Abstract
The regulation of aquatic carbon cycles by temperature is a significant uncertainty in our understanding of how watersheds will respond to climate change. Aquatic ecosystems transport substantial quantities of carbon to the atmosphere and ocean, yet we have limited understanding of how temperature modifies aquatic ecosystem metabolic processes and contributions to carbon cycles at watershed to global scales. We propose that geomorphology controls the distribution and quality of organic material that forms the metabolic base of aquatic ecosystems, thereby controlling the response of aquatic ecosystem metabolism to temperature across landscapes. Across 23 streams and four years during summer baseflow, we estimated variation in the temperature sensitivity of ecosystem respiration (R) among streams draining watersheds with different geomorphic characteristics across a boreal river basin. We found that geomorphic features imposed strong controls on temperature sensitivity; R in streams draining flat watersheds was up to six times more temperature sensitive than streams draining steeper watersheds. Further, our results show that this association between watershed geomorphology and temperature sensitivity of R was linked to the carbon quality of substrates that changed systematically across the geomorphic gradient. This suggests that geomorphology will control how carbon is transported, stored, and incorporated into river food webs as the climate warms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Aquatic ecosystems play an important role in watershed carbon (C) cycles and process a substantial amount of C from the terrestrial environment1,2,3,4; however, there are critical uncertainties in our understanding of how aquatic ecosystem metabolism will respond to climate induced shifts in temperature and hydrology. Temperature is a primary control of organism metabolism so changes to aquatic thermal regimes will influence how C is processed through aquatic food webs. Furthermore, climate-driven changes in terrestrial productivity and litter decomposition rates will likely affect the delivery, quantity, and quality of terrestrial organic matter that supports aquatic food webs. Shifting precipitation patterns have already altered hydrological regimes5,6. This can further impact aquatic-terrestrial linkages through changes to dominant water sources (e.g., snow vs. rain), timing of seasonal flows6, soil flow paths7, and drought or flood conditions, which all affect the delivery of terrestrial-derived organic matter to aquatic ecosystems8. Therefore, not only will climate-driven temperature and hydrological change influence thermal characteristics of aquatic systems, but also the quantity and quality of organic matter that fuels ecosystem metabolism and supports recipient food webs.
One expectation for how ecosystem metabolic processes respond to changing temperature has been formalized as the Metabolic Theory of Ecology9,10 (MTE). MTE postulates that temperature dependence of most biochemical processes, including those involved in ecosystem C cycling, is described by a universal relationship due to the consistent biochemistry involved in metabolism across organisms and ecosystems10,11,12. This relationship can be characterized by Arrhenius kinetics, in which temperature sensitivity of metabolic rates is expressed as enzyme activation energies (Et) according to Eq. (1):
where R (T) is the rate of respiration at temperature T (°K). Et is the activation energy of respiration in eV and describes the slope of the of R with temperature, i.e., the temperature dependence of respiration. Kb is the Boltzmann constant (8.62 × 10-5 J °K−1), and Rref is respiration at a standard temperature (Tref).
It remains unclear whether the temperature response of important C cycling processes, such as R, is universal13 or whether there are meaningful departures from this value as a result of differences in resource supply14,15, resource quality16,17, nutrient availability18,19,20 or community composition21. For example, there may be fundamental differences in how aquatic and terrestrial ecosystem C cycles respond to warming temperatures, which poses challenges for applying terrestrial-derived relationships to the temperature scaling of R in aquatic ecosystems. A global meta-analysis showed that the temperature sensitivity of aquatic R was higher and more variable than terrestrial R13, possibly reflecting greater variation in the quality of organic substrates that support aquatic R. Further, a recent study of the temperature sensitivity of metabolic rates in streams across the US showed that the greater sensitivity of R to temperature than gross primary production (GPP) could decrease net ecosystem production (NEP) in streams as global temperatures increase22. This higher and more variable temperature response of R has been supported by several studies in aquatic ecosystems23,24,25,26,27, but reasons for this variation are not fully understood.
Aquatic R tends to be supported by C substrates that vary more widely in their bioavailability than those in terrestrial ecosystems8,13,28,29,30, which could affect not only the gross rate of R but also how it responds to changes in temperature. Theoretical and experimental work in terrestrial systems has shown that breakdown rates of recalcitrant C sources are often more sensitive to increasing temperature than the breakdown of more labile C16,31, a result supported by microcosm experiments in aquatic systems27,28. While terrestrial R is often tightly coupled to labile substrates from gross primary production, aquatic R tends to be more often fueled by terrestrial organic matter8,29, which varies more in its lability than the high-quality substrates produced by aquatic autotrophs16,32. This difference in temperature dependence of R between terrestrial and aquatic systems could reflect greater variation in the quality and timing of C sources in aquatic systems, which are typically mediated by watershed scale controls on aquatic-terrestrial connectivity33, hydrology34, and light35,36,37.
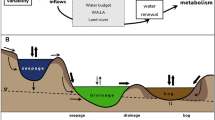
Geomorphic features of watersheds may, therefore, ultimately control the temperature dependence of aquatic ecosystem metabolism because they constrain both the thermal regime38 and the quantity and quality of organic matter loaded to aquatic ecosystems. Specifically, features such as watershed slope, valley form, and network structure39 control hydrological interaction with floodplains and hyporheic zones, in-channel storage40,water residence time, and thermal regimes38 that in turn influence carbon retention, opportunities for microbial decomposition, and connectivity with floodplain soils and wetlands39,40,41,42. Further, other physical features of watersheds, such as their size, have been shown to alter the production and temperature response of organic matter by autotrophs within river networks through controlling light availability37,43,44. Therefore, by affecting how controlling factors are arranged on the riverine landscape and affecting processing time, geomorphic features should also constrain variation in the response of R to temperature across river networks25.
We estimated how growing season aquatic R responded to variation in water temperature among tributaries of a boreal river basin in southwest Alaska that fall along a distinct geomorphic gradient. We analyzed daily dynamics in in situ dissolved oxygen concentrations with a statistical process model45 to estimate rates of R and its sensitivity to changes in water temperature. This approach enabled a whole-stream assessment of how R responds to temperature and avoids some of the scaling limitations imposed by mesocosm experiments, assumptions of cross-system studies, or experiments involving artificial warming, methods that have been employed in most previous assessments of the temperature response of R24,26,27. In addition, we also evaluated support for a model that partitioned R into a base component (Rb) that reflects “background” C pools that do not show diel changes in availability, and a primary production-derived component45,46 (Rp) that responds to hourly changes in primary production. This model allowed us to estimate an integrated temperature sensitivity (Et) of Rb and Rp (Eb and Ep, respectively), which have been shown to reflect diel change in the quality of C substrates45.
We hypothesized that Et would vary predictably with watershed geomorphic features such as slope, size, and elevation through their influence on primary production, accumulation and residence time of carbon in watersheds, and aquatic-terrestrial coupling as reflected in the quantity and quality of organic substrates available to support aquatic R.
Results and Discussion
We found wide variation in the temperature sensitivity (Et) of R across 23 streams and four years within the Wood River basin in southwest Alaska (Fig. 1). Posterior estimates of Et values were consistently well constrained and varied between 0 and 1.94 (average = 0.33 eV, S.D = 0.33; Q10, 5–15 °C ~1.7, Supplementary Tables 1 and 7). Our analyses show that a model allowing for a variable, stream-specific response of R to temperature performed substantially better in most cases than a model that set the temperature sensitivity at a single theoretical value (Supplementary Table 7). On average, R estimated at the ecosystem scale, was less sensitive to temperature than predicted by MTE11 and mesocosm experiments in this system27 (0.71 eV), but fell within the range of short-term temperature dependence for rivers estimated by other whole-stream studies (Song et al.22: median = 0.70 eV; 0.4–8.68 eV; Yvon-Durocher et al.13: median 0.53, 0.24–0.89 eV).
Temperature sensitivity corresponds with watershed slope. Left panel: Map of sites in Wood River basin. Points represent streams included in study and size of point is scaled to the magnitude of estimated temperature sensitivity (Et). Map of state of Alaska shows location of study area. Map was generated using the World Imagery dataset from Esri ArcMap 10.5.0.6491. Sources of satellite imagery cited on image include: “Esri, DigitalGlobe, GeoEye, Earthstar Geographic, USDA, USGS, AeroGrid, ICN, and the GIS user community”. Right panel: (A) Distribution of estimated Et values - Orange represents streams included in current study and green represents streams for which we have watershed slope data in the broader Wood River Basin and could estimate an Et value, (B) The relationship of temperature sensitivity with watershed slope (Rc2 = 0.46). Size of points scale with amount of rain over the course of and one day prior to metabolism measurements.
Simulations showed that our median posterior estimates of Eb were robust to changes in the range of diel temperature in the model (Supplementary Methods, Supplementary Fig. 4) and that Eb could be estimated independently from k20, the parameter governing stream reaeration rate (Supplementary Fig. 6, Supplementary Table 8). Specifically, simulations showed that posteriors of Eb estimates were well constrained when diel temperature variation was more than 1.0 °C, but temperature variation of less than 0.5 °C per day substantially increased uncertainty as indicated by wider posteriors. This was especially true for simulations in which we set temperature sensitivity to be low (0.32 vs 1.0 eV, Supplementary Fig. 4). However, streams in our study had average diel temperature ranges from 0.9 °C (2 streams) to 5.8 °C (mean = 2.9 °C per day), suggesting that our model was not biased under normal thermal conditions or even during storm events when diel temperature variation was reduced (1.6–1.91 °C). In addition, we evaluated whether our estimates of Eb were biased by or correlated with stream reaeration rates. For example, we may expect that streams draining steep watersheds have lower diel variation in temperature as a result of shorter residence time and would also have high reaeration rates, which decreases diel changes in O2 and, thus, could bias our estimates of Et since it also depends on the daily range of those two variables. However, we found no systematic bias in the median or variation of Eb estimates with estimates of k20 (Supplementary Fig. 6, Supplementary Table 8).
Et was clearly associated with watershed geomorphic features across all years of the study (Fig. 1, Supplementary Fig. 5). We used a linear mixed modeling approach to quantify how aspects of watershed geomorphology influenced Et and to account for among-year variation in Et, and found that the best geomorphic predictor of Et was watershed slope (R2c = 0.46, Fig. 1; Supplementary Table 2, Supplementary Methods). Consistent with previous experimental results in this system27, we found that R was considerably more sensitive to temperature in streams draining flatter watersheds than in streams draining steeper watersheds and not as responsive to other geomorphic features such as watershed area or substrate size (Supplementary Table 2). Watershed slope likely influenced Et in these streams through its effects on the quantity and quality of C on the landscape47. In general, steep watersheds tend to accumulate little C in soils, wetlands or stream channels, while flatter watersheds accumulate C in soils and peat, within stream channels in slow-moving transient storage zones, and tend to be more hydrologically connected to carbon and nutrient sources in their floodplains39,40,48. There is also a tendency for R and food webs in steeper watersheds in this system to be supported more by autochthonous production of organic matter45,48 which is typically more labile than the terrestrially-derived organic matter loaded from flat watersheds dominated by peat bogs and deep terrestrial soils.
Our results support the expectation that watershed slope controlled the characteristics of organic matter in streams. We found that DOC concentration was highest and its quality was lowest (i.e., had the highest C:N ratios) in streams draining flat watersheds (Fig. 2; Supplementary Fig. 8). Furthermore, regression models including C:N as a predictor of temperature sensitivity (Et) among streams were better supported by the data than models ignoring substrate quality (Supplementary Tables 3 and 4, Fig. 2). Other measures of C substrate quantity and quality (DOC, C:N, chlorophyll a) were also included in the best models for individual years (Supplementary Tables 3 and 4). In 2013, to supplement C:N as a metric of substrate quality, we also measured SUVA (specific ultra-violet absorbance), an index of aromatic content of organic matter49. SUVA was positively correlated with C:N ratio (r = 0.48, n = 11), indicating that high C:N ratios were indeed related to the presence of lower quality C substrates in streams draining flatter watersheds. Average summer temperature was also included in some of the top models, which suggests that either higher temperatures may increase decomposition rates and lead to more recalcitrant C pools or that microbes acclimated to different thermal regimes have distinct thermal responses50. These results suggest that although there may be some underlying universal physiological temperature response11, geomorphic context influences how ecosystem metabolism responds to rising temperatures through impacting C pools and stream temperature.
While watershed slope was shown to be an important control on Et, there remained additional unexplained variation (~50%). Several other factors could explain some of this variation, including more local scale geomorphic features that could alter residence time and deposition of organic C, seasonal succession of benthic communities, the extent of watershed forest cover and its species composition, the biomass of the organisms performing R10,17, and the history of hydrological events prior to the time we measured metabolism. Our results suggest that precipitation regimes affected temperature dependence of R in these streams, and that watershed geomorphology may have modified the response. Processing of organic carbon in streams and rivers varies with precipitation and hydrology patterns that influence the source, residence time, and decomposition kinetics of DOM51. For example, during periods of increased precipitation there would be increased hydraulic connectivity of streams with the surrounding riparian environment altering the relative amount and character of terrestrial C sources available to stream decomposers52 and the size of the active soil layer contributing to stream DOC fluxes7,53. Further, we expect that watershed and floodplain morphology mediate the degree to which streams become connected to floodplain organic matter sources during high flow events41,54,55. Our data suggest some of the variation in Et that we observed was due to precipitation, and the degree to which this was important varied across the watershed slope gradient Specifically, we found that rainfall during metabolism measurements (plus one-day prior) decreased Et in steeper streams, but may have slightly increased Et in flatter watersheds (Fig. 1, Supplementary Table 5).
One possible explanation for this is that precipitation events altered the amount and quality of organic matter in streams51, but to a larger degree in flatter watersheds. Work in this basin has demonstrated that DIC and food webs reflect increasing terrestrial inputs in response to storms in flatter watersheds48, which could increase the short-term temperature sensitivity of R, an expectation supported by our data. First, we found that rainfall prior to collection of DOC and C:N samples increased C:N slightly more in flat streams than in steep streams (Supplementary Fig. 1) and regression models suggested that an interaction between slope and precipitation for DOC and C:N was important (Supplementary Table 5). Second, we more directly evaluated the influence of a large precipitation event on temperature sensitivity, and the quantity (DOC) and quality (C:N) of C in streams draining a flat (2.6 degrees) and a steep (25.6 degrees) watershed. We found that Et, DOC, and C:N all increased in both streams immediately following a 64 mm rain event, but to a larger degree in the flatter stream (Fig. 3, Table 1). Taken together, these results indicate that watershed geomorphology (therefore the types and amount of organic matter on the landscape) and hydrology (the degree to which streams are connected to fringing riparian areas and hyporheic flow56) interact to alter carbon inputs and the sensitivity of its breakdown to temperature in streams. Whether this interaction is robust, and if it indeed reflects loading of DOM with differing lability or other factors such as bacterial community shifts, remains to be determined. Our metabolism deployments were relatively short, which may have also led to unexplained residual variation in these relationships if stream rates were highly variable within the summer base flow period during which we measured them. However, previous work in this system showed relatively consistent conditions and metabolic rates during the period over which we measured metabolism (between snow melt and prior to the arrival of spawning salmon57) and we observed consistent results among years for individual streams (Supplementary Fig. 5, Supplementary Table 7). In any case, further work is needed to test these hypotheses and assess the temporal and spatial scales over which these interactions occur in river networks.
Interestingly, our average estimated Et values were lower than expected from mesocosm experiments in this system27 (Supplementary Fig. 3), but similar to values estimated in other stream ecosystems using whole-stream approaches22. There are several possible explanations for these differences. First, we used in situ diel oxygen and temperature changes rather than experimental mesocosm warming to estimate Et, which reflects respiration of the whole stream ecosystem. Mesocosm experiments by nature include only a portion of the benthos, whereas ecosystem metabolism measurements represent respiration occurring in the stream channel as well as in the hyporheic and fringing riparian zones. If diel variation in temperature and oxygen is lower in these subsurface areas than in the stream channel58, as we have seen in this system, this could lower the ecosystem-scale temperature sensitivity relative to what would be measured for surface sediments that experience larger daily swings in temperature and oxygen59. In addition, the magnitude and concentration of groundwater flux has been shown to influence rates of R60 and may also impact estimates of Et. Because these are gravel bed streams with shallow flow paths resuting from a clay lens that restricts deep groundwater flow (D.E. Schindler, pers. observation), we have assumed groundwater input is negligible and have not accounted for it within our modeling framework. How changes in groundwater translate into changes in temperature sensitivity is not known but is likely an additional important avenue for research. Second, it is possible that more labile C sources from autocthonous sources become limiting over the duration of mesocosm experiments, thereby increasing the temperature response of R through altering substrate quality. Few studies have used a whole ecosystem approach to estimating temperature dependence of aquatic R23,24,36, but the one study that used an approach similar to ours (i.e., modeling temperature sensitivity responses to local temperature fluctuation) found an even greater range of values than we observed within boreal forest streams and across biomes in their study22 (0.4–8.68 eV). Third, our model only accounted for diel variation in photosynthetic biomass pools (associated with Rp), not microbial or allochthonous biomass pools. We assumed the latter remained constant over a day (i.e., “background R”45), which could resulted in observed deviation from expectations of MTE10. In any case, this in situ approach produces a more accurate estimate of whole-stream temperature sensitivity than mesocosm experiments, and these differences further emphasize the need to assess and constrain these responses at this scale.
It is also important to consider how the short-term temperature response we measured here, and which has typically been measured in other aquatic ecosystem studies, translates to a longer-term response and fits into our understanding of how aquatic ecosystem metabolic rates will respond to global change. While a substantial body of work in soils and terrestrial ecosystems explores how short-term warming responses inform our understanding of long-term effects of climate warming on R, much less work exists in aquatic ecosystems13. Ours and other studies report that the temperature sensitivity of R is more variable among aquatic than terrestrial ecosystems13, which suggests that predicting long-term warming responses of aquatic metabolism may depend less on understanding their “inherent” temperature response than on understanding how dynamic biomass and resource pools that affect the temperature response respond to long-term warming trends. Further, although the temperature dependence we have estimated here represents the response to diel temperature change, the modeling framework we use here provides a means to use increasingly available metabolic data captured across seasons and years61,62 to understand the temperature response of aquatic R over longer time scales. Last, recent work has suggested that the temperature dependence of GPP may also be highly variable and higher than predicted by MTE, which can impact net ecosystem production and emissions as climate warms22,44. Here we modeled GPP as temperature invariant, but our results suggest that the temperature response of GPP may respond similarly to watershed physical features given their control on light and thermal regimes35,63. Further work is required to understand longer term temperature responses in aquatic systems, such as those that represent changes across seasons or years13, but our results take an initial step towards characterizing how the response of R to temperature may vary spatially across river networks.
In summary, our results suggest that physical features of watersheds provide a means with which to scale the thermal sensitivity of stream metabolic rates across river networks. We show that watershed geomorphology controls the basic drivers of aquatic R and its sensitivity to thermal variation (DOC, C:N, and stream temperature), which implies that basic descriptors such as watershed slope can be used to scale across more complex river basins. While our study was limited in both space (across a single river basin) and time (summer base flow), our modeling approach paired with increasingly available metabolism62 as well as GIS or remotely sensed data on watershed geomorphic features and vegetation can be easily expanded temporally and spatially to generate estimates of how carbon processing will respond to increasing temperatures at the watershed scale. How these relationships with geomorphology vary across seasonal changes in temperature, at longer time scales, or in developed regions where geomorphic characteristics have more subtle effects on C storage and quality64 remains to be quantified, but will be a fruitful area for future research. In the case of our study of a boreal river system, responses during the growing season may well be representative of most of the annual scale respiration given low temperatures and long periods of ice and snow cover.
Climate change has broad implications for thermal and hydrological regimes in stream ecosystems, and is already altering aquatic-terrestrial linkages through melting permafrost65, and “browning”66,67. Our study highlights how watershed geomorphic features control organic matter transport to aquatic ecosystems and influence how aquatic ER responds to variation in temperature. These results can help guide the development of a framework for scaling climate and land use64 driven changes to aquatic carbon dynamics from single streams to river basins based on simple geomorphic characteristics of landscapes.
Methods
Field site
This study was conducted in 23 2nd–4th order streams of the Wood River basin (59°20’N, 158°40’W) in the Bristol Bay region of southwest Alaska during the summers of 2010–2013. The majority of the Wood River basin lies within the Wood-Tikchik State Park and contains five large, interconnected lakes fed by numerous small streams that drain through the Wood River into Bristol Bay. This region is one of the fastest warming on Earth (Maurer et al. 2007), and is characterized by extensive peatlands with the potential to release substantial amounts of C to the atmosphere with ongoing climate warming. The Wood River basin has considerable variation in geomorphic conditions among sub-watersheds that produces a distinct gradient in watershed slope and elevation among streams38 (Supplementary Table 6).
Metabolism & temperature sensitivity
Water temperature and [O2] were recorded at a single station near the outflow of each stream for 3–8 days each year during the summer base flow period at 10-min intervals with a YSI 6600 V2 sonde equipped with an optical dissolved oxygen (ROx) sensor. Sensor [O2] measurements were calibrated in an oxygen-saturated bath, and cross-checked with [O2] measurements on a subset (n = 10 streams, 3 replicates per stream) as determined by Winkler titrations at the time of sonde deployment or retrieval. Irradiance at the water surface was measured directly in 10-min intervals at one of two stations no more than 30 km away with a HOBO universal weather station PAR sensor (Onset Computer Corp., Bourne, MA, USA).
Mean width and depth were measured at 10 transects over 200 meters upstream of the sensor (every ~20 m). At each transect we measured width using a laser range finder (Laser Technology Incorporated, Englewood, CO) and measured depth at five points across the stream channel. All depth measurements were averaged to generate a mean depth value for each stream. Our objective was to assess spatial variation across multiple streams during the short summer baseflow period between snow melt and the arrival of spawning salmon in late July/early August each year. Therefore, we rotated multiple sondes around 11–16 streams each year (23 unique streams in the four-year dataset), which meant that we were only able to capture a short period within this time frame with each deployment. We aimed to deploy sondes for at least three consecutive days, but in some cases storm events also interrupted our ability to estimate metabolism and estimates reflect shorter time periods. Across years, all metabolism measurements were taken within the same time frame (late June to early August) and during a similar time period for each individual stream. Further, the order and timing of these deployments were random across the watershed slope gradient, thereby removing potential bias between timing and geomorphic drivers (Supplementary Fig. 7).
We estimated the metabolic parameters and temperature sensitivity of ecosystem respiration by fitting a process model of ecosystem metabolism to daily changes in stream dissolved oxygen concentration, water temperature, and irradiance data68, as developed and reported by Jankowski69. We fit the model to these data to estimate rates of gross primary production (GPP), ecosystem respiration (ER), and gas exchange45,68,69. The model simulates changes in stream oxygen concentrations through estimating light-dependent oxygen production via photosynthesis, temperature-dependent oxygen consumption via respiration, and oxygen exchange between the stream and the atmosphere dependent on the gas transfer velocity and concentration gradient:
where [O2] is the dissolved oxygen concentration (mg m−3), [O2,sat] is the dissolved oxygen concentration at atmospheric equilibrium, R is the instantaneous respiration rate, P is the instantaneous rate of photosynthesis (both in units of mg O2 m−2 h−1), and D is the average depth (m). The first term in the above equation is the net effect of gas exchange, which is the gas transfer velocity, k, times the O2 concentration gradient.
To estimate temperature sensitivity of R, we modified the respiration portion of the equation based on Arrhenius kinetics and to reflect respiration derived from GPP. We used the model as developed by Jankowski69 and Schindler et al.45 that allows for variation in the substrate supporting heterotrophic respiration, which could influence the overall temperature sensitivity of oxygen consumption rates27,70. This model considers the potential for two substrate pools to support R: respiration of ambient carbon substrates or “background R” (Rb, Eq. 1), and respiration of a pool of labile organic matter produced via recent photosynthesis46 (Rp;).
Rp was modeled as an exponentially declining fraction of photosynthesis that occurred in 20 previous time steps, formulated as:
According to this formulation we assume that carbon produced by some portion of n previous time steps of photosynthesis was directly consumed and respired by heterotrophs. The slope of an exponential decay function (β) describes the rate at which photosynthetically-derived organic matter is metabolized or washed out of the system. The greater the value of β, the lower the influence of photosynthetically-derived carbon on R.
Rb and Rp were considered to have different sensitivities to temperature. We set the E value for Rp at the theoretical E value for photosynthesis, Ep = 0.32 eV (Allen et al. 2005) and then estimated Eb for Rb from the data. Where the model converged on a clearly defined β, (i.e., there was strong evidence for two-sources of respiration, Supplementary Table 7) we use a model-integrated value of E, Et, which accounted for the proportional contribution of Rp and Rb to the overall estimate of R as follows:
where pb and pp (equal to 1-pb) are the proportions of Rb and Rp integrated over a 24-hour period. In the cases where the model did not converge on β, we assumed that there was no evidence for a two-stage R (Rb and Rp) and estimated Et with a single source model (Rb only, Eb = Et). We also examined whether estimating temperature sensitivity using a model that considered respiration rates of either one or two pools of carbon (daily change in respiration as a function of carbon pools45). This typically did not change the estimate for Eb. When it did, however, the estimate of Eb increased slightly since Ep captured some of the temperature dependency of R. In all cases, however, we report a model-integrated value of Et which was weighted according the proportional contribution from Rb and Rp.
Environmental data
To evaluate the influence of environmental characteristics on Et, we collected samples at the time of sonde deployment to measure stream chemical and physical conditions. Water samples for total nitrogen (TN), total phosphorus (TP), dissolved organic C (DOC), total dissolved nitrogen (TDN) and their C:N ratiowere taken from streams at the time of sonde deployment. Seasonal water temperature data used in regressions were taken from Lisi et al. who measured stream temperatures at the mouth of each stream at 60–90-min intervals throughout each summer in our study from June – September. Samples for the analysis of specific ultraviolet absorbance (SUVA) were taken once from streams in July 2013. To measure discharge, we either measured flow and depth at a cross-section of the stream using a Swoffer flow meter (Swoffer Instruments, Inc; Federal Way, WA) at the start and end of each deployment, where we had continuous water level loggers (Hobo U2O-001-04; Onset Computer Corp, Bourne, MA) we used stream-specific rating curves to convert water level to discharge. Watershed geomorphic characteristics for streams in this study were taken from Lisi et al.38 who demonstrated that watershed slope, elevation, stream particle size, watershed area, and lake area explained 84% of geomorphic variation among Wood River streams. Watershed slope and elevation captured the majority of this variation. However, they were correlated with one another, so they were tested individually in this study (Supplementary Table 2).
Data analysis
We evaluated the influence of watershed geomorphic features and stream environmental conditions on the temperature sensitivity of stream respiration by regressing Et estimated from the metabolism model described above against watershed geomorphic and stream chemistry variables. We tested the controls on Et across all years in our dataset (2010–2013) and within each year independently (Supplementary Tables 2–4). When evaluating environmental and geomorphic controls across all years, we used a mixed effects modeling framework to account for repeated measurements in individual streams and across years by including a random effect of year and stream on the intercept as below:
We considered several alternative models to explain variation in Et among streams in this river basin including several stream-specific environmental parameters as fixed effects (DOC, C:N, TN, TP, chlorophyll a, average temperature, seasonal and daily temperature range, discharge) to evaluate how these influenced the temperature sensitivity of R. Models to compare fixed effects were fit by maximum likelihood and compared with AICc. For year-specific assessment of environmental controls on Et, we used step-wise multiple regressions and model selection by AICc to compare the influence of all of the same environmental parameters listed above on Et. We report R2 values for the fixed and random components of these models according to Nakagawa and Schielzeth71. We used a similar mixed model approach to evaluate the influence of hydrological changes on temperature sensitivity by testing the effect cumulative rainfall during the period of metabolism measurements and one day prior and its interaction with watershed slope.
We used the lme4 package72 to do the mixed effects analysis and all analyses were done in R (R Core Team 2014).
Change history
27 February 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Kling, G. W., Kipphut, G. W. & Miller, M. C. Lakes and streams as gas conduits to the atmosphere: Implications for tundra carbon budgets. Science 251, 298–301 (1991).
Richey, J. E., Melack, J. M., Aufdenkampe, A. K., Ballester, V. M. & Hess, L. L. Outgassing from Amazonian rivers and wetlands as large tropical source of atmospheric CO2. Nature 416, 617–620 (2002).
Raymond, P. A. et al. Global carbon dioxide emissions from inland waters. Nature 503, 355–359 (2013).
Cole, J. J. et al. Plumbing the global carbon cycle: integrating inland waters into the terrestrial carbon budget. Ecosystems 10, 171–184 (2007).
Stewart, I. T. Changes in snowpack and snowmelt runoff for key mountain regions. Hydrol. Process. 23, 78–94 (2009).
Berghuijs, W. R., Woods, Ra & Hrachowitz, M. A precipitation shift from snow towards rain leads to a decrease in streamflow. Nat. Clim. Change 4, 583–586 (2014).
Tank, S. E., Fellman, J. B., Hood, E. & Kritzberg, E. S. Beyond respiration: Controls on lateral carbon fluxes across the terrestrial-aquatic interface. Limnol. Oceanogr. Lett. 3, 76–88 (2018).
Drake, T. W., Raymond, P. A. & Spencer, R. G. M. Terrestrial carbon inputs to inland waters: A current synthesis of estimates and uncertainty. Limnol. Oceanogr. Lett. 3, 132–142 (2018).
Brown, J., Gillooly, J., Allen, A., Savage, V. & West, G. Toward a metabolic theory of ecology. Ecology 85, 130–137 (2004).
Allen, A., Gillooly, J. & Brown, J. Linking the global carbon cycle to individual metabolism. Funct. Ecol. 202–213, https://doi.org/10.1111/j.1365-2435.2005.00952.x (2005).
Gillooly, J., Brown, J., West, G., VM, S. & EL, C. Effects of Size and Temperature on Metabolic Rate. Science 293, 2248–2251 (2001).
Enquist, B. J. et al. Scaling metabolism from organisms to ecosystems. Nature 423, 639–642 (2003).
Yvon-Durocher, G. et al. Reconciling the temperature dependence of respiration across timescales and ecosystem types. Nature 487, 472–476 (2012).
Fissore, C., Giardina, C. P. & Kolka, R. K. Reduced substrate supply limits the temperature response of soil organic carbon decomposition. Soil Biol. Biochem. 67, 306–311 (2013).
Crowther, T. W. et al. Quantifying global soil carbon losses in response to warming. Nature 540, 104–108 (2016).
Davidson, E. & Janssens, I. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440 (2006).
Cross, W. F., Hood, J. M., Benstead, J. P., Huryn, A. D. & Nelson, D. Interactions between temperature and nutrients across levels of ecological organization. Glob. Change Biol. 21, 1025–1040 (2015).
López-Urrutia, Á. & Morán, X. Resource limitation of bacterial production distorts the temperature dependence of oceanic carbon cycling. Ecology 88, 817–822 (2007).
Berggren, M., Laudon, H. & Jonsson, A. Nutrient constraints on metabolism affect the temperature regulation of aquatic bacterial growth efficiency. 894–902, https://doi.org/10.1007/s00248-010-9751-1 (2010)
Thomas, M. K. et al. Temperature-nutrient interactions exacerbate sensitivity to warming in phytoplankton. Glob. Change Biol. 23, 3269–3280 (2017).
Allison, S. D., Wallenstein, M. D. & Bradford, Ma Soil-carbon response to warming dependent on microbial physiology. Nat. Geosci. 3, 336–340 (2010).
Song, C. et al. Continental-scale decrease in net primary productivity in streams due to climate warming. Nat. Geosci. 11, 415–420 (2018).
Demars, B. O. L. et al. Temperature and the metabolic balance of streams. Freshw. Biol. 56, 1106–1121 (2011).
Perkins, D. M. et al. Consistent temperature dependence of respiration across ecosystems contrasting in thermal history. Glob. Change Biol. 18, 1300–1311 (2012).
AcuñA, V., Wolf, A., Uehlinger, U. & Tockner, K. Temperature dependence of stream benthic respiration in an Alpine river network under global warming. Freshw. Biol. 53, 2076–2088 (2008).
Welter, J. R. et al. Does N2-fixation amplify the temperature dependence of ecosystem metabolism? Ecology 96, 603–610 (2015).
Jankowski, K., Schindler, D. E. & Lisi, P. J. Temperature sensitivity of community respiration rates in streams is associated with watershed geomorphic features. Ecology 95, 2707–2714 (2014).
Jane, S. F. & Rose, K. C. Carbon quality regulates the temperature dependence of aquatic ecosystem respiration. Freshw. Biol., https://doi.org/10.1111/fwb.13168 (2018).
del Giorgio, P. A., Cole, J. J. & Cimbleris, A. Respiration rates in bacteria exceed phytoplankton production in unproductive aquatic systems. nature 385, 148–151 (1997).
del Giorgio, P. A. & Williams, P. Respiration in Aquatic Ecosystems. (Oxford University Press, 2005).
Wang, Q., Liu, S. & Tian, P. Carbon quality and soil microbial property control the latitudinal pattern in temperature sensitivity of soil microbial respiration across Chinese forest ecosystems. Glob. Change Biol. 24, 2841–2849 (2018).
Bosatta, E. & Ågren, G. Soil organic matter quality interpreted thermodynamically. Soil Biol. Biochem. 31, 1889–1891 (1999).
Vannote, R., Minshall, G., Cummins, K., Sedell, J. & Cushing, C. The river continuum concept. Can. J. Fish. Aquat. Sci. 37, 130–137 (1980).
Poole, G. C. Stream hydrogeomorphology as a physical science basis for advances in stream ecology. J. North Am. Benthol. Soc. 29, 12–25 (2010).
Finlay, J. C. et al. Light-mediated thresholds in stream-water nutrient composition in a river network. Ecology 92, 140–50 (2011).
Huryn, A. D., Benstead, J. P. & Parker, S. M. Seasonal changes in light availability modify the temperature dependence of ecosystem metabolism in an arctic stream. Ecology 95, 2826–2839 (2014).
Koenig, L. E. et al. Emergent productivity regimes of river networks. Limnol. Oceanogr. Lett., https://doi.org/10.1002/lol2.10115 (2019).
Lisi, P. J., Schindler, D. E., Bentley, K. T. & Pess, G. R. Association between geomorphic attributes of watersheds, water temperature, and salmon spawn timing in Alaskan streams. Geomorphology 185, 78–86 (2013).
Lininger, K. B., Wohl, E. & Rose, J. R. Geomorphic Controls on Floodplain Soil Organic Carbon in the Yukon Flats, Interior Alaska, From Reach to River Basin Scales. Water Resour. Res. 54, 1934–1951 (2018).
Battin, T. et al. Biophysical controls on organic carbon fluxes in fluvial networks. Nat. Geosci. 1, 95–100 (2008).
Montgomery, D. R. Process Domains and the River Continuum. J. Am. Water Resour. Assoc. 35, 397–410 (1999).
Aufdenkampe, A. K. et al. Riverine coupling of biogeochemical cycles between land, oceans, and atmosphere, https://doi.org/10.1890/100014 (2011).
Finlay, J. C. Stream size and human influences on ecosystem production in river networks. Ecosphere 2, art87 (2011).
Demars, B. O. L. et al. Impact of warming on CO2 emissions from streams countered by aquatic photosynthesis. Nat. Geosci. 9, 758–761 (2016).
Schindler, D. E., Jankowski, K., A’mar, Z. T. & Holtgrieve, G. W. Two-stage metabolism inferred from diel oxygen dynamics in aquatic ecosystems. Ecosphere 8, e01867 (2017).
Sadro, S., Holtgrieve, G. W., Solomon, C. T. & Koch, G. R. Widespread variability in overnight patterns of ecosystem respiration linked to gradients in dissolved organic matter, residence time, and productivity in a global set of lakes. Limnol. Oceanogr. 59, 1666–1678 (2014).
Catalán, N. et al. Biodegradation kinetics of dissolved organic matter chromatographic fractions in an intermittent river: Biodegradation Dynamics of DOM Fractions. J. Geophys. Res. Biogeosciences 122, 131–144 (2017).
Smits, A. P., Schindler, D. E., Holtgrieve, G. W., Jankowski, K. J. & French, D. W. Watershed geomorphology interacts with precipitation to influence the magnitude and source of CO2 emissions from Alaskan streams: Controls on Boreal Stream CO2 Emissions. J. Geophys. Res. Biogeosciences 122, 1903–1921 (2017).
Chin, Y., Aiken, G. R. & O’Loughlin, E. Molecular weight, polydispersity, and spectroscopic properties of aquatic humic substances. Environ. Sci. 28, 1853–1858 (1994).
Crowther, T. W. & Bradford, M. A. Thermal acclimation in widespread heterotrophic soil microbes. Ecol. Lett. 16, 469–477 (2013).
Raymond, P. A., Saiers, J. E. & Sobczak, W. V. Hydrological and biogeochemical controls on watershed dissolved organic matter transport: pulse-shunt concept. Ecology 97, 5–16 (2016).
Webster, J. R. & Benfield, E. F. Vascular plant breakdown in freshwater ecosystems. Annu. Rev. Ecol. Syst. 17, 567–594 (1986).
Ledesma, J. L. J., Grabs, T., Bishop, K. H., Schiff, S. L. & Köhler, S. J. Potential for long-term transfer of dissolved organic carbon from riparian zones to streams in boreal catchments. Glob. Change Biol. 21, 2963–2979 (2015).
Fisher, S. G., Heffernan, J. B., Sponseller, Ra & Welter, J. R. Functional ecomorphology: Feedbacks between form and function in fluvial landscape ecosystems. Geomorphology 89, 84–96 (2007).
Harvey, J. & Gooseff, M. River corridor science: Hydrologic exchange and ecological consequences from bedforms to basins: River corridors from bedforms to basins. Water Resour. Res. 51, 6893–6922 (2015).
Harvey, J. et al. How Hydrologic Connectivity Regulates Water Quality in River Corridors. JAWRA J. Am. Water Resour. Assoc. 55, 369–381 (2019).
Holtgrieve, G. W. & Schindler, D. E. Marine-derived nutrients, bioturbation, and ecosystem metabolism: reconsidering the roleof salmon in streams. Ecology 92, 373–385 (2011).
Helton, A. M., Poole, G. C., Payn, R. A., Izurieta, C. & Stanford, J. A. Scaling flow path processes to fluvial landscapes: An integrated field and model assessment of temperature and dissolved oxygen dynamics in a river-floodplain-aquifer system: Scaling flow paths to floodplains. J. Geophys. Res. Biogeosciences 117, n/a–n/a (2012).
Comer-Warner, S. A. et al. Thermal sensitivity of CO2 and CH4 emissions varies with streambed sediment properties. Nat. Commun. 9 (2018).
Hall, R. O. & Tank, J. Correcting whole-stream estimates of metabolism for groundwater input. Limnol. Oceanogr. Methods 3, 222–229 (2005).
Bernhardt, E. S. et al. The metabolic regimes of flowing waters: Metabolic regimes. Limnol. Oceanogr. 63, S99–S118 (2018).
Appling, A. P. et al. The metabolic regimes of 356 rivers in the United States. Sci. Data 5, 180292 (2018).
Lisi, P. J., Schindler, D. E., Cline, T. J., Scheuerell, M. D. & Walsh, P. B. Watershed geomorphology and snowmelt control stream thermal sensitivity to air temperature: Water source and stream thermal regimes. Geophys. Res. Lett. 42, 3380–3388 (2015).
Wohl, E., Hall, R. O. Jr., Lininger, K. B., Sutfin, N. A. & Walters, D. M. Carbon dynamics of river corridors and the effects of human alterations. Ecol. Monogr. 87, 379–409 (2017).
Drake, T. W. et al. The Ephemeral Signature of Permafrost Carbon in an Arctic Fluvial Network. J. Geophys. Res. Biogeosciences 123, 1475–1485 (2018).
Monteith, D. T. et al. Dissolved organic carbon trends resulting from changes in atmospheric deposition chemistry. Nature 450, 537–540 (2007).
Williamson, C. E. et al. Ecological consequences of long-term browning in lakes. Sci. Rep. 5 (2016).
Holtgrieve, G. W., Schindler, D. E., Branch, Ta & A’mar, Z. T. Simultaneous quantification of aquatic ecosystem metabolism and reaeration using a Bayesian statistical model of oxygen dynamics. Limnol. Oceanogr. 55, 1047–1063 (2010).
Jankowski, K. J. Watershed controls on biogeochemical processes in aquatic ecosystems. (University of Washington, 2014).
Sinsabaugh, R. L. & Follstad Shah, J. J. Integrating resource utilization and temperature in metabolic scaling of riverine bacterial production. Ecology 91, 1455–1465 (2010).
Nakagawa, S. & Schielzeth, H. A general and simple method for obtaining R 2 from generalized linear mixed-effects models. Methods Ecol. Evol. 4, 133–142 (2013).
Bates, D., Maechler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Acknowledgements
We thank Gordon Holtgrieve, Mike Brett, Claire Horner-Devine, and Jeff Houser for helpful reviews on this manuscript. Teresa Z’Amar provided technical assistance with updating and implementing the Bayesian model. Funding was provided by EPA STAR and ARCS Foundation fellowships and the US Army Corps of Engineers’ Upper Mississippi River Restoration Program to K.J Jankowski; NSF and the Harriet Bullitt Professorship to D.E. Schindler; and the UW Alaska Salmon Program. We thank the Wood-Tikchik State Park for enabling our research.
Author information
Authors and Affiliations
Contributions
K.J.J. and D.E.S. designed the study and collected field data, K.J.J. analyzed data and performed analyses, K.J.J. and D.E.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jankowski, K.J., Schindler, D.E. Watershed geomorphology modifies the sensitivity of aquatic ecosystem metabolism to temperature. Sci Rep 9, 17619 (2019). https://doi.org/10.1038/s41598-019-53703-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53703-3
- Springer Nature Limited
This article is cited by
-
Climate and landform interact to control the source and transport of nitrate in Pacific Northwest rivers
Communications Earth & Environment (2024)
-
Dissolved oxygen isotope modelling refines metabolic state estimates of stream ecosystems with different land use background
Scientific Reports (2022)
-
Turbidity Structures the Controls of Ecosystem Metabolism and Associated Metabolic Process Domains Along a 75-km Segment of a Semiarid Stream
Ecosystems (2022)