Abstract
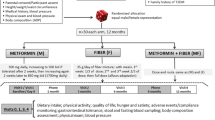
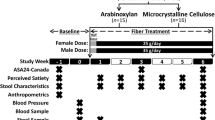
Fecal microbial transplantation (FMT) from lean donors to patients with obesity has been associated with metabolic benefits, yet results so far have been inconsistent. In this study, we tested the application of daily fiber supplementation as an adjunct to FMT therapy to modulate cardiometabolic outcomes. We performed a double-blind randomized trial in patients with severe obesity and metabolic syndrome receiving oral FMT, to test high-fermentable (HF) and low-fermentable (LF) fiber supplements (NCT03477916). Seventy participants were randomized to the FMT-HF (n = 17), FMT-LF (n = 17), HF (n = 17) and LF (n = 19) groups. The primary outcome was the assessment of change in insulin sensitivity from baseline to 6 weeks using the homeostatic model assessment (HOMA2-IR/IS). After 6 weeks, only patients in the FMT-LF group had significant improvements in HOMA2-IR (3.16 ± 3.01 at 6 weeks versus 3.77 ± 3.57 at baseline; P = 0.02). No difference in HOMA2-IR was observed over this period for those in the FMT-HF group (3.25 ± 1.70 at 6 weeks versus 3.17 ± 1.72 at baseline; P = 0.8), the HF group (3.49 ± 1.43 at 6 weeks versus 3.26 ± 1.33 at baseline; P = 0.8) or the LF group (3.76 ± 2.01 at 6 weeks versus 3.56 ± 1.81 at baseline; P = 0.8). Interventions were safe and well-tolerated with no treatment-attributed serious adverse events. We provide proof of concept for the use of a single-dose oral FMT combined with daily low-fermentable fiber supplementation to improve insulin sensitivity in patients with severe obesity and metabolic syndrome.




Similar content being viewed by others
Data availability
The data generated or analyzed during this study are included in this article along with the supplementary information files. The raw sequencing data have been deposited into the Sequence Read Archive (SRA) of the NCBI (http://www.ncbi.nlm.nih.gov/sra) under BioProject PRJNA708262. All other relevant data related to the current study are freely available from the corresponding author (K.L.M.) upon request, which does not include confidential patient information. Source data are provided with this paper.
References
Hruby, A. & Hu, F. B. The epidemiology of obesity: a big picture. Pharmacoeconomics 33, 673–689 (2015).
Twells, L. K., Gregory, D. M., Reddigan, J. & Midodzi, W. K. Current and predicted prevalence of obesity in Canada: a trend analysis. CMAJ Open 2, 18–26 (2014).
Twells, L. K., Janssen, I. & Kuk, J. L. Canadian Adult Obesity Clinical Practice Guidelines: Epidemiology of Adult Obesity https://obesitycanada.ca/guidelines/epidemiology (2020).
Mehta, A., Marso, S. P. & Neeland, I. J. Liraglutide for weight management: a critical review of the evidence. Obes. Sci. Pract. 3, 3–14 (2016).
Dansinger, M. L., Gleason, J. A., Griffith, J. L., Selker, H. P. & Schaefer, E. J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA 293, 43–53 (2005).
le Roux, C. W. et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes: a randomised, double-blind trial. Lancet 389, 1399–1409 (2017).
Klebanoff, M. J. et al. Cost-effectiveness of bariatric surgery in adolescents with obesity. JAMA Surg. 152, 136–141 (2017).
Wolf, A. M. & Colditz, G. A. Current estimates of the economic cost of obesity in the United States. Obes. Res. 6, 97–106 (2012).
Parekh, P. J., Balart, L. A. & Johnson, D. A. The influence of the gut microbiome on obesity, metabolic syndrome and gastrointestinal disease. Clin. Transl. Gastroenterol. 6, e91 (2015).
Pradhan, B., Datzkiw, D. & Aich, P. Gut microbiota and health: a review with focus on metabolic and immunological disorders and microbial remediation. Biomed. Rev. 27, 1–17 (2016).
Dovrolis, N., Filidou, E. & Kolios, G. Systems biology in inflammatory bowel diseases: on the way to precision medicine. Ann. Gastroenterol. 32, 233–246 (2019).
Meijnikman, A. S., Gerdes, V. E., Nieuwdorp, M. & Herrema, H. Evaluating causality of gut microbiota in obesity and diabetes in humans. Endocr. Rev. 39, 133–153 (2018).
de Groot, P. F., Frissen, M. N., de Clercq, N. C. & Nieuwdorp, M. Fecal microbiota transplantation in metabolic syndrome: history, present and future. Gut Microbes 8, 253–267 (2017).
Bakker, G. J. & Nieuwdorp, M. Fecal microbiota transplantation: therapeutic potential for a multitude of diseases beyond Clostridium difficile. Microbiol. Spectr. 5, https://doi.org/10.1128/microbiolspec.BAD-0008-2017 (2017).
Vrieze, A. et al. Transfer of intestinal microbiota from lean donors increases insulin. Gastroenterology 143, 913–916 (2012).
Kootte, R. S. et al. Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 26, 611–619 (2017).
de Groot, P. et al. Donor metabolic characteristics drive effects of faecal microbiota transplantation on recipient insulin sensitivity, energy expenditure and intestinal transit time. Gut 69, 502–512 (2020).
Zhang, Z. et al. Impact of fecal microbiota transplantation on obesity and metabolic syndrome: a systematic review. Nutrients 11, 2291 (2019).
Ianiro, G., Bibbo, S., Gasbarrini, A. & Cammarota, G. Therapeutic modulation of gut microbiota: current clinical applications and future perspectives. Curr. Drug Targets 15, 762–770 (2014).
Chis, R., Sheth, P. M. & Petrof, E. O. in Mechanisms Underlying Host–Microbiome Interactions in Pathophysiology of Human Diseases. Physiology in Health and Disease (eds Sun, J. & Dudeja, P.) Ch. 11 (Springer, 2018).
Allegretti, J. et al. The current landscape and lessons from fecal microbiota transplantation for inflammatory bowel disease: past, present, and future. Inflamm. Bowel Dis. 23, 1710–1717 (2017).
Sbahi, H. & Di Palma, J. A. Faecal microbiota transplantation: applications and limitations in treating gastrointestinal disorders. BMJ Open Gastroenterol. 3, e000087 (2016).
Stripling, J. & Rodriguez, M. Current evidence in delivery and therapeutic uses of fecal microbiota transplantation in human diseases: Clostridium difficile disease and beyond. Am. J. Med. Sci. 356, 424–432 (2018).
Marotz, C. A. & Zarrinpar, A. Treating obesity and metabolic syndrome with fecal microbiota transplantation. Yale J. Biol. Med. 89, 383–388 (2016).
Aron-Wisnewsky, J., Clement, K. & Nieuwdorp, M. Fecal microbiota transplantation: a future therapeutic option for obesity/diabetes?. Curr. Diab. Rep. 19(8), 51 (2019).
Porras, D. et al. Intestinal microbiota modulation in obesity-related non-alcoholic fatty liver disease. Front. Physiol. 9, 1813 (2018).
Leshem, A., Horesh, N. & Elinav, E. Fecal microbial transplantation and its potential application in cardiometabolic syndrome. Front. Immunol. 10, 1341 (2019).
Zou, J. et al. Fiber-mediated nourishment of gut microbiota protects against diet-induced obesity by restoring IL-22-mediated colonic health. Cell Host Microbe 23, 41–53 (2018).
Dahiya, D. K. et al. Gut microbiota modulation and its relationship with obesity using prebiotic fibers and probiotics: a review. Front. Microbiol. 8, 563 (2017).
Lee, P., Yacyshyn, B. R. & Yacyshyn, M. B. Gut microbiota and obesity: an opportunity to alter obesity through faecal microbiota transplant (FMT). Diabetes Obes. Metab. 21, 479–490 (2019).
Finkelstein, E. A. et al. Obesity and severe obesity forecasts through 2030. Am. J. Prev. Med. 42, 563–570 (2012).
Gill, S. K., Rossi, M., Bajka, B. & Whelan, K. Dietary fibre in gastrointestinal health and disease. Nat. Rev. Gastroenterol. Hepatol. 18, 101–116 (2021).
Makki, K., Deehan, E. C., Walter, J. & Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 23, 705–715 (2018).
Walter, J., Maldonado-Gómez, M. X. & Martínez, I. To engraft or not to engraft: an ecological framework for gut microbiome modulation with live microbes. Curr. Opin. Biotechnol. 49, 129–139 (2018).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Ellulu, M. S., Patimah, I., Khaza’ai, H., Rahmat, A. & Abed, Y. Obesity and inflammation: the linking mechanism and the complications. Arch. Med. Sci. 13, 851–863 (2017).
Gribble, F. M. & Reimann, F. Function and mechanisms of enteroendocrine cells and gut hormones in metabolism. Nat. Rev. Endocrinol. 15, 226–237 (2019).
Wang, T.-Y. et al. A comparative study of microbial community and functions of type 2 diabetes mellitus patients with obesity and healthy people. Appl. Microbiol. Biotechnol. 104, 7143–7153 (2020).
Naderpoor, N. et al. Faecal microbiota are related to insulin sensitivity and secretion in overweight or obese adults. J. Clin. Med. 8, 452 (2019).
Atzeni, A. et al. Gut microbiota profile and changes in body weight in elderly subjects with overweight/obesity and metabolic syndrome. Microorganisms 9, 346 (2021).
Louis, P., Hold, G. L. & Flint, H. J. The gut microbiota, bacterial metabolites and colorectal cancer. Nat. Rev. Microbiol. 12, 661–672 (2014).
Hiippala, K. et al. Novel Odoribacter splanchnicus strain and its outer membrane vesicles exert immunoregulatory effects in vitro. Front. Microbiol. 11, 2906 (2020).
Iida, N. et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science 342, 967–970 (2013).
Chassard, C., Delmas, E., Robert, C. & Bernalier-Donadille, A. The cellulose-degrading microbial community of the human gut varies according to the presence or absence of methanogens. FEMS Microbiol. Ecol. 74, 205–213 (2010).
Abenavoli, L. et al. Gut microbiota and obesity: a role for probiotics.Nutrients 11, 2690 (2019).
Guidance Document: Fecal Microbiota Therapy Used in the Treatment of Clostridium difficile Infection Not Responsive to Conventional Therapies. Health Canada Guidance Document 11. https://www.canada.ca/en/health-canada/services/drugs-health-products/biologics-radiopharmaceuticals-genetic-therapies/applications-submissions/guidance-documents/regulation-fecal-microbiota-therapy-treatment-difficile-infections.html (Health Canada Publications, 2020)
Kao, D. et al. Effect of oral capsule- vs colonoscopy-delivered fecal microbiota transplantation on recurrent Clostridium difficile infection: a randomized clinical trial. JAMA 318, 1985–1993 (2017).
Bindels, L. B., Walter, J. & Ramer-Tait, A. E. Resistant starches for the management of metabolic diseases. Curr. Opin. Clin. Nutr. Metab. Care 18, 559–565 (2015).
Martínez, I., Kim, J., Duffy, P. R., Schlegel, V. L. & Walter, J. Resistant starches types 2 and 4 have differential effects on the composition of the fecal microbiota in human subjects. PLoS ONE 5, e15046 (2010).
Holscher, H. D. et al. Fiber supplementation influences phylogenetic structure and functional capacity of the human intestinal microbiome: follow-up of a randomized controlled trial. Am. J. Clin. Nutr. 101, 55–64 (2015).
Upadhyaya, B. et al. Impact of dietary resistant starch type 4 on human gut microbiota and immunometabolic functions. Sci. Rep. 6, 28797 (2016).
Babiker, R. et al. Effects of gum arabic ingestion on body mass index and body fat percentage in healthy adult females: two-arm randomized, placebo controlled, double-blind trial. Nutr. J. 11, 111 (2012).
Tan, W. S., Chia, P. F., Ponnalagu, S., Karnik, K. & Henry, C. J. The role of soluble corn fiber on glycemic and insulin response. Nutrients 12, 961 (2020).
National Health and Nutrition Examination Survey (NHANES). Anthropometry Procedures Manual https://www.cdc.gov/nchs/data/nhanes/nhanes_11_12/Anthropometry_Procedures_Manual.pdf (2011).
National Institutes of Health, National Heart, Lung, and Blood Institute, North American Association for the Study of Obesity. The Practical Guide: Identification, Evaluation, and Treatment of Overweight and Obesity in Adults (2000).
Laffin, M. et al. Prebiotic supplementation following ileocecal resection in a murine model is associated with a loss of microbial diversity and increased inflammation. Inflamm. Bowel Dis. 24, 101–110 (2018).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857 (2019).
Oksanen, J. et al. Vegan: Community Ecology Package. https://cran.r-project.org/web/packages/vegan/index.html (version 2.5-6).
Paradis, E. et al. Analysis of Phyogenetics and Evolution. ape http://ape-package.ird.fr/ (accessed 15 December 2019).
Acknowledgements
We thank the Center of Excellence for Gastrointestinal Inflammation and Immunity Research (CEGIIR) for the infrastructure and support provided throughout the project. We also thank M. Emberg for his support with FMT preparation, R. Odsen for his help with trial coordination, and T. Dang for his help with fecal 16s rRNA extraction. We acknowledge S. Jamieson and D. Turner-Crerar as members of the University of Alberta Quality Management in Clinical Research Department (QMCR) for providing safety oversight. V.M. is the recipient of scholarships from the Canadian Institutes of Health Research (CIHR), Alberta Innovates, and the Clinical Investigator Program. Last, we thank the W. Garfield Weston Foundation (RES0048879) for the funding that made this project possible. The funders had no role in the study design, data collection and analysis, decision to publish or the preparation of the paper.
Author information
Authors and Affiliations
Contributions
K.L.M. conceived the project. K.L.M., J.W., Z.Z., E.C.D., D.H.K., N.H. and V.M. designed the proof-of-concept study. K.L.M., J.W. and E.C.D. designed the fiber mixture. K.L.M. and D.H.K. supervised the clinical aspect of the study. D.H.K. supervised the FMT donor program. D.W.B. and S.K. helped identify patients enrolled in the bariatric clinic for study inclusion. V.M. and K.K.S. recruited, coordinated and conducted clinical visits. N.H. performed study randomization and created the fiber sachets. V.M. performed the analysis and interpretation of anthropometric, biochemical, immunologic and enteroendocrine results. E.C.D. conducted the analysis and interpretation of dietary data. Z.Z. performed the fecal microbiome sequencing and bioinformatics analysis. All authors discussed the study findings, interpretations and approved the final version of the paper.
Corresponding author
Ethics declarations
Competing interests
J.W. has received research funding and consulting fees from industry sources involved in the manufacture and marketing of fibers, and is a co-owner of Synbiotics Health, a developer of synbiotic products. There are no conflicts pertaining to this manuscript. All other authors have no competing interests.
Additional information
Peer review information Nature Medicine thanks Bright Offorha and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Jennifer Sargent was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Changes in anthropometric parameters across intervention groups from baseline (BL) to six (T6) and twelve (T12) weeks.
a, Weight. b, Waist circumference. c, Systolic Blood Pressure (SBP). d, Diastolic Blood Pressure (DBP). Values are reported as mean ± SEM or as percent change from BL to T6 and BL to T12. Box-and-whisker plots represent the distribution of each group from baseline to week 12 by intervention group. The median is represented by the middle line while the upper and lower borders of the box plot identify the 75th and 25th percentile, respectively. The whiskers correspond to the minimal and maximal values. Pairwise comparisons between groups were conducted using Mann-Whitney U-tests without adjustment for multiple comparisons. Paired time points were compared to baseline values using the Wilcoxon signed-rank test. FMT-HF (n = 15), FMT and high-fermentable fiber group. HF (n = 15), high-fermentable fiber group. FMT-LF (n = 14), FMT and low-fermentable fiber group. LF (n = 17), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 2 Changes in lipid parameters across intervention groups from baseline (BL) to six (T6) and twelve (T12) weeks.
a, Total Cholesterol. b, High-density lipoprotein (HDL-C). c, Low-density lipoprotein (LDL-C). d, Triglycerides (Tg). Values are reported as mean ± SEM or as percent change from BL to T6 and BL to T12. Box-and-whisker plots represent the distribution of each group from baseline to week 12 by intervention group. The median is represented by the middle line while the upper and lower borders of the box plot identify the 75th and 25th percentile, respectively. The whiskers correspond to the maximal and minimal values. Pairwise comparisons between groups were conducted using Mann-Whitney U-tests without adjustment for multiple comparisons. Paired time points were compared to baseline values using the Wilcoxon signed-rank test. FMT-HF (n = 15 patients), FMT and high-fermentable fiber group. HF (n = 15 patients), high-fermentable fiber group. FMT-LF (n = 14 patients) FMT and low-fermentable fiber group. LF (n = 17 patients), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 3 Changes in HOMA2-IR across intervention groups baseline (BL) to six (T6) and twelve (T12) weeks.
a, HOMA2-IR. Values are reported as mean ± SEM or as percent change from BL to T6 and from BL to T12. Points represent raw unadjusted values at baseline, week 6, and week 12 following and/or fiber supplementation. Box-and-whisker plots represent the distribution of each group from baseline to week 12 by intervention group. The median is represented by the middle line while the upper and lower borders of the box plot identify the 75th and 25th percentile, respectively. The whiskers correspond to the maximal and minimal values. Pairwise comparisons between groups were conducted using Mann-Whitney U-tests without adjustment for multiple comparisons. Paired time points were compared to baseline values using the Wilcoxon signed-rank test. FMT-HF (n = 15 patients), FMT and high-fermentable fiber group. HF (n = 15 patients), high-fermentable fiber group. FMT-LF (n = 14 patients), FMT and low-fermentable fiber group. LF (n = 17 patients), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 4 Composite scores of 5-level EQ-5D (EQ-5D-5L) survey domains from baseline to 12 weeks.
a, Mobility domain. b, Self-Care domain. c, Usual Activity domain. d, Pain and Discomfort domain. e, Anxiety and Depression domain., f, Visual Analogue Health Scale. Data reported as mean ± SEM. Pairwise comparisons between two groups were conducted using Mann-Whitney U-tests and Kruskal-Wallis tests for four group comparisons. No adjustment was made for multiple comparisons. FMT-HF (n = 15), FMT and high-fermentable fiber group. HF (n = 15), high-fermentable fiber group. FMT-LF (n = 14), FMT and low-fermentable fiber group. LF (n = 17), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 5 Responses to Hunger and Satiety SLIM from baseline to 12 weeks.
a, Hunger and satiety SLIM scoring upon waking. b, Hunger and satiety SLIM scoring 1-1.5 hours after a meal. c, Hunger and satiety SLIM scoring 2-2.5 hours after a meal. Data reported as mean ± SEM. Results were analyzed by fitting a random effects mixed model analysis with Dunnett’s test controlling for multiple comparisons. FMT-HF (n = 15), FMT and high-fermentable fiber group. HF (n = 15), high-fermentable fiber group. FMT-LF (n = 14), FMT and low-fermentable fiber group. LF (n = 17), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 6 Responses to Gastrointestinal Tolerance questionnaire from baseline to 12 weeks.
a, Stomach aches. b, Bloating. c, Flatulence. d, Overall Well-being. e, Consistency. f, Frequency. Data reported as mean ± SEM. Pairwise comparisons between two groups were conducted using Mann-Whitney U-tests and Kruskal-Wallis tests for four group comparisons. Paired data was analyzed by fitting a mixed model. No adjustments were made for multiple comparisons. FMT-HF (n = 15), FMT and high-fermentable fiber group. HF (n = 15), high-fermentable fiber group. FMT-LF (n = 14), FMT and low-fermentable fiber group. LF (n = 17), low-fermentable fiber group. All tests were two-tailed with a p-value <0.05 identifying statistical significance.
Extended Data Fig. 7 Donor composition at Phylum level of stool and oral capsules.
Bar chart represents proportion of ASV counts at the phylum level.
Extended Data Fig. 8 Family level microbial composition of FMT recipients and paired donors from baseline (BL) to week 6 (T6).
a, BL FMT-LF composition. b, Composition of matched FMT-donor in patients receiving FMT and low-fermentable fiber. c, T6 FMT-LF composition. d, BL FMT-HF composition. e, Composition of matched FMT-donor in patients receiving FMT and high-fermentable fiber. f, T6 FMT-HF composition.
Extended Data Fig. 9 Evidence of fecal microbial engraftment in FMT-LF patients from baseline to week 12.
a, Recipient Chao1 index from baseline to week 12 versus donor FMT. Box-and-whisker plots represent the distribution of each group’s Chao1 index from baseline to week 12. The median is represented by the middle line while the upper and lower borders of the box plot identify the 75th and 25th percentile, respectively. The whiskers correspond to the maximal and minimal values. Between-group differences were assessed by linear regression, and pairwise comparisons were conducted using unpaired t-tests. P-values were adjusted by Benjamin-Hochberg false discovery rate (FDR) method. b, Principal-coordinate analysis (PCoA) of ASVs demonstrating that at baseline, recipients have significantly different microbial clustering patterns than donors. At week 6, no significant differences in microbial structure were observed between recipients and donors. At week 12, after fiber cessation, significant differences in microbial structure between recipients and donors are again demonstrated. Significant differences were assessed by PERMANOVA based on unweighted UniFrac distances with FDR correction (* q < 0.05). c-f, PCoA grouping of patients and donors (baseline, week 2, week 6, and week 12) demonstrating substantial increases in similarity between recipient and donor microbial structure over time. g, Evidence of donor specific ASV engraftment obtained if taxa were shared between donor and recipients at week 6 but not present in recipients at baseline ASVs with the highest number of post-FMT patients received showing evidence of acquisition of the donor’s taxa. FMT-LF (n = 14 patients), FMT and low-fermentable fiber group. All tests were two-tailed with a q-value <0.05 identifying statistical significance.
Extended Data Fig. 10 Lack evidence for fecal microbial engraftment in FMT-HF patients from baseline to week 12.
a, Recipient Chao1 index from baseline to week 6 versus donor FMT. Box-and-whisker plots represent the distribution of each group’s Chao1 index from baseline to week 12. The median is represented by the middle line while the upper and lower borders of the box plot identify the 75th and 25th percentile, respectively. The whiskers correspond to the maximal and minimal values. Between-group differences were assessed by linear regression, and pairwise comparisons were conducted using unpaired t-tests. P-values were adjusted by Benjamin-Hochberg false discovery rate (FDR) method. b, Principal-coordinate analysis (PCoA) of ASVs demonstrating that at baseline, week 6 and week 12, recipients have significantly different microbial clustering patterns than donors. Significant differences were assessed by PERMANOVA based on unweighted UniFrac distances with FDR correction (* q < 0.05). c, PCoA grouping of patients and donors (baseline, week 2, week 6, and week 12) demonstrating minor increases in similarity between recipient and donor microbial structure over time. FMT-HF (n = 15 patients), FMT and high-fermentable fiber group. All tests were two-tailed with a q-value <0.05 identifying statistical significance.
Supplementary information
Supplementary Information
Supplementary Tables 1–5.
Source data
Source Data Fig. 2
Statistical Source Data for Glycemic Parameters (Excel)
Source Data Fig. 3
Statistical Source Data for GLP-1 and Insulin for oral glucose tolerance test Parameters (Excel)
Source Data Extended Fig. 1
Statistical Source Data for Anthropometric Parameters (Excel)
Source Data Extended Fig. 2
Statistical Source Data for Lipid Parameters (Excel)
Source Data Extended Fig. 3
Statistical Source Data for HOMA2 Parameters (Excel)
Source Data Extended Fig. 4
Statistical Source Data for 5-level EQ-5D (EQ-5D-5L) Questionnaire (Excel)
Source Data Extended Fig. 5
Statistical Source Data for Hunger and Satiety Questionnaire (Excel)
Source Data Extended Fig. 6
Statistical Source Data for Gastrointestinal Tolerance Questionnaire (Excel)
Rights and permissions
About this article
Cite this article
Mocanu, V., Zhang, Z., Deehan, E.C. et al. Fecal microbial transplantation and fiber supplementation in patients with severe obesity and metabolic syndrome: a randomized double-blind, placebo-controlled phase 2 trial. Nat Med 27, 1272–1279 (2021). https://doi.org/10.1038/s41591-021-01399-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-021-01399-2
- Springer Nature America, Inc.
This article is cited by
-
Effects of dietary fibre on metabolic health and obesity
Nature Reviews Gastroenterology & Hepatology (2024)
-
Impact of the gut microbiota and associated metabolites on cardiometabolic traits, chronic diseases and human longevity: a Mendelian randomization study
Journal of Translational Medicine (2023)
-
Clec7a drives gut fungus-mediated host lipid deposition
Microbiome (2023)
-
Parabacteroides distasonis ameliorates insulin resistance via activation of intestinal GPR109a
Nature Communications (2023)
-
The potential of tailoring the gut microbiome to prevent and treat cardiometabolic disease
Nature Reviews Cardiology (2023)