Abstract
Two novel New Delhi metallo-β-lactamase-1 (NDM-1)-positive plasmids containing a complete composite transposon, Tn125, from two respective Acinetobacter towneri isolates were characterized. Plasmid pNDM-GJ01 (30,293 bp) isolated from A. towneri G165 did not show homology to any known plasmid structure, except for the transposon Tn125 containing bla NDM-1. A novel repB gene and two XRE-type transcriptional regulators were found in pNDM-GJ01. Plasmid pNDM-GJ02 (62,011 bp) isolated from A. towneri G295 showed the highest homology to pBJAB0715 (41% coverage, 99% nucleotide identity). In addition to the bla NDM-1-harbouring transposon Tn125, pNDM-GJ02 also had an IS26-composite transposon, which contains ISCR1 and two class 1 integrons carrying different cassette arrays. Both clinical isolates were highly resistant to β-lactams and susceptible to tigecycline and colistin. Ten other resistance genes were detected in G295, and one other resistance gene was detected in G165. No transconjugant was obtained from any of the donors by broth and filter mating. The emergence of these two novel plasmids carrying NDM-1 in Acinetobacter spp., pNDM-GJ01 and pNDM-GJ02, suggests Tn125 mobile integration.
Similar content being viewed by others
Introduction
The emergence of the bla NDM-1 gene, which confers gram-negative bacteria with antibiotic resistance, has gained worldwide attention owing to its recent and rapid spread1,2,3. New Delhi metallo-β-lactamase-1 (NDM-1)-positive plasmids have accelerated the process of spread because of their potential for mobility and the fact that they carry multiple resistance genes. Pfeifer et al. first reported the genetic context of bla NDM-1 in a clinical Acinetobacter baumannii 161/07 isolate discovered in a German hospital in 20074. The bla NDM-1 gene was integrated on a new transposon structure (Tn125) flanked by two insertion elements of ISAba125. Subsequently, Tn125 harbouring bla NDM-1 was found on chromosomes in several multiple-resistant Acinetobacter spp. isolates throughout Europe5, 6. Furthermore, in 2011, Hu et al. reported a bla NDM-1-bearing plasmid, pNDM-BJ01 (GenBank: JQ001791), isolated from a clinical A. lwoffii strain in China7. This plasmid carried a complete Tn125 transposon and showed high horizontal transfer ability7. Since then, several NDM-1-positive plasmids have been isolated from Acinetobacter spp. in China, which showed a similar structure to pNDM-BJ018, 9. These pNDM-BJ01-like plasmids have contributed to the spread of bla NDM-1 in Acinetobacter spp. Here, we report the complete sequences of two novel plasmids, pNDM-GJ01 and pNDM-GJ02, isolated from clinical Acinetobacter towneri strains that do not share any similar structure with pNDM-BJ01 except for Tn125 harbouring the bla NDM-1 gene. Both plasmids were characterized and their complete sequences were elucidated.
Results
Species identification
Strains G165 and G295 were identified as Kocuria varians (55%) and Acinetobacter sp. (90%) respectively by Vitek® 2 system. Both isolates were identified as Acinetobacter sp. by 16S-23S rRNA gene intergenic spacer region sequence comparisons, and identified as Acinetobacter towneri by analyzing results of the partial rpoB sequence10, 11.
Antimicrobial data
The two clinical strains showed similar susceptibility profiles: they were resistant to broad-spectrum cephalosporins, carbapenems, fluoroquinolones, and sulfamethoxazole. However, they showed a slight difference in susceptibility to aminoglycosides: G165 was resistant to amikacin, and was intermediate to gentamicin and tobramycin; G295 was resistant to amikacin and tobramycin, and was intermediate to gentamicin. Furthermore, both strains were susceptible to tigecycline and colistin, as well as to tetracycline and aztreonam (Table 1)12.
Metallo-beta lactamase (MBL) detection with Etest MBL strips (bioMérieux, France) was positive for both strains. Apart from bla NDM-1, 10 other resistance genes, including bla PSE-1, bla VIM-11, bla IMP-1, bla OXA-58, sul1, sul2, tetA, aacC1 gene, aacC3, and aacA4, were detected in G295, whereas only one other resistance gene, tetA, was detected in G165.
Transconjugation assays
No transconjugants were obtained for the two plasmids, in spite of using three kinds of recipients and applying both methods repeatedly. Subsequent plasmid analysis showed that these two plasmids did not carry the element required to initiate the transconjugation.
pNDM-GJ01 characteristics
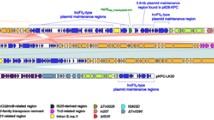
Plasmid pNDM-GJ01 from the isolate G165 was 30,293 bp long and contained 35 predicted coding sequences with an average GC content of 43.4% (Fig. 1). Three different functional regions were predicted: variable region I containing the complete composite transposon Tn125, a replication region, and variable region II containing multiple truncated transposable elements (Fig. 1). Plasmid pNDM-GJ01 was believed to contain a novel structure, as a BLAST search showed that it had no identical structure to previously released plasmids in NCBI, with the exception of Tn125. In addition, the plasmid pNDM-GJ01 could not be classified using the PCR-based replicon typing method (PBRT)13.
Circular maps of plasmids pNDM-GJ01. Starting from the outside, the first outer circle contains the coordinates of the complete plasmid circle and open reading frames (ORFs). The white arrows represent the direction of transcription. In addition, coding sequences, with known gene names reported in the NCBI database, are indicated by grey arrows. The second circle indicates the functional sequence blocks. The third circle indicates the GC content of plasmids, and the innermost circle indicates the GC-skew of the plasmids.
The complete composite transposon Tn125 was found in variable region I. The Tn125 sequence was relatively conserved without loss of the downstream segment, showing 99% sequence similarity with that of pNDM-BJ01 (Fig. 1). The gene aphA6 was not detected in the 5′-flanking region of Tn125 as in pNDM-BJ01, but the resolvase was retained in the 3′-flanking region7. The GC content of Tn125 was 61% and that of the whole plasmid was 43%, which indicated that the emergence of the NDM-1-harbouring novel plasmid is related to Tn125 integration and acquisition. Moreover, the target site duplications of Tn125 were not found in the plasmid; the adjacent base of the left inverted repeat (IRL) sequence of upstream ISba125 was CCT and the adjacent base of the right inverted repeat (IRR) sequence of downstream ISba125 was GTT (Fig. 2). The replication region contained a DEAD/DEAH box helicase gene, the plasmid-partitioning gene parA, and the repB gene (Fig. 1). The repB gene showed high identity with many sequences encoding an initiator RepB protein from the genome of Acinetobacter spp. published in the NCBI database: it showed 99% amino acid identity with a hypothetical protein from A. towneri strain KCTC 12419 (NCBI Reference Sequence: WP_004968874.1) and 98% amino acid identity with an initiator RepB protein from Acinetobacter sp. YZS-X1–1 (NCBI Reference Sequence: WP_034171688.1), which was isolated from the same region as strain G165 (Guangzhou, China). These novel classes of replication proteins are of great significance for plasmid replication and stability13, 14. The variable region contained multiple truncated transposable elements and regulatory factors (Fig. 1). An unknown insertion sequence with the downstream fragment missing and IS1006 may be involved in multiple transposition events. Two regulators (XRE family transcriptional regulator and Cro/Cl family transcriptional regulator) were also found in this region, which is related to the conjugation of plasmids and genomic islands15.
Features of transposon Tn125 and its target site duplications. Arrows indicate genes and their transcription orientations, and all regions are not drawn to scale. Similar structures and high sequence homology are shown by the dotted line. The 3-bp Tn125 target sites in each plasmid are indicated by thin arrows. Un1 and Un2 indicate unknown reading frames encoding hypothetical proteins, and res indicates a gene encoding resolvase.
pNDM-GJ02 characteristics
Plasmid pNDM-GJ02 from the isolate G295 was 62,011 bp long and contained 58 predicted coding sequences with an average GC content of 45.4% (Fig. 3). Four different functional regions were predicted: variable region I, variable region II, variable region III, and a replication region (Fig. 3). Plasmid pNDM-GJ02 showed the highest similarity to the plasmid pBJAB0715 (41% coverage and 99% nucleotide identity; GenBank: CP003848.1). Based on these results, we concluded that the emergence of pNDM-GJ02 has involved multiple recombination events (Fig. 3). Plasmid pNDM-GJ02 also could not be classified using the PBRT method13.
In variable region I, the complete composite transposon Tn125 was detected. The Tn125 sequence was also relatively conserved without loss of the downstream segment, showing 99% sequence similarity with that of pNDM-BJ01 (Fig. 3). Tn125 was inserted into the gene from pBJAB0715 (GenBank: CP003848.1), which encoded a protein of unknown function. The GC content of Tn125 was 61% and that of the whole plasmid was 45%, which indicated that the emergence of the NDM-1-harbouring novel plasmid was related to Tn125 integration and acquisition. The 3-bp (ATT) target site duplications of Tn125 were found in this plasmid (Fig. 2).
In variable region II, the composite transposon flanked by IS26 was found to be bracketed by two copies of the same insertion sequence with opposite directions (Fig. 3). The class 1 integron was found in both the upstream and downstream regions of the transposon. The gene cassette of the upstream integron was aar-3-aacA4-qacEdelta1-sul1, and the integrase gene was truncated by the IS26 insertion sequence. The downstream integron had a typical structure of 5ʹ- and 3′-conserved regions and contained a bla PSE-1 gene cassette. A specific type of transposon (ISCR1) was found between the two integrons, which has previously been shown to be able to transfer its adjacent sequences and structures (Fig. 3)16.
Variable region III comprised multiple transposable elements, regulatory factors, and transport proteins. The resolvase TnpR and the transposase TnpA were detected in the variable region, of which the 5′-flanking region showed a transport structure encoding an MFS family transport permease, channel protein, and outer membrane protein. In addition, genes encoding multiple regulation proteins were identified in this region, including a universal stress protein, antitoxin components belonging to the toxin and antitoxin system, and AraC family transcription factors (Fig. 3).
The replication region contained replication-related functional genes as well as several transposable elements, which may be related to the multiple recombination events of the plasmid. The rep gene was not found, and only a gene encoding a novel partitioning protein, parB, was observed in this region. Moreover, we also found genes encoding multiple enzymes associated with plasmid replication and distribution, including an ATP enzyme, DNA polymerase V, and deoxyribonuclease 7 subunits (xseA).
Discussion
Two novel plasmids, pNDM-GJ01 and pNDM-GJ02, containing NDM-1 were found from two A. towneri isolates in China. The plasmids showed a novel structure and contained a complete composite transposon, Tn125. A BLAST search showed no homology with any known plasmid structure in the NCBI database to pNDM-GJ01, except for the transposon Tn125 containing bla NDM-1. A region showing high sequence similarity to the plasmid pBJAB0715 (41% coverage, 99% nucleotide identity) and two complete composite transposons (Tn125, IS26) were found in pNDM-GJ02. It was concluded that the emergence of novel plasmids carrying NDM-1 in Acinetobacter spp. is the result of Tn125 mobile integration involving multiple recombination events.
In Acinetobacter spp., transposon Tn125 appeared to be the main vehicle for dissemination of bla NDM-1 5. In Enterobacteriaceae, the complete Tn125 and multiple residual Tn125-like structures were found in NDM-1-positive plasmids, they appeared with related mobile factors, such as IS26, IS903, ISKpn14, IS1, ISEc33, and Tn3, which participated in the transfer and integration of bla NDM-1 between different plasmids17,18,19,20,21. These results suggest that the bla NDM-1 gene is spread by transposable elements between different plasmids, and is then further spread in multiple bacteria via plasmids. Over the past two years, an increasing number of reports associated with Enterobacteriaceae carrying NDM-1 have begun to emerge continuously in China22,23,24,25. Given the trend of the spread of the bla NDM-1 gene in Enterobacteriaceae, the potential threat of these dangerous resistant bacteria has not been eliminated. Therefore, further effort should be devoted to detecting the prevalence and spread of bla NDM-1 in China.
Materials and Methods
Ethics statement
All volunteers provided written, informed consent to participate in this study, which was reviewed and approved by the ethics committee of the Academy of Military Medical Sciences, China. All experiments were performed in accordance with relevant guidelines and regulations.
Bacterial isolates and patient history
A. towneri G165 and G295 were isolated in 2011 as part of the continuous surveillance on the infection status of NDM-1-producing bacteria in China26. Both samples were collected from outpatients. The first case involved a 50-year-old female patient suffering from recurring urinary tract infection. The second case involved a 30-year-old male patient with suspected chronic appendicitis. Both patients received antibiotics and had not travelled to India. Species identification of the isolates was carried out using a Vitek® 2 system (bioMérieux, France), 16S–23S rRNA gene intergenic spacer sequencing, and partial rpoB sequence analysis10, 11.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was performed by the microbroth dilution method according to the Clinical and Laboratory Standards Institute (CLSI) guidelines. CLSI 2016 breakpoints were applied (M100-S26)12.
Detection of MBLs was performed with Etest MBL strips (AB bioMérieux, St. Louis, MO, USA) containing IMP (IP) and IMP + EDTA (IPI). Strips were used according to instructions provided by the manufacturer.
Molecular detection of β-lactamase production
The putative carbapenemase, ESBL, and AmpC resistance genes were screened by PCR27,28,29,30. Aminoglycoside resistance genes were also detected by PCR, using the oligonucleotides listed in Supplementary Data Table S1 31. Positive PCR results were confirmed by direct sequencing. Furthermore, all original contigs from the plasmids were searched against the nucleotide database from NCBI to find resistance genes.
Transconjugation assays
The horizontal transfer capability of the bla NDM-1 gene was assessed by broth and filter mating using a standard Escherichia coli J53 azide-resistant strain, Shigella flexneri 2a 301, and Salmonella Paratyphi A CMCC 50973 as the recipients2. The donor/recipient ratio was 10:1 and the temperature was 30 °C. MacConkey agar containing 100 mg/L sodium azide and 0.5 mg/L meropenem was used to select for E. coli J53 transconjugants. Both SS agar and XLD media (BD Difco, USA) with 0.5 mg/L meropenem were chosen to select for S. flexneri and S. Paratyphi transconjugants. Putative transconjugants were confirmed by detection of bla NDM-1 with PCR as described above.
Plasmid DNA sequencing, annotation, and analysis
Plasmid DNA isolation and purification was performed with the BAC/PAC DNA Kit (Omega Biotek, Norcross, GA, USA) according to the manufacturer’s protocol. Single-end pyrosequencing reads of plasmids were generated using the Ion Torrent™ sequencing platform32. Raw reads were first assembled into contigs using Newbler version 2.9, followed by gap filling by local assembly. To ensure accuracy, the raw reads were mapped onto the assembled complete genomes to detect the mis-assembly and low quality regions. Then the genomes were revised and the low quality regions were further confirmed by overlap PCR followed by Sanger sequencing. Each assembled genome was annotated with the Rapid Annotations using Subsystems Technology (RAST) server and verified with the Basic Local Alignment Search Tool (BLAST) against the non-redundant NCBI database (http://blast.ncbi.nlm.nih.gov/Blast.cgi)33.
Nucleotide accession numbers
The complete sequences of pNDM-GJ01 and pNDM-GJ02 have been deposited in GenBank (accession numbers KT965092 and KT965093, respectively).
References
Kumarasamy, K. K. et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. The Lancet. Infectious diseases 10, 597–602, doi:10.1016/s1473-3099(10)70143-2 (2010).
Walsh, T. R., Weeks, J., Livermore, D. M. & Toleman, M. A. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: an environmental point prevalence study. The Lancet. Infectious diseases 11, 355–362, doi:10.1016/s1473-3099(11)70059-7 (2011).
Berrazeg, M. et al. New Delhi Metallo-beta-lactamase around the world: an eReview using Google Maps. Euro surveillance: bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin 19 (2014).
Pfeifer, Y. et al. Molecular characterization of blaNDM-1 in an Acinetobacter baumannii strain isolated in Germany in 2007. The Journal of antimicrobial chemotherapy 66, 1998–2001, doi:10.1093/jac/dkr256 (2011).
Poirel, L. et al. Tn125-related acquisition of blaNDM-like genes in Acinetobacter baumannii. Antimicrobial agents and chemotherapy 56, 1087–1089, doi:10.1128/aac.05620-11 (2012).
Bonnin, R. A. et al. Dissemination of New Delhi metallo-beta-lactamase-1-producing Acinetobacter baumannii in Europe. Clinical microbiology and infection: the official publication of the European Society of Clinical Microbiology and Infectious Diseases 18, E362–365, doi:10.1111/j.1469-0691.2012.03928.x (2012).
Hu, H. et al. Novel plasmid and its variant harboring both a bla(NDM-1) gene and type IV secretion system in clinical isolates of Acinetobacter lwoffii. Antimicrobial agents and chemotherapy 56, 1698–1702, doi:10.1128/aac.06199-11 (2012).
Fu, Y. et al. Epidemiological characteristics and genetic structure of blaNDM-1 in non-baumannii Acinetobacter spp. in China. The Journal of antimicrobial chemotherapy 67, 2114–2122, doi:10.1093/jac/dks192 (2012).
Zhang, W. J. et al. Complete sequence of the bla(NDM-1)-carrying plasmid pNDM-AB from Acinetobacter baumannii of food animal origin. The Journal of antimicrobial chemotherapy 68, 1681–1682, doi:10.1093/jac/dkt066 (2013).
Chang, H. C. et al. Species-level identification of isolates of the Acinetobacter calcoaceticus-Acinetobacter baumannii complex by sequence analysis of the 16S–23S rRNA gene spacer region. Journal of clinical microbiology 43, 1632–1639 (2005).
La Scola, B., Gundi, V. A., Khamis, A. & Raoult, D. Sequencing of the rpoB gene and flanking spacers for molecular identification of Acinetobacter species. Journal of clinical microbiology 44, 827–832 (2006).
Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing: Twenty-sixth Informational Supplement M100-S26. CLSI, Wayne, PA, USA 2016.
Bertini, A. et al. Characterization and PCR-based replicon typing of resistance plasmids in Acinetobacter baumannii. Antimicrobial agents and chemotherapy 54, 4168–4177, doi:10.1128/aac.00542-10 (2010).
Robinson, A. et al. Essential biological processes of an emerging pathogen: DNA replication, transcription, and cell division in Acinetobacter spp. Microbiology and Molecular Biology Reviews 74, 273–297 (2010).
López-Fuentes, E., Torres-Tejerizo, G., Cervantes, L. & Brom, S. Genes encoding conserved hypothetical proteins localized in the conjugative transfer region of plasmid pRet42a from Rhizobium etli CFN42 participate in modulating transfer and affect conjugation from different donors. Frontiers in microbiology 5 (2014).
Toleman, M. A., Bennett, P. M. & Walsh, T. R. ISCR elements: novel gene-capturing systems of the 21st century? Microbiology and Molecular Biology Reviews 70, 296–316 (2006).
Sekizuka, T. et al. Complete sequencing of the bla(NDM-1)-positive IncA/C plasmid from Escherichia coli ST38 isolate suggests a possible origin from plant pathogens. PloS one 6, e25334, doi:10.1371/journal.pone.0025334 (2011).
Villa, L., Poirel, L., Nordmann, P., Carta, C. & Carattoli, A. Complete sequencing of an IncH plasmid carrying the blaNDM-1, blaCTX-M-15 and qnrB1 genes. The Journal of antimicrobial chemotherapy 67, 1645–1650, doi:10.1093/jac/dks114 (2012).
Poirel, L., Bonnin, R. A. & Nordmann, P. Analysis of the resistome of a multidrug-resistant NDM-1-producing Escherichia coli strain by high-throughput genome sequencing. Antimicrobial agents and chemotherapy 55, 4224–4229, doi:10.1128/aac.00165-11 (2011).
Ho, P. L. et al. Complete sequencing of pNDM-HK encoding NDM-1 carbapenemase from a multidrug-resistant Escherichia coli strain isolated in Hong Kong. PloS one 6, e17989, doi:10.1371/journal.pone.0017989 (2011).
Ho, P. L. et al. Identification and characterization of a novel incompatibility group X3 plasmid carrying bla NDM-1 in Enterobacteriaceae isolates with epidemiological links to multiple geographical areas in China. Emerging microbes & infections 1, e39, doi:10.1038/emi.2012.37 (2012).
Wang, X. et al. An outbreak of a nosocomial NDM-1-producing Klebsiella pneumoniae ST147 at a teaching hospital in mainland China. Microbial drug resistance (Larchmont, N.Y.) 20, 144–149, doi:10.1089/mdr.2013.0100 (2014).
Zhou, G. et al. NDM-1-producing strains, family Enterobacteriaceae, in hospital, Beijing, China. Emerging infectious diseases 20, 340–342, doi:10.3201/eid2002.121263 (2014).
Chen, Z. et al. First report in China of Enterobacteriaceae clinical isolates coharboring blaNDM-1 and blaIMP-4 drug resistance genes. Microbial drug resistance (Larchmont, N.Y.) 21, 167–170, doi:10.1089/mdr.2014.0087 (2015).
Liu, Y. et al. First description of NDM-1-, KPC-2-, VIM-2- and IMP-4-producing Klebsiella pneumoniae strains in a single Chinese teaching hospital. Epidemiology and infection 143, 376–384, doi:10.1017/s0950268814000995 (2015).
Wang, X. et al. High rate of New Delhi metallo-beta-lactamase 1-producing bacterial infection in China. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 56, 161–162, doi:10.1093/cid/cis782 (2013).
Dijkshoorn, L., Nemec, A. & Seifert, H. An increasing threat in hospitals: multidrug-resistant Acinetobacter baumannii. Nature Reviews Microbiology 5, 939–951 (2007).
Hujer, K. M. et al. Analysis of Antibiotic Resistance Genes in Multidrug-Resistant Acinetobacter sp. Isolates from Military and Civilian Patients Treated at the Walter Reed Army Medical Center. Antimicrobial agents and chemotherapy 50, 4114–4123 (2006).
Denisuik, A. J. et al. Molecular epidemiology of extended-spectrum β-lactamase-, AmpC β-lactamase-and carbapenemase-producing Escherichia coli and Klebsiella pneumoniae isolated from Canadian hospitals over a 5 year period: CANWARD 2007–11. Journal of Antimicrobial Chemotherapy 68, i57–i65 (2013).
Österblad, M. et al. Carbapenemase-producing Enterobacteriaceae in Finland: the first years (2008–11). Journal of Antimicrobial Chemotherapy 67, 2860–2864 (2012).
Akers, K. S. et al. Aminoglycoside resistance and susceptibility testing errors in Acinetobacter baumannii-calcoaceticus complex. Journal of clinical microbiology 48, 1132–1138 (2010).
Merriman, B., Torrent, I., Rothberg, J. M. & Team, D. Progress in ion torrent semiconductor chip based sequencing. Electrophoresis 33, 3397–3417 (2012).
Aziz, R. K. et al. The RAST Server: rapid annotations using subsystems technology. BMC genomics 9, 75 (2008).
Acknowledgements
This work received support from the Beijing Natural Science Foundation (grant number 7164289), National Natural Science Foundation of China (grant number 81373077).
Author information
Authors and Affiliations
Contributions
H.L.Y. and Y.J. designed experiments; Z.D.Y., L.W., Y.Z. and D.D.R. carried out experiments; H.S.M., H.X.M., A.D. and L.N.W. analyzed experimental results; H.Y. and T.Y.G. analyzed sequencing data; W.Y. and W.S.S. collected hospital patient information; Z.D.Y. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zou, D., Huang, Y., Liu, W. et al. Complete sequences of two novel bla NDM-1-harbouring plasmids from two Acinetobacter towneri isolates in China associated with the acquisition of Tn125 . Sci Rep 7, 9405 (2017). https://doi.org/10.1038/s41598-017-09624-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-09624-0
- Springer Nature Limited
This article is cited by
-
Molecular mechanism of plasmid-borne resistance to sulfonamide antibiotics
Nature Communications (2023)
-
Abundance of mobile genetic elements in an Acinetobacter lwoffii strain isolated from Transylvanian honey sample
Scientific Reports (2020)
-
What do we know about plasmids carried by members of the Acinetobacter genus?
World Journal of Microbiology and Biotechnology (2020)