Abstract
This study aimed to provide insights into the relationship between lipid levels and new-onset diabetes (NOD) in 14,864 Chinese hypertensive patients without diabetes (6056 men and 8808 women) aged 45–75 years from the China Stroke Primary Prevention Trial (CSPPT, led by Nanfang Hospital, Guangzhou, China). NOD (defined as fasting plasma glucose (FPG) ≥ 7.0 mmol/L at the end of study or self-reported physician diagnosis of diabetes or self-reported use of hypoglycemic agents during follow-up) was analyzed using multivariate analysis. Follow-up was censored on August 24, 2014. Among the 14,864 subjects, 1615 developed NOD (10.9%, men = 10.8% and women = 10.9%). Increased triglycerides (TG) [odds ratio (OR) = 1.18; 95% confidence interval (CI): 1.13–1.25, P < 0.001], TG/HDL (OR = 1.12; 95%CI: 1.08–1.17, P < 0.001), and decreased high density lipoprotein (HDL) (OR = 0.79; 95%CI: 0.67–0.93, P = 0.005) were associated with NOD, independently from age, gender, body mass index, clinical center, systolic blood pressure, diastolic blood pressure, FPG, smoking, and drinking. Compared to subjects with the methylenetetrahydrofolate reductase (MTHFR) 677 CC and TT genotypes, those with the CT genotype had a higher risk of NOD (OR = 1.54; 95%CI: 1.30–1.81, P for interaction = 0.044) in subjects with high TG. These results suggested that TG and TG/HDL were independent risk factors for NOD in this Chinese hypertensive population. HDL was a protective factor for NOD.
Similar content being viewed by others
Introduction
China, the most populous country in the world, is experiencing an increase in diabetes morbidity rates each year. A recent national cross-sectional survey showed that the overall prevalence of diabetes was estimated to be 11.6% in adult Chinese1. Previous studies found that impaired fasting glucose (IFG) or diabetes was positively associated with the risk of coronary heart disease (CHD)2, 3. In addition, diabetes is a major risk factor for ischemic heart disease (IHD) and stroke, which, in 2010, collectively contributed to an estimated global mortality of 12.9 million people4. In recent years, the consequences of unhealthy living habits have been shown to lead to a rapid increase in risk factors of diabetes and CHD, such as dyslipoproteinemia.
Several large randomized trials reported that baseline fasting blood glucose (FBG) levels are predictive for new-onset type 2 diabetes (NOD)5, 6. Indeed, Sattar et al.7 confirmed that high triglyceride levels (TG) (≥1.69 mmol/L) and low high-density lipoprotein (HDL) cholesterol (≤1.04 mmol/L for men and ≤1.29 mmol/L for women) are also predictors of diabetes. Tirosh et al.8 indicated that TG levels may identify people at an increased risk for diabetes, even in apparently healthy young men, independently of traditional risk factors, confirming that dyslipidemia may play an important role in identifying people at risk for NOD.
Most studies of the associations between reduced HDL-cholesterol or elevated TG levels and NOD risk are from European and American populations7,8,9 and there are very few studies on East Asian populations, especially hypertensive populations. In addition, the gender effect of higher HDL on NOD is unclear. Two studies indicated that higher HDL had a protective effect against NOD only among females10, 11. While Meisinger et al.12 showed that higher HDL was inversely associated with diabetes in both males and females.
Presently, there are no studies investigating the association between lipid levels and NOD in a Chinese hypertensive population. Therefore, the present study aimed to provide insights into the relationship between lipid levels and NOD in a Chinese hypertensive population, providing a better understanding of the relationship between HDL and NOD among males and females.
Results
Characteristics of the subjects
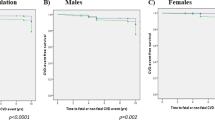
Follow-up was censored on August 24, 2014, and 14,864 subjects could be analyzed (71.8% of the original CSPPT study). Among them, 1615 (657 men and 958 women) developed NOD, for an incidence of 10.9%. The baseline characteristics of the participants are presented in Table 1. Individuals who developed NOD had significantly higher baseline fasting plasma glucose (FPG), body mass index (BMI), TG, and total cholesterol (TC), as well as lower HDL than those who did not develop diabetes (all P < 0.05). Females who developed NOD tended to be slightly older (59.8 vs. 59.1 years, P = 0.01) and to have higher baseline systolic blood pressure (SBP) (169.4 vs. 167.8 mmHg, P = 0.02) than non-diabetics, but these differences were not observed in males.
Association between lipid levels and NOD
Both continuous and categorical TG, TG/HDL, and HDL levels were significantly associated with incident diabetes (all P < 0.001). They were independent predictors of diabetes after adjusting for other covariables. The incidence of NOD was significantly higher in subjects with high TG levels compared to those with normal TG levels (13.9% vs. 9.2%, P < 0.001) and with high TG/HDL ratio compared to those with normal (17.2% vs. 10.4%, P < 0.001), while significantly lower when HDL levels were elevated (9.8% vs. 12.8%, P < 0.001) (Table 2). Compared to subjects with normal TG levels (<1.7 mmol/L), those with high TG levels (≥1.7 mmol/L) had a higher risk of NOD [odds ratio (OR) = 1.35; 95%CI confidence interval (95%CI): 1.20–1.51). Similarly, high TG/HDL ratio (≥2.8) had a higher risk of NOD (OR = 1.46; 95%CI: 1.22–1.75), while high HDL levels [≥1.0 (males)/1.3 (females) mmol/L] were negatively associated with NOD (OR = 0.82; 95%CI: 0.72–0.92) compared to subjects with low HDL levels [<1.0 (males)/1.3 (females) mmol/L]. TC was not associated with NOD (P = 0.10) when only adjusted for age, but after adjusting for other covariables, TC was negatively associated with NOD (P = 0.03). This suggests that TC alone is a protective factor of NOD, but that its effect is easily influenced by other factors.
Subgroup analyses of factors influencing the association between TG and NOD
We further explored the role of other covariables on the association between lipid profiles and NOD. Table 3 shows the results of a subgroup analysis assessing the risk of NOD associated with TG. TG was positively associated with NOD, and it was more significant in patients from the Anqing district (Anqing: OR = 1.27, 95%CI: 1.13–1.43; Lianyungang: OR = 1.16, 95%CI: 1.10–1.23, P for interaction = 0.026). Compared to subjects with the MTHFR 677 CC and TT genotypes, those with the CT genotype had a higher risk of NOD (OR = 1.54, 95%CI, 1.30–1.81, P for interaction = 0.044) in subjects with high TG levels. The effect of TG on NOD showed no difference in the following subgroups: age, gender, treatment group (enalapril vs. enalapril-folic acid), smoking, drinking, BMI, SBP, DBP, folic acid, and homocysteine (Hcy) levels (all P for interaction >0.10) (Table 3).
Subgroup analyses of factors influencing the association between TC and NOD
We then analyzed the relationship between TC and NOD. The data suggest that TC was protective in the enalapril-folic acid group (enalapril-folic acid: OR = 0.91, 95%CI: 0.85–0.98; enalapril: OR = 0.98, 95%CI: 0.92–1.05, P for interaction = 0.034). Compared to subjects who ever smoked or currently smoke, those who never smoked had a significant protective effect of NOD (OR = 0.92, 95%CI: 0.87–0.98, P for interaction = 0.036). TC appeared to be negatively associated with NOD, but the stratified analyses show that in each subgroup presented above, TC is not associated with NOD (P for interaction >0.05). In addition, the effect of TC on NOD showed no differences within the subgroups of age, clinical center, gender, alcohol, BMI, SBP, DBP, MTHFR C677T, folic acid, and Hcy (all P for interaction >0.05) (Table 4).
Subgroup analyses of factors influencing the association between HDL and NOD
HDL was negatively associated with NOD, and was more significant for patients from the Anqing district (Anqing: OR = 0.42, 95%CI: 0.29–0.61; Lianyungang: OR = 0.94; 95%CI: 0.78–1.13, P for interaction <0.001). This was also true for individuals with high folic acid levels at baseline (≥8.1 ng/ml: OR = 0.65, 95%CI: 0.51–0.83; <8.1 ng/ml: OR = 0.95, 95%CI: 0.76–1.19, P for interaction = 0.027). In addition, stratified analyses were performed by MTHFR C677T genotypes (CC, CT, and TT), age, gender, treatment group (enalapril vs. enalapril-folic acid), smoking, drinking, BMI, SBP, DBP, and Hcy level, but there were no significant interactions in any of the subgroups (all P > 0.05), including gender (P = 0.199) (Table 5).
Subgroup analyses of factors influencing the association between TG/HDL and NOD
We further explored the role of other covariables on the association between lipid profiles and NOD. Table 6 shows the results of a subgroup analysis assessing the risk of NOD with TG/HDL. TG/HDL was positively associated with NOD, and it was more significant in patients from the Anqing district (Anqing: OR = 1.18, 95%CI: 1.09–1.28; Lianyungang: OR = 1.10, 95%CI: 1.05–1.16, P for interaction = 0.028). The effect of TG/HDL on NOD showed no difference in the following subgroups: age, gender, treatment group (enalapril vs. enalapril-folic acid), smoking, drinking, BMI, SBP, DBP, folic acid, MTHFR C677T genotypes and Hcy (homocysteine) levels (all P for interaction >0.10) (Table 6).
Subgroup analyses of factors influencing the association between statins and NOD
We explored the role of other covariables on the association between statins and NOD. After removing the patients taking statins, Table 7 shows that basically the same associations were still observed.
Discussion
Previous studies showed that baseline lipid levels are important risk determinants of NOD among Caucasian populations7,8,9, but the association between lipid levels and NOD in Asian populations is unclear. Therefore, this study aimed to provide insights into the relationship between lipid levels and NOD in a Chinese hypertensive population. Results showed that TG and TG/HDL were independent risk factor for NOD in this Chinese hypertensive population. HDL had a protective effect for NOD.
The central pathophysiological feature in the development of type 2 diabetes mellitus from dyslipidemia is not clear. Despite the controversy, accumulating evidence indicates that both low HDL and high TG levels are early manifestations of insulin resistance (IR) and later diabetes, and that they can actively add to β-cell failure and participate in NOD onset13. In addition, HDL may also increase glucose disposal through direct effects in the skeletal muscle, the major site of glucose catabolism in the body14. Ginsberg et al.15 indicated that plasma TG may play a role in insulin resistance. Boden et al.16 suggested that elevated free fatty acids (FFA) may contribute to hyperglycemia by antagonizing the effects of insulin on endogenous glucose production and affecting insulin secretion. On the other hand, internal glycerol and fatty acids can be converted to glucose in the liver. A study revealed increased insulin levels in subjects with hypertension despite normal glucose levels. Nevertheless, the specific mechanisms linking lipids and NOD need to be further investigated.
In the present study, serum TG levels were a strong predictor of NOD in both genders, independent of the other risk factors. The association of fasting TG with NOD has been documented previously8, 17, 18, but these results were mainly reported using TG levels that were pooled with additional risk factors for diabetes or cardiovascular diseases8. Nevertheless, these previous studies support the present study, in which high TG levels significantly increased NOD risk by 18% if analyzed continuously or by 35% for high TG levels (≥1.7 mmol/l).
Low HDL is known to be an important predictor for the development of diabetes7, and certain agents known to raise HDL improve glucose metabolism and prevent diabetes14, but the protective effect among gender is still controversial. The present study confirmed that HDL protected against NOD. Continuous HDL levels decreased NOD risk by 21%, but without differences between genders, which suggests that the protective effect of HDL on NOD is not gender-specific in a Chinese hypertensive population.
The high TG and low HDL profile is the classical profile associated with the metabolic syndrome and other blood lipid abnormalities19, which have been associated with NOD20. Squillace et al.21 showed that high TG/HDL ratio increased the risk of NOD, independently of other traditional risk factors, supporting the results of the present study.
There are very few studies that observed the effect of TC on NOD. Mozaffarian et al.22. found that a lower total cholesterol to HDL cholesterol ratio (−4.7%; P < 0.001) was associated with a substantially lower incidence of diabetes. In the present study, TC had no effect on NOD when adjusted for age only, but after adjustment for multiple covariables, TC was negatively associated with NOD. This indicates that TC alone could have a protective effect against the development of NOD, but this effect is easily influenced by other factors. More studies are needed to confirm the relationship between TC and NOD.
Generally, physicians suggest lifestyle interventions to patients whose TG levels fall between 1.70 and 2.25 mmol/L, or lifestyle intervention combined with fibrate therapy to those whose TG levels falling between 2.26 and 4.5 mmol/L23, 24. From the present study, controlling the levels of TG for Chinese hypertensive people seems to be of great importance. TG levels can be easily changed by diet, therefore, diet and/or exercise may help to decrease the risk of NOD for hypertensive patients by decreasing TG levels. Studies are necessary to evaluate the impact of diet and exercise on NOD.
Statins are known to be associated with a higher risk of NOD25,26,27,28,29. In the present study, the sensitivity analysis showed that statins had no impact on NOD. These discrepancies could be due to the specific population being studied, including factors such as diet, lifestyle, and genetics. Additional studies are necessary to examine this point, but the impact of statins on adiponectin levels could be involved30.
There are potential limitations of our results. First, TC, HDL, and TG levels were only measured at baseline, thus the potential bias resulting from changes in TG and HDL over time cannot be ignored. Additionally, although laboratory parameters do not include measurements such as circulating insulin levels, they constitute a set of routine tests that are typically available to the practicing physician but not routinely used in screening. Thirdly, no adjustment could be done for changes in drugs during follow-up because of missing data. It has been shown that some classes of antihypertensive were associated with increased NOD risk31. Nevertheless, a review underlined that controlling the risk of NOD should not compromise blood pressure control32 and all subjects of the present study were taking angiotensin-converting enzyme inhibitors at baseline. Finally, diabetes onset can be affected by or result from diet and physical exercise, but we were not able to control for diet and exercise.
In conclusion, TG and TG/HDL were independent risk factors for NOD in this Chinese hypertensive population. HDL presented a protective effect for NOD. NOD was independent from TC.
Material and Methods
Study population
The subjects were from a randomized, double-blind, controlled trial (the CSPPT study)33 conducted from May 19, 2008, to August 24, 2013. This trial consisted of men and women aged 45–75 years old and with hypertension (defined as seated resting systolic blood pressure of >140 mmHg or diastolic blood pressure of >90 mmHg) at both the screening and recruitment visits, or who were taking at least one antihypertensive medication. A total of 20,702 people were included in the CSPPT and underwent a baseline examination in 2008. Fasting blood samples were collected for lipid analysis and genotyping of the MTHFR polymorphism. Eligible participants were randomly assigned in a 1:1 ratio to one of two treatment groups: a daily oral dose of one tablet containing 10 mg of enalapril and 0.8 mg of folic acid (the enalapril-folic acid group) or a daily oral dose of one tablet containing 10 mg of enalapril only (the enalapril-only group)33. The complete eligibility criteria and interventions are described in the CSPPT paper33.
The present post hoc study focused on those subjects with valid FPG values at baseline and at the end of study. Participants lacking lipid values were excluded. Those who had a self-reported diabetes history, patients whose FPG > 7 mmol/L at baseline or were taking hypoglycemic agents or lipid lowering therapy were also excluded. In addition, subjects with missing glucose values at the end of study, dead, or lost to follow-up were excluded as well. Thus, the present study included 14,864 non-diabetic subjects (6056 men and 8808 women) (Fig. 1).
The present study was approved by the ethics committee of the Nanfang Hospital, Guangzhou, China. The patients provided a written informed consent under the premises of the original CSPPT trial, including the possibility of post hoc analyses. This trial was registered with Clinicaltrials.gov (#NCT00794885). All methods were performed in accordance with the relevant guidelines and regulations.
Data collection
Under the premises of the CSPPT trial33, all participants completed a detailed questionnaire assessing demographic, nutritional, lifestyle, and medical parameters. Height, weight, waist circumference, hip circumference, and blood pressure were recorded by trained medical staff. BMI was calculated as weight in kilograms (kg) divided by height in meters squared (m2). Serum Hcy, fasting TC, TG, HDL, and FPG levels at baseline and end of study were measured using automatic clinical analyzers (Beckman Coulter, Brea, CA, USA) at the National Clinical Research Center for Kidney Disease, Nanfang Hospital, Guangzhou, China. Smoking was recorded as never, former, or current. Alcohol drinking was recorded as never, former, and current.
NOD was defined as FPG ≥ 7.0 mmol/L at the end of study or self-reported physician diagnosis of diabetes or self-reported use of hypoglycemic agents during follow-up34, 35. The original CSPPT trial was conducted from May 19, 2008, to August 24, 2014. Follow-up was censored on August 24, 2014.
Statistical analysis
All continuous data were evaluated for normality using plots. Normally distributed continuous variables were presented as mean ± standard deviation and analyzed using the Student t test. Non-normally distributed variables (Hcy and folate levels) were presented as median (interquartile range) and analyzed using the Kruskal-Wallis test. Categorical variables were presented as number and frequencies, and analyzed using the chi-square test. Logistic regression models were used to predict incident diabetes. The first model was adjusted for age only. The second model was adjusted for age and other confounders. Lipid-level variables were modeled as both continuous and binary: high TG levels and normal (≥1.7 vs. <1.7 mmol/L); low HDL levels and high [(<1.0 (males)/1.3 (females) vs. ≥1.0 (males)/1.3 (females)]; and normal TC levels and high (<5.2 vs. ≥5.2 mmol/L).
In the stratified analyses, the effects of lipid-level variables (both as continuous and binary) on the risks of NOD were estimated using logistic regression models among subgroups classified according to age (≥60 and <60), clinical center (Anqing and Lianyungang), gender (males and females), treatment group (enalapril and enalapril-folic acid), smoking status (never, ever, and current), alcohol (never, ever, and current), baseline BMI (<24, ≥24 and <28, and ≥28 kg/m2), SBP (<140, ≥140 and <160, ≥160 and <180, and ≥180 mmHg), DBP (<90, ≥90 and <100, ≥100 and <110, and ≥110 mmHg), folic acid (<8.1 and ≥8.1 ng/ml), Hcy (<10 and ≥10 μmol/l), and MTHFRC677T polymorphism (CC, CT, and TT). The results were presented as OR with 95%CI. An OR > 1 indicated an increased risk of NOD, while an OR < 1.0 indicates reduced risk. Possible confounding factors were taken into account, such as gender, clinical center, systolic and diastolic blood pressure, BMI, smoking, alcohol, and FPG. A double-tailed P-value < 0.05 was considered statistically significant in all analyses. Empowerstats (http://www.EmpowerStats.com.cn) and R (http://www.R-project.org/. version 3.2) were used to perform all statistical analyses.
References
Xu, Y. et al. Prevalence and control of diabetes in Chinese adults. JAMA 310, 948–959 (2013).
Blake, D. R. et al. Impaired glucose tolerance, but not impaired fasting glucose, is associated with increased levels of coronary heart disease risk factors: results from the Baltimore Longitudinal Study on Aging. Diabetes 53, 2095–2100 (2004).
Levitzky, Y. S. et al. Impact of impaired fasting glucose on cardiovascular disease: the Framingham Heart Study. J Am Coll Cardiol 51, 264–270 (2008).
Lozano, R. et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2095–2128 (2012).
LaRosa, J. C. et al. Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med 352, 1425–1435 (2005).
Pedersen, T. R. et al. High-dose atorvastatin vs usual-dose simvastatin for secondary prevention after myocardial infarction: the IDEAL study: a randomized controlled trial. JAMA 294, 2437–2445 (2005).
Sattar, N. et al. Can metabolic syndrome usefully predict cardiovascular disease and diabetes? Outcome data from two prospective studies. Lancet 371, 1927–1935 (2008).
Tirosh, A. et al. Changes in triglyceride levels over time and risk of type 2 diabetes in young men. Diabetes Care 31, 2032–2037 (2008).
Wilson, P. W. et al. Prediction of incident diabetes mellitus in middle-aged adults: the Framingham Offspring Study. Arch Intern Med 167, 1068–1074 (2007).
Hadaegh, F. et al. Lipid ratios and appropriate cut off values for prediction of diabetes: a cohort of Iranian men and women. Lipids Health Dis 9, 85 (2010).
Qin, X. et al. Relationship of MTHFR gene 677C –> T polymorphism, homocysteine, and estimated glomerular filtration rate levels with the risk of new-onset diabetes. Medicine (Baltimore) 94, e563 (2015).
Meisinger, C. et al. Sex differences in risk factors for incident type 2 diabetes mellitus: the MONICA Augsburg cohort study. Arch Intern Med 162, 82–89 (2002).
Van Linthout, S., Spillmann, F., Schultheiss, H. P. & Tschope, C. High-density lipoprotein at the interface of type 2 diabetes mellitus and cardiovascular disorders. Curr Pharm Des 16, 1504–1516 (2010).
Drew, B. G. et al. High-density lipoprotein modulates glucose metabolism in patients with type 2 diabetes mellitus. Circulation 119, 2103–2111 (2009).
Ginsberg, H. N., Zhang, Y. L. & Hernandez-Ono, A. Regulation of plasma triglycerides in insulin resistance and diabetes. Arch Med Res 36, 232–240 (2005).
Boden, G. & Shulman, G. I. Free fatty acids in obesity and type 2 diabetes: defining their role in the development of insulin resistance and beta-cell dysfunction. Eur J Clin Invest 32(Suppl 3), 14–23 (2002).
Dotevall, A., Johansson, S., Wilhelmsen, L. & Rosengren, A. Increased levels of triglycerides, BMI and blood pressure and low physical activity increase the risk of diabetes in Swedish women. A prospective 18-year follow-up of the BEDA study. Diabet Med 21, 615–622 (2004).
Sanyal, D., Ghosh, S., Mukherjee, P., Mukherjee, S. & Chowdhury, S. Dyslipidemia, metabolic syndrome, and liver enzymes in impaired glucose tolerance and new onset untreated, type 2 diabetes Indian subjects. Indian J Endocrinol Metab 16, S434–435 (2012).
Quispe, R. et al. Relationship of the triglyceride to high-density lipoprotein cholesterol (TG/HDL-C) ratio to the remainder of the lipid profile: The Very Large Database of Lipids-4 (VLDL-4) study. Atherosclerosis 242, 243–250 (2015).
Katsiki, N., Rizzo, M., Mikhailidis, D. P. & Mantzoros, C. S. New-onset diabetes and statins: throw the bath water out, but, please, keep the baby! Metabolism 64, 471–475 (2015).
Squillace, N. et al. Triglyceride/HDL ratio and its impact on the risk of diabetes mellitus development during ART. J Antimicrob Chemother 71, 2663–2669 (2016).
Mozaffarian, D. et al. Trans-palmitoleic acid, metabolic risk factors, and new-onset diabetes in U.S. adults: a cohort study. Ann Intern Med 153, 790–799 (2010).
Catapano, A. L., Chapman, J., Wiklund, O. & Taskinen, M. R. The new joint EAS/ESC guidelines for the management of dyslipidaemias. Atherosclerosis 217, 1 (2011).
Cugnet-Anceau, C. & Moret, M. & Moulin, P. [Hypertriglyceridemia: therapeutic strategy]. Rev Prat 61, 1110–1116 (2011).
Yoon, D. et al. Statins and risk for new-onset diabetes mellitus: A real-world cohort study using a clinical research database. Medicine (Baltimore) 95, e5429 (2016).
Barylski, M. et al. Statins and new-onset diabetes. Curr Pharm Des 20, 3657–3664 (2014).
Chrysant, S. G. New onset diabetes mellitus induced by statins: current evidence. Postgrad Med 129, 430–435 (2017).
Banach, M. et al. Statin therapy and new-onset diabetes: molecular mechanisms and clinical relevance. Curr Pharm Des 19, 4904–4912 (2013).
Casula, M. et al. Statin use and risk of new-onset diabetes: A meta-analysis of observational studies. Nutr Metab Cardiovasc Dis 27, 396–406 (2017).
Chrusciel, P. et al. Impact of statin therapy on plasma adiponectin concentrations: A systematic review and meta-analysis of 43 randomized controlled trial arms. Atherosclerosis 253, 194–208 (2016).
Huang, C. Y. et al. A retrospective longitudinal cohort study of antihypertensive drug use and new-onset diabetes in Taiwanese patients. Biomed Res Int 2013, 287696 (2013).
Alderman, M. H. New onset diabetes during antihypertensive therapy. Am J Hypertens 21, 493–499 (2008).
Huo, Y. et al. Efficacy of folic acid therapy in primary prevention of stroke among adults with hypertension in China: the CSPPT randomized clinical trial. JAMA 313, 1325–1335 (2015).
Genuth, S. et al. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 26, 3160–3167 (2003).
Qin, X. et al. Effect of folic acid supplementation on risk of new-onset diabetes in adults with hypertension in China: Findings from the China Stroke Primary Prevention Trial (CSPPT). J Diabetes 8, 286–294 (2016).
Acknowledgements
We thank the team of the CSPPT, Xiping X, MD, PhD (National Clinical Research Center for Kidney Disease, the State Key Laboratory for Organ Failure Research), the Renal Department (Nanfang Hospital, Southern Medical University, Guangzhou, China), and AUSA Research Institute (Shenzhen AUSA Pharmed Co. Ltd., Shenzhen, China), and the former employee of AUSA Research Institute (Shenzhen AUSA Pharmed Co. Ltd., Shenzhen, China). This research was supported by the National Science and Technology Major Projects Specialized for “Major New Drugs Innovation and Development” during the 12th Five-Year Plan Period: China Stroke Primary Prevention Trial (grant #zx09101105); the Major State Basic Research Development Program of China (973 program) (#2012 CB517703); the Clinical Center (grant #zx09401013); Projects of National Natural Science Foundation of China (grants #81473052, 81441091, 81402735 and 81560079); the National Clinical Research Center for Kidney Disease, Nanfang Hospital, Nanfang Medical University, Guangzhou, China; State Key Laboratory for Organ Failure Research, Nanfang Hospital, Nanfang Medical University, Guangzhou, China; and the Department of Development and Reform, Shenzhen Municipal Government (grant #SFG 20201744).
Author information
Authors and Affiliations
Contributions
Leliang Li participated in literature search, study design, data collection, data analysis, data interpretation, and wrote the manuscript. Ping Li, Juan Yang, Xiao Huang, Huihui Bao, Chunyan Zhang, Yun Song, Min Zhao and Meng Ji carried out the data collection and analysis, and provided the critical revision. Yi Wang, Geng Qian, Genfu Tang, Shanqun Jiang, Qiang Dong, Yan Zhang, Jianping Li and Xiping Xu conceived of the study, and participated in its design and coordination. Binyan Wang, Yong Huo and Xiaoshu Cheng participated in study design and provided the critical revision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, L., Li, P., Yang, J. et al. Lipid levels and new-onset diabetes in a hypertensive population: the China Stroke Primary Prevention Trial. Sci Rep 7, 7014 (2017). https://doi.org/10.1038/s41598-017-07355-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-07355-w
- Springer Nature Limited
This article is cited by
-
Plasma apolipoprotein concentrations and incident diabetes in subjects with prediabetes
Cardiovascular Diabetology (2022)