Abstract
Cell death is a fundamental progress that regulates cell number, tissue homeostasis and organ size in development. The c-Jun N-terminal kinase (JNK) pathway has been evolutionarily conserved from fly to human, and plays essential roles in regulating cell death. To characterize additional genes that regulate JNK signaling, we performed a genetic screen in Drosophila and identified dGLYAT, a novel gene whose function was previously unknown, as a modulator of JNK-mediated cell death. We found that loss of dGLYAT suppressed JNK activation and cell death triggered by over-expression of Egr or Hep, or depletion of puc or lgl in development, suggesting dGLYAT regulates both ectopic and physiological functions of JNK pathway. Furthermore, we showed that loss of dGLYAT inhibits JNK-mediated ROS production, suggesting dGLYAT regulates multiple functions of JNK signaling in vivo.
Similar content being viewed by others
Introduction
The glycine N-acyltransferase (GLYAT) family, which includes hGLYAT, hGLYATL1, hGLYATL2 and hGLYATL3 in human1,2,3,4,5,6, encodes a characteristic GNAT (Gcn5-related N-Acetyltransferase) domain that is evolutionarily conserved from invertebrate to mammals7, 8. GLYAT proteins are specifically localized in the mitochondria9, and play pivotal roles in catalyzing the formation of Primary Fatty Acid Amides (PFAMs)6, 10, a family of bioactive lipids essential for many biological processes6, 10, 11. Anandamide, a member of PFAMs, was shown to activate JNK signaling and promote reactive oxygen species (ROS) formation12,13,14, yet a direct role of GLYAT in JNK signaling and cell death has not been reported. CG34010, whose function has not been previously investigated, encodes a Drosophila homolog of GLYAT, and is referred to as dGLYAT hereafter.
The c-Jun N-terminal kinase (JNK) signaling pathway is highly conserved from fly to mammals15, 16, and plays essential roles in regulating cellular activities including cell proliferation, differentiation, migration and apoptosis17, 18. In Drosophila, ectopic expression of the tumor necrosis factor (TNF) ortholog Eiger (Egr) triggers cell death through the JNK pathway19. Egr is recognized by its receptor Grindelwald (Grnd), which acts through the TNF receptor-associated factor2 (dTRAF2) and the Bendless (Ben)/dUev1a ubiquitin conjugating enzyme complex to initiate a kinase cascade reaction including the JNK kinase kinase dTAK1 (MAP3K), the JNK kinase hemipterous (Hep) and Basket (Bsk), the fly JNK, through phosphorylation19,20,21,22,23,24. The activation of JNK signaling could be reflected by the expression of its target gene puckered (puc), which encodes a JNK phosphatase that negatively regulates JNK activity and thus establishes a negative feedback loop25,26,27. In addition, activated JNK signaling promotes the production of Reactive Oxygen Species (ROS)28, 29.
To identify additional factors that regulate JNK-mediated cell death, we have been performing genetic screens in Drosophila for modifiers of Egr-triggered JNK-dependent cell death, and have characterized Ben, dUev1a and Wallenda (Wnd) as components of this evolutionary conserved pathway24, 30, 31. In this study, we characterized dGLYAT as an essential regulator of JNK signaling in Drosophila. Firstly, loss of dGLYAT suppresses ectopic Egr or Hep-induced JNK-dependent cell death in development. Secondly, depletion of dGLYAT blocks ectopic Egr or Hep-triggered JNK pathway activation. Furthermore, dGLYAT is required for physiological JNK activation-induced cell death, which is triggered by depletion of puc or lgl. Finally, loss of dGLYAT impedes activated JNK signaling-induced ROS production. Thus, these data not only represent the first in vivo function of dGLYAT in Drosophila development, but also suggest a role of GLYAT in regulating JNK signaling in mammals.
Result and Discussion
Loss of dGLYAT suppresses ectopic Egr-induced cell death in eye development
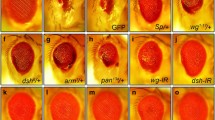
Compared with the control (Fig. 1a), ectopic expression of TNF ortholog Egr in the developing eye driven by GMR-Gal4 (GMR > Egr) triggers JNK-dependent cell death and produces a small eye phenotype in adults (Fig. 1b)22, 30,31,32,33,34,35. We found the GMR > Egr eye phenotype was significantly suppressed by a mutation in CG34010 (Fig. 1d), a novel gene whose function was previously unknown. CG34010 encodes a Drosophila ortholog of glycine N-acyltransferase (GLYAT), and is referred to as dGLYAT hereafter. The mutant, PBac{PB}CG34010 c02982, has a piggyBac insertion into the second exon and generates a truncated protein that deletes most of the coding region including the critical Gcn5-related N-acetyltransferases (GNAT) domain. Thus, the mutant is most probably a null allele for dGLYAT. Interestingly, the mutant is homozygously viable, and does not produce any discernible phenotype, suggesting it is not essential for normal development. Furthermore, RNAi-mediated depletion of dGLYAT also suppressed the GMR > Egr induced small eye phenotype (Fig. 1e), compared with the expression of a UAS-GFP transgene that served as a negative control (Fig. 1c), confirming that dGLYAT is required for ectopic Egr-triggered morphological defect. Expression of RNAi-mediated depletion of Bsk, the Drosophila JNK ortholog, served as a positive control (Fig. 1f). Consistently, GMR > Egr-triggered cell death, indicated by acridine orange (AO) staining, posterior to the morphogenetic furrow (MF) in 3rd instar eye discs (Fig. 1h), was significantly impeded by loss of dGLYAT or Bsk (Fig. 1j–l), but remained unaffected by the expression of GFP (Fig. 1i). The statistics of adult eye sizes (Fig. 1m) and apoptotic cell numbers in larval eye discs (Fig. 1n) were shown. Taken together, the above data suggest that dGLYAT is physiologically required for ectopic Egr-induced cell death in eye development.
Loss of dGLYAT suppresses ectopic Egr-induced cell death in eye development. Light micrographs of Drosophila adult eyes (a–f) and fluorescent micrographs of third instar eyes discs (g–l) are shown. Compared with the GMR-Gal4 controls (a,g), GMR > Egr induces a small eye phenotype in adults (b) and massive cell death in 3rd instar larval eye discs with AO staining (h). Both phenotypes are suppressed by mutation (d,j) or RNAi-mediated depletion (e,k) of dGLYAT, but remains unaffected by expressing GFP (c,i). Expression of bsk-IR serves as a positive control (f,l). (m) Statistics of eye sizes shown in (a–e) (a, n = 15; b, n = 11; c, n = 26; d, n = 20; e, n = 13; f, n = 14). (n) Statistics of AO-positive cell numbers shown in (g–l) (g, n = 5; h, n = 6; i, n = 12; j, n = 7; k, n = 7; l, n = 8). n.s., P > 0.05; ****P < 0.0001; ***P < 0.001.
Loss of dGLYAT impedes ectopic Hep-induced cell death in eye development
GMR > Egr triggers cell death via two independent pathways, the caspase pathway and the JNK pathway34. To examine whether dGLYAT is required for caspase-mediate cell death, we overexpressed Drosophila p53 (Dp53), a pro-apoptotic gene that triggers caspase-mediated cell death36,37,38,39, in the eye by GMR-Gal4. We found that GMR > Dp53-triggered small eye phenotype was not suppressed by loss of dGLYAT (Figure S1), suggesting dGLYAT is not involved in caspase-mediated cell death. To investigate the role of dGLYAT in JNK-mediated cell death, we expressed a constitutive active form of the Drosophila JNK kinase Hemipterous (Hep) in the developing eye. GMR > HepCA induces JNK-mediated cell death in eye discs (Fig. 2h) and produces a small eye phenotype in adults (Fig. 2b)31, 33. Both phenotypes were significantly suppressed by loss of dGLYAT or depletion of Bsk, but not the expression of GFP (Fig. 2c–f,i–n). Thus, dGLYAT is necessary for ectopic Hep-induced JNK-mediated cell death in eye development.
Loss of dGLYAT suppresses ectopic Hep-induced cell death in eye development. Light micrographs of Drosophila adult eyes (a–f) and fluorescent micrographs of third instar eyes discs (g–l) are shown. Compared with the GMR-Gal4 controls (a,g), GMR > Hep induces a small eye phenotype in adults (b) and extensive cell death in 3rd instar larval eye discs (h). Both phenotypes are suppressed by mutation (d,j) or RNAi-mediated depletion (e,k) of dGLYAT, but remains unaffected by expressing GFP (c,i). Expression of bsk-IR serves as a positive control (f,l). (m) Statistics of eye sizes shown in (a–e) (a, n = 12; b, n = 51; c, n = 29; d, n = 16; e, n = 26; f, n = 20). (n) Statistics of AO-positive cell numbers shown in (g–l) (g, n = 5; h, n = 8; i, n = 8; j, n = 11; k, n = 8; l, n = 8). n.s., P > 0.05; ****P < 0.0001.
Loss of dGLYAT inhibits JNK activation in eye discs
The above data suggest that dGLYAT is necessary for JNK-mediated cell death in eye development, yet it remains unknown whether dGLYAT is required for JNK pathway activation. To address this, we checked the expression of puc-LacZ, a well-known readout of JNK signaling in Drosophila 25, 40, 41. We found that GMR > Egr induced strong puc-LacZ expression posterior to the morphogenetic furrow (MF) in the eye disc (Fig. 3b), which was remarkably inhibited by loss of dGLYAT. Again, expression of Bsk and GFP were served as a positive and negative controls, respectively (Fig. 3c–f). Hence, dGLYAT is necessary for JNK signaling activation in eye development.
Loss of dGLYAT suppresses ectopic Egr-induced puc expression in eye discs. Light micrographs of Drosophila third instar eye discs with X-Gal staining are shown. Compared with the GMR-Gal4 control (a), ectopic Egr-induced puc-LacZ expression in eye discs (b) is suppressed by mutation (d) or RNAi-mediated depletion (e) of dGLYAT. Expression of GFP (c) and Bsk-IR (f) acts as a negative and a positive control (a, n = 4; b, n = 6; c, n = 8; d, n = 6; e, n = 5; f, n = 9).
dGLYAT modulates JNK-mediated cell death in other tissues
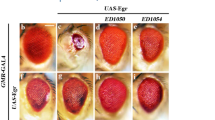
To investigate whether dGLYAT modulates JNK-mediated cell death in other tissues, we examined the interaction between dGLYAT and JNK signaling in the developing wing, another important tissue frequently used for genetics studies. Ectopic expression of Egr driven by ptc-Gal4 (ptc > Egr) was able to induce extensive cell death along the anterior/posterior (A/P) compartment boundary in 3rd instar larval wing discs (Fig. 4h) and produce loss of the anterior cross vein (ACV) phenotype in adult wings (Fig. 4b)22, 42. We found that both phenotypes were significantly suppressed by loss of dGLYAT or Bsk, but not the expression of GFP (Fig. 4c–f,i–n). Thus, dGLYAT is also required for ectopic Egr-triggered cell death in wing development.
Loss of dGLYAT suppresses ectopic Egr-induced cell death in wing development. Light micrographs of Drosophila adult wings (a–f) and fluorescent micrographs of third instar wing discs (g–l) are shown. The lower panels are high magnification of boxed area in upper panels (a–f). Compared with the ptc-Gal4 controls (a,g), ptc > Egr induces a loss-of-ACV phenotype in adult wings (b) and extensive cell death along the A/P boundary in third instar larval wing discs (h), while remains unaffected by expressing GFP as a negative control (c,i). Both phenotypes are suppressed partially by mutation in dGLYAT (d,j) and strongly by RNAi-mediated knocking-down of dGLYAT (e,k). Expression of Bsk-IR acts as a positive control (f,l) (m) Statistics of the loss-of-ACV phenotype in (a–f) (a, n = 50; b, n = 56; c, n = 90; d, n = 88; e, n = 113; f, n = 80), (n) Statistics of the AO-positive cell number in (g–l) (g, n = 17; h, n = 12; i, n = 12; j, n = 21; k, n = 12; l, n = 10). n.s., P > 0.05; ****P < 0.0001.
Ectopic expression of Hep driven by ptc-Gal4 (ptc > Hep) induces JNK-mediated cell death in wing discs and generates a loss-of-ACV phenotype in adults22, 32, both of which were blocked by loss of dGLYAT (Fig. 5). Consistently, ptc > Hep-induced puc-LacZ activation along the A/P boundary in 3rd instar larval wing discs (Fig. 6b)23, 26, 30, 31 was significantly inhibited by depleting dGLYAT or Bsk, but not the expression of GFP (Fig. 6c–f). Collectively, these data indicate that dGLYAT modulates JNK-mediated cell death in a non-tissue specific manner.
Loss of dGLYAT suppresses ectopic Hep-induced cell death in wing development. Light micrographs of Drosophila adult wings (a–f) and fluorescent micrographs of third instar wing discs (g–l) are shown. The lower panels are high magnification of boxed area in upper panels (a–d). Compared with the ptc-Gal4 controls (a,g), ptc > Hep induces a loss-of-ACV phenotype in adult wings (b) and extensive cell death along the A/P boundary in third instar larval wing discs (h), while remains unaffected by expressing GFP as a negative control (c,i). Both phenotypes are suppressed partially by mutation in dGLYAT (d,j) and strongly by RNAi-mediated knocking-down of dGLYAT (e,k). (m) Statistics of the loss-of-ACV phenotype in (a–f) (a, n = 70; b, n = 176; c, n = 110; d, n = 93; e, n = 146; f, n = 98). (n) Statistics of the AO-positive cell number in (g–l) (g, n = 16; h, n = 5; i, n = 7; j, n = 9; k, n = 10; l, n = 6). n.s., P > 0.05; ****P < 0.0001; **P < 0.01.
Loss of dGLYAT suppresses ectopic Hep-induced puc transcription in wing discs. Light micrographs of Drosophila third instar wing discs with X-Gal staining are shown. Compared with the ptc-Gal4 control (a), ptc > Hep-induced puc-LacZ expression in wing disc (b) is suppressed by mutation (d) or RNAi-mediated depletion of dGLYAT (e). Expression of GFP (c) and Bsk-IR (f) acts as a negative and a positive control (a, n = 8; b, n = 10; c, n = 5; d, n = 10; e, n = 11; f, n = 9).
To investigate whether expression of dGLYAT is able to trigger JNK activation and cell death, we drove dGLYAT expression in the developing eye or wing by GMR-Gal4 or ptc-Gal4, respectively. We found that ectopic expression of dGLYAT did not trigger JNK signaling activation (Figure S2f) or cell death (Figures S2d and S2j) in the imaginal discs, and produced wild-type like eyes (Figure. S2b) and wings (Figure S2h) in the adult. Thus, expression of dGLYAT by itself is not sufficient to trigger JNK activation and cell death. Consistently, expression of Bsk, the fly JNK ortholog, or dTRAF2 that acts upstream of dTAK1, is not sufficient to induce JNK activation and cell death23. It remains to be explored whether expression of an activated form of dGLYAT, or co-expression of dGLYAT with its co-factor(s), is able to induce JNK activation and cell death in development.
dGLYAT is required for physiological activation of JNK signaling
The above data suggest that dGLYAT is important for ectopically activated JNK signaling-induced cell death, yet it remains unclear whether dGLYAT modulates the physiological functions of JNK signaling. To address this question, we knocked down puc, a negative regulator of JNK signaling, by the ptc-Gal4 driver. Depletion of puc induced robust cell death in third instar larval wing discs, as detected by AO staining (Fig. 7a and b). Intriguingly, the phenotype was significantly impeded by expressing a dGLYAT RNAi, but not GFP (Fig. 7c and d), suggesting dGLYAT is essential for physiologically activated JNK-induced cell death.
dGLYAT modulates loss of puc-induced cell death in wing discs. Light micrographs of Drosophila third instar wing discs (a–d) are shown. Compared with ptc-Gal4 controls (a), depletion of puc along the A/P boundary induces cell death in wing discs (b), which is suppressed by expressing a dGLYAT-IR (d), but remains unaffected by expressing GFP (c). (e) Statistics of the AO-positive cell numbers shown in (a–d) (a, n = 8; b, n = 8; c, n = 6; d, n = 9), n.s., P > 0.05; ****P < 0.0001.
It has been reported that loss of cell polarity in wing disc epithelial results in JNK-mediated cell death43, 44. Consistently, knockdown the cell polarity gene lethal giant larva (lgl) by ptc-Gal4 promotes JNK-mediated cell death along the A/P boundary in third instar larval wing discs (Fig. 8b)30, 31. This phenotype was significantly suppressed in heterozygous dGLYAT mutant or by the expression of a dGLYAT RNAi (Fig. 8c–e). Therefore, dGLYAT modulates the physiological function of JNK signaling in development.
dGLYAT modulates loss of lgl-induced cell death in wing discs. Light micrographs of Drosophila wing discs are shown. Compared with the ptc-Gal4 control (a), depletion of lgl induces cell death along the A/P boundary (b), which is suppressed by mutation (c) or RNAi-mediated depletion (d) of dGLYAT. (e) Statistics of the AO positive cell numbers in (a–e) (a, n = 6; b, n = 10; c, n = 13; d, n = 7). ****P < 0.0001.
dGLYAT regulates JNK-mediated ROS activation
Activated JNK signaling not only triggers cell death, but also stimulates Reactive Oxygen Species (ROS) in a feed-back loop29. To examine whether dGLYAT is crucial for JNK-mediated ROS activation, we detected ROS level in third instar larval eye discs by CellROX staining29. Consistent with previous study28, 29, 45, ectopic expression of Egr (GMR > Egr) promoted abundant ROS production (Fig. 9a and b), which was considerably suppressed by mutation or RNAi-mediated depletion (Fig. 9c and d) of dGLYAT, suggesting dGLYAT regulates JNK-mediated ROS activation in vivo.
Loss of dGLYAT suppresses ectopic Egr-induced ROS activation in eye discs. Light micrographs of Drosophila eye discs are shown. Compared with the GMR-Gal4 control (a), GMR > Egr-induced ROS positive staining (b) is suppressed by mutation (c) or RNAi-mediated depletion (d) of dGLYAT (a, n = 8; b, n = 6; c, n = 5; d, n = 6).
Intriguingly, PFAMs, the catalytic products of hGLYATs, were reported to play a role in ROS activation13, 14, 46. However, the underlying mechanism has remained unknown, and a connection with JNK signaling has not been examined. Given the fact that both JNK pathway and its role in ROS have been highly conserved from Drosophila to human, it is plausible that GLYATs also regulate JNK signaling in mammals.
Summary
dGLYAT contains a conserved GNAT domain and is supposed to function as an Acyl-CoA N-acyltransferase, yet its in vivo function has not been previously explored. In the present study, we identified dGLYAT as a crucial modulator of JNK pathway in vivo by using Drosophila as a model organism. We showed that loss of dGLYAT blocks not only ectopic Egr- or Hep-induced JNK activation and cell death, but also depletion-of-puc or lgl-triggered physiological JNK activation and cell death in development. In addition, loss of dGLYAT impedes JNK-dependent ROS activation. Thus, dGLYAT regulates multiple physiological functions of JNK signaling in vivo, yet the molecular mechanism by which dGLYAT regulates JNK pathway remains unknown, and should be addressed by further investigations.
Materials and Methods
Drosophila Genetics and Stocks
All stocks were raised on a standard cornmeal and agar medium at 25 °C unless otherwise indicated. For experiments involving tub-Gal80ts, eggs were allowed to develop at 25 °C for 2–3 days, then transferred to 29 °C for 2 days to inactivate Gal80.
ptc-Gal442, GMR-Gal447, UAS-EgrRegg119, UAS-GFP and UAS-HepCA 48, UAS-Dp5333, tub-Gal80ts and puc E6933 have been used previously; UAS-puc-IR (V3018) was obtained from Vienna Drosophila RNAi Center; UAS-bsk-IR (NIG5680R-2) was obtained from National Institute of Genetics (NIG-FLY); PBac{PB}CG34010 c02982 was obtained from Harvard (the Exelixis Collection); UAS-dGLYAT-IR was obtained from Tsinghua Fly Center.
Acridine orange staining
Eye and wing discs were dissected from 3rd instar larvae in 1 × PBS (phosphate-buffered saline) and incubated in 1 × 10−5 M acridine orange (AO) for 5 minutes at room temperature34.
X-gal staining
Wing and eye discs were dissected from third instar larvae in PBST (1 × PBS pH 7.0, 0.1% Triton X-100) and stained for β-galactosidase activity as described49.
Microscopy and phenotype analysis
Flies of indicated genotypes were collected and frozen in −80 °C. Wings were dissected and mounted on the slide in the alcohol/glycerol (1:1) medium, and flies were mounted in the alcohol on 3% agarose plate. Image of wings were captured with Olympus microscope BX51, and light image of eyes were captured with Olympus stereo microscope SZX1632.
Statistical analysis
Results are presented in bar graphs and box graphs created with GraphPad Prism 6. For loss-of-ACV phenotype, statistics were analyzed by chi-square test. For AO staining and area of eye size, one-way ANOVA with Bonferroni’s multiple comparison tests are used to calculate statistical significance. P-values are included in the relevant figure legends.
ROS staining
The level of ROS was detected by CellROX (Life Technologies). Eye discs were dissected from third instar larvae in Drosophila cell media, incubated in 5 μM CellRox for 15 minutes, rinsed in PBS, fixed in 3.7% formaldehyde for 5 minutes, and mounted in 80% glycerol for imaging45.
References
Schachter, D. & Taggart, J. V. Glycine N-acylase: purification and properties. J Biol Chem 208, 263–275 (1954).
Waluk, D. P., Schultz, N. & Hunt, M. C. Identification of glycine N-acyltransferase-like 2 (GLYATL2) as a transferase that produces N-acyl glycines in humans. FASEB J 24, 2795–2803, doi:10.1096/fj.09-148551 (2010).
Matsuo, M. et al. Designation of enzyme activity of glycine-N-acyltransferase family genes and depression of glycine-N-acyltransferase in human hepatocellular carcinoma. Biochem Biophys Res Commun 420, 901–906, doi:10.1016/j.bbrc.2012.03.099 (2012).
Waluk, D. P., Sucharski, F., Sipos, L., Silberring, J. & Hunt, M. C. Reversible lysine acetylation regulates activity of human glycine N-acyltransferase-like 2 (hGLYATL2): implications for production of glycine-conjugated signaling molecules. J Biol Chem 287, 16158–16167, doi:10.1074/jbc.M112.347260 (2012).
van der Sluis, R., Badenhorst, C. P., van der Westhuizen, F. H. & van Dijk, A. A. Characterisation of the influence of genetic variations on the enzyme activity of a recombinant human glycine N-acyltransferase. Gene 515, 447–453, doi:10.1016/j.gene.2012.12.003 (2013).
Jeffries, K. A. et al. Glycine N-acyltransferase-like 3 is responsible for long-chain N-acylglycine formation in N18TG2 cells. J Lipid Res 57, 781–790, doi:10.1194/jlr.M062042 (2016).
Dyda, F., Klein, D. C. & Hickman, A. B. GCN5-related N-acetyltransferases: a structural overview. Annu Rev Biophys Biomol Struct 29, 81–103, doi:10.1146/annurev.biophys.29.1.81 (2000).
Majorek, K. A., Kuhn, M. L., Chruszcz, M., Anderson, W. F. & Minor, W. Structural, functional, and inhibition studies of a Gcn5-related N-acetyltransferase (GNAT) superfamily protein PA4794: a new C-terminal lysine protein acetyltransferase from pseudomonas aeruginosa. J Biol Chem 288, 30223–30235, doi:10.1074/jbc.M113.501353 (2013).
Webster, L. T., Siddiqui, U. A., Lucas, S. V., Strong, J. M. & Mieyal, J. J. Identification of separate acyl- CoA:glycine and acyl-CoA:L-glutamine N-acyltransferase activities in mitochondrial fractions from liver of rhesus monkey and man. J Biol Chem 251, 3352–3358 (1976).
Jeffries, K. A., Dempsey, D. R., Behari, A. L., Anderson, R. L. & Merkler, D. J. Drosophila melanogaster as a model system to study long-chain fatty acid amide metabolism. FEBS Lett 588, 1596–1602, doi:10.1016/j.febslet.2014.02.051 (2014).
Farrell, E. K. et al. Primary fatty acid amide metabolism: conversion of fatty acids and an ethanolamine in N18TG2 and SCP cells. J Lipid Res 53, 247–256, doi:10.1194/jlr.M018606 (2012).
Sarker, K. P. et al. ASK1-p38 MAPK/JNK signaling cascade mediates anandamide-induced PC12 cell death. J Neurochem 85, 50–61 (2003).
Siegmund, S. V., Uchinami, H., Osawa, Y., Brenner, D. A. & Schwabe, R. F. Anandamide induces necrosis in primary hepatic stellate cells. Hepatology 41, 1085–1095, doi:10.1002/hep.20667 (2005).
Siegmund, S. V. et al. Fatty acid amide hydrolase determines anandamide-induced cell death in the liver. J Biol Chem 281, 10431–10438, doi:10.1074/jbc.M509706200 (2006).
Davis, R. J. Signal transduction by the JNK group of MAP kinases. Cell 103, 239–252, doi:10.1016/S0092-8674(00)00116-1 (2000).
Weston, C. R. & Davis, R. J. The JNK signal transduction pathway. Curr Opin Genet Dev 12, 14–21 (2002).
Weston, C. R. & Davis, R. J. The JNK signal transduction pathway. Curr Opin Cell Biol 19, 142–149, doi:10.1016/j.ceb.2007.02.001 (2007).
Varfolomeev, E. E. & Ashkenazi, A. Tumor necrosis factor: an apoptosis JuNKie? Cell 116, 491–497 (2004).
Igaki, T. et al. Eiger, a TNF superfamily ligand that triggers the Drosophila JNK pathway. The EMBO journal 21, 3009–3018, doi:10.1093/emboj/cdf306 (2002).
Noselli, S. & Agnes, F. Roles of the JNK signaling pathway in Drosophila morphogenesis. Curr Opin Genet Dev 9, 466–472, doi:10.1016/S0959-437X(99)80071-9 (1999).
Andersen, D. S. et al. The Drosophila TNF receptor Grindelwald couples loss of cell polarity and neoplastic growth. Nature 522, 482–486, doi:10.1038/nature14298 (2015).
Wu, C. et al. Toll pathway modulates TNF-induced JNK-dependent cell death in Drosophila. Open Biol 5, 140171, doi:10.1098/rsob.140171 (2015).
Xue, L. et al. Tumor suppressor CYLD regulates JNK-induced cell death in Drosophila. Dev Cell 13, 446–454, doi:10.1016/j.devcel.2007.07.012 (2007).
Ma, X. et al. Bendless modulates JNK-mediated cell death and migration in Drosophila. Cell Death Differ 21, 407–415, doi:10.1038/cdd.2013.154 (2014).
Martin-Blanco, E. et al. puckered encodes a phosphatase that mediates a feedback loop regulating JNK activity during dorsal closure in Drosophila. Genes Dev 12, 557–570, doi:10.1101/gad.12.4.557 (1998).
Agnes, F., Suzanne, M. & Noselli, S. The Drosophila JNK pathway controls the morphogenesis of imaginal discs during metamorphosis. Development 126, 5453–5462 (1999).
Adachi-Yamada, T. & O’Connor, M. B. Morphogenetic apoptosis: a mechanism for correcting discontinuities in morphogen gradients. Dev Biol 251, 74–90 (2002).
Santabarbara-Ruiz, P. et al. ROS-Induced JNK and p38 Signaling Is Required for Unpaired Cytokine Activation during Drosophila Regeneration. PLoS Genet 11, e1005595, doi:10.1371/journal.pgen.1005595 (2015).
Fogarty, C. E. et al. Extracellular Reactive Oxygen Species Drive Apoptosis-Induced Proliferation via Drosophila Macrophages. Curr Biol 26, 575–584, doi:10.1016/j.cub.2015.12.064 (2016).
Ma, X. et al. dUev1a modulates TNF-JNK mediated tumor progression and cell death in Drosophila. Dev Biol 380, 211–221, doi:10.1016/j.ydbio.2013.05.013 (2013).
Ma, X. et al. Wallenda regulates JNK-mediated cell death in Drosophila. Cell Death Dis 6, e1737, doi:10.1038/cddis.2015.111 (2015).
Wang, X. J. et al. APLP1 promotes dFoxO-dependent cell death in Drosophila. Apoptosis 20, 778–786, doi:10.1007/s10495-015-1097-1 (2015).
Zhang, S. et al. The canonical Wg signaling modulates Bsk-mediated cell death in Drosophila. Cell Death Dis 6, e1713, doi:10.1038/cddis.2015.85 (2015).
Ma, X. et al. NOPO modulates Egr-induced JNK-independent cell death in Drosophila. Cell Res 22, 425–431, doi:10.1038/cr.2011.135 (2012).
Dorstyn, L. et al. The role of cytochrome c in caspase activation in Drosophila melanogaster cells. J Cell Biol 156, 1089–1098, doi:10.1083/jcb.200111107 (2002).
Ollmann, M. et al. Drosophila p53 is a structural and functional homolog of the tumor suppressor p53. Cell 101, 91–101, doi:10.1016/S0092-8674(00)80626-1 (2000).
Hofmann, T. G. et al. Regulation of p53 activity by its interaction with homeodomain-interacting protein kinase-2. Nat Cell Biol 4, 1–10, doi:10.1038/ncb715 (2002).
Vousden, K. H. & Lane, D. P. p53 in health and disease. Nat Rev Mol Cell Biol 8, 275–283, doi:10.1038/nrm2147 (2007).
Fan, Y. et al. Dual roles of Drosophila p53 in cell death and cell differentiation. Cell Death Differ 17, 912–921, doi:10.1038/cdd.2009.182 (2010).
Moreno, E., Yan, M. & Basler, K. Evolution of TNF signaling mechanisms: JNK-dependent apoptosis triggered by Eiger, the Drosophila homolog of the TNF superfamily. Curr Biol 12, 1263–1268 (2002).
McEwen, D. G. & Peifer, M. Puckered, a Drosophila MAPK phosphatase, ensures cell viability by antagonizing JNK-induced apoptosis. Development 132, 3935–3946, doi:10.1242/dev.01949 (2005).
Wang, X. et al. FoxO mediates APP-induced AICD-dependent cell death. Cell Death Dis 5, e1233, doi:10.1038/cddis.2014.196 (2014).
Vidal, M., Larson, D. E. & Cagan, R. L. Csk-deficient boundary cells are eliminated from normal Drosophila epithelia by exclusion, migration, and apoptosis. Dev Cell 10, 33–44, doi:10.1016/j.devcel.2005.11.007 (2006).
Elsum, I., Yates, L., Humbert, P. O. & Richardson, H. E. The Scribble-Dlg-Lgl polarity module in development and cancer: from flies to man. Essays Biochem 53, 141–168, doi:10.1042/bse0530141 (2012).
Liu, D., Shaukat, Z., Saint, R. B. & Gregory, S. L. Chromosomal instability triggers cell death via local signalling through the innate immune receptor Toll. Oncotarget 6, 38552–38565, doi:10.18632/oncotarget.6035 (2015).
Zaccagnino, P. et al. N-arachidonylglycine causes ROS production and cytochrome c release in liver mitochondria. Free Radic Biol Med 47, 585–592, doi:10.1016/j.freeradbiomed.2009.05.038 (2009).
Li, W. Z., Li, S. L., Zheng, H. Y., Zhang, S. P. & Xue, L. A broad expression profile of the GMR-GAL4 driver in Drosophila melanogaster. Genet Mol Res 11, 1997–2002, doi:10.4238/2012.August.6.4 (2012).
Kanda, H., Igaki, T., Kanuka, H., Yagi, T. & Miura, M. Wengen, a member of the Drosophila tumor necrosis factor receptor superfamily, is required for Eiger signaling. J Biol Chem 277, 28372–28375, doi:10.1074/jbc.C200324200 (2002).
Xue, L. & Noll, M. Drosophila female sexual behavior induced by sterile males showing copulation complementation. Proc Natl Acad Sci USA 97, 3272–3275, doi:10.1073/pnas.060018897 (2000).
Acknowledgements
We thank the Bloomington Drosophila Stock Center, Vienna Drosophila Research Center, National Institute of Genetics (NIG-FLY) and Tsinghua Fly Center for fly stocks, members of the Xue laboratory for comments and discussion. This work is supported by the National Natural Science Foundation of China (31371490, 31571516) and Shanghai Committee of Science and Technology (09DZ2260100, 14JC1406000).
Author information
Authors and Affiliations
Contributions
P.R. and L.X. conceived and designed the experiments. P.R. performed the experiments and analyzed the data, P.R., W.L. and L.X. wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ren, P., Li, W. & Xue, L. GLYAT regulates JNK-mediated cell death in Drosophila . Sci Rep 7, 5183 (2017). https://doi.org/10.1038/s41598-017-05482-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-05482-y
- Springer Nature Limited
This article is cited by
-
dGLYAT modulates Gadd45-mediated JNK activation and cell invasion
Cell Division (2022)
-
Toll signaling promotes JNK-dependent apoptosis in Drosophila
Cell Division (2020)