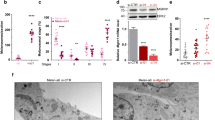
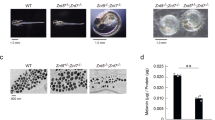
Abstract
Dozens of genes contribute to the wide variation in human pigmentation. Many of these genes encode proteins that localize to the melanosome—the organelle, related to the lysosome, that synthesizes pigment—but have unclear functions1,2. Here we describe MelanoIP, a method for rapidly isolating melanosomes and profiling their labile metabolite contents. We use this method to study MFSD12, a transmembrane protein of unknown molecular function that, when suppressed, causes darker pigmentation in mice and humans3,4. We find that MFSD12 is required to maintain normal levels of cystine—the oxidized dimer of cysteine—in melanosomes, and to produce cysteinyldopas, the precursors of pheomelanin synthesis made in melanosomes via cysteine oxidation5,6. Tracing and biochemical analyses show that MFSD12 is necessary for the import of cysteine into melanosomes and, in non-pigmented cells, lysosomes. Indeed, loss of MFSD12 reduced the accumulation of cystine in lysosomes of fibroblasts from patients with cystinosis, a lysosomal-storage disease caused by inactivation of the lysosomal cystine exporter cystinosin7,8,9. Thus, MFSD12 is an essential component of the cysteine importer for melanosomes and lysosomes.




Similar content being viewed by others
Data availability
Fig. 3a and Extended Data Fig. 3a were generated from FANTOM5 expression data, accessed via the Human Protein Atlas20,21 from https://www.proteinatlas.org/about/assays+annotation#fantom. Raw values from this accession are included in the source data for Fig. 3a and Extended Data Fig. 3a. Unique biological materials in the form of plasmids available from Addgene. Unique biological materials in the form of cell lines are available from the authors by request. Source data are provided with this paper.
References
Basrur, V. et al. Proteomic analysis of early melanosomes: identification of novel melanosomal proteins. J. Proteome Res. 2, 69–79 (2003).
Sturm, R. A. Molecular genetics of human pigmentation diversity. Hum. Mol. Genet. 18 (R1), R9–R17 (2009).
Adhikari, K. et al. A GWAS in Latin Americans highlights the convergent evolution of lighter skin pigmentation in Eurasia. Nat. Commun. 10, 358 (2019).
Crawford, N. G. et al. Loci associated with skin pigmentation identified in African populations. Science 358, eaan8433 (2017).
D’Alba, L. & Shawkey, M. D. Melanosomes: biogenesis, properties, and evolution of an ancient organelle. Physiol. Rev. 99, 1–19 (2019).
Prota, G. Melanins and Melanogenesis (Academic, 1992).
Gahl, W. A., Bashan, N., Tietze, F., Bernardini, I. & Schulman, J. D. Cystine transport is defective in isolated leukocyte lysosomes from patients with cystinosis. Science 217, 1263–1265 (1982).
Jonas, A. J., Smith, M. L. & Schneider, J. A. ATP-dependent lysosomal cystine efflux is defective in cystinosis. J. Biol. Chem. 257, 13185–13188 (1982).
Town, M. et al. A novel gene encoding an integral membrane protein is mutated in nephropathic cystinosis. Nat. Genet. 18, 319–324 (1998).
Watabe, H., Kushimoto, T., Valencia, J. C. & Hearing, V. J. in Current Protocols in Cell Biology (eds J. S. Bonifacino et al.) (John Wiley & Sons, 2005).
Abu-Remaileh, M. et al. Lysosomal metabolomics reveals V-ATPase- and mTOR-dependent regulation of amino acid efflux from lysosomes. Science 358, 807–813 (2017).
Chen, W. W., Freinkman, E., Wang, T., Birsoy, K. & Sabatini, D. M. Absolute quantification of matrix metabolites reveals the dynamics of mitochondrial metabolism. Cell 166, 1324–1337 (2016).
Ray, G. J. et al. A PEROXO-tag enables rapid isolation of peroxisomes from human cells. iScience 23, 101109 (2020).
Bruder, J. M. et al. Melanosomal dynamics assessed with a live-cell fluorescent melanosomal marker. PLoS ONE 7, e43465 (2012).
Diment, S., Eidelman, M., Rodriguez, G. M. & Orlow, S. J. Lysosomal hydrolases are present in melanosomes and are elevated in melanizing cells. J. Biol. Chem. 270, 4213–4215 (1995).
Bissig, C., Rochin, L. & van Niel, G. PMEL amyloid fibril formation: the bright steps of pigmentation. Int. J. Mol. Sci. 17, 1438 (2016).
Reddy, V. S., Shlykov, M. A., Castillo, R., Sun, E. I. & Saier, M. H. Jr. The major facilitator superfamily (MFS) revisited. FEBS J. 279, 2022–2035 (2012).
Bloom, J. L. & Falconer, D. S. ‘Grizzled’, a mutant in linkage group X of the mouse. Genet. Res. 7, 159–167 (1966).
Potterf, S. B. et al. Cysteine transport in melanosomes from murine melanocytes. Pigment Cell Res. 12, 4–12 (1999).
Kawaji, H., Kasukawa, T., Forrest, A., Carninci, P. & Hayashizaki, Y. The FANTOM5 collection, a data series underpinning mammalian transcriptome atlases in diverse cell types. Sci. Data 4, 170113 (2017).
Uhlén, M. et al. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Pisoni, R. L., Acker, T. L., Lisowski, K. M., Lemons, R. M. & Thoene, J. G. A cysteine-specific lysosomal transport system provides a major route for the delivery of thiol to human fibroblast lysosomes: possible role in supporting lysosomal proteolysis. J. Cell Biol. 110, 327–335 (1990).
Gahl, W. A., Thoene, J. G. & Schneider, J. A. Cystinosis. N. Engl. J. Med. 347, 111–121 (2002).
Oshima, R. G., Rhead, W. J., Thoene, J. G. & Schneider, J. A. Cystine metabolism in human fibroblasts. Comparison of normal, cystinotic, and gamma-glutamylcysteine synethetase-deficient cells. J. Biol. Chem. 251, 4287–4293 (1976).
Sato, H., Tamba, M., Ishii, T. & Bannai, S. Cloning and expression of a plasma membrane cystine/glutamate exchange transporter composed of two distinct proteins. J. Biol. Chem. 274, 11455–11458 (1999).
Wilbrandt, W. & Rosenberg, T. The concept of carrier transport and its corollaries in pharmacology. Pharmacol. Rev. 13, 109–183 (1961).
Behnke, J., Eskelinen, E.-L., Saftig, P. & Schröder, B. Two dileucine motifs mediate late endosomal/lysosomal targeting of transmembrane protein 192 (TMEM192) and a C-terminal cysteine residue is responsible for disulfide bond formation in TMEM192 homodimers. Biochem. J. 434, 219–231 (2011).
Lloyd, J. B. Disulphide reduction in lysosomes. The role of cysteine. Biochem. J. 237, 271–272 (1986).
Mego, J. L. Role of thiols, pH and cathepsin D in the lysosomal catabolism of serum albumin. Biochem. J. 218, 775–783 (1984).
Tsui, C. K. et al. CRISPR–cas9 screens identify regulators of antibody-drug conjugate toxicity. Nat. Chem. Biol. 15, 949–958 (2019).
Gahl, W. A. et al. Cysteamine therapy for children with nephropathic cystinosis. N. Engl. J. Med. 316, 971–977 (1987).
Thoene, J. G., Oshima, R. G., Crawhall, J. C., Olson, D. L. & Schneider, J. A. Cystinosis. Intracellular cystine depletion by aminothiols in vitro and in vivo. J. Clin. Invest. 58, 180–189 (1976).
Mujahid, N. et al. A UV-independent topical small-molecule approach for melanin production in human skin. Cell Rep. 19, 2177–2184 (2017).
Djoumbou-Feunang, Y. et al. CFM-ID 3.0: significantly improved ESI-MS/MS prediction and compound identification. Metabolites 9, 72 (2019).
Wishart, D. S. et al. HMDB: the Human Metabolome Database. Nucleic Acids Res. 35, D521–D526 (2007).
Guan, X., Hoffman, B., Dwivedi, C. & Matthees, D. P. A simultaneous liquid chromatography/mass spectrometric assay of glutathione, cysteine, homocysteine and their disulfides in biological samples. J. Pharm. Biomed. Anal. 31, 251–261 (2003).
Martin, G. B. et al. Development of a mass spectrometry method for the determination of a melanoma biomarker, 5-S-cysteinyldopa, in human plasma using solid phase extraction for sample clean-up. J. Chromatogr. A 1156, 141–148 (2007).
Ito, S. & Prota, G. A facile one-step synthesis of cysteinyldopas using mushroom tyrosinase. Experientia 33, 1118–1119 (1977).
Graham, J. M. Isolation of lysosomes from tissues and cells by differential and density gradient centrifugation. Curr. Protoc. Cell Biol. Ch. 3, Unit 3.6 (2001).
Goldman, R. & Kaplan, A. Rupture of rat liver lysosomes mediated by l-amino acid esters. Biochim. Biophys. Acta 318, 205–216 (1973).
Verdon, Q. et al. SNAT7 is the primary lysosomal glutamine exporter required for extracellular protein-dependent growth of cancer cells. Proc. Natl Acad. Sci. USA 114, E3602–E3611 (2017).
Acknowledgements
We thank P. Budde, M. Abu-Remaileh, W. W. Chen, L. Bar-Peled and J. G. Bryan, as well as all current members of the Sabatini laboratory for helpful insights. This work was supported by grants from the Leo Foundation (LF18057) and NIH (R01 CA103866, R01 CA129105 and R01 AI047389), fellowship support from the NIH (NRSA F31 CA228241-01) to C.H.A., Marshall Plan Foundation to A.K.T., HHMI XROP to B.C., and NSF (2016197106) to K.J.C. D.M.S. is an investigator of the Howard Hughes Medical Institute and an American Cancer Society Research Professor.
Author information
Authors and Affiliations
Contributions
C.H.A. and D.M.S. initiated the project and designed the research plan. C.H.A. performed the experiments and analysed the data with help from A.K.T., B.C. and K.J.C. The LC–MS platform was operated by S.H.C., T.K. and C.A.L., who also had a critical role in method development for LC–MS-based assays for cysteine and cysteinyldopas. C.H.A. wrote the manuscript and D.M.S. edited it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Edmund Kunji and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 MelanoIP analysis detects changes in Tyr dependent melanosomal metabolites.
a, Schematic of melanin synthesis. The common pathway elements for eumelanin and pheomelanin synthesis have a grey backdrop. The brown and red backdrops highlight unique portions of eumelanin and pheomelanin synthesis, respectively. Enzymes proposed to catalyse each step are shown in green. Synthetic intermediates annotated and validated in biological samples in this study are in blue. b, Follow-up analysis with standard validated m/z and internal normalization of ‘proteogenic amino acids’ highlighted in untargeted metabolite profiling of wild-type and Tyr-knockout melanosomes (Fig. 1d). Amino acids are presented in order of increasing retention time (n = 3 independently prepared extracts, aP = 3.9 × 10−2, bP = 2.0 × 10−3, cP = 6.5 × 10−3, dP = 3.8 × 10−2, eP = 1.9 × 10−2). Error bars are mean ± s.e.m., P values by two-sided Student’s t-test.
Extended Data Fig. 2 In vitro synthesis and biological detection of cysteinyldopas.
a, Cysteinyldopas were synthesized according to an adapted protocol from Ito and Prota, 197738. Two species, distinguished by retention time, were generated at the expected m/z for cysteinyldopas. It has been shown that 5′-cysteinyldopa is produced in greater abundance than 2’-cysteinyldopa in this reaction. Taking MS1 peak intensity to approximate abundance, we putatively annotate the ‘Minor Isomer’ as 2’ substituted, and the ‘Major Isomer’ as 5′ substituted. b, Mirror plot of ddMS2 data comparing 2’- and 5′-cysteinyldopa in synthetic cysteinyldopas. c,d, Mirror plots of ddMS2 peaks displaying similarities in ddMS2 spectra of 2’- and 5′-cysteinyldopa species in biological samples (B16F10 extracts) and synthetic standards.
Extended Data Fig. 3 MFSD12 maintains lysosomal cystine in non-pigmented cells.
a, FANTOM5 CAGE profiling data accessed via Human Protein Atlas20,21. Six representative pigmentation genes, including MFSD12, are shown. b, Metabolite profiling of LysoIP samples from HEK 293T cells comparing lysosomes from wild-type and MFSD12-knockout cells. ‘Accumulates upon inhibition of:’ has been previously reported11 (n = 4 independently prepared metabolite extracts, aP = 7.0 × 10−4, bP = 3.0 × 10−3, cP = 4.1 × 10−2, dP = 4.2 × 10−2, eP = 1.7 × 10−4). c, Lentiviral shRNA knock-down of MFSD12 quantified via qPCR and normalized to ACTB levels (n = 3 assays on independently prepared cDNA libraries, aP = 1.97 × 10−3, bP = 3.0 × 10−3). Error bars are mean ± s.e.m., P values by two-sided Student’s t-test.
Extended Data Fig. 4 MFSD12 mediated cysteine transport is cysteine specific.
a, Test of lysosomal counter-transport. Lysosomes were purified by differential centrifugation and incubated with water or 1 mM cysteine methyl ester before washing, resuspension, and incubated for 5 min with 20 μM cysteine and trace amounts of [35S]cysteine (n = 3 independently performed assays per condition, aP = 2.5 × 10−3, bP = 3.3 × 10−2, NS = not significant). b, Lysosomal import of [14C]cystine. Lysosomes were purified by differential centrifugation and incubated for 10 min with 1 μM [14C]cystine, either untreated (Unreduced) or pre-treated with 10 mM DTT (Reduced, n = 6 independently performed assays per condition, aP = 2.1 × 10−7, bP = 1.3 × 10−6, cP = 3.8 × 10−8). c, Competition for [35S]cysteine transport. Lysosomes were purified by differential centrifugation and incubated for 10 min with 20 μM cysteine and trace amounts of [35S]cysteine with 500 μM competitor where indicated (n = 3 independently performed assays per condition, P values compare competition condition versus water control condition (red), aP = 2.7 × 10−4, bP = 2.5 × 10−4, cP = 3.2 × 10−4). Error bars are mean ± s.e.m., P values by two-sided Student’s t-test.
Supplementary information
Supplementary Figure 1
Uncropped blots for Fig 1c.
Rights and permissions
About this article
Cite this article
Adelmann, C.H., Traunbauer, A.K., Chen, B. et al. MFSD12 mediates the import of cysteine into melanosomes and lysosomes. Nature 588, 699–704 (2020). https://doi.org/10.1038/s41586-020-2937-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2937-x
- Springer Nature Limited
This article is cited by
-
Tumor-suppressive miR-4732-3p is sorted into fucosylated exosome by hnRNPK to avoid the inhibition of lung cancer progression
Journal of Experimental & Clinical Cancer Research (2024)
-
Recycled melanoma-secreted melanosomes regulate tumor-associated macrophage diversification
The EMBO Journal (2024)
-
Integrative functional genomic analyses identify genetic variants influencing skin pigmentation in Africans
Nature Genetics (2024)
-
Lysosomal cystine export regulates mTORC1 signaling to guide kidney epithelial cell fate specialization
Nature Communications (2023)
-
Dietary supplementation of cystinotic mice by lysine inhibits the megalin pathway and decreases kidney cystine content
Scientific Reports (2023)