 was expressed as described in41, while MtbClpX-His6 was expressed at 16 °C in E. coli BL21 (DE3) codon+ RIL cells, following the addition of 0.5 mM IPTG. ClpP1 and ClpP2 (from either Msm or Mtb) were expressed, with either a C-terminal His10 tag, essentially as described in39 or alternatively with a His6-Ub tag (which was subsequently cleaved) as described44. All His6-tagged fusion proteins were purified using Ni-NTA-agarose beads as described previously43, while His10-tagged fusion proteins were purified essentially as described45. Untagged ClpP1 and ClpP2 (wild type and specific point mutants) were also generated using the Ub-fusion system40 and purified essentially as described, using a combination of IMAC and preparative grade size exclusion chromatography (SEC).
Protein analysis by electrophoresis
For the analysis of protein purity and protein turnover, samples were separated by 16.5% Tricine SDS-PAGE46. To analyse the oligomerisation/native structure of wild type and mutant ClpP1 complexes, 5 µg of purified protein was separated using 4–16% Native-PAGE Novex Bis-Tris gels (Invitrogen) essentially as described29 and visualised by staining with Coomassie Brilliant Blue (CBB) R250.
Processing and peptidase assays
For processing assays, either full length Msm or Mtb ClpP1 was incubated together with full length ClpP2 (from either Msm or Mtb) at 30 °C in Buffer XP (25 mM Tris-HCl pH 7.5, 100 mM KCl, 100 mM NaCl, 20 mM MgCl2, 10% (v/v) glycerol, 0.025% (v/v) Triton X-100, 1 mM DTT) in the absence or presence of the activator, z-Leu-Leu-H (z-LL). Processing of ClpP (2.8 µM) was initiated, either by addition of the activator (0.5 mM), or equimolar amounts of the partner ClpP protein. The reaction was stopped (at the indicated time points), by the addition of sample buffer followed by incubation at 95 °C for 5 min. Processing of MsmClpP was analysed by 16.5% Tris-tricine SDS-PAGE while processing of MtbClpP was analysed by 15% glycine SDS-PAGE. To monitor the peptidase activity of wild type and mutant ClpPs, peptide degradation assays were performed using fluorescently labelled peptides essentially as described29. Similarly, the turnover of GFP-EcSsrA, GFP-MtbSsrA or FITC-casein (by 1 µM EcΔNClpX, MtbClpX or MtbClpC1, respectively) was and monitored by fluorescence as described29. All peptide and protein degradation assays were performed in the presence of 0.5 mM z-LL (unless otherwise stated).
Crystallisation of MsmClpP1 and diffraction data collection
Purified MsmClpP1 (in 50 mM Tris-HCl pH 8.0, 200 mM KCl) was concentrated using a Centricon-30 centrifugal concentrator (Amicon) up to 30 mg/mL. MsmClpP1 crystallization experiments were performed using the vapor diffusion method. Initial high-throughput crystallization experiments were performed in house or at the CSIRO Collaborative Crystallization Centre (www.csiro.au/C3; Melbourne, Australia). For crystal optimization experiments, drops were set in 24-well plates by mixing 1 µL of protein with 1 µL of well condition and drops were equilibrated at 20 °C against a reservoir volume of 500 µl. Small hexagonal crystals were obtained in 1.8M sodium malonate pH 6.4. After two rounds of optimisation, which included changing pH, malonate concentration and protein concentration a significant improvement in the quality of the crystals was obtained. Larger hexagonal crystals (with approximate dimensions 0.3 mm × 0.2 mm × 0.1 mm) were obtained from solutions consisting of 2.1–3.4M sodium malonate pH 6.6–6.8 and a protein concentration of 5.5 mg/mL.
The crystals were cryoprotected using 3M sodium malonate pH 6.5–6.9 and flash cooled in liquid nitrogen. Diffraction data was collected at 100 K at the protein crystallography beamline MX2 at the Australian synchrotron using an ADSC Quantum 315r detector. 1° oscillation images were collected for a total of 180° using a crystal-to-detector distance of 260 mm. Diffraction data were integrated and scaled with HKL200047.
Structure determination and refinement
The crystal structure of MsmClpP1 was solved by molecular replacement using BALBES48 using the structure of M. tuberculosis ClpP as a search model (PDB: 2CBY, sequence identity 96%). The model was built using Coot49 and refined using phenix.refine50 and TLS (translation/libration/screw) refinement51. Most of the structure could be unambiguously assigned in the electron density map except residues 1–10 at the N- terminus of each chain, and the loop region between residues 125–129, which in chains A, B, C and E were difficult to model because of poor density. The final model was validated using Molprobity52. Table 1 provides the statistics for the X-ray data collection and final refined model. All structural figures were generated with PyMOL. Superposition of molecules was carried out using the Secondary Structure Matching (SSM) option from the program Coot49.
References
Gur, E., Ottofueling, R. & Dougan, D. A. Machines of destruction - AAA+ proteases and the adaptors that control them. Subcell Biochem 66, 3–33, https://doi.org/10.1007/978-94-007-5940-4_1 (2013).
Olivares, A. O., Baker, T. A. & Sauer, R. T. Mechanistic insights into bacterial AAA+ proteases and protein-remodelling machines. Nat Rev Microbiol 14, 33–44, https://doi.org/10.1038/nrmicro.2015.4 (2016).
Alhuwaider, A. A. H. & Dougan, D. A. AAA+ Machines of Protein Destruction in Mycobacteria. Front Mol Biosci 4, 49, https://doi.org/10.3389/fmolb.2017.00049 (2017).
Elsholz, A. K. W., Birk, M. S., Charpentier, E. & Turgay, K. Functional Diversity of AAA+ Protease Complexes in Bacillus subtilis. Front Mol Biosci 4, 44, https://doi.org/10.3389/fmolb.2017.00044 (2017).
Mahmoud, S. A. & Chien, P. Regulated Proteolysis in Bacteria. Annu Rev Biochem 87, 677–696, https://doi.org/10.1146/annurev-biochem-062917-012848 (2018).
Striebel, F., Kress, W. & Weber-Ban, E. Controlled destruction: AAA+ ATPases in protein degradation from bacteria to eukaryotes. Curr Opin Struct Biol 19, 209–217, https://doi.org/10.1016/j.sbi.2009.02.006 (2009).
Maurizi, M. R., Clark, W. P., Kim, S. H. & Gottesman, S. Clp P represents a unique family of serine proteases. The Journal of biological chemistry 265, 12546–12552 (1990).
Wang, J., Hartling, J. A. & Flanagan, J. M. The structure of ClpP at 2.3 A resolution suggests a model for ATP-dependent proteolysis. Cell 91, 447–456 (1997).
Liu, K., Ologbenla, A. & Houry, W. A. Dynamics of the ClpP serine protease: a model for self-compartmentalized proteases. Crit Rev Biochem Mol Biol 49, 400–412, https://doi.org/10.3109/10409238.2014.925421 (2014).
Kim, Y. I. et al. Molecular determinants of complex formation between Clp/Hsp100 ATPases and the ClpP peptidase. Nat Struct Biol 8, 230–233, https://doi.org/10.1038/84967 (2001).
Martin, A., Baker, T. A. & Sauer, R. T. Distinct static and dynamic interactions control ATPase-peptidase communication in a AAA+ protease. Mol Cell 27, 41–52, https://doi.org/10.1016/j.molcel.2007.05.024 (2007).
Leung, E. et al. Activators of cylindrical proteases as antimicrobials: identification and development of small molecule activators of ClpP protease. Chemistry & biology 18, 1167–1178, https://doi.org/10.1016/j.chembiol.2011.07.023 (2011).
Martin, A., Baker, T. A. & Sauer, R. T. Pore loops of the AAA+ ClpX machine grip substrates to drive translocation and unfolding. Nat Struct Mol Biol 15, 1147–1151, https://doi.org/10.1038/nsmb.1503 (2008).
Brotz-Oesterhelt, H. et al. Dysregulation of bacterial proteolytic machinery by a new class of antibiotics. Nat Med 11, 1082–1087, https://doi.org/10.1038/nm1306 (2005).
Kirstein, J. et al. The antibiotic ADEP reprogrammes ClpP, switching it from a regulated to an uncontrolled protease. EMBO Mol Med 1, 37–49, https://doi.org/10.1002/emmm.200900002 (2009).
Lee, B. G. et al. Structures of ClpP in complex with acyldepsipeptide antibiotics reveal its activation mechanism. Nat Struct Mol Biol 17, 471–478, https://doi.org/10.1038/nsmb.1787 (2010).
Li, D. H. et al. Acyldepsipeptide antibiotics induce the formation of a structured axial channel in ClpP: A model for the ClpX/ClpA-bound state of ClpP. Chemistry & biology 17, 959–969, https://doi.org/10.1016/j.chembiol.2010.07.008 (2010).
Balogh, D. et al. Insights into ClpXP proteolysis: heterooligomerization and partial deactivation enhance chaperone affinity and substrate turnover in Listeria monocytogenes. Chem Sci 8, 1592–1600, https://doi.org/10.1039/c6sc03438a (2017).
Cohen, S. E., McKnight, B. M. & Golden, S. S. Roles for ClpXP in regulating the circadian clock in Synechococcus elongatus. Proc Natl Acad Sci USA 115, E7805–E7813, https://doi.org/10.1073/pnas.1800828115 (2018).
Dahmen, M., Vielberg, M. T., Groll, M. & Sieber, S. A. Structure and mechanism of the caseinolytic protease ClpP1/2 heterocomplex from Listeria monocytogenes. Angewandte Chemie 54, 3598–3602, https://doi.org/10.1002/anie.201409325 (2015).
Akopian, T. et al. The active ClpP protease from M. tuberculosis is a complex composed of a heptameric ClpP1 and a ClpP2 ring. EMBO J 31, 1529–1541, https://doi.org/10.1038/emboj.2012.5 (2012).
Lavey, N. P., Shadid, T., Ballard, J. D. & Duerfeldt, A. S. Clostridium difficile ClpP Homologues are Capable of Uncoupled Activity and Exhibit Different Levels of Susceptibility to Acyldepsipeptide Modulation. ACS. Infect Dis 5, 79–89, https://doi.org/10.1021/acsinfecdis.8b00199 (2019).
Wood, N. A. et al. Initial Characterization of the Two ClpP Paralogs of Chlamydia trachomatis Suggests Unique Functionality for Each. Journal of bacteriology 201, https://doi.org/10.1128/JB.00635-18 (2019).
Schmitz, K. R., Carney, D. W., Sello, J. K. & Sauer, R. T. Crystal structure of Mycobacterium tuberculosis ClpP1P2 suggests a model for peptidase activation by AAA+ partner binding and substrate delivery. Proc Natl Acad Sci USA 111, E4587–4595, https://doi.org/10.1073/pnas.1417120111 (2014).
Leodolter, J., Warweg, J. & Weber-Ban, E. The Mycobacterium tuberculosis ClpP1P2 Protease Interacts Asymmetrically with Its ATPase Partners ClpX and ClpC1. PloS one 10, e0125345, https://doi.org/10.1371/journal.pone.0125345 (2015).
Personne, Y., Brown, A. C., Schuessler, D. L. & Parish, T. Mycobacterium tuberculosis ClpP proteases are co-transcribed but exhibit different substrate specificities. PloS one 8, e60228, https://doi.org/10.1371/journal.pone.0060228 (2013).
Kang, S. G., Maurizi, M. R., Thompson, M., Mueser, T. & Ahvazi, B. Crystallography and mutagenesis point to an essential role for the N-terminus of human mitochondrial ClpP. J Struct Biol 148, 338–352, https://doi.org/10.1016/j.jsb.2004.07.004 (2004).
Benaroudj, N., Raynal, B., Miot, M. & Ortiz-Lombardia, M. Assembly and proteolytic processing of mycobacterial ClpP1 and ClpP2. BMC Biochem 12, 61, https://doi.org/10.1186/1471-2091-12-61 (2011).
Brodie, E. J., Zhan, H., Saiyed, T., Truscott, K. N. & Dougan, D. A. Perrault syndrome type 3 caused by diverse molecular defects in CLPP. Sci Rep 8, 12862, https://doi.org/10.1038/s41598-018-30311-1 (2018).
Gersch, M., List, A., Groll, M. & Sieber, S. A. Insights into structural network responsible for oligomerization and activity of bacterial virulence regulator caseinolytic protease P (ClpP) protein. The Journal of biological chemistry 287, 9484–9494, https://doi.org/10.1074/jbc.M111.336222 (2012).
Schmitz, K. R. & Sauer, R. T. Substrate delivery by the AAA+ ClpX and ClpC1 unfoldases activates the mycobacterial ClpP1P2 peptidase. Molecular microbiology 93, 617–628, https://doi.org/10.1111/mmi.12694 (2014).
Ingvarsson, H. et al. Insights into the inter-ring plasticity of caseinolytic proteases from the X-ray structure of Mycobacterium tuberculosis ClpP1. Acta Crystallogr D Biol Crystallogr 63, 249–259, https://doi.org/10.1107/S0907444906050530 (2007).
Sprangers, R., Gribun, A., Hwang, P. M., Houry, W. A. & Kay, L. E. Quantitative NMR spectroscopy of supramolecular complexes: dynamic side pores in ClpP are important for product release. Proc Natl Acad Sci USA 102, 16678–16683, https://doi.org/10.1073/pnas.0507370102 (2005).
Kim, J. et al. Structures, Functions, and Interactions of ClpT1 and ClpT2 in the Clp Protease System of Arabidopsis Chloroplasts. Plant Cell 27, 1477–1496, https://doi.org/10.1105/tpc.15.00106 (2015).
Bai, L. et al. Structural analysis of the dodecameric proteasome activator PafE in Mycobacterium tuberculosis. Proc Natl Acad Sci U S A 113, E1983–1992, https://doi.org/10.1073/pnas.1512094113 (2016).
Hu, K. et al. Proteasome substrate capture and gate opening by the accessory factor PafE from Mycobacterium tuberculosis. The Journal of biological chemistry 293, 4713–4723, https://doi.org/10.1074/jbc.RA117.001471 (2018).
Delley, C. L. et al. Bacterial proteasome activator bpa (rv3780) is a novel ring-shaped interactor of the mycobacterial proteasome. PloS one 9, e114348, https://doi.org/10.1371/journal.pone.0114348 (2014).
Muller, A. U. & Weber-Ban, E. The Bacterial Proteasome at the Core of Diverse Degradation Pathways. Front Mol Biosci 6, 23, https://doi.org/10.3389/fmolb.2019.00023 (2019).
Truscott, K. N. et al. A presequence- and voltage-sensitive channel of the mitochondrial preprotein translocase formed by Tim23. Nat Struct Biol 8, 1074–1082, https://doi.org/10.1038/nsb726 (2001).
Catanzariti, A. M., Soboleva, T. A., Jans, D. A., Board, P. G. & Baker, R. T. An efficient system for high-level expression and easy purification of authentic recombinant proteins. Protein Sci 13, 1331–1339, https://doi.org/10.1110/ps.04618904 (2004).
Vasudevan, D., Rao, S. P. & Noble, C. G. Structural basis of mycobacterial inhibition by cyclomarin A. The Journal of biological chemistry 288, 30883–30891, https://doi.org/10.1074/jbc.M113.493767 (2013).
Dougan, D. A., Reid, B. G., Horwich, A. L. & Bukau, B. ClpS, a substrate modulator of the ClpAP machine. Mol Cell 9, 673–683 (2002).
Dougan, D. A., Weber-Ban, E. & Bukau, B. Targeted delivery of an ssrA-tagged substrate by the adaptor protein SspB to its cognate AAA+ protein ClpX. Mol Cell 12, 373–380 (2003).
Ninnis, R. L., Spall, S. K., Talbo, G. H., Truscott, K. N. & Dougan, D. A. Modification of PATase by L/F-transferase generates a ClpS-dependent N-end rule substrate in Escherichia coli. EMBO J 28, 1732–1744, https://doi.org/10.1038/emboj.2009.134 (2009).
Lowth, B. R. et al. Substrate recognition and processing by a Walker B mutant of the human mitochondrial AAA+ protein CLPX. J Struct Biol 179, 193–201, https://doi.org/10.1016/j.jsb.2012.06.001 (2012).
Schagger, H. & von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166, 368–379 (1987).
Otwinowski, Z. & Minor, W. In Methods in Enzymology Vol. Volume 276, 307–326 (Academic Press, 1997).
Long, F., Vagin, A. A., Young, P. & Murshudov, G. N. BALBES: a molecular-replacement pipeline. Acta Crystallogr D Biol Crystallogr 64, 125–132, https://doi.org/10.1107/S0907444907050172 (2008).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr D Biol Crystallogr 60, 2126–2132, https://doi.org/10.1107/S0907444904019158 (2004).
Afonine, P. V. et al. Joint X-ray and neutron refinement with phenix.refine. Acta Crystallogr D Biol Crystallogr 66, 1153–1163, https://doi.org/10.1107/S0907444910026582 (2010).
Painter, J. & Merritt, E. A. Optimal description of a protein structure in terms of multiple groups undergoing TLS motion. Acta Crystallogr D Biol Crystallogr 62, 439–450, https://doi.org/10.1107/S0907444906005270 (2006).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr D Biol Crystallogr 66, 12–21, https://doi.org/10.1107/S0907444909042073 (2010).
Acknowledgements
D.A.D. was supported by an Australian Research Council (ARC) Australian Research Fellowship (DP110103936) and B.H. was supported by an ARC Future Fellowship (FT130100580). The work was supported by a Victoria India Doctoral Scholarship (to J.N.), La Trobe University Postgraduate Research Scholarships (to J.E.Z. and A.A.T.) and La Trobe University Full Fee Research Scholarships (to J.N and A.A.T.). We thank Dr. Pierre Genevaux (Université de Toulouse, France) for providing pET15(b)/MtbclpX, Dr. Dileep Vasudevan (Institute of Life Sciences, Bhubaneswar, India) for providing pET30(a)/MtbclpC1, Nilushika Silva for technical assistance in cloning of MsmClpP1in and MsmClpP2in. Crystals were analysed at the Australian Synchrotron. The atomic coordinates and structure factors for 6PBU have been deposited in the Protein Data Bank (http://wwpdb.org/). Preparation of a draft manuscript was supported by a “write-up” award from the Understanding Disease Research Focus Area at La Trobe University to J.N.
Author information
Authors and Affiliations
Contributions
The experiments were conceived and designed by D.A.D., B.H. and K.N.T., performed by J.N., J.P., J.Z., A.A.T. and B.H. Data analysis was carried out by J.N., B.H., J.P., K.N.T. and D.A.D. The paper was written by D.A.D., J.N., B.H., J.P. and K.N.T. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nagpal, J., Paxman, J.J., Zammit, J.E. et al. Molecular and structural insights into an asymmetric proteolytic complex (ClpP1P2) from Mycobacterium smegmatis. Sci Rep 9, 18019 (2019). https://doi.org/10.1038/s41598-019-53736-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53736-8
- Springer Nature Limited
This article is cited by
-
Bioprospecting the microbiome of Red Sea Atlantis II brine pool for peptidases and biosynthetic genes with promising antibacterial activity
Microbial Cell Factories (2022)
-
ClpP inhibitors are produced by a widespread family of bacterial gene clusters
Nature Microbiology (2022)
-
Polymerase delta-interacting protein 38 (PDIP38) modulates the stability and activity of the mitochondrial AAA+ protease CLPXP
Communications Biology (2020)