Abstract
Semiochemicals play a pivotal role in the location, evaluation, and utilization of hosts by herbivorous insects. Mixtures of host plant-derived compounds are often required to elicit appropriate levels of response to olfactory stimuli. In multiple-choice bioassays, we characterized the response of adult Drosophila suzukii to foliage- and fruit-based synthetic compounds tested alone and in association with grape and tart cherry juices, and assessed whether synergistic interactions among olfactory stimuli are involved in the olfactory-driven behavior of D. suzukii. Our results established (1) significant attraction of females (but not males) to β-cyclocitral and isoamyl acetate when tested singly, (2) the presence of a synergistic interaction between β-cyclocitral and cherry juice only for females, and (3) the presence of a synergistic interaction between β-cyclocitral and isoamyl acetate but only in the case of males. Our findings increase our understanding of male and female D. suzukii olfactory responses to synthetic compounds and fruit juices as sources of attractants. Combinations of foliage- and fruit-based compounds may be needed to increase SWD attraction.
Similar content being viewed by others
Introduction
Chemical cues are considered to play a pivotal role in the location, evaluation, and utilization of hosts by herbivorous insects1,2. Numerous studies have shown that, rather than single constituents, mixtures of host plant-derived compounds are often required to elicit appropriate levels of response to olfactory stimuli3,4. Furthermore, synergistic interactions among the components of an odor mixture have been found to contribute to the attraction of some species of insects to their host plants5,6,7.
Native to Southeast Asia, the invasive Spotted Wing Drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilidae)8,9, has reached world-wide pest status resulting from its recent invasion of much of North America10,11,12, South America13,14 and some regions of Europe15,16. Its polyphagous habits, preference for ripening fruits, and the female’s ability to pierce soft-skinned fruits during egg-laying make D. suzukii a pest of great economic significance17,18. Current management options for D. suzukii in fruit crops rely heavily on insecticide applications19,20,21,22. While early detection and effective monitoring tools are critical to implementation of pest control tactics against D. suzukii23, effective semiochemically-based monitoring represents a challenge for pest managers given the fly’s ability to reproduce in different host plant species24,25, its high reproductive potential26, and a general lack of species-specific attractants23,27.
While foraging, adult D. suzukii seek essential resources suitable for feeding, mating, and oviposition, and volatiles play key roles during this process28. Chemical ecology research conducted across the globe has resulted in the identification of volatiles emitted from fermenting materials that are attractive to D. suzukii. Synthetic lures have been developed based on blends of four compounds isolated from fermentation headspace of wine and vinegar29. The four-component blend has shown to attract more adult D. suzukii than apple cider vinegar29 and has also shown to detect adults earlier than apple cider vinegar when evaluated in areas that harbor wild blackberry (Rubus) species and in commercial blueberry orchards30. Given these advantages, commercial food-based lures are currently being used to monitor SWD populations28,31,32. However, lures that are based on fermentation materials attract a comparatively high number of other Drosophilid species (and other non-target insects) hindering trap performance and increasing sorting time26,31,33.
A promising alternative to the use of fermentation-derived attractants is the identification of volatiles emitted by foliage and/or host fruits. Keesey et al.34 reported a positive behavioral response of male and female D. suzukii towards the strawberry leaf terpenoid β-cyclocitral. Similar results were found under laboratory conditions when β-cyclocitral was evaluated in 60 ml plastic containers. EAG recordings demonstrated that the antennal response of D. suzukii to β-cyclocitral is species-specific, given the lack of EAG detection to the same compound by D. melanogaster and D. biarmipes34.
Attraction of adult D. suzukii to fruit volatiles emitted by intact blackberry (Rubus fruticosus L), blueberry (Vaccinium corymbosum L.), cherry (Prunus cerasus L.), raspberry (Rubus idaeus L.), and strawberry (Fragaria ananassa Duchesne) has been reported by Revadi et al.35. Further evidence that fruit volatiles are important in D. suzukii host location was provided by Abraham et al.36, who documented fly attraction to fruit extracts of highbush blueberry, cherry, raspberry, and strawberry, and to selected volatile compounds derived from raspberry fruit. Using behavioral and electrophysiological assays, Revadi et al.35 found that isoamyl acetate, a fruit-based compound, elicited a positive behavioral response in D. suzukii35. This compound also attracts other Drosophilids37. More recently, a quaternary blend made of ethyl octanoate, acetoin, acetic acid, and ethyl acetate, compounds present in headspace volatiles collected from fermented apple juice, was reported to be 2–4 times more attractive and 2–3 times more selective than the widely used ACV and commercially available SWD lures under field conditions38.
To date, the role of synthetic or natural sources of plant-based odor in the olfactory response of male and female D. suzukii is little understood. The main goals of this study were (1) to quantify the response of male and female D. suzukii to the leaf compound β-cyclocitral and to the fruit compound isoamyl acetate tested singly against mineral oil (solvent control), and tested against each other, (2) to assess the types of interactions between β-cyclocitral and isoamyl acetate when tested alone or in combination, and (3) to assess whether β-cyclocitral and isoamyl acetate would increase the response of adult D. suzukii to grape and tart cherry juices.
Results
Experiment 1
Significant interactions between main effects (sex and treatment) were detected for the comparison of (1) β-cyclocitral against control (mineral oil) and (2) β-cyclocitral against isoamyl acetate, but not for the comparison of isoamyl acetate against control (Table 1). When tested singly, β-cyclocitral and isoamyl acetate were not attractive to D. suzukii males, compared to mineral oil (t-tests t18 = 1.4; P = 0.167 and t14 = 1.2; P = 0.236 for β-cyclocitral and isoamyl acetate, respectively) (Fig. 1A,B). In contrast, both chemical compounds were attractive to females (t-tests t18 = 4.1; P < 0.001 and t14 = 2.2; P = 0.043 for β-cyclocitral and isoamyl acetate, respectively) (Fig. 1A,B). When tested singly against each other, β-cyclocitral and isoamyl acetate were similarly attractive to males (t-test t12 = 1.7; P = 0.126) and isoamyl acetate was significantly more attractive to females than β-cyclocitral (t-test t12 = 3.0; P = 0.010) (Fig. 1C).
Response of male and female D. suzukii to (A) β-cyclocitral and (B) isoamyl acetate when compared singly against mineral oil (control), and to (C) β-cyclocitral and isoamyl acetate, tested against each other. For each sex, different letters above bars denote significant differences according to t-tests at P = 0.05.
Experiment 2
A 2-way ANOVA detected a significant effect of “treatment” and “sex” on the response of D. suzukii to the treatments evaluated, and the interaction term was non- significant (Table 1). A significant effect of olfactory treatment was detected for both males (ANOVA F3,92 = 3.4; P = 0.021) and females (ANOVA F3,92 = 3.2; P = 0.025). While the level of response of males to isoamyl acetate or β -cyclocitral alone did not differ significantly from the response to mineral oil, and synergistic effect was noted when both compounds were presented in combination (Fig. 2). Females showed a significant preference for isoamyl acetate over β-cyclocitral, and the level of response to the combination of isoamyl acetate and β-cyclocitral did not differ from the response exhibited to isoamyl acetate alone (Fig. 2).
Experiment 3
Male and female D. suzukii response to synthetic compounds in association with grape juice
For both β-cyclocitral and isoamyl acetate the outcomes of the 2-way ANOVAS revealed a significant effect of treatment, a non-significant effect of sex, and non-significant interaction terms (Table 1). Grape juice was attractive to males (ANOVA F3,28 = 6.8; P < 0.01) and the level of response was not significantly enhanced by the addition of β-cyclocitral (Fig. 3A). For females, grape juice alone was attractive and the addition of β-cyclocitral to grape juice significantly increased the response (ANOVA F3,28 = 16.8; P < 0.001) compared to grape juice alone. The effect was additive (Table 2). β-cyclocitral was attractive to females, but not to males, when compared against water (Fig. 3A).
Response of male and female D. suzukii to Concord grape juice either, alone or in association with (A) β-cyclocitral, and (B) isoamyl acetate. Mineral oil was used as control. For each sex, different letters above bars denote significant differences according to ANOVA and Fisher-protected LSD tests at P = 0.05.
For the tests that involved isoamyl acetate, grape juice alone was attractive both to males (ANOVA F3,22 = 3.4; P = 0.04) and females (ANOVA F3,22 = 10.0; P < 0.001) when compared to the control (Fig. 3B). The addition of isoamyl acetate to grape juice did not increase significantly the level of response of males or females compared to grape juice alone. Isoamyl acetate was not attractive to males or females (Fig. 3B).
Male and female D. suzukii response to synthetic compounds in association with cherry juice
The outcomes of the 2-way ANOVAS revealed a significant effect of treatment and sex (main factors) and a significant treatment x sex interaction (Table 1) indicating dissimilar responses by males and females to the olfactory treatments when cherry juice was included in the evaluations. For males, no significant preference for cherry juice either, alone or in combination with β-cyclocitral, or for β-cyclocitral alone was recorded (ANOVA F3,32 = 1.2; P = 0.320). For females, cherry juice and β-cyclocitral interacted synergistically (Table 2), resulting in significantly increased attraction (ANOVA F3,32 = 17.4; P < 0.001) (Fig. 4A). β-cyclocitral was attractive to females when compared to the control.
Response of male and female D. suzukii to tart cherry juice either, alone or in association with (A) β-cyclocitral, and (B) isoamyl acetate. Mineral oil was used as control. For each sex, different letters above bars denote significant differences according to ANOVA and Fisher-protected LSD tests at P = 0.05.
Cherry juice and isoamyl acetate either, alone or in combination, were not attractive to males (ANOVA F3,26 = 1.2; P = 0.343). In contrast, female D. suzukii showed a significant preference for cherry juice and for cherry juice + isoamyl acetate when compared to isoamyl acetate alone and the control (ANOVA F3,26 = 10.2; P < 0.001) (Fig. 4B).
Discussion
The results of the behavioral experiments demonstrated: (1) positive responses of D. suzukii females (but not males) to isoamyl acetate and β-cyclocitral when presented singly, (2) a significant preference of females (but not males) for isoamyl acetate over β-cyclocitral, (3) a significant increase in the level of response of females to grape juice when β-cyclocitral was added and a female-specific synergistic interaction between β-cyclocitral and cherry juice, and (4) a male-specific synergistic interaction between β-cyclocitral and isoamyl acetate when these compounds were presented in association.
Drosophila suzukii is known to colonize host plants in the early stages of fruit ripening. Therefore, it is reasonable to hypothesize that adults use chemical cues emitted from both foliage and fruit during host location28,36. The synthetic compounds evaluated in this study, β-cyclocitral and isoamyl acetate, are present in various host34,35 and non-host plants (e.g., Saglam et al.39) of D. suzukii, and they were selected based on reported positive physiological and behavioral responses of adult D. suzukii (e.g.34,35).
The results from the first experiment involving synthetic compounds indicated positive responses of D. suzukii females (but not males) to β-cyclocitral and to isoamyl acetate when tested singly against the solvent mineral oil. Previously, Keesey et al.34 demonstrated that both male and female D. suzukii are sensitive physiologically and respond behaviorally to this compound. In turn, isoamyl acetate is present in the aroma of many fruit species and it was found in the headspace of the yeast Hanseniospora uvanum40, a species found in the alimentary canal of adult D. suzukii41. In the present study, isoamyl acetate was significantly more attractive to females than β-cyclocitral when these compounds were tested against each other. Females used in all experiments were sexually mature and presumably mated therefore those individuals were considered to have a high oviposition drive. Males, however, did not discriminate between the leaf and fruit compounds a result that suggests that both types of compounds may play a role in male attraction to host plants. Keesey et al.34 hypothesized that β-cyclocitral is used as a possible long-range cue in selectively attracting D. suzukii to the vicinity of a fruiting plants. The synergistic effect of β-cyclocitral and isoamyl acetate noted for males, but not for females, suggests that both compounds may be involved in host and mate finding and courtship behaviors28. Whether age or mating status influence the response of males and females to β-cyclocitral remains to be determined.
Isoamyl acetate did not increase significantly the response of males and females to either fruit juice. It is conceivable that this result may be due to redundancy, as isoamyl acetate was found to be present in several commercial fruit juices (J.C. Piñero et al. unpub. data). However, when β-cyclocitral was added to cherry juice it synergistically enhanced the response of females to the binary combination, a result that suggests that both foliage and fruit compounds may be needed to elicit high levels of response in females seeking for food or oviposition resources.
A variety of insect herbivores are indeed attracted to specific sorts of volatile chemicals, usually in an appropriate blend, emanating from their respective hosts. Within the sensory modality of olfaction, a quantitative dependent synergism between the fermentation materials acetic acid and acetoin in the response of D. suzukii was documented by Cha et al.33. Similar results were seen with the closely related D. melanogaster42, suggesting that the synergistic relationship between acetic acid and acetoin may be conserved in the melanogaster group. Synergistic interactions among components of host-plant odor have been reported in the Oriental fruit moth, Grapholita molesta (Busk)7 and in the grapevine moth, Lobesia botrana (Denis & Schiffermüller)43 (both Tortricidae). For the former species, Piñero and Dorn7 identified a bioactive 5-compound mixture composed of two constituents, green leaf volatiles and aromatic compounds, and reported that benzaldehyde and benzonitrile must be present in association with three distinct green leaf volatiles to produce an attractant effect on G. molesta females. In turn, Tasin et al.43 reported a strong synergistic effect of a blend of the grape volatiles β-caryophyllene, (E)-β-farnesene and (E)-4,8-dimethyl-1,3,7-nonatrieneon on female L. botrana attraction, and omission of any one compound from this 3-component blend almost abolished attraction.
Collectively, our findings (1) increase our understanding of male and female D. suzukii foraging behavior in response to synthetic compounds and natural juices as potential sources of attractants, and (2) support the conclusion that olfactory synergisms play a role in host finding behavior in D. suzukii, and such effects seem to apply differently to males and females. Improved efficacy of D. suzukii kairomonal lures is desirable to improve monitoring systems and potentially for the development of more effective semiochemically-based management.
Methods
Behavioral bioassays
All studies were conducted from 28 June to 7 August 2017 at the University of Hawaii Agricultural Experiment Station in Kainaliu, Hawaii Island, and from 10 May to 15 July 2018 at the University of Massachusetts, Amherst, Massachusetts. All experiments were conducted using screened cages (60 cm3) positioned inside a shade house (Kainaliu) and in the laboratory (Amherst). Four equidistant wires (15 cm in length) were suspended at each of the four corners of the cages, to hang the sources of volatiles.
Insects
Adult D. suzukii stemmed from colonies established at the Daniel K. Inouye U.S. Pacific Basin Agricultural Research Center, United States Department of Agriculture (USDA) Agricultural Research Service (ARS), Hilo, Hawaii Island, and at the University of Massachusetts (Amherst) campus. Fruit fly rearing followed procedures described by Follett et al.44. In short, immature stages were reared in 275-ml plastic containers with ventilated lids on diet that consisted of agar (13.5 g), water (1,500 ml), cornmeal (37.5 g), sugar (60 g), nutritional yeast (21 g), methyl paraben (1 g, dissolved in 10 ml of 90% ethanol), and propionic acid (12 ml). Rearing conditions were 24 °C (±2 °C) and a photoperiod of 12:12 (L:D) h. Upon adult emergence, adults were transferred to adult cages as needed to maintain a relatively constant adult density. Flies were provided water (one cup) and a 20% sugar/water solution (one cup) ad libitum. All flies were tested when they were 2–3 d old.
Sources of volatiles
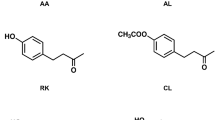
The synthetic compounds tested were β-cyclocitral (≥95%) and isoamyl acetate (≥95%) (Sigma-Aldrich Chemical Co., Milwaukee, WI). β-cyclocitral is a volatile isoprenoid present in green plant foliage including strawberry34, a host plant of D. suzukii. Isoamyl acetate is a compound present in ripening fruit35. Each compound was evaluated at 10−2 concentration based on Bolton et al. (unpub. data). Concord grape and tart cherry juices (Knudsen Just Juice, Knudsen & Sons, Inc., Chico, CA) were purchased from a local store.
For each experiment, 200 µL of each material (either, a particular synthetic compound at the 10−2 concentration in mineral oil, mineral oil alone, or undiluted fruit juice) were pipetted onto clear 1.5 ml centrifuge tubes. Prior to treatment application, the lids of the centrifuge tubes were removed, a 3 cm wire was wrapped around their neck, and a thin coating of Tangletrap insect coating (Tanglefoot Company, Grand Rapids, MI) was applied to the outer surface of the tubes to capture alighting flies. For all experiments, Tangletrap-coated tubes were placed in all four corners of the cage.
For experiment 1, which compared the flies’ response to two olfactory treatments (synthetic compound versus mineral oil as control), two centrifuge tubes loaded with a particular compound were hung from each of the two diagonally opposite corners in the cage and the other two corners received mineral oil. For experiments 2, 3, and 4, which involved evaluations of materials tested singly or in combination, two centrifuge tubes were fastened together using wire. One tube received a particular juice type (or synthetic compound), and the second tube was loaded with a particular synthetic compound. To account for the additional surface area, for the treatments that received only one treatment (either, a synthetic compound, a particular fruit juice, or the solvent mineral oil), a second, empty Tangletrap-coated tube was attached to the treated tube. Under this approach, each corner of the cage had two centrifuge tubes.
Experimental approach
For experiments 1, 3 and 4, observations took place between 7:00 a.m. and 12:00 p.m. in shade house (Kainaliu, HI). For experiment 2, trials were conducted from 8:00 to 12:00 pm in the laboratory (Amherst, MA). The ranges of temperature and relative humidity values during the observations were 20–26 °C and 65–85%, respectively for Kainaliu (HI), and 21–24 °C and 65–75%, respectively, for Amherst. For each trial, 10 males and 10 females were introduced into each cage using two glass vials (diam: 1.5 cm; height: 7 cm). The lid was removed to allow adult D. suzukii to disperse within the cage and acclimatize to cage conditions. Observations started about 20 min after fly release, as soon as the olfactory treatments were prepared and deployed. Each trial lasted 4 hours, time during which the number of males and females responding (i.e., landing on a centrifuge tube) was recorded. All responders were removed using an aspirator.
Experiment 1
This series of three tests evaluated the response of male and female D. suzukii to (1) β-cyclocitral and (2) isoamyl acetate, tested singly against mineral oil (solvent control) and (3) β-cyclocitral versus isoamyl acetate. Trials were replicated 10–11 times.
Experiment 2
This experiment examined the type of interactions existing between β-cyclocitral and isoamyl acetate in terms of attractiveness to male and female D. suzukii. The olfactory treatments that were evaluated were: (1) isoamyl acetate alone, (2) β-cyclocitral alone, (3) isoamyl acetate in association with β-cyclocitral, and (4) mineral oil (control). Trials were replicated 24 times.
Experiment 3
For this set of evaluations, we aimed at identifying the type of interactions existing between β-cyclocitral and isoamyl acetate and two types of fruit juices. More specifically, we quantified the response of male and females to (1) Concord grape juice, either alone or in combination with β-cyclocitral versus β-cyclocitral alone and mineral oil (control), and (2) grape juice, either alone or in combination with isoamyl acetate versus isoamyl acetate alone and mineral oil. The aforementioned approach was repeated in a separate set of trials using tart cherry juice instead of grape juice. Each trial was replicated 8 times.
Data analyses
For all experiments, the number of adult D. suzukii responding to the olfactory treatments over the 4-hour period were compared by two-way Analysis of Variance (ANOVA) with “treatment” and “sex” as main factors. Because a significant interaction was detected in 4/8 analyses (see Results section), we then subsequently compared, for each sex, the level of male and female response to the olfactory treatments using t-tests (Experiment 1) and one-way ANOVAS (Experiments 2 and 3). Data were transformed using √(x + 0.5) prior to analysis to stabilize variances. Means were separated, whenever appropriate, by a Fisher’s Least Significant Differences test at the 5% probability level. In addition, for experiment 3 we performed comparisons of ratios of interaction (ROIs)45,46 to examine type of interactions (additive or synergistic) among single (a particular plant volatile, a particular fruit juice) vs. 2-component odor treatments. In our case, a ROI = [(A + B) + control]/[(A) + (B)], where (A) represents adult SWD response to a particular plant volatile alone, (B) is the response of adults to a particular fruit juice alone, (A + B) denotes attraction to the combination of a particular plant volatile and a particular fruit juice, and control represents SWD responses recorded in vials having mineral oil. ROI values equal to 1 indicate additive effects. ROI values significantly greater than 1 indicate synergistic interactions between chemicals, and values significantly less than 1 denote inhibitory45,46. Analyses were done in STATISTICA 1347. All figures show untransformed data.
References
Smith, B. H. & Getz, W. M. Nonpheromonal olfactory processing in insects. Annu. Rev. Entomol. 39, 351–375, https://doi.org/10.1146/annurev.en.39.010194.002031 (1994).
Schoonhoven, L. M., Jermy, T. & van Loon, J. J. A. Insect-Plant Biology: from Physiology to Evolution. (Chapman and Hall, London, UK https://trove.nla.gov.au/version/45451272, 1998).
Carlsson, M. A. & Hansson, B. S. Plasticity and coding mechanisms in the insect antennal lobe. In Blomquist, G. J., Vogt, R. V. (Eds), Insect Pheromone Biochemistry and Molecular Biology. (Elsevier Academic Press, San Diego, CA, USA, 699–728, 2003).
Bruce, T. J. A. Insect host location: a volatile situation. Trends Plant Sci 10, 269–274, https://doi.org/10.1016/j.tplants.2005.04.003 (2005).
Visser, J. H. Host odour perception in phytophagous insects. Annu. Rev. Entomol. 31, 121–144, https://doi.org/10.1146/annurev.en.31.010186.001005 (1986).
Visser, J. H. & de Jong, R. Olfactory coding in the perception of semiochemicals. J. Chem. Ecol. 14, 2005–2018, https://doi.org/10.1007/BF01014246 (1988).
Piñero, J. C. & Dorn, S. Synergism between aromatic compounds and green leaf volatiles derived from the host plant underlies female attraction in the oriental fruit moth. Ent. Exp. Appl. 125, 185–194, https://doi.org/10.1111/j.1570-7458.2007.00614.x (2007).
Kanzawa, T. Studies on Drosophila suzukii Mats. Kofu, Yamanashi agricultural experiment station 49 pp. Abstract in Review of Appl. Entomol. 29, 622 https://www.cabdirect.org/cabdirect/abstract/19410501073 (1939).
Asplen, M. K. et al. Invasion biology of spotted wing Drosophila (Drosophila suzukii): a global perspective and future priorities. J. Pest Sci. 88, 469–494, 10.1007%2Fs10340-015-0681-z (2015).
Arriaga, J. T. Phytosanitary Alert System: Detection of spotted winged drosophila (Drosophila suzukii Matsumura) in the Municipality of Los Reyes, State of Michoacan, Mexico. North American Plant Protection Organization’s (NAPPO), https://www.pestalerts.org/oprDetail.cfm?oprID=507 (2011).
Hauser, M. A historic account of the invasion of Drosophila suzukii (Matsumura) (Diptera: Drosophilae) in the continental United States, with remarks on their identification. Pest Manag. Sci. 67, 1352–1357, https://doi.org/10.1002/ps.2265 (2011).
Lasa, R. & Tadeo, E. Invasive Drosophilid Pests Drosophila suzukii and Zaprionus indianus (Diptera: Drosophilidae) in Veracruz, Mexico. Fla Entomol. 98, 987–988, https://doi.org/10.1653/024.098.0332 (2015).
Deprá, M., Poppe, J. L., Schmitz, H. J., De Toni, D. C. & Valente, V. L. S. The first records of the invasive pest Drosophila suzukii in the South American continent. J. Pest Sci. 87, 379–383, https://doi.org/10.1007/s10340-014-0591-5 (2014).
Sandatino, M. V. et al. Primer registro de Drosophila suzukii (Diptera: Drosophilidae) asociado al cultivo de arándanos (Vaccinium spp.) de Argentina. Rev. Soc. Entomol. Argent. 74, 183–185, https://www.redalyc.org/articulo.oa?id=322043152009 (2015).
Cini, A., Loriatti, C. & Anfora, G. A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull. Insectol. 65, 149–160, http://www.bulletinofinsectology.org/pdfarticles/vol65-2012-149-160cini.pdf (2012).
Calabria, G., Máca, J., Bächli, G., Serra, L. & Pascual, M. First records of the potential pest species Drosophila suzukii (Diptera: Drosophilidae) in Europe. J Appl. Entomol. 136, 139–147, https://doi.org/10.1111/j.1439-0418.2010.01583.x (2012).
Goodhue, R. E., Bolda, M., Farnsworth, D., Williams, J. C. & Zalom, F. G. Spotted wing drosophila infestation of California strawberries and raspberries: economic analysis of potential revenue losses and control costs. Pest Manag. Sci. 67, 1396–1402, https://doi.org/10.1002/ps.2259 (2011).
Farnsworth, D. et al. Economic analysis of revenue losses and control costs associated with the spotted wing drosophila, Drosophila suzukii (Matsumura), in the California raspberry industry. Pest Manag. Sci. 73, 1083–1090, https://doi.org/10.1002/ps.4497 (2017).
Haviland, D. R. & Beers, E. H. Chemical Control Programs for Drosophila suzukii that Comply with International Limitations on Pesticide Residues for Exported Sweet Cherries. J Integr. Pest Manag. 3, F1–F6, https://doi.org/10.1603/IPM11034 (2012).
Van Timmeren, S. & Isaacs, R. Control of spotted wing drosophila, Drosophila suzukii, by specific insecticides and by conventional and organic crop protection programs. Crop Prot. 54, 126–33, https://doi.org/10.1016/j.cropro.2013.08.003 (2013).
Diepenbrock, L. M., Rosensteel, D. O., Hardin, J. A., Sial, A. A. & Burrack, H. J. Season-long programs for control of Drosophila suzukii in southeastern US blueberries. Crop Prot. 81, 76–84, https://doi.org/10.1016/j.cropro.2015.12.012 (2016).
Diepenbrock, L. M., Hardin, J. A. & Burrack, H. J. Season-long programs for control of Drosophila suzukii in southeastern US blackberries. Crop Prot. 98, 149–156, https://doi.org/10.1016/j.cropro.2017.03.022 (2017).
Tonina, L. et al. Comparison of attractants for monitoring Drosophila suzukii in sweet cherry orchards in Italy. J. Appl. Entomol. 142, 18–25, https://doi.org/10.1111/jen.12416 (2018).
Burrack, H. J., Fernandez, G. E., Spivey, T. & Kraus, D. A. Variation in selection and utilization of host crops in the field and laboratory by Drosophila suzukii Matsumura (Diptera: Drosophilidae), an invasive frugivore. Pest Mgmt. Sci. 69, 1173–1180, https://doi.org/10.1002/ps.3489 (2013).
Kenis, M. et al. Non-crop plants used as hosts by Drosophila suzukii in Europe. J. Pest Sci. 89, 735–748, https://doi.org/10.1007/s10340-016-0755-6 (2016).
Wiman, N. G. et al. Integrating temperature-dependent life table data into a matrix projection model for Drosophila suzukii population estimation. PLOS ONE 9(9), e106909, https://doi.org/10.1371/journal.pone.0106909 (2014).
Burrack, H. J. et al. Multistate comparison of attractants for monitoring Drosophila suzukii (Diptera: Drosophilidae) in blueberries and caneberries. Environ. Entomol. 44, 704–712, https://doi.org/10.1093/ee/nvv022 (2015).
Cloonan, K. R., Abraham, J., Angeli, S., Syed, Z. & Rodriguez-Saona., C. Advances in the chemical ecology of the spotted wing Drosophila (Drosophila suzukii) and its applications. J. Chem. Ecol. 44, 922–939, https://doi.org/10.1007/s10886-018-1000-y (2018).
Cha, D. H. et al. A four-component synthetic attractant for Drosophila suzukii (Diptera: Drosophilidae) isolated from fermented bait headspace. Pest Manag. Sci. 70, 324–331, https://doi.org/10.1002/ps.3568 (2014).
Cha, D. H. et al. Simpler is better: fewer non-target insects trapped with a four-component chemical lure vs. a chemically more complex food-type bait for Drosophila suzukii. Ent. Exp. Appl. 154, 251–260, https://doi.org/10.1111/eea.12276 (2015).
Frewin, A. J., Renkema, J., Fraser, H. & Hallett, R. H. Evaluation of attractants for monitoring Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Entomol. 110, 1156–1163, https://doi.org/10.1093/jee/tox081 (2017).
Jaffe, B. D. et al. Multistate comparison of attractants and the impact of fruit development stage on trapping Drosophila suzukii (Diptera: Drosophilidae) in raspberry and blueberry. J. Econ. Entomol. 47, 935–945, https://doi.org/10.1093/ee/nvy052 (2018).
Cha, D. H., Landolt, P. J. & Adams, T. B. Effect of chemical ratios of a microbial-based feeding attractant on trap catch of Drosophila suzukii (Diptera: Drosophilidae). Environ. Entomol. 46, 907–915, https://doi.org/10.1093/ee/nvx079 (2017).
Keesey, I. A., Knaden, M. & Hansson, B. S. Olfactory specialization in Drosophila suzukii supports an ecological shift in host preference from rotten to fresh fruit. J. Chem. Ecol. 41, 121–128, https://doi.org/10.1007/s10886-015-0544-3 (2015).
Revadi, S. et al. Olfactory responses of Drosophila suzukii females to host plant volatiles. Physiol. Entomol. 40, 54–64, https://doi.org/10.1111/phen.12088 (2015).
Abraham, J. et al. Behavioral and antennal responses of Drosophila suzukii (Diptera: Drosophilidae) to volatiles from fruit extracts. Environ. Entomol. 44, 356–367, https://doi.org/10.1093/ee/nvv013 (2015).
Stökl, J. et al. A deceptive pollination system targeting drosophilids through olfactory mimicry of yeast. Curr. Biol. 20, 1846–1852, https://doi.org/10.1016/j.cub.2010.09.033 (2010).
Feng, Y., Bruton, R., Park, A. & Zhang, A. Identification of attractive blend for spotted wing drosophila, Drosophila suzukii, from apple juice. J. Pest Sci. 91, 1251–1267, https://doi.org/10.1007/s10340-018-1006-9 (2018).
Saglam, H., Gozler, T., Kivcak, B., Demirci, B. & Baser, K. H. C. Volatile compounds from Haplophyllum myrtifolium. Chem. Nat. Comp. 37, 442–444, https://doi.org/10.1023/A:1014467225815 (2001).
Scheidler, N. H., Liu, C., Hamby, K. A., Zalom, F. G. & Syed, Z. Volatile codes: Correlation of olfactory signals and reception in Drosophila-yeast chemical communication. Sci. Rep. 5, 14059, https://doi.org/10.1038/srep14059 (2015).
Hamby, K. A., Hernandez, A., Boundy-Mills, K. & Zalom, F. G. Associations of yeasts with spotted-wing drosophila (Drosophila suzukii; Diptera: Drosophilidae) in cherries and raspberries. Appl. Environ. Microbiol. 78, 4869–4873, https://doi.org/10.1128/AEM.00841-12 (2012).
Becher, P. G. et al. Yeast, not fruit volatiles, mediate Drosophila melanogaster attraction, oviposition and development. Funct. Ecol. 26, 822–828, https://doi.org/10.1111/j.1365-2435.2012.02006.x (2012).
Tasin, M. et al. Synergism and redundancy in a plant volatile blend attracting grapevine moth females. Phytochem. 68, 203–209, https://doi.org/10.1016/j.phytochem.2006.10.015 (2007).
Follett, P. A., Swedman, A. & Price, D. K. Postharvest irradiation treatment for quarantine control of Drosophila suzukii (Diptera: Drosophilidae) in fresh commodities. J. Econ. Entomol. 107, 964–969, https://doi.org/10.1603/EC14006 (2014).
Hammack, L. Corn volatiles as attractants for northern and western corn rootworm beetles (Coleoptera: Chrysomelidae: Diabrotica spp.). J. Chem Ecol. 22, 1237–1253, https://doi.org/10.1007/BF02266963 (1996).
Piñero, J. C. & Prokopy, R. J. Field evaluation of plant odor and pheromonal combinations for attracting plum curculios. J. Chem. Ecol. 29, 2735–2748, https://doi.org/10.1023/B:JOEC.0000008017.16911.aa (2003).
TIBCO Software Inc. Statistica (data analysis software system), version 13, http://statistica.io (2017).
Acknowledgements
We thank Nicole Foley and Natalie DiDomenico (University of Massachusetts, Amherst) for technical assistance. This material is based upon work supported by the National Institute of Food and Agriculture (NIFA), U.S. Department of Agriculture (USDA) under project number MAS 00522, the Center for Agriculture, Food and the Environment and the Stockbridge School of Agriculture at University of Massachusetts Amherst. Additional support was provided by an USDA – Agricultural Research Service 1890 Faculty Research Fellowship to J.P. (project # 58-2040-7-002). The contents are solely the responsibility of the authors and do not necessarily represent the official views of the USDA or NIFA.
Author information
Authors and Affiliations
Contributions
J.C.P., B.A.B., and P.A.F. conceived the experiments; J.C.P. and G.B. conducted the experiments; P.A.F. and B.A.B. provided experimental insects; J.C.P. and P.A.F. analyzed the results. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Piñero, J.C., Barrett, B.A., Bolton, L.G. et al. β-cyclocitral synergizes the response of adult Drosophila suzukii (Diptera: Drosophilidae) to fruit juices and isoamyl acetate in a sex-dependent manner. Sci Rep 9, 10574 (2019). https://doi.org/10.1038/s41598-019-47081-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47081-z
- Springer Nature Limited
This article is cited by
-
Yeast species affects feeding and fitness of Drosophila suzukii adults
Journal of Pest Science (2020)