Abstract
This phase 2 study evaluated the safety and efficacy of perioperative chemotherapy with S-1 plus oxaliplatin (SOX) for stage III colorectal cancer (CRC). Patients with stage III CRC received surgery after neoadjuvant chemotherapy (NAC; SOX 4 cycles) and adjuvant chemotherapy (AC; SOX 4 cycles). The primary endpoints were response rate and safety. We enrolled 30 patients. Their median age was 62 years (range: 43–87 years); 53% were women. They received a median of 4 cycles (range: 1–4) of NAC and a median 4 cycles (range: 0–4) of AC. Five patients interrupted NAC treatment because of toxicity (grade 3 diarrhoea [n = 1], grade 3 ileus [n = 1], and grade 3–4 thrombocytopenia [n = 3]). Patients’ responses were complete responses: n = 2 (6.6%), partial responses: n = 21 (70%), stable disease: n = 6 (20.0%), and progressive disease: n = 1 (3.3%; response rate: 73.3%). Curative resection was performed in 29 patients. No patients showed anastomotic leakage. Five-year overall survival and disease-free survival were 83.3% and 76.7%, respectively (median follow-up time: 48 months). NAC using SOX regimen is safe and effective, and may lead to reduced local recurrence and distant metastasis. Long-term outcomes are awaited to evaluate further the efficacy of this strategy (UMIN000006790).
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is second-most common cancer in the world. Among patients with resectable CRC, the 5-year survival rate has improved to more than 80%1. However, the survival rate of patients with stage III CRC is still not satisfactory. To improve outcomes, postoperative adjuvant chemotherapy (AC) is usually recommended to suppress distant micrometastasis; it has shown a 63–74% improvement in 5-year survival over surgery alone2. To improve outcomes further, some researchers investigated the effectiveness of administering cytotoxic therapy as neoadjuvant chemotherapy (NAC)2. Potentially, NAC can provide in vivo therapeutic responses, tumour downstaging, and early treatment for micrometastatic disease. However, NAC for CRC has several potential drawbacks, such as possible disease progression, surgery postponement due to adverse event (AEs), and increased postoperative complications. Although NAC is promising, data demonstrating its safety and efficacy are still scarce.
FOLFOX and CAPOX has been used in clinical trials of NAC for stage III CRC3,4,5,6. Although CAPOX and SOX reportedly have equivalent effects in metastatic CRC7,8, clinical trials that use SOX therapy as NAC have not been reported for stage III CRC. AEs are thought to differ between SOX and CAPOX, as S-1 and capecitabine have different metabolic pathways9. AEs associated with NAC over a limited administration period are unknown. This trial evaluated the efficacy and safety of NAC for clinical lymph-node-positive CRC. Here, we present the short-term results of this trial, with respect to response rates, toxicities and surgical complications.
Results
Patient characteristics
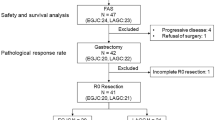
We enrolled 30 patients who were treated between March 2012 and September 2016. Their median age was 62 years (range: 43–87); 53% were women. Table 1 summarizes their baseline characteristics. Their median body mass index was 21.6 kg/m2. Their ECOG-PS scores were 0: 40%, 1: 60%. The median length of the primary tumour at the baseline was 50 mm (range: 20–102 mm); distribution was cT2: n = 1, cT3: n = 12, and cT4: n = 17 (cT4a: n = 12, cT4b: n = 5).
Treatment compliance and toxicity of NAC and AC
Of the 30 patients enrolled in this study, 25 patients completed NAC. The patients received median 4 (1–4) cycles of NAC and median 4 (0–4) cycles of AC. Their total median dose of oxaliplatin was 635 (122–1040) mg/m2 (NAC; 494 [122–520] mg/m2, AC; 230 [0–520] mg/m2). Five patients interrupted NAC due to AEs (grade 3 diarrhoea [n = 1], grade 3 ileus [n = 1], grade 3–4 thrombocytopenia [n = 3]). AC was administered to 21 patients (17: SOX: n = 17, S-1 only: n = 4). Eight patients did not received AC.
AEs during NAC are shown in Table 2. The most common AEs were thrombocytopenia (n = 16, 53%) and neutropenia (n = 11, 37%) for haematological events; and peripheral neuropathy (n = 25, 83%) and fatigue (n = 18, 60%) for non-haematological events. Two patients had grade 4 AEs (diarrhoea and thrombocytopenia).
Effect of chemotherapy
The response rate to NAC in this study, according to RECIST ver.1.1, was 76.6% (complete response: 6.6%; partial response: 70.0%; stable disease: 20.0%; and progressive disease: 3.3%). The disease control rate was 96.7%. One patient who was not responsive was shown to have para-aortic lymph node (LN) metastasis on computed tomography (CT) after 4 cycles of SOX. The median tumour shrinkage rate was 33% (range: −42–100%) after NAC; 23 of the 30 patients achieved a > 30% reduction in tumour size. The waterfall plot (Fig. 1) shows the rate of change in primary tumour diameters at the end of NAC compared with baseline.
Surgical outcomes
Of the 30 patients who received NAC, 29 underwent curative surgery (Table 3). One patient received a pancreaticoduodenectomy in addition to right hemicolectomy. Procedures were laparoscopic: 55.2%, and open: 44.8%. Median surgical time was 335 (72–729) minutes. Median estimated blood loss was 129 (0–2210) g. Twelve patients (41.3%) suffered postoperative complications; 2 patients, respectively, experienced pancreatic fistula and lower-leg compartment syndrome (Clavien–Dindo grade ≥ 3). Although no anastomotic leakage was observed, 13 patients received ileostomies or colostomies.
Pathological assessment
Three (10.3%) of the 29 patients who underwent resections showed pCR, and 12 (41.4%) exhibited good tumour regression (grade 2/3). One patient’s primary tumour disappeared but his LN metastasis remained. Table 4 shows the relationships between clinical stage before chemotherapy and pathological stage. After NAC, the frequency of T downstaging was 69.0% (20/29) and disappearance of LN metastasis was 65.5% (19/29).
Prognosis
Five-year OS and DFS were 83.3% and 76.7%, respectively (median follow-up time 48 months); OS and DFS curves are shown in Fig. 2. Of the 29 patients who underwent R0 resection, 7 patients had relapsed. Lung recurrence was seen in 2 patients, liver recurrence in 1 patient, LN recurrence in 3 patients, and local recurrence in 2 patients (colon: 0, rectum: 2).
Discussion
Whereas surgery removes obvious tumours, NAC provides the advantage of anticancer drug delivery, to treat distant micrometastases that have not been detected, potentially reducing the risk of recurrence10,11,12. Use of NAC can also help in estimating response to chemotherapy, and thus in selecting the optimal adjuvant therapy. NAC is used to reduce tumour size and render initially unresectable tumours resectable13,14.
This trial evaluated the efficacy and safety of SOX as NAC for patients with clinical node-positive CRC. Because port-free chemotherapy is suitable for perioperative patients, and can avoid complications associated with central venous ports15, the SOX regimen was chosen over the FOLFOX regimen. S-1 can lead to less hand-foot syndrome (HFS) than capecitabine16 and several studies have demonstrated the efficacy of SOX for metastatic CRC17,18, As the AVOID trial demonstrated that adding dexamethasone could help prevent oxaliplatin-induced hypersensitivity reactions19, we mixed dexamethasone (6.6 mg) into the oxaliplatin.
However, this is the first report of SOX therapy as a preoperative chemotherapy. Hong et al. had shown that patients treated with SOX had greater overall objective responses than did those treated with CAPOX, and that S-1 was more advantageous than capecitabine in reducing HFS frequency7, They also showed clear noninferiority of SOX and CAPOX with respect to progression-free survival. Thrombocytopenia is an important haematological AE of oxaliplatin. However, whereas 22% of patients treated with SOX reportedly suffer grade 3–4 thrombocytopenia7, the incidence of grade 3–4 thrombocytopenia in this study is 13.8%—lower than previous studies. This probably reflects our study’s drug-withdrawal period for surgery. AE rates in this trial differed from reported rates for CAPEOX as NAC3,4,5. SOX appears to be especially good for patients in occupations that require manual dexterity because of the low HFS incidence.
In this study, the response rate was 76.6%, and tumour control rate was 96.7%, with 29 patients undergoing curative resections. Two patients showed perioperative complications grade ≥ 3 (Clavien–Dindo classification), but these were not thought to be directly related to NAC. Overall survival and relapse-free survival were 83.3% and 76.7%, respectively (median follow-up time 48 months). These results are not inferior to those of other reported clinical trials3,4,5,6. SOX therapy is a better regimen for those who do not want to have a CV port or those suffering from severe HFS.
Preoperative chemoradiotherapy (CRT) combined with total TME has improved local control and sphincter preservation rates in patients with locally advanced rectal cancer20. The reported 3-year DFS of CRT for patients with locally advanced rectal cancer is 71.6–75.9%21,22,23, which is equivalent to the results of the present study. However, CRT has been recently shown to increase perioperative complications, such as local infections and anastomotic leakage6. Furthermore, irradiated patients may develop later complications of bowel, bladder, and sexual function24. However, NAC for CRC also has some potential disadvantages, including the increased possibility of postsurgical complications. Although NAC is theoretically promising, data to support its safety and efficacy is still inadequate.
This trial had some limitations. First, the study cohort was rather small. Second, preoperative staging is not always accurate, which could have led to overtreatment of patients who may not need NAC. To increase dose intensity, we recommend examining results of other SOX studies and setting the administration method for the phase 3 study. Despite these potential limitations, we believe this strategy is promising as NAC for stage III CRC.
Conclusions
This research confirmed the feasibility of NAC using SOX regimen for stage III CRC. However, the results of the phase 3 trial will better indicate whether this regimen reduces local recurrence and distant metastasis rates.
Patients and Methods
This study was a single-arm, open-label, prospective, phase 2 study to evaluate the response rate and safety of NAC with SOX therapy for stage III CRC. The study protocol was approved by the institutional review board of Fukuoka University Hospital (No. 11-056). This study is registered with UMIN-CTR, UMIN000006790 (first registration date: 28/Nov/2011). Informed consent was obtained from all patients prior to study entry. All procedures were performed in accordance with the Declaration of Helsinki.
Eligibility criteria
All patients had pathologically confirmed adenocarcinoma of the colon and rectum without evidence of distant metastasis. The inclusion criteria for this study were stage III CRC (any T and N1–N3). The stage was assessed by colonoscopy, computed tomography (CT), or magnetic resonance imaging (MRI). The definition of LN metastasis was determined by CT or MRI as follows: colon: minimum LN diameter > 10 mm as determined by CT25,26,27; rectum: LN > 5 mm in short-axis diameter with an irregular border or mixed signal intensity as determined by MRI28,29. Patients had Eastern Cooperative Oncology Group Performance Status (ECOG-PS) of 0 or 1 and preserved organ function, and had provided written informed consent. Exclusion criteria included distant metastasis or synchronous tumours, history of neurologic or psychiatric disorders, cardiovascular disease, previous history of chemotherapy or pelvic radiotherapy, or another cancer diagnosis within the past 5 years.
Treatment protocol
Patients with stage III CRC received NAC (4 cycles of SOX) before surgery and AC (4 cycles of SOX) after surgery. Four cycles of SOX were administered before surgery, because many clinical trials of CAPOX-based NAC use four cycles of treatment3,4,30.
Neoadjuvant treatment
The treatment schema is shown in Fig. 3. In principle, patients received 4 cycles of NAC, consisting of SOX: S-1 (80 mg/m2/day) from Days 1 to 14, and oxaliplatin (85 mg/m2) on Day 1, every 3 weeks. To prevent oxaliplatin-induced hypersensitivity reaction, dexamethasone and oxaliplatin were co-injected19. Toxicity was assessed according to the National Cancer Institute Common Terminology for Adverse Events, version 4.0. We used the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines to determine objective responses of target lesions.
Surgery
After four cycles of neoadjuvant SOX therapy, tumour resectability was reassessed using CT. Patients for whom surgery was suitable underwent complete mesocolic excisions or total mesorectal excisions (TME) at 4–6 weeks after their last NAC courses. Combined resections were performed when cancer was intraoperatively suspected to have invaded other organs. Selection of an open or laparoscopic approach was at the surgeon’s discretion. Postoperative morbidities were assessed according to the Clavien–Dindo classification.
Adjuvant chemotherapy
Patients for whom NAC was effective (i.e., had no progressive disease) and tolerable, and who had R0 resections received postoperative AC with four cycles of SOX.
Study end points
The primary end points of this study were objective response rate and safety. The secondary end points include NAC completion rate, relative dose intensity, incidence of AEs, postoperative complications, pathologic tumour response, and 5-year overall survival (OS) and disease-free survival (DFS). Pathologic tumour responses were graded according to the Japanese Classification of Colorectal Carcinoma31 as 1a: denaturation and necrosis of cancer cells in less than a third of the cancer tissue; 1b: denaturation and necrosis in less than two thirds of the cancer cells, plus fusion in more than one third of the cancer; 2: significant denaturation, necrosis, fusion, and loss in more than two thirds of the cancer; and 3: pathologic complete response (pCR) with no cancer cells observed in either the primary or regional LNs.
Sample size and statistical analysis
This study aims to reduce the risk of recurrence of stage III CRC, using pre- and postoperative SOX therapy. Therefore, the response rate to SOX therapy in preoperative chemotherapy was regarded as the primary endpoint. In clinical trials for metastatic CRC, response rates for FOLFOX, CAPOX, and SOX were 41–49%32,33,34, 27–55%32,33,34,35, and 50%36, respectively. However, according to an integrated analysis of results of randomized controlled trials of FOLFOX and CAPOX, CAPOX therapy is inferior to FOLFOX therapy in response rates (odds ratio: 0.74)37. The planned sample size was determined by defining expected and threshold response rates as 50% and 30%, respectively. By setting alpha and beta errors at 0.05 and 0.2 (respectively), statistical analysis calculated the required number of patients to be 30 patients. We evaluated OS and DFS by the Kaplan–Meier method, using SPSS software, version 22.0 (IBM Japan Ltd., Tokyo, Japan).
References
Jemal, A. et al. Global cancer statistics. CA: a cancer journal for clinicians 61, 69–90, https://doi.org/10.3322/caac.20107 (2011).
O’Connell, M. J. et al. Controlled trial of fluorouracil and low-dose leucovorin given for 6 months as postoperative adjuvant therapy for colon cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 15, 246–250, https://doi.org/10.1200/jco.1997.15.1.246 (1997).
Hasegawa, J. et al. Neoadjuvant capecitabine and oxaliplatin (XELOX) combined with bevacizumab for high-risk localized rectal cancer. Cancer chemotherapy and pharmacology 73, 1079–1087, https://doi.org/10.1007/s00280-014-2417-9 (2014).
Nishimura, J. et al. Phase II trial of capecitabine plus oxaliplatin (CAPOX) as perioperative therapy for locally advanced rectal cancer. Cancer chemotherapy and pharmacology 82, 707–716, https://doi.org/10.1007/s00280-018-3663-z (2018).
Kamiya, T. et al. Early results of multicenter phase II trial of perioperative oxaliplatin and capecitabine without radiotherapy for high-risk rectal cancer: CORONA I study. European journal of surgical oncology: the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology 42, 829–835, https://doi.org/10.1016/j.ejso.2016.02.014 (2016).
Deng, Y. et al. Modified FOLFOX6 With or Without Radiation Versus Fluorouracil and Leucovorin With Radiation in Neoadjuvant Treatment of Locally Advanced Rectal Cancer: Initial Results of the Chinese FOWARC Multicenter, Open-Label, Randomized Three-Arm Phase III Trial. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 34, 3300–3307, https://doi.org/10.1200/jco.2016.66.6198 (2016).
Hong, Y. S. et al. S-1 plus oxaliplatin versus capecitabine plus oxaliplatin for first-line treatment of patients with metastatic colorectal cancer: a randomised, non-inferiority phase 3 trial. The Lancet. Oncology 13, 1125–1132, https://doi.org/10.1016/s1470-2045(12)70363-7 (2012).
Kim, S. T. et al. S-1 plus oxaliplatin versus capecitabine plus oxaliplatin for the first-line treatment of patients with metastatic colorectal cancer: updated results from a phase 3 trial. BMC cancer 14, 883, https://doi.org/10.1186/1471-2407-14-883 (2014).
Adjei, A. A. A review of the pharmacology and clinical activity of new chemotherapy agents for the treatment of colorectal cancer. British journal of clinical pharmacology 48, 265–277 (1999).
Nordlinger, B. et al. Combination of surgery and chemotherapy and the role of targeted agents in the treatment of patients with colorectal liver metastases: recommendations from an expert panel. Annals of oncology: official journal of the European Society for Medical Oncology 20, 985–992, https://doi.org/10.1093/annonc/mdn735 (2009).
Nordlinger, B. et al. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet (London, England) 371, 1007–1016, https://doi.org/10.1016/s0140-6736(08)60455-9 (2008).
Leichman, L. Neoadjuvant chemotherapy for disseminated colorectal cancer: changing the paradigm. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 24, 3817–3818, https://doi.org/10.1200/jco.2006.07.2546 (2006).
Ellis, L. M., Curley, S. A. & Grothey, A. Surgical resection after downsizing of colorectal liver metastasis in the era of bevacizumab. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 23, 4853–4855, https://doi.org/10.1200/jco.2005.23.754 (2005).
Nordlinger, B. et al. Does chemotherapy prior to liver resection increase the potential for cure in patients with metastatic colorectal cancer? A report from the European Colorectal Metastases Treatment Group. European journal of cancer (Oxford, England: 1990) 43, 2037–2045, https://doi.org/10.1016/j.ejca.2007.07.017 (2007).
Yoshida, Y. et al. Administration of chemotherapy via the median cubital vein without implantable central venous access ports: port-free chemotherapy for metastatic colorectal cancer patients. International journal of clinical oncology 20, 332–337, https://doi.org/10.1007/s10147-014-0703-5 (2015).
Lee, J. L. et al. A randomised multicentre phase II trial of capecitabine vs S-1 as first-line treatment in elderly patients with metastatic or recurrent unresectable gastric cancer. British journal of cancer 99, 584–590, https://doi.org/10.1038/sj.bjc.6604536 (2008).
Yamada, Y. et al. Leucovorin, fluorouracil, and oxaliplatin plus bevacizumab versus S-1 and oxaliplatin plus bevacizumab in patients with metastatic colorectal cancer (SOFT): an open-label, non-inferiority, randomised phase 3 trial. The Lancet. Oncology 14, 1278–1286, https://doi.org/10.1016/s1470-2045(13)70490-x (2013).
Zang, D. Y. et al. Phase II study with oxaliplatin and S-1 for patients with metastatic colorectal cancer. Annals of oncology: official journal of the European Society for Medical Oncology 20, 892–896, https://doi.org/10.1093/annonc/mdn721 (2009).
Yoshida, Y. et al. A single-arm Phase II validation study of preventing oxaliplatin-induced hypersensitivity reactions by dexamethasone: the AVOID trial. Drug design, development and therapy 9, 6067–6073, https://doi.org/10.2147/dddt.S94901 (2015).
Kapiteijn, E. et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. The New England journal of medicine 345, 638–646, https://doi.org/10.1056/NEJMoa010580 (2001).
Allegra, C. J. et al. Neoadjuvant 5-FU or Capecitabine Plus Radiation With or Without Oxaliplatin in Rectal Cancer Patients: A Phase III Randomized Clinical Trial. Journal of the National Cancer Institute 107, https://doi.org/10.1093/jnci/djv248 (2015).
Hong, Y. S. et al. Oxaliplatin, fluorouracil, and leucovorin versus fluorouracil and leucovorin as adjuvant chemotherapy for locally advanced rectal cancer after preoperative chemoradiotherapy (ADORE): an open-label, multicentre, phase 2, randomised controlled trial. The Lancet. Oncology 15, 1245–1253, https://doi.org/10.1016/s1470-2045(14)70377-8 (2014).
Gerard, J. P. et al. Clinical outcome of the ACCORD 12/0405 PRODIGE 2 randomized trial in rectal cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 30, 4558–4565, https://doi.org/10.1200/jco.2012.42.8771 (2012).
Marijnen, C. A. et al. Impact of short-term preoperative radiotherapy on health-related quality of life and sexual functioning in primary rectal cancer: report of a multicenter randomized trial. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 23, 1847–1858, https://doi.org/10.1200/jco.2005.05.256 (2005).
Acunas, B. et al. Preoperative CT staging of colon carcinoma (excluding the recto-sigmoid region). Eur J Radiol 11, 150–153, https://doi.org/10.1016/0720-048x(90)90166-9 (1990).
Cademartiri, F., Luccichenti, G., Rossi, A. & Pavone, P. Spiral hydro-CT in the evaluation of colo-sigmoideal cancer. La Radiologia medica 104, 295–306 (2002).
Dighe, S., Swift, I. & Brown, G. CT staging of colon cancer. Clinical radiology 63, 1372–1379, https://doi.org/10.1016/j.crad.2008.04.021 (2008).
Kim, J. H., Beets, G. L., Kim, M.-J., Kessels, A. G. & Beets-Tan, R. G. High-resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? European journal of radiology 52, 78–83 (2004).
Brown, G. et al. Morphologic predictors of lymph node status in rectal cancer with use of high-spatial-resolution MR imaging with histopathologic comparison. Radiology 227, 371–377, https://doi.org/10.1148/radiol.2272011747 (2003).
Uehara, K. et al. Phase II trial of neoadjuvant chemotherapy with XELOX plus bevacizumab for locally advanced rectal cancer. Japanese journal of clinical oncology 41, 1041–1044, https://doi.org/10.1093/jjco/hyr084 (2011).
Colon, J. S. f. C. o. t. & Rectum. (Kanehara Tokyo, Japan, 2009).
Ducreux, M. et al. Capecitabine plus oxaliplatin (XELOX) versus 5-fluorouracil/leucovorin plus oxaliplatin (FOLFOX-6) as first-line treatment for metastatic colorectal cancer. International journal of cancer 128, 682–690, https://doi.org/10.1002/ijc.25369 (2011).
Hochster, H. S. et al. Safety and efficacy of oxaliplatin and fluoropyrimidine regimens with or without bevacizumab as first-line treatment of metastatic colorectal cancer: results of the TREE Study. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 26, 3523–3529, https://doi.org/10.1200/jco.2007.15.4138 (2008).
Cassidy, J. et al. Randomized phase III study of capecitabine plus oxaliplatin compared with fluorouracil/folinic acid plus oxaliplatin as first-line therapy for metastatic colorectal cancer. Journal of clinical oncology 26, 2006–2012 (2008).
Cassidy, J. et al. XELOX (capecitabine plus oxaliplatin): active first-line therapy for patients with metastatic colorectal cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 22, 2084–2091, https://doi.org/10.1200/jco.2004.11.069 (2004).
Yamada, Y. et al. Phase I/II study of oxaliplatin with oral S-1 as first-line therapy for patients with metastatic colorectal cancer. British journal of cancer 98, 1034–1038, https://doi.org/10.1038/sj.bjc.6604271 (2008).
Arkenau, H. T. et al. Efficacy of oxaliplatin plus capecitabine or infusional fluorouracil/leucovorin in patients with metastatic colorectal cancer: a pooled analysis of randomized trials. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 26, 5910–5917, https://doi.org/10.1200/jco.2008.16.7759 (2008).
Acknowledgements
We thank Marla Brunker, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
N.A. and Y.Y. wrote the main manuscript. S.H. provided analytical oversight and revised the manuscript for important intellectual content. A.K., S.R., and D.K. collected the data. All authors have read and approved the final version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aisu, N., Yoshida, Y., Komono, A. et al. Phase 2 study of perioperative chemotherapy with SOX and surgery for stage III colorectal cancer (SOS3 study). Sci Rep 9, 16568 (2019). https://doi.org/10.1038/s41598-019-53096-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53096-3
- Springer Nature Limited