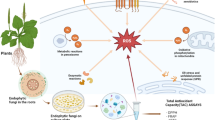
Abstract
Tuta absoluta (L.) (Lepidoptera: Gelechiidae), a major pest of solanaceous plant species, causes serious losses in the agriculture sector around the globe. For better pest management, entomopathogenic fungi such as Beauveria bassiana and Purpureocillium lilacinum, play an efficient role in suppressing the pest population. The present study was carried out to analyse the effects post fungal infections through proteome profiling using an Orbitrap Fusion Tribrid mass spectrometer. A total of 2,201 proteins were identified from the fourth instar larvae of T. absoluta, of which 442 and 423 proteins were significantly dysregulated upon infection with P. lilacinum and B. bassiana respectively. The potential proteins related to immune systems as well as detoxification processes showed significant alterations after post fungal infection. Studies on T. absoluta proteomics and genomics as well as the consequences of entomopathogenic fungal infection on the immune response of this insect could provide an initial framework for exploring more fungus-host interactions for the development of better strategies for integrated pest management.
Measurement(s) | Proteome |
Technology Type(s) | Mass Spectrometry |
Factor Type(s) | Dosage |
Sample Characteristic - Organism | Tuta absoluta |
Similar content being viewed by others
Background & Summary
Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae) is an oligophagous pest causing serious damage to solanaceous crop plants, especially tomato (Solanum lycopersicum L.)1,2. With the increase in global trading and lack of quarantine measures, T. absoluta has spread to almost every tomato producing country, thus threatening 87% of tomato production across the globe3,4,5. As this pest has a high fecundity rate with overlapping generations, which often leads to selection pressure, T. absoluta has developed resistance to numerous insecticides6,7. To overcome the problem of chemical insecticides and to eradicate pest infestation, entomopathogenic microbes (biological control) can play a significant role in reducing insect populations and avoiding damage from chemical residues, which have detrimental effects on humans and the environment8. Among entomopathogenic microbes, entomopathogenic fungi can be considered promising agents to control various insect pests. The generalist entomopathogenic fungi, B. bassiana and P. lilacinum cause pathogenicity to insect hosts by breaching their cuticle and degrading insect cuticular proteins, chitin, and lipids, which are hydrocarbons, in nature9,10,11,12,13,14. After invasion, the hyphae of the fungal pathogen perforate the haemocoel of the insect body and proliferate throughout the insect host to form hyphal bodies after replication15,16. During the process of invasion, entomopathogenic fungi secrete many secondary metabolites that are toxic and immunosuppressive and often lead to the decline of the host defence system, leading to the death of insects17,18. To combat the process of colonization by B. bassiana, the insect reciprocates by triggering the melanization process, producing antimicrobial peptides (AMPs), detoxifying enzymes and reactive oxygen species19,20,21. The evolutionary process has favored to the development of insects to develop a more potent and efficient immune system in insects to combat invading pathogens and parasites in their surroundings22,23. These pathogens trigger innate immune responses after invasion, which comprise cellular and humoral immune reactions.
The present study was carried out to identify proteins in T. absoluta and their alterations upon entomopathogenic fungal infection with an advanced and robust proteomic technique using an Orbitrap Fusion Tribrid mass spectrometer. A comprehensive analysis was carried out to elucidate the potential proteins involved in the host immune response and detoxification by the T. absoluta larvae. These potential proteins have shown significant alterations after entomopathogenic fungal infection. The list of potential proteins is provided in Supplementary Tables 1 & 2 and were obtained after 48 h of infection with B. bassiana and P. lilacinum.
The raw data were obtained from mass spectrometry analysis against the protein databases of Manduca sexta and Plutella xylostella, and a total of 2,201 proteins were identified in T. absoluta. Among these, 1,440 proteins mapped to M. sexta, whereas 878 proteins were mapped from the P. xylostella protein database (http://www.insect-genome.com/data/detail.php?id=19) (Supplementary data file 1). Dynamic changes in the expression of proteins indicate that B. bassiana and P. lilacinum were potent and altered the protein expression in fourth instar larvae of T. absoluta 48 h post-infection. The pathogenicity of the entomopathogenic fungus B. bassiana resulted in the dysregulation (upregulation and downregulation) of 452 proteins, while P. lilacinum revealed 455 proteins in fourth instar larvae of T. absoluta. Upon infection with the entomopathogenic fungi, P. lilacinum and B. bassiana, there was a significant dysregulation at the protein expression level in T. absoluta. The distribution of proteins is shown by volcano plots, where upregulated and downregulated proteins are highlighted in red and blue, respectively (Fig. 1A,B; P. lilacinum-FC 18 and B. bassiana-FC21). Using a threshold of a 2-fold change (FC) difference and <0.05 FDR (adjusted p value), 442 proteins were identified as dysregulated in P. lilacinum-infected larvae when compared with control samples (Supplementary data file 2). Similarly, B. bassiana-infected samples revealed 423 significantly dysregulated proteins in comparison to control samples (FC > 2, FDR < 0.05 p) (supplementary data file 3). Heatmaps for significantly dysregulated proteins show clustering of the two types of treated samples separately from the control samples (Fig. 2A,B).
Volcano plots showing the distribution of proteins in (A) Tuta absoluta infected with P. lilacinum (FC18) when compared with the control sample. (B) Tuta absoluta Infected with B. bassiana (FC21) when compared with control sample, dysregulated proteins were highlighted in red (overexpressed) and blue (downregulated).
Clustering of treated samples and control sample separately in heatmap (A) P. lilacinum (FC18) infected larvae and control. (B) B. bassiana (FC21) infected larvae and control. Red colors indicate the intensity of expression of the protein in the sample and green color represents low expression level.
Gene ontology enrichment analysis showed that both entomopathogenic fungi affected the biological, cellular and molecular processes in T. absoluta. These differentially expressed proteins were involved in various metabolic activities, including oxidation, hydrolysis and conjugation, catalytic activity, etc., which are activated by enzymes with the resultant release of respective metabolites24,25. The bioinformatic analysis revealed many proteins and their interactions with other associated members. The data obtained will provide significant insights into understanding the mechanism of the pathogenicity of B. bassiana and P. lilacinum and their interaction with T. absoluta larvae.
Methods
Insect culture rearing
Adults of T. absoluta were procured from the Germplasm Division, ICAR-NBAIR-Bangalore, India and released into a large sterile breeding chamber with tomato seedlings. The insect culture was maintained under laboratory conditions at 27 ± 2 °C and 55% R.H. The moths were supplied with 10% honey solution as a source of food. Eggs were collected from the breading chamber and reared in rearing jars containing tomato leaves for growth and development. The early 4th instar larvae were collected from rearing jars for treatment with the entomopathogenic fungi B. bassiana and P. lilacinum.
Entomopathogenic fungal strain collection and identification
B. bassiana and P. lilacinum were isolated from soil samples from the agricultural fields of Pune University, Pune, India and were identified on the basis of morphology and sequencing with phylogenetic analysis. Sequences have been submitted to the National Center for Biotechnology Information (NCBI) GenBank with accession numbers MT672601 (Purpureocillium lilacinum) and MT672634. (Beauveria bassiana).
Preparation of fungal conidial suspension for infection
In vitro cultures of B. bassiana and P. lilacinum were raised following a standard procedure on potato dextrose agar plates. The inoculated plates were incubated and maintained at a temperature of 25 ± 1 °C with 70 ± 5% relative humidity for 2 weeks. The spores and hyphae were scraped from a fully grown colony and transferred to sterile water containing 0.01% (vol/vol) Tween-20. The conidia were dispersed by vortexing, and the hyphae were removed by filtering the suspension with a sterilized muslin cloth. The concentration of conidia was measured using a haemocytometer. A final concentration of 1 × 107 conidia mL−1 with sterile 0.01% Tween-20 solution was used for the experiments.
Infection of T. absoluta larvae with B. bassiana and P. lilacinum strains
The fourth instar larvae of T. absoluta were infected with B. bassiana and the P. lilacinum strain separately at a concentration of 1 × 107 conidia mL−1 by the dipping method for 15 sec. For each treatment, 100 larvae were immersed in the conidial suspension and then released onto tomato leaves after removing extra spores. The larvae in the control set were treated with sterile distilled water containing 0.01% Tween-20.
Sample preparation for proteomic analysis
Whole fourth instar larvae were collected from both infected and control sets 48 h post infection. One hundred larvae from each group were ground to a fine powder in liquid nitrogen using a prechilled mortar and pestle. To this, 500 µl extraction buffer (0.5 M Tris-HCl (pH 7.5); 50 mM EDTA; 0.5 M sucrose; 0.1 M KCL and 2% β-mercaptoethanol) was added and vortexed for 10 min. The homogenate was centrifuged at 10,000 rpm for 10 min, and the supernatant was transferred to a new tube. To the supernatant, an equal amount of water-saturated phenol was added, and the mixture was vortexed well for 10 min, and centrifuged at 10,000 rpm for 10 min. The phenol phase was retracted and precipitated with 5 volumes of 0.1 M ammonium acetate. After 24 h, the precipitate was centrifuged at 10,000 rpm for 10 min, and the supernatant was discarded. To the pellet, 200 µl of 0.1 M ammonium acetate was added, the mixture was vortexed and again centrifuged at 10,000 rpm for 5 mins. This process was repeated twice. Then, the obtained pellets were washed with 200 µl of 100% acetone, air dried, suspended in 8 M urea and stored at −70 °C for further use. The entire process of centrifugation and precipitation was carried out at 4 °C. The total proteins from three biological replicates were pooled and dried in a speed vac concentrator and stored at −80 °C until till further use.
Preparation of samples for mass spectrometry
Dried samples were reconstituted in 50 mM triethylammonium bicarbonate buffer and processed for in solution digestion. Briefly, the protein concentration of the samples was measured by a PierceTM BCA Protein Assay Kit. Based on the protein concentration, equal protein amounts (~100 µg) from each sample were taken. Reduction of the samples was carried out using a final concentration of 5 mM dithiothreitol (DTT) at 60 °C for 30 mins followed by alkylation by 10 mM iodoacetamide (IAA) for 30 min in the dark at room temperature. Samples were digested using Promega sequencing grade modified Trypsin at a ratio of 1:20 (enzyme: protein) at 37 °C overnight. Peptides were then acidified by 1% formic acid (FA) and were cleaned by a Sep-Pak C18 cartridge. Eluted samples were dried in a Speed Vac and stored at −80 °C until further analysis.
LC–MS/MS analysis
Data acquisition for digested samples was carried out on a Thermo ScientificTM Orbitrap Fusion Tribrid mass spectrometer (Thermo Fisher Scientific, Bremen, Germany) interfaced with a Thermo Scientific EASY-nLC 1000 (Thermo Fisher Scientific, Germany). Peptides were reconstituted in 0.1% formic acid and loaded onto a trap column (Thermo ScientificTM AcclaimTM PepMapTM 100 C18 LC Column, 75 µm × 2 cm, 3 µm). Peptides were resolved on an analytical column (Thermo ScientificTM AcclaimTM PepMapTM 100 C18 LC Column, 75 µm × 50 cm, 2 µm) at a flow rate of 300 nl/min using an optimized linear gradient of 8–32% solvent B (0.1% formic acid in 100% ACN) over 100 min. The total run time for each replicate, including sample loading and column reconditioning, was 120 min. One microgram of peptide was used in each mass spectrometer run. The MS data acquisition was carried out from the 350–1600 m/z range using an Orbitrap mass analyser. The AGC target was set to 400,000 with an ion injection time of 50 ms and dynamic exclusion of 30 sec. Precursor ions were fragmented using high energy collision dissociation (HCD) (NCE 32%) and analysed using an Orbitrap mass analyser with a resolution of 15000. For MS/MS scans, the AGC target was set to 100,000 with an ion injection time of 100 ms. Samples were analysed in technical triplicates.
Data Records
The mass spectrometry proteomics data have been deposited in the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) using the PRIDE partner repository with the dataset identifier PXD02937426,27,28.
Technical Validation
Data analysis
The MS/MS raw data searches were carried out using SEQUEST search algorithms using Proteome Discoverer 2.2 (Thermo Fisher Scientific, Bremen, Germany) against RefSeq protein databases of Manduca sexta and Plutella xylostella. The search parameters involved carbamidomethylation at cysteine residues (+57.021 Da) as a fixed modification and oxidation of methionine (+15.995 Da) as a dynamic modification. The precursor and fragment mass tolerances were set to 10 ppm and 0.05 Da, respectively. Trypsin was specified as protease, and a maximum of two missed cleavages were allowed. An FDR of 1% was applied as a cut-off value for reporting identified peptides. All 9 raw files pertaining to the three samples were searched against databases. In the peptide and protein quantifier node of the proteome discoverer, we specified the total peptide intensity of each sample for normalization. This node calculates the sum of abundance values of all peptides identified in each raw file referred to as the channel hereafter. The channel with the highest total abundance is then used as a reference to normalize all other channels by a constant factor per channel. In the end, the total abundance was the same for all channels. The software groups the abundance values of each peptide across three technical replicates. For statistical analysis we used Perseus proteomics software (version 1.6.2.2), where Student’s T test was performed to calculate p values. A fold change cut-off of 1.5 was applied to determine significantly dysregulated proteins.
Gene ontology analysis
Gene ontology analysis of differentially expressed proteins was performed by the Blast2GO® software package (BioBam Bioinformatics Solutions, Spain). The protein sequences were imported into Blast2GO® in Fasta format and aligned against the nonredundant (nr) NCBI database using the inbuilt Blast function (blastp) with an e-value cut-off of x10−3. The resulting hits were matched against the Gene Ontology Annotation database, and Gene Ontology was assigned to each hit on the basis of protein sequences using the mapping and annotation functions. The blast hits obtained were also subjected to Gene Ontology assignment based on protein domains and families using the InterProScan. The Gene Ontology results from the two steps were then merged together, followed by the generation of relevant graphs. The summary statistics of the annotation are provided in Table 1. The full list includes enriched GO terms for biological processes (BP), cellular components (CC) and molecular functions (MF) in T. absoluta infected with P. lilacinum and B. bassiana using blast2GO analysis along with the genes for each term. The topmost enriched GO terms for P. lilacinum and B. bassiana are summarized in Fig. 3 (A: Biological Processes; B: Cellular Compartments; C: Molecular Functions). Of the top enriched biological process terms, proteolysis, ion transport, nitrogen compound transport, organic substance catabolic process, and carbohydrate derivative metabolic process were enriched by when infection with P. lilacinum, whereas cellular protein modification process, catabolic process, and organic cyclic compound biosynthetic process were enriched after B. bassiana infection. From the uppermost enriched cellular component terms, the endomembrane system was enriched with P. lilacinum treatment, whereas in B. bassiana most of the proteins were localized to the endoplasmic reticulum. The top enriched molecular function was common to both datasets, although the enrichment score varied.
Gene ontology of dysregulated genes upon EPF infection of P. lilacinum and B. bassiana Bar chart showing the summary of Top 10 blast hits infected with P. lilacinum and B. bassiana datasets (A). Bar charts depicting the top enriched terms for Biological Processes (B), Cellular Compartment (C), and Molecular Functions (D). The detailed results are shown in Table 1.
Furthermore, with the protein sequences and abundance values obtained from the mass spectrometry analysis, we used the abundance ratio column for the classification of upregulated and downregulated genes. The genes whose abundance ratio was greater than 1 with respect to the control were classified as upregulated genes, and those whose abundance ratio was less than 1 were considered downregulated genes. In addition, gene enrichment analysis of biological processes (upregulated and downregulated) of T. absoluta proteins was also performed separately after entomopathogenic fungal infection. B. bassiana (FC21) showed 213 upregulated genes and 210 downregulated genes and the genes involved in organic substance transport, protein localization to organelles etc., were upregulated, whereas those involved in cytoskeleton organization, actin filament-based processes etc., were downregulated (Supplementary Fig. 1i & ii). Similarly, P. lilacinum (FC18) infection resulted in 203 upregulated genes and 242 downregulated genes. Genes involved in organic substance transport, nucleotide-containing small molecule metabolic processes etc., were upregulated whereas cytoskeleton organisation, actin filament-based process etc. are downregulated (Supplementary Fig. 1iii & iv). Most importantly, through KEGG pathway analysis, we found that some of the proteins were involved in melanin biosynthesis (Supplementary Fig. 2). Melanin is known as an important defence molecule in invertebrate immunity29,30.
Code availability
Perseus proteomics software version 1.6.2.2 (https://www.perseus-framework.org) was used for statistical analysis, where p values were calculated by Students t test. The volcano plots and heatmaps were also generated from the Perseus proteomics tools. The differentially expressed proteins were further subjected to Gene Ontology analysis using the Blast2GO® software package (BioBam Bioinformatics Solutions, Spain) (https://www.biobam.com/omicsbox). These annotated genes were used for enrichment analysis of the gene ontologies of upregulated and downregulated genes by clusterProfiler version 4.2.2 (https://bioconductor.org).
References
Desneux, N. et al. Biological invasion of European tomato crops by Tuta absoluta: Ecology, geographic expansion and prospects for biological control. Journal of Pest Science 83 (2010).
Biondi, A., Guedes, R. N. C., Wan, F. H. & Desneux, N. Ecology, Worldwide Spread and Management of the Invasive South American Tomato Pinworm, Tuta absoluta: Past, Present, and Future. Annual Review of Entomology 63, 239–258, https://doi.org/10.1146/annurev-ento-031616-034933 (2018).
Campos, M. R., Biondi, A., Adiga, A., Guedes, R. N. C. & Desneux, N. From the Western Palaearctic region to beyond: Tuta absoluta 10 years after invading Europe. Journal of Pest Science 90 (2017).
Desneux, N., Luna, M. G., Guillemaud, T. & Urbaneja, A. The invasive South American tomato pinworm, Tuta absoluta, continues to spread in Afro-Eurasia and beyond: the new threat to tomato world production. Journal of Pest Science 84, 403–408, https://doi.org/10.1007/s10340-011-0398-6 (2011).
Olaniyi, J. O., Akanbi, W. B., Adejumo, T. a. & Akande, O. G. Growth, fruit yield and nutritional quality of tomato varieties. African Journal of Food Science 4 (2010).
Guedes, R. N. C. & Siqueira, H. A. A. The tomato borer Tuta absoluta: Insecticide resistance and control failure. CABI Reviews: Perspectives in Agriculture, Veterinary Science, Nutrition and Natural Resources 15, 1–7, https://doi.org/10.1079/PAVSNNR20127055 (2012).
Guedes, R. N. C. et al. Insecticide resistance in the tomato pinworm Tuta absoluta: patterns, spread, mechanisms, management and outlook. Journal of Pest Science 92, 1329–1342, https://doi.org/10.1007/s10340-019-01086-9 (2019).
Paraïso, A. Comparative study of the efficacy of entomopathogenic fungi, chemicals and botanical pesticides in the management of cotton pests and their natural enemies in Benin. International Journal of Science and Advanced Technology 3 (2013).
Zhang, S. et al. CYP52X1, representing new cytochrome P450 subfamily, displays fatty acid hydroxylase activity and contributes to virulence and growth on insect cuticular substrates in entomopathogenic fungus Beauveria bassiana. Journal of Biological Chemistry 287 (2012).
Pedrini, N., Ortiz-Urquiza, A., Huarte-Bonnet, C., Zhang, S. & Keyhani, N. O. Targeting of insect epicuticular lipids by the entomopathogenic fungus Beauveria bassiana: Hydrocarbon oxidation within the context of a host-pathogen interaction. Frontiers in Microbiology 4 (2013).
Fang, W. et al. Expressing a fusion protein with protease and chitinase activities increases the virulence of the insect pathogen Beauveria bassiana. Journal of Invertebrate Pathology 102 (2009).
Fang, W. et al. Cloning of Beauveria bassiana chitinase gene Bbchit1 and its application to improve fungal strain virulence. Applied and Environmental Microbiology 71 (2005).
st. Leger, R. J., Cooper, R. M. & Charnley, A. K. Cuticle degrading enzymes of entomopathogenic fungi: Regulation of production of chitinolytic enzymes. Journal of General Microbiology 132 (1986).
st. Leger, R. J., Charnley, A. K. & Cooper, R. M. Cuticle-degrading enzymes of entomopathogenic fungi: Synthesis in culture on cuticle. Journal of Invertebrate Pathology 48 (1986).
Lewis, M. W., Robalino, I. v. & Keyhani, N. O. Uptake of the fluorescent probe FM4-64 by hyphae and haemolymph-derived in vivo hyphal bodies of the entomopathogenic fungus Beauveria bassiana. Microbiology (N Y) 155 (2009).
Wanchoo, A., Lewis, M. W. & Keyhani, N. O. Lectin mapping reveals stage-specific display of surface carbohydrates in in vitro and haemolymph-derived cells of the entomopathogenic fungus Beauveria bassiana. Microbiology (N Y) 155 (2009).
Trienens, M. & Rohlfs, M. Insect-fungus interference competition - The potential role of global secondary metabolite regulation, pathway-specific mycotoxin expression and formation of oxylipins. Fungal Ecology 5 (2012).
Han, Y. et al. Functional analysis of arabidopsis mutants points to novel roles for glutathione in coupling H2O2 to activation of salicylic acid accumulation and signaling. Antioxidants and Redox Signaling 18 (2013).
Strand, M. R. Insect Hemocytes and Their Role in Immunity. in Insect Immunology, https://doi.org/10.1016/B978-012373976-6.50004-5 (2008).
Lemaitre, B. & Hoffmann, J. The host defense of Drosophila melanogaster. Annual Review of Immunology 25, 697–743, https://doi.org/10.1146/annurev.immunol.25.022106.141615 (2007).
Christophides, G. K. et al. Immunity-related genes and gene families in Anopheles gambiae. Science (1979) 298 (2002).
Lemaitre, B. The road to Toll. Nature Reviews Immunology 4(7), 521–527, https://doi.org/10.1038/nri1390 (2004).
Pal, S. & Wu, L. P. Lessons from the fly: Pattern recognition in Drosophila melanogaster. Advances in Experimental Medicine and Biology 653 (2009).
Che-Mendoza, A., Penilla, R. P. & Rodriguez, D. A. Insecticide resistance and glutathione S-transferases in mosquitoes: A review. African Journal of Biotechnology 8, 1386-1397 (2009).
Lü, D., Xu, P., Hou, C., Gao, K. & Guo, X. Label-free LC-MS/MS proteomic analysis of the hemolymph of silkworm larvae infected with Beauveria bassiana. Journal of Invertebrate Pathology 166 (2019).
Deutsch, E. W. et al. The ProteomeXchange consortium in 2020: Enabling “big data” approaches in proteomics. Nucleic Acids Research 48 (2020).
Perez-Riverol, Y. et al. The PRIDE database resources in 2022: A hub for mass spectrometry-based proteomics evidences. Nucleic Acids Research 50 (2022).
Bali, G. K. et al. An insight in proteome profiling of Tuta absoluta larvae after entomopathogenic fungal infection. Pride Archive https://identifiers.org/pride.project:PXD029374 (2022).
Cerenius, L., Lee, B. L. & Söderhäll, K. The proPO-system: pros and cons for its role in invertebrate immunity. Trends in Immunology 29, 263–271, https://doi.org/10.1016/j.it.2008.02.009 (2008).
Kanost, M. R. & Gorman, M. J. Phenoloxidases in Insect Immunity. in Insect Immunology https://doi.org/10.1016/B978-012373976-6.50006-9 (2008).
Acknowledgements
Ms. G K Bali is thankful for the university fellowship received from the Savitribai Phule Pune University for her research work and RSP wishes to acknowledge UGC CAS Phase III, DST-PURSE for the financial assistance.
Author information
Authors and Affiliations
Contributions
G.K.B., R.S.P. and S.K.S. designed the study; R.S.P. contributed to the project coordination. G.K.B., R.V. and B.R. collected the sample and maintained in the laboratory under optimal conditions; G.K.B. performed the experiments. G.K.B., F.A.B., W.A.M. and N.J. analyzed and evaluated the data. G.K.B., V.K.C. and M.K. wrote the paper which was critically evaluated and edited by R.S.P. and S.K.S. The research funding was procured by R.S.P. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bali, G.K., Singh, S.K., Chauhan, V.K. et al. An insight in proteome profiling of Tuta absoluta larvae after entomopathogenic fungal infection. Sci Data 9, 507 (2022). https://doi.org/10.1038/s41597-022-01593-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-022-01593-y
- Springer Nature Limited
This article is cited by
-
Morphology, phylogeny, and pathogenicity of Simplicillium obclavatum (Hypocreales: Cordycipitaceae) against tomato leafminer, Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae)
International Journal of Tropical Insect Science (2023)