Abstract
Chinese cavefishes are a bizarre and interesting vertebrate taxa, but one with relatively little research. China holds the highest global cavefish diversity, accounting for about one-third of known species. Sinocyclocheilus is the largest genus of cavefishes in the world and is endemic to the south of China. The distribution of Sinocyclocheilus species is very narrow, and sometimes they inhabit just a single cave; this feature increases the vulnerability to extinction. With this study we provide the first comprehensive dataset related to the morphometrics of eight Sinocyclocheilus species. In addition to enhancing our knowledge on these poorly known species we aim to provide a dataset useful for future comparative analyses aiming to better understand the adaptive ability of cavefishes.
Measurement(s) | morphometry |
Technology Type(s) | digital camera • computational modeling technique |
Factor Type(s) | species • Museum collection |
Sample Characteristic - Organism | Sinocyclocheilus |
Sample Characteristic - Environment | groundwater |
Sample Characteristic - Location | China |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.9963206
Similar content being viewed by others
Background & Summary
Cavefishes are one of the least studied vertebrate taxa globally1,2. These fish are restricted to groundwater environments (from here the name stygo-fauna)3,4, habitats which are generally difficult to explore, even for specialists5,6. Cavefishes show a range of adaptations to subterranean environments7,8, namely habitats characterized by particular features such as the general lack of light and limited availability of food resources9,10. Such adaptations may involve changes in fish behaviour and physiology, but also the development (or regression) of various organs and body shapes2,11,12. These adaptations are most evident in obligate cave species (stygobites) as they are normally not able to exit caves and thus, high adaptation degree to subterranean environments is beneficial for their fitness13. Contrary to this facultative cave-dwellers (stygophiles) show many fewer specialist adaptations to subterranean environments as they show a mixed lifestyle, alternating between hypogean and epigean phases14,15.
China hosts over 150 cavefish species, accounting for about one-third of known species worldwide16. South China Karst hosts the majority of Chinese cavefishes, and most of them are endemic to small areas or even to single caves, condition increasing their conservation concerns16,17,18. Chinese cavefishes mainly belong to the order Cypriniformes; only one species is ascribed to the order Siluriformes8. The genus Sinocyclocheilus, with more than 70 known species, is the worldwide largest known group of cavefishes and is endemic to South China16,19. Its diversity occurs in a relatively narrow area, suggesting high adaptability of these fishes to the subterranean environment16,20; indeed, more than half of Sinocyclocheilus species are stygobites8. Four different clades are recognized within Sinocyclocheilus fishes: “jii”, “cyphotergous,”, “tingi” and “angularis”8,21. In all four clades some fishes develop the humpback, a peculiar morphological shape thought to serve as energy storage in environments where food supply is not constant8. In the “angularis” clade a further bizarre onward projection (the so called “horn”) is also present, but its function is still unknown16,22.
Sinocyclocheilus fishes are an important component of both Chinese and worldwide biodiversity16,19,20; yet, almost no effort is dedicated to their protection1,18. Cave species are often very susceptible to environmental changes23,24,25 and their narrow distribution increases their vulnerability to extinction risk26,27,28. However a lack of clear information on the distribution or ecology of this taxa impedes effective conservation management and prioritisation1,29,30. Although the genetic and genomic of Chinese cavefishes were the topic of several scientific papers (Refs21,31 among them), no other studies on their ecology, behavior or life history exist.
In the present work, we report the most comprehensive dataset on the morphology of eight Chinese cavefishes belonging to the genus Sinocyclocheilus (S. brevibarbatus, S. brevis, S. huanjiangensis, S. jii, S. lateristritus, S. mashanensis, S. microphthalmus and S. qiubeiensis). At present information on these species is virtually absent, and the limited available data lies only in Chinese literature19. Our goal is therefore to raise awareness of Chinese cavefishes, providing useful data to be employed in future comparative analyses with other cavefishes; in this way, divergences and similarities in adaptive abilities across different species worldwide could be assessed. Our study will contribute in improving species knowledge, an important step towards species protection29. To do that, we started with sharing the information related to the specimens present in the collection of the Institute of Zoology of the Chinese Academy of Sciences in Beijing (China), which holds the biggest collection of Chinese cavefishes. Specimens from different species and populations are present in this collection and they were often used in taxonomic and phylogenetic studies.
Methods
Experimental design
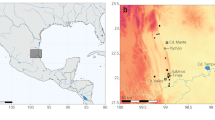
We examined specimens belonging to 8 species of Chinese cavefishes from the collection of the Institute of Zoology of the Chinese Academy of Sciences in Beijing (China). The examined species inhabit groundwater environments in the Provinces of Guangxi (N species = 6) and Yunnan (N species = 2) (Fig. 1). We built up a large database including date and locality of fish collection, the description of their body organs and morphometrics. When precise coordinates were present, we provide a specific code (species initials + a number) to distinguish between different populations (Table 1). According to the standard methodology used to record fishes’ morphology32, we identified multiple landmarks from which measurements were taken (Fig. 2). These points correspond to the following body parts: A (snout tip); B (nostril); C (eye); D (top end of the head); E (farthest backward end of the head); F (beginning of the forward pectoral fin base); G (end of the forward pectoral fin base); H (farthest end of the forward pectoral fin lobe); I (beginning of dorsal fin base); J (beginning of the backward pectoral fin base); K (end of dorsal fin base); L (farthest end of the backward pectoral fin lobe); M (farthest end of the dorsal fin lobe); N (beginning of the anal fin base); O (end of the anal fin base); P (farthest end of the anal fin lobe); Q (top beginning of caudal fin); R (low beginning of caudal fin); S (middle point between Q and R); T (median end of the caudal fin lobe); U (farthest end of the top caudal fin lobe); V (farthest end of the low caudal fin lobe); W (end of the backward pectoral fin base). Alongside measurements involving the above listed points (see below), we also recorded data from additional parts of fishes’ body (identified with dashed lines, Fig. 2): Snout (distance between the mouth tip and the beginning of the eye); Eye (eye diameter); Eyeball (eyeball diameter); Mouth width (length between the two mouth angles); Mouth length (length of the lower jaw).
Map of the area from where the specimens were collected. Each symbol corresponds to a distinct population of the following species: Sinocyclocheilus brevibarbatus (blue), S. brevis (yellow), S. huanjiangensis (green), S. jii (violet), S. lateristritus (brown), S. mashanensis (red), S. microphthalmus (orange), S. qiubeiensis (pink). Maps were created with the program QGIS37 using data from http://www.ngcc.cn/ngcc//html/1/.
Reference adopted in fishes’ body measurements. In the figure two individuals of (a) S. microphtalmus and (b) S. jii showing all the landmarks and dashed lines used to record measurements on (a) the lateral and (b) the ventral side of the fish body (see the main text for a detailed description of the recorded measurements). Shaded area indicates the fish humpback.
Specimens sampling
We first described the shape of three body organs: the eye, the mouth and the caudal fin. For the eye, we considered three different categories according to the eye ball’s development degree: “Developed” when is fully developed; “Reduced” when is small and poorly developed; “Absent” when the fish lack eyes (Fig. 3). We then described the mouth position according to where the opening occurs: “Terminal” if it opens at the tip of the fish head; “Subterminal” if it opens close to the tip head but downward; “Inferior” if it opens downward; “Superior” if it opens upward (Fig. 3). We finally described the caudal fin according to its shape. We used five different shape categories: “Rounded”, “Truncate”, “Emarginate”, “Forked”, “Lunate” (Fig. 3).
Reference showing the different shapes of the considered cavefish organs. Eye: Developed, Reduced, Absent. Mouth: Terminal, Subterminal, Inferior, Superior. Caudal fin: Rounded, Truncate, Emarginate, Forked, Lunate. Images for mouth and caudal fin are modified from38.
After the first descriptive part, we recorded measurements of the fishes’ body parts. Measurements were taken using a digital calliper and analysing pictures of specimens. Digital calliper was used to record measurements hardly visible form pictures; in the following table, morphometrics recorded using this methodology are indicated with the symbol “*”. Pictures were taken using a digital camera and placing fishes on a light background with a ruler as a scale. Files were then analysed with the software ImageJ. Once the scale was settled, the distance between two points (Fig. 2) was measured with a straight line; the same method was used to evaluate the length of dashed lines (Fig. 2).
The recorded measures were the following:
-
Eye*;
-
Eye_ball*;
-
Snout*;
-
Mouth width*;
-
Mouth length*;
-
AD: linear distance between the snout tip and the top end of the head;
-
B_height: head height measured at the nostril;
-
C_height: head height measured at the eye;
-
D_height: head height measured at the upper end;
-
DI: linear distance between the top end of the head and the beginning of the dorsal fin;
-
AE: maximum head length, measured from the snout tip until the farthest backward end of the head;
-
FG: length of the forward pectoral fin base;
-
FH: maximum extension of the forward pectoral fin;
-
IM: maximum extension of the dorsal fin;
-
IK: length of the dorsal fin base;
-
I_depth: body depth measured at the beginning of the dorsal fin base;
-
JL: maximum extension of the backward pectoral fin;
-
JW: length of the backward pectoral fin base*;
-
K_depth: body depth measured at the end of the dorsal fin base;
-
NO: length of the anal fin base;
-
O_depth: body depth measured at the end of the anal fin base;
-
NP: maximum extension of the anal fin;
-
QR: caudal fin height at its base;
-
QU: maximum extension of the top part of the caudal fin;
-
ST: caudal fin mid length;
-
RV: maximum extension of the lower part of the caudal fin;
-
AS: standard length;
-
AT: total length.
Besides the above mentioned fish standard lengths, we recorded the measurement of a specific body part characterizing Chinese cavefishes: the humpback area8. This peculiar structure develops on the fish back, between the head and the dorsal fin (Fig. 2), and it is used to store energy, a practical adaptation to food deprived environments8,9. The humpback area (DID) is located above the DI segment (shaded area in Fig. 2a) and was delimited connecting back D from I following the animal shape.
Data Records
The dataset (Morphometrics of eight Chinese cavefishes33) consists of:
-
1.
451 specimens belonging to eight Sinocyclocheilus species of Chinese cavefishes (S. brevibarbatus N = 34, S. brevis N = 31, S. huanjiangensis N = 42, S. jii N = 140, S. lateristritus N = 44, S. mashanensis N = 16, S. microphthalmus N = 101, S. qiubeiensis N = 43).
-
2.
Description of three organs: eye, mouth and caudal fin.
-
3.
Measurements of 28 fish body parts (27 in four species because their eye diameter equals the eye ball diameter).
-
4.
NA means no specific data existing. Preserved specimens were not always integer or in some cases, after their fixation in alcohol, their original shape was not well conserved. This was also used in the category “Eye” when eye diameter equals the diameter of the eye ball. Furthermore, NA was used in the “Population” column to indicate that precise coordinates were not present.
Detailed explanation of dataset Morphometrics of eight Chinese cavefishes33 is given in Table 1.
Technical Validation
Studied specimens belong to the fish collection of National Zoological Museum, the Institute of Zoology, Chinese Academy of Sciences (ASIZB)34; with an appropriated request, the same fishes can be further studied. Blinded fish measurements were performed to further reduce any possible bias35. The whole dataset was double-checked for any possible error. Outliers were identified in two ways: before by visual check (i.e., plotting the data), and then using three times the standard deviation from the data mean (+/−) as cut-off. Successively, the relative measurement was taken again to check whether the outlier was due to measurement mistakes.
Usage Notes
Dataset is provided in CSV format, ready to be used with statistic programs like R (http://www.R-project.org/) and PAST. Precise coordinates of collection points are not shown to increase species protection36. Data were collected with instruments allowing high precision (0.01 mm). Prior to any analyses, we suggest to log-transform the measures to improve linearity and reduce skewness.
Code availability
No code was used in this study.
References
Zhao, Y.-H., Gozlan, R. E. & Zhang, C.-G. Out of sight out of mind: current knowledge of Chinese cave fishes. J. Fish. Biol 79, 1545–1562 (2011).
Trajano, E., Bichuette, M. & Kapoor, B. G. Biology of Subterranean Fishes. (Science Publishers, 2010).
Romero, A. The biology of hypogean fishes. (Springer Science + Business Media, 2001).
Gibert, J., Stanford, J. A., Dole-Olivier, M.-J. & Ward, J. V. In Groundwater Ecology. (eds Gibert, J., Danielopol, D. L & Stanford, J. A.) Ch. 1 (Academic Press, 1994).
Buzzacott, P. L., Zeigler, E., Denoble, P. & Vann, R. American cave diving fatalities 1969–2007. Int. J. Aqua. Res. Educ. 3, 162–177 (2009).
Zagmajster, M., Culver, D. C., Christman, M. C. & Sket, B. Evaluating the sampling bias in pattern of subterranean species richness: combining approaches. Biodivers. Conserv. 19, 3035–3048 (2010).
Poulson, T. L. Cave adaptation in Amblyopsid fishes. American Midland Naturalist 70, 257–290 (1963).
Li, M., Zhao, Y. & Yang, J. In Encyclopedia of Caves 3rd edn. (eds White, W., Culver, D. C. & Pipan, T) Ch. 27 (Academic Press, 2019).
Culver, D. C. & Pipan, T. The biology of caves and other subterranean habitats. (Oxford University Press, 2009).
Romero, A. The Evolution of Cave. Life. Am. Sci. 99, 144–151 (2011).
Parzefall, J. A review of morphological and behavioural changes in the cave molly, Poecilia mexicana, from Tabasco, Mexico. Environ. Biol. Fishes 62, 263–275 (2001).
Culver, D. C. & Pipan, T. Paradigms of the evolution of cave life. Acta Carsol 44, 415–425 (2015).
Brandon, R. N. Adaptation and evolutionary theory. Stud. Hist. Philos. Sci. 9, 181–206 (1978).
Trajano, E. & de Carvalho, M. R. Towards a biologically meaningful classification of subterranean organisms: a critical analysis of the Schiner-Racovitza system from a historical perspective, difficulties of its application and implications for conservation. Subterr. Biol 22, 1–26 (2017).
Lunghi, E., Manenti, R. & Ficetola, G. F. Do cave features affect underground habitat exploitation by non-troglobite species? Acta Oecol. 55, 29–35 (2014).
Zhao, Y. et al. In Cave Life - Drivers of Diversity and Diversification. (ed. Wynne, J. J.) (Nova Science Publisher, in press).
Manenti, R. et al. The stenoendemic cave-dwelling planarians (Platyhelminthes, Tricladida) of the Italian Alps and Apennines: conservation issues. J. Nat. Conserv. 45, 90–97 (2018).
Zhao, Y. & Zhang, C. Threatened fishes of the world: Sinocyclocheilus anophthalmus (Chen and Chu, 1988) (Cyprinidae). Environ. Biol. Fishes 86, 163 (2009).
Zhao, Y. & Zhang, C. Endemic Fishes of Sinocyclocheilus (Cypriniformes: Cyprinidae) in China - Species diversity, cave adaptation, systematics and zoogeography. (Science Press, 2009).
Xing, Y., Zhang, C., Fan, E. & Zhao, Y. Freshwater fishes of China: species richness, endemism, threatened species and conservation. Divers. Distrib. 22, 358–370 (2016).
Xu-Fang, L., Cao, L. & Zhang, C.-G. Molecular phylogeny of the Sinocyclocheilus (Cypriniformes: Cyprinidae) fishes in northwest part of Guangxi, China. Environ. Biol. Fishes 92, 371–379 (2011).
Huang, J., Gluesenkamp, A., Fenolio, D., Wu, Z. & Zhao, Y. Neotype designation and redescription of Sinocyclocheilus cyphotergous (Dai) 1988, a rare and bizarre cavefish species distributed in China (Cypriniformes: Cyprinidae). Environ. Biol. Fishes 100, 1483–1488 (2017).
Manenti, R., Barzaghi, B., Tonni, G., Ficetola, G. F. & Melotto, A. Even worms matter: cave habitat restoration for a planarian species increased environmental suitability but not abundance. Oryx, 1–6 (2018).
Suwannapoom, C. et al. A striking new genus and species of cave-dwelling frog (Amphibia: Anura: Microhylidae: Asterophryinae) from Thailand. PeerJ 6, e4422 (2018).
Bland, L. M. Global correlates of extinction risk in freshwater crayfish. Anim. Conserv. 20, 532–542 (2017).
Carrascal, L. M., Moreno, Á. C., Delgado, A., Suárez, V. & Trujillo, D. Habitat suitability-density relationship in an endangered woodland species: the case of the Blue Chaffinch (Fringilla polatzeki). PeerJ 5, e3771 (2017).
Cejuela Tanalgo, K., Tabora, J. A. G. & Hughes, A. C. Bat cave vulnerability index (BCVI): A holistic rapid assessment tool to identify priorities for effective cave conservation in the tropics. Ecol. Indic. 89, 852–860 (2018).
Williams, S. E. et al. Ecological specialization and population size in a biodiversity hotspot: How rare species avoid extinction. Proc. Natl. Acad. Sci. USA 106, 19737–19741 (2009).
Hochkirch, A. The insect crisis we can’t ignore. Nature 539, 141 (2016).
Bressi, N. Underground and unknown: updated distribution, ecological notes and conservation guidelines on the Olm Proteus anguinus anguinus in Italy (Amphibia, Proteidae). Ital. J. Zool. 71, 55–59 (2004).
Yang, J. et al. The Sinocyclocheilus cavefish genome provides insights into cave adaptation. BMC Biol. 14, 1 (2016).
Zhao, Y., Watanabe, K. & Zhang, C. Sinocyclocheilus donglanensis, a new cavefish (Teleostei: Cypriniformes) from Guangxi, China. Ichthyol. Res. 53(121), 128 (2006).
Lunghi, E., Zhao, Y., Sun, X. & Zhao, Y. Morphometrics of eight Chinese cavefish species. figshare, https://doi.org/10.6084/m9.figshare.c.4621676 (2019).
Leviton, A. E., Gibbs, R. H. J., Heal, E. & Dawson, C. W. Standards in Herpetology and Ichthyology: part I. Standard symbolic codes for institutional resource collections in Herpetology and Ichthyology. Copeia 1985, 802–832 (1985).
MacCoun, R. & Perlmutter, S. Hide results to seek the truth. Nature 526, 187–189 (2015).
Lunghi, E., Corti, C., Manenti, R. & Ficetola, G. F. Consider species specialism when publishing datasets. Nat. Ecol. Evol 3, 319 (2019).
QGIS Development Team. QGIS Geographic Information System, version 3.8.1. Open Source Geospatial Foundation Project (2019).
Hastings, P. A., Walker, H. J. & Galland, G. R. Fishes. A Guide to Their Diversity. (University of California Press, 2014).
Acknowledgements
We thank A. C. Hughes to have checked the spelling of our manuscript. This study was conducted under the auspices of the Chinese Cavefish Working Group and supported by a grant from National Natural Science Foundation of China (NSFC- 31972868), a grant (No. Y229YX5105) from the Key Laboratory of the Zoological Systematics and Evolution of the Chinese Academy of Sciences, a grant (P.PROJ.GEN > FH01_1819) from Ocean Park Conservation Foundation, Hong Kong, and a grant (NSFC-31471961) from the National Nature Science Foundation of China. Enrico Lunghi is supported by the Chinese Academy of Sciences President’s International Fellowship Initiative for postdoctoral researchers.
Author information
Authors and Affiliations
Contributions
E.L. conceived the study, prepared tables, figures and first draft of the manuscript; E.L., Yan.Z. and X.S. examined and measured fish specimens; E.L. and Yah.Z. revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Lunghi, E., Zhao, Y., Sun, X. et al. Morphometrics of eight Chinese cavefish species. Sci Data 6, 233 (2019). https://doi.org/10.1038/s41597-019-0257-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-019-0257-5
- Springer Nature Limited
This article is cited by
-
Photographic database of the European cave salamanders, genus Hydromantes
Scientific Data (2020)






