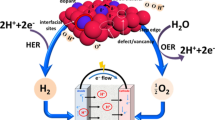
Abstract
Single-atom catalysts (SACs) are advantageous because every active atom is exposed at the surface, ensuring maximum utilization of catalytically active metals. To optimize the effectiveness of SACs, every atomic site needs to contribute to an accelerated reaction and retain this performance over extended use. The state-of-the-art approach for optimizing the catalytic properties of these atomic sites is through metal–support interactions. In this Review, we present the concept of co-catalytic interactions, in which both the single atom and the support are directly involved in catalysis by binding intermediates to enhance and alter the reaction mechanism. The power of this concept is highlighted for a range of important electrocatalytic reactions. First, we investigate the role of single atoms and supports in the reaction mechanism and explore the SAC designs that have successfully enhanced performance. We then discuss the synthetic targets and strategies for producing SACs that achieve co-reactant, functional group or intermediate binding for co-catalyst metal–support interactions. Finally, we offer a perspective on the future of SAC research and on the opportunities in co-catalytic metal–support interactions to further elevate electrocatalytic performance.





Similar content being viewed by others
References
Mitchell, S. & Pérez-Ramírez, J. Single atom catalysis: a decade of stunning progress and the promise for a bright future. Nat. Commun. 11, 4302 (2020).
Wang, A., Li, J. & Zhang, T. Heterogeneous single-atom catalysis. Nat. Rev. Chem. 2, 65–81 (2018).
Alinezhad, A. et al. Direct growth of highly strained Pt islands on branched Ni nanoparticles for improved hydrogen evolution reaction activity. J. Am. Chem. Soc. 141, 16202–16207 (2019).
Poerwoprajitno, A. R., Gloag, L., Cheong, S., Gooding, J. J. & Tilley, R. D. Synthesis of low- and high-index faceted metal (Pt, Pd, Ru, Ir, Rh) nanoparticles for improved activity and stability in electrocatalysis. Nanoscale 11, 18995–19011 (2019).
Gloag, L. et al. Synthesis of hierarchical metal nanostructures with high electrocatalytic surface areas. Sci. Adv. 9, eadf6075 (2023).
Ding, J., Ji, Y., Li, Y. & Hong, G. Monoatomic platinum-embedded hexagonal close-packed nickel anisotropic superstructures as highly efficient hydrogen evolution catalyst. Nano Lett. 21, 9381–9387 (2021).
Cai, C. et al. Ultrahigh oxygen evolution reaction activity achieved using Ir single atoms on amorphous CoOx nanosheets. ACS Catal. 11, 123–130 (2021).
Dong, Q. et al. Ultrahigh mass activity for the hydrogen evolution reaction by anchoring platinum single atoms on active {100} facets of TiC via cation defect engineering. Adv. Funct. Mater. 33, 2210665 (2023).
Hu, P. et al. Electronic metal-support interactions in single-atom catalysts. Angew. Chem. Int. Ed. 53, 3418–3421 (2014).
Li, Z. et al. Metal-support interactions in designing noble metal-based catalysts for electrochemical CO2 reduction: recent advances and future perspectives. Nano Res. 14, 3795–3809 (2021).
Yang, J., Li, W., Wang, D. & Li, Y. Electronic metal–support interaction of single‐atom catalysts and applications in electrocatalysis. Adv. Mater. 32, 2003300 (2020).
Zheng, W. et al. Tailoring bond microenvironments and reaction pathways of single‐atom catalysts for efficient water electrolysis. Angew. Chem. Int. Ed. 61, e202208667 (2022).
Chen, Y. et al. Isolating single and few atoms for enhanced catalysis. Adv. Mater. 34, 2201796 (2022).
Luo, Z., Zhao, G., Pan, H. & Sun, W. Strong metal–support interaction in heterogeneous catalysts. Adv. Energy Mater. 12, 2201395 (2022).
Cheng, N. et al. Platinum single-atom and cluster catalysis of the hydrogen evolution reaction. Nat. Commun. 7, 13638 (2016).
Liu, D. et al. Three-dimensional controlled growth of monodisperse sub-50 nm heterogeneous nanocrystals. Nat. Commun. 7, 10254 (2016).
Li, Z. et al. Iridium single-atom catalyst on nitrogen-doped carbon for formic acid oxidation synthesized using a general host–guest strategy. Nat. Chem. 12, 764–772 (2020).
Ge, R. et al. Selective electrooxidation of biomass‐derived alcohols to aldehydes in a neutral medium: promoted water dissociation over a nickel‐oxide‐supported ruthenium single‐atom catalyst. Angew. Chem. Int. Ed. 61, e202200211 (2022).
Huang, L. et al. Shape-control of Pt–Ru nanocrystals: tuning surface structure for enhanced electrocatalytic methanol oxidation. J. Am. Chem. Soc. 140, 1142–1147 (2018).
Chen, C. et al. Ruthenium‐based single‐atom alloy with high electrocatalytic activity for hydrogen evolution. Adv. Energy Mater. 9, 1803913 (2019).
Zhai, P. et al. Engineering single-atomic ruthenium catalytic sites on defective nickel-iron layered double hydroxide for overall water splitting. Nat. Commun. 12, 4587 (2021).
Ge, J. et al. Dual-metallic single Ru and Ni atoms decoration of MoS2 for high-efficiency hydrogen production. Appl. Catal. B 298, 120557 (2021).
Li, J. et al. Elucidating the critical role of ruthenium single atom sites in water dissociation and dehydrogenation behaviors for robust hydrazine oxidation‐boosted alkaline hydrogen evolution. Adv. Funct. Mater. 32, 2109439 (2022).
Hung, S.-F. et al. A metal-supported single-atom catalytic site enables carbon dioxide hydrogenation. Nat. Commun. 13, 819 (2022).
O’Mara, P. B. et al. Cascade reactions in nanozymes: spatially separated active sites inside Ag-core–porous-Cu-shell nanoparticles for multistep carbon dioxide reduction to higher organic molecules. J. Am. Chem. Soc. 141, 14093–14097 (2019).
Meng, D. et al. Highly selective tandem electroreduction of CO2 to ethylene over atomically isolated nickel–nitrogen site/copper nanoparticle catalysts. Angew. Chem. Int. Ed. 60, 25485–25492 (2021).
Fang, S. et al. Uncovering near-free platinum single-atom dynamics during electrochemical hydrogen evolution reaction. Nat. Commun. 11, 1029 (2020).
Shi, Y. et al. Electronic metal–support interaction modulates single-atom platinum catalysis for hydrogen evolution reaction. Nat. Commun. 12, 3021 (2021).
Yan, Q.-Q. et al. Reversing the charge transfer between platinum and sulfur-doped carbon support for electrocatalytic hydrogen evolution. Nat. Commun. 10, 4977 (2019).
Kuang, P. et al. Pt single atoms supported on N‐doped mesoporous hollow carbon spheres with enhanced electrocatalytic H2‐evolution activity. Adv. Mater. 33, 2008599 (2021).
Liu, Y. et al. Unraveling the function of metal–amorphous support interactions in single‐atom electrocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 134, e202114160 (2022).
DeRita, L. et al. Structural evolution of atomically dispersed Pt catalysts dictates reactivity. Nat. Mater. 18, 746–751 (2019).
Yang, J. et al. The electronic metal–support interaction directing the design of single atomic site catalysts: achieving high efficiency towards hydrogen evolution. Angew. Chem. Int. Ed. 60, 19085–19091 (2021).
Zhang, Y. et al. The effect of coordination environment on the activity and selectivity of single-atom catalysts. Coord. Chem. Rev. 461, 214493 (2022).
Hu, C. et al. Main‐group metal single‐atomic regulators in dual‐metal catalysts for enhanced electrochemical CO2 reduction. Small 18, 2201391 (2022).
Zhang, Z. et al. Selectively anchoring single atoms on specific sites of supports for improved oxygen evolution. Nat. Commun. 13, 2473 (2022).
Chang, S. H. et al. Functional links between stability and reactivity of strontium ruthenate single crystals during oxygen evolution. Nat. Commun. 5, 4191 (2014).
Poerwoprajitno, A. R. et al. Formation of branched ruthenium nanoparticles for improved electrocatalysis of oxygen evolution reaction. Small 15, 1804577 (2019).
Gloag, L. et al. Three-dimensional branched and faceted gold–ruthenium nanoparticles: using nanostructure to improve stability in oxygen evolution electrocatalysis. Angew. Chem. Int. Ed. 57, 10241–10245 (2018).
Gloag, L. et al. A cubic-core hexagonal-branch mechanism to synthesize bimetallic branched and faceted Pd–Ru nanoparticles for oxygen evolution reaction electrocatalysis. J. Am. Chem. Soc. 140, 12760–12764 (2018).
Wan, W. et al. Mechanistic insight into the active centers of single/dual-atom Ni/Fe-based oxygen electrocatalysts. Nat. Commun. 12, 5589 (2021).
Yin, J. et al. Iridium single atoms coupling with oxygen vacancies boosts oxygen evolution reaction in acid media. J. Am. Chem. Soc. 142, 18378–18386 (2020).
Zhu, Y. et al. Iridium single atoms incorporated in Co3O4 efficiently catalyze the oxygen evolution in acidic conditions. Nat. Commun. 13, 7754 (2022).
Li, S. et al. Oxygen-evolving catalytic atoms on metal carbides. Nat. Mater. 20, 1240–1247 (2021).
Zheng, Y. et al. Molecule-level g-C3N4 coordinated transition metals as a new class of electrocatalysts for oxygen electrode reactions. J. Am. Chem. Soc. 139, 3336–3339 (2017).
Xu, H., Cheng, D., Cao, D. & Zeng, X. C. A universal principle for a rational design of single-atom electrocatalysts. Nat. Catal. 1, 339–348 (2018).
Han, X. et al. Atomically dispersed binary Co‐Ni sites in nitrogen‐doped hollow carbon nanocubes for reversible oxygen reduction and evolution. Adv. Mater. 31, 1905622 (2019).
Fang, C. et al. Synergy of dual-atom catalysts deviated from the scaling relationship for oxygen evolution reaction. Nat. Commun. 14, 4449 (2023).
Yu, Z. et al. Iridium–iron diatomic active sites for efficient bifunctional oxygen electrocatalysis. ACS Catal. 12, 9397–9409 (2022).
Shah, K. et al. Cobalt single atom incorporated in ruthenium oxide sphere: a robust bifunctional electrocatalyst for HER and OER. Angew. Chem. Int. Ed. 61, e202114951 (2022).
Li, Y. et al. Reversely trapping isolated atoms in high oxidation state for accelerating the oxygen evolution reaction kinetics. Angew. Chem. Int. Ed. 62, e202309341 (2023).
Zhang, F.-F. et al. Iridium oxide modified with silver single atom for boosting oxygen evolution reaction in acidic media. ACS Energy Lett. 6, 1588–1595 (2021).
Li, J. et al. Experimental proof of the bifunctional mechanism for the hydrogen oxidation in alkaline media. Angew. Chem. Int. Ed. 56, 15594–15598 (2017).
Wang, Y. et al. Spectroscopic verification of adsorbed hydroxy intermediates in the bifunctional mechanism of the hydrogen oxidation reaction. Angew. Chem. Int. Ed. 60, 5708–5711 (2021).
Huang, Z. et al. A highly efficient pH‐universal HOR catalyst with engineered electronic structures of single Pt sites by isolated Co atoms. Adv. Funct. Mater. 33, 2306333 (2023).
Sahoo, S., Dekel, D. R., Maric, R. & Alpay, S. P. Atomistic insights into the hydrogen oxidation reaction of palladium-ceria bifunctional catalysts for anion-exchange membrane fuel cells. ACS Catal. 11, 2561–2571 (2021).
Wang, L. et al. Stabilizing low‐valence single atoms by constructing metalloid tungsten carbide supports for efficient hydrogen oxidation and evolution. Angew. Chem. Int. Ed. 135, e202311937 (2023).
Ma, M. et al. Single‐atom molybdenum engineered platinum nanocatalyst for boosted alkaline hydrogen oxidation. Adv. Energy Mater. 12, 2103336 (2022).
Chen, L. et al. Platinum–ruthenium single atom alloy as a bifunctional electrocatalyst toward methanol and hydrogen oxidation reactions. ACS Appl. Mater. Interfaces 14, 27814–27822 (2022).
Wang, X. et al. Embedding oxophilic rare-earth single atom in platinum nanoclusters for efficient hydrogen electro-oxidation. Nat. Commun. 14, 3767 (2023).
Bender, M. T., Yuan, X., Goetz, M. K. & Choi, K.-S. Electrochemical hydrogenation, hydrogenolysis, and dehydrogenation for reductive and oxidative biomass upgrading using 5-hydroxymethylfurfural as a model system. ACS Catal. 12, 12349–12368 (2022).
Yang, Y. & Mu, T. Electrochemical oxidation of biomass derived 5-hydroxymethylfurfural (HMF): pathway, mechanism, catalysts and coupling reactions. Green Chem. 23, 4228–4254 (2021).
Poerwoprajitno, A. R. et al. Faceted branched nickel nanoparticles with tunable branch length for high‐activity electrocatalytic oxidation of biomass. Angew. Chem. Int. Ed. 59, 15487–15491 (2020).
Chen, W. et al. Activity origins and design principles of nickel-based catalysts for nucleophile electrooxidation. Chem 6, 2974–2993 (2020).
Tao, Y. et al. CuxCo3–xO4 spinel nanofibers for selective oxidation of 5-hydroxymethylfurfural into fuel additives. ACS Appl. Nano Mater. 5, 16564–16572 (2022).
Nam, D.-H., Taitt, B. J. & Choi, K.-S. Copper-based catalytic anodes to produce 2,5-furandicarboxylic acid, a biomass-derived alternative to terephthalic acid. ACS Catal. 8, 1197–1206 (2018).
Xu, C., Paone, E., Rodríguez-Padrón, D., Luque, R. & Mauriello, F. Recent catalytic routes for the preparation and the upgrading of biomass derived furfural and 5-hydroxymethylfurfural. Chem. Soc. Rev. 49, 4273–4306 (2020).
Li, C. & Na, Y. Recent advances in photocatalytic oxidation of 5‐hydroxymethylfurfural. ChemPhotoChem 5, 502–511 (2021).
Lee, D. K. et al. The impact of 5‐hydroxymethylfurfural (HMF)‐metal interactions on the electrochemical reduction pathways of HMF on various metal electrodes. ChemSusChem 14, 4563–4572 (2021).
Ji, K. et al. Electrocatalytic hydrogenation of 5‐hydroxymethylfurfural promoted by a Ru1Cu single‐atom alloy catalyst. Angew. Chem. Int. Ed. 61, e202209849 (2022).
Tilley, R. D. & Gooding, J. J. Electrocatalysis: understanding platinum migration. Nat. Energy 1, 16174 (2016).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Papanikolaou, K. G., Darby, M. T. & Stamatakis, M. Engineering the surface architecture of highly dilute alloys: an ab initio Monte Carlo approach. ACS Catal. 10, 1224–1236 (2020).
Zheng, K., Fung, V., Yuan, X., Jiang, D. & Xie, J. Real time monitoring of the dynamic intracluster diffusion of single gold atoms into silver nanoclusters. J. Am. Chem. Soc. 141, 18977–18983 (2019).
Lu, S. et al. Unveiling the structural transformation and activity origin of heteroatom-doped carbons for hydrogen evolution. Proc. Natl Acad. Sci. USA 120, e2300549120 (2023).
Ren, M. et al. Interlayer palladium-single-atom-coordinated cyano-group-rich graphitic carbon nitride for enhanced photocatalytic hydrogen production performance. ACS Catal. 12, 5077–5093 (2022).
Zhang, E. et al. Engineering the local atomic environments of indium single‐atom catalysts for efficient electrochemical production of hydrogen peroxide. Angew. Chem. Int. Ed. 61, e202117347 (2022).
Zhang, L. et al. Atomic layer deposited Pt–Ru dual-metal dimers and identifying their active sites for hydrogen evolution reaction. Nat. Commun. 10, 4936 (2019).
Xie, Y. et al. Direct oxygen–oxygen cleavage through optimizing interatomic distances in dual single‐atom electrocatalysts for efficient oxygen reduction reaction. Angew. Chem. Int. Ed. 62, e202301833 (2023).
Li, R. & Wang, D. Superiority of dual‐atom catalysts in electrocatalysis: one step further than single‐atom catalysts. Adv. Energy Mater. 12, 2103564 (2022).
Zhou, Y. et al. Dual‐metal interbonding as the chemical facilitator for single‐atom dispersions. Adv. Mater. 32, 2003484 (2020).
Zhu, P. et al. Regulating the FeN4 moiety by constructing Fe–Mo dual-metal atom sites for efficient electrochemical oxygen reduction. Nano Lett. 22, 9507–9515 (2022).
Kakati, N. et al. Anode catalysts for direct methanol fuel cells in acidic media: do we have any alternative for Pt or Pt–Ru? Chem. Rev. 114, 12397–12429 (2014).
Cuesta, A. At least three contiguous atoms are necessary for CO formation during methanol electrooxidation on platinum. J. Am. Chem. Soc. 128, 13332–13333 (2006).
Tripković, A. V. et al. Methanol electrooxidation on supported Pt and PtRu catalysts in acid and alkaline solutions. Electrochim. Acta 47, 3707–3714 (2002).
Wang, Q. et al. Manipulating the surface composition of Pt–Ru bimetallic nanoparticles to control the methanol oxidation reaction pathway. Chem. Commun. 56, 2419–2422 (2020).
Zhao, L. et al. The oxidation of methanol on PtRu(111): a periodic density functional theory investigation. J. Phys. Chem. C 119, 20389–20400 (2015).
You, G. et al. PtPd(111) surface versus PtAu(111) surface: which one is more active for methanol oxidation? ACS Catal. 8, 132–143 (2018).
Kim, H. et al. Palladium single‐atom catalysts supported on C@C3N4 for electrochemical reactions. ChemElectroChem 6, 4757–4764 (2019).
Kim, Y.-T. et al. Fine size control of platinum on carbon nanotubes: from single atoms to clusters. Angew. Chem. Int. Ed. 45, 407–411 (2006).
Awoke, Y. A. et al. The synergistic effect Pt1-W dual sites as a highly active and durable catalyst for electrochemical methanol oxidation. Electrochim. Acta 432, 141161 (2022).
Sun, S. et al. Single-atom catalysis using Pt/graphene achieved through atomic layer deposition. Sci. Rep. 3, 1775 (2013).
Poerwoprajitno, A. R. et al. A single-Pt-atom-on-Ru-nanoparticle electrocatalyst for CO-resilient methanol oxidation. Nat. Catal. 5, 231–237 (2022).
Zhang, Z. et al. Single-atom catalyst for high-performance methanol oxidation. Nat. Commun. 12, 5235 (2021).
Poerwoprajitno, A. R. et al. Tuning the Pt–Ru atomic neighbors for active and stable methanol oxidation electrocatalysis. Chem. Mater. 35, 10724–10729 (2023).
Liu, J. et al. Tackling CO poisoning with single-atom alloy catalysts. J. Am. Chem. Soc. 138, 6396–6399 (2016).
Chen, X., Granda-Marulanda, L. P., McCrum, I. T. & Koper, M. T. M. How palladium inhibits CO poisoning during electrocatalytic formic acid oxidation and carbon dioxide reduction. Nat. Commun. 13, 38 (2022).
Liang, M. et al. Modulating reaction pathways of formic acid oxidation for optimized electrocatalytic performance of PtAu/CoNC. Nano Res. 15, 1221–1229 (2022).
Xiong, Y. et al. Single-atom Rh/N-doped carbon electrocatalyst for formic acid oxidation. Nat. Nanotechnol. 15, 390–397 (2020).
Shen, T. et al. Engineering Ir atomic configuration for switching the pathway of formic acid electrooxidation reaction. Adv. Funct. Mater. 32, 2107672 (2022).
Peng, Z. & Yang, H. PtAu bimetallic heteronanostructures made by post-synthesis modification of Pt-on-Au nanoparticles. Nano Res. 2, 406–415 (2009).
Liu, M. et al. Ligand‐mediated self‐terminating growth of single‐atom Pt on Au nanocrystals for improved formic acid oxidation activity. Adv. Energy Mater. 12, 2103195 (2022).
Duchesne, P. N. et al. Golden single-atomic-site platinum electrocatalysts. Nat. Mater. 17, 1033–1039 (2018).
Li, J., Liang, X., Cai, L., Huang, S. & Zhao, C. Modification of palladium nanocrystals with single atom platinum via an electrochemical self-catalysis strategy for efficient formic acid electrooxidation. ACS Appl. Mater. Interfaces 14, 8001–8009 (2022).
Wang, H. et al. PdBi single‐atom alloy aerogels for efficient ethanol oxidation. Adv. Funct. Mater. 31, 2103465 (2021).
Wang, H. et al. Pd metallene aerogels with single-atom W doping for selective ethanol oxidation. ACS Nano 16, 21266–21274 (2022).
Zhang, G. et al. Tuning the selective ethanol oxidation on tensile‐trained Pt(110) surface by Ir single atoms. Small 18, 2202587 (2022).
Luo, S. et al. A tensile‐strained Pt–Rh single‐atom alloy remarkably boosts ethanol oxidation. Adv. Mater. 33, 2008508 (2021).
Nguyen, T. N., Salehi, M., van Le, Q., Seifitokaldani, A. & Dinh, C. T. Fundamentals of electrochemical CO2 reduction on single-metal-atom catalysts. ACS Catal. 10, 10068–10095 (2020).
Zhang, L., Zhao, Z.-J. & Gong, J. Nanostructured materials for heterogeneous electrocatalytic CO2 reduction and their related reaction mechanisms. Angew. Chem. Int. Ed. 56, 11326–11353 (2017).
Thevenon, A., Rosas-Hernández, A., Fontani Herreros, A. M., Agapie, T. & Peters, J. C. Dramatic HER suppression on Ag electrodes via molecular films for highly selective CO2 to CO reduction. ACS Catal. 11, 4530–4537 (2021).
Zhang, Y.-J., Sethuraman, V., Michalsky, R. & Peterson, A. A. Competition between CO2 reduction and H2 evolution on transition-metal electrocatalysts. ACS Catal. 4, 3742–3748 (2014).
Cheng, M., Clark, E. L., Pham, H. H., Bell, A. T. & Head-gordon, M. Quantum mechanical screening of single-atom bimetallic alloys for the selective reduction of CO2 to C1 hydrocarbons. ACS Catal. 6, 7769–7777 (2016).
Jiang, K. et al. Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction. Energy Environ. Sci. 11, 893–903 (2018).
Zhang, N. et al. A supported Pd2 dual‐atom site catalyst for efficient electrochemical CO2 reduction. Angew. Chem. Int. Ed. 60, 13388–13393 (2021).
Jiang, J.-C. et al. Rational design of copper-based single-atom alloy catalysts for electrochemical CO2 reduction. Nano Res. 15, 7116–7123 (2022).
Wang, Y. et al. CO electroreduction on single-atom copper. Sci. Adv. 9, eade3557 (2023).
Yan, Z. et al. Recent progress in electrocatalytic conversion of CO2 to valuable C2 products. Adv. Mater. Interfaces 10, 2300186 (2023).
Morales-Guio, C. G. et al. Improved CO2 reduction activity towards C2+ alcohols on a tandem gold on copper electrocatalyst. Nat. Catal. 1, 764–771 (2018).
Li, F. et al. Cooperative CO2-to-ethanol conversion via enriched intermediates at molecule–metal catalyst interfaces. Nat. Catal. 3, 75–82 (2020).
Yin, Z. et al. Hybrid catalyst coupling single-atom Ni and nanoscale Cu for efficient CO2 electroreduction to ethylene. J. Am. Chem. Soc. 144, 20931–20938 (2022).
Chen, L., Tang, C., Zheng, Y., Skúlason, E. & Jiao, Y. C3 production from CO2 reduction by concerted *CO trimerization on a single-atom alloy catalyst. J. Mater. Chem. A 10, 5998–6006 (2022).
Back, S., Yeom, M. S. & Jung, Y. Active sites of Au and Ag nanoparticle catalysts for CO2 electroreduction to CO. ACS Catal. 5, 5089–5096 (2015).
Liu, W., Tkatchenko, A. & Scheffler, M. Modeling adsorption and reactions of organic molecules at metal surfaces. Acc. Chem. Res. 47, 3369–3377 (2014).
Hsu, C.-S. et al. Activating dynamic atomic-configuration for single-site electrocatalyst in electrochemical CO2 reduction. Nat. Commun. 14, 5245 (2023).
Hu, Y. et al. Atomic modulation of single dispersed Ir species on self-supported NiFe layered double hydroxides for efficient electrocatalytic overall water splitting. ACS Catal. 13, 11195–11203 (2023).
Deng, X., Alfonso, D., Nguyen-Phan, T.-D. & Kauffman, D. R. Resolving the size-dependent transition between CO2 reduction reaction and H2 evolution reaction selectivity in sub-5 nm silver nanoparticle electrocatalysts. ACS Catal. 12, 5921–5929 (2022).
Nie, X., Esopi, M. R., Janik, M. J. & Asthagiri, A. Selectivity of CO2 reduction on copper electrodes: the role of the kinetics of elementary steps. Angew. Chem. Int. Ed. 52, 2459–2462 (2013).
Wang, C. et al. Combining Fe nanoparticles and pyrrole-type Fe-N4 sites on less-oxygenated carbon supports for electrochemical CO2 reduction. Nat. Commun. 14, 5108 (2023).
Wang, D. et al. Oxygen‐bridged copper–iron atomic pair as dual‐metal active sites for boosting electrocatalytic NO reduction. Adv. Mater. 35, e2304646 (2023).
Li, Y. et al. Single‐atom iron catalyst with biomimetic active center to accelerate proton spillover for medical‐level electrosynthesis of H2O2 disinfectant. Angew. Chem. Int. Ed. 62, e202306491 (2023).
Yang, J. et al. Regulating the tip effect on single‐atom and cluster catalysts: forming reversible oxygen species with high efficiency in chlorine evolution reaction. Angew. Chem. Int. Ed. 61, e202200366 (2022).
Speck, F. D. et al. Atomistic insights into the stability of Pt single-atom electrocatalysts. J. Am. Chem. Soc. 142, 15496–15504 (2020).
Bai, X. et al. Dynamic stability of copper single-atom catalysts under working conditions. J. Am. Chem. Soc. 144, 17140–17148 (2022).
Yang, Q., Yang, C.-C., Lin, C.-H. & Jiang, H.-L. Metal-organic-framework-derived hollow N-doped porous carbon with ultrahigh concentrations of single Zn atoms for efficient carbon dioxide conversion. Angew. Chem. Int. Ed. 58, 3511–3515 (2019).
Ye, C., Zhang, N., Wang, D. & Li, Y. Single atomic site catalysts: synthesis, characterization, and applications. Chem. Commun. 56, 7687–7697 (2020).
Zhang, H., Jin, X., Lee, J.-M. & Wang, X. Tailoring of active sites from single to dual atom sites for highly efficient electrocatalysis. ACS Nano 16, 17572–17592 (2022).
Fang, J., Chen, Q., Li, Z., Mao, J. & Li, Y. The synthesis of single-atom catalysts for heterogeneous catalysis. Chem. Commun. 59, 2854–2868 (2023).
Zheng, X., Li, B., Wang, Q., Wang, D. & Li, Y. Emerging low-nuclearity supported metal catalysts with atomic level precision for efficient heterogeneous catalysis. Nano Res. 15, 7806–7839 (2022).
Yang, X. et al. Review of emerging atomically precise composite site‐based electrocatalysts. Adv. Energy Mater. https://doi.org/10.1002/aenm.202301737 (2023).
Qi, Z., Zhou, Y., Guan, R., Fu, Y. & Baek, J. Tuning the coordination environment of carbon‐based single‐atom catalysts via doping with multiple heteroatoms and their applications in electrocatalysis. Adv. Mater. 35, 2210575 (2023).
Gao, Y., Liu, B. & Wang, D. Microenvironment engineering of single/dual‐atom catalysts for electrocatalytic application. Adv. Mater. 35, 2209654 (2023).
Shi, B. et al. Fe single-atom catalyst for cost-effective yet highly efficient heterogeneous Fenton catalysis. ACS Appl. Mater. Interfaces 14, 53767–53776 (2022).
Ji, D. et al. Atomically transition metals on self‐supported porous carbon flake arrays as binder‐free air cathode for wearable zinc−air batteries. Adv. Mater. 31, 1808267 (2019).
Mou, K. et al. Highly efficient electroreduction of CO2 on nickel single‐atom catalysts: atom trapping and nitrogen anchoring. Small 15, 1903668 (2019).
Tian, H. et al. High durability of Fe–N–C single atom catalysts with carbon vacancies towards oxygen reduction reaction in alkaline media. Adv. Mater. 35, 2210714 (2023).
Chen, X. et al. MOF-derived isolated Fe atoms implanted in N-doped 3D hierarchical carbon as an efficient ORR electrocatalyst in both alkaline and acidic media. ACS Appl. Mater. Interfaces 11, 25976–25985 (2019).
Sun, K. et al. Nature‐inspired design of molybdenum–selenium dual‐single‐atom electrocatalysts for CO2 reduction. Adv. Mater. 34, 2206478 (2022).
Yu, L.-Q. et al. Universal method to fabricate transition metal single-atom-anchored carbon with excellent oxygen reduction reaction activity. ACS Appl. Mater. Interfaces 13, 13534–13540 (2021).
Liu, S. et al. Chemical vapor deposition for atomically dispersed and nitrogen coordinated single metal site catalysts. Angew. Chem. Int. Ed. 59, 21698–21705 (2020).
Li, X. et al. Chemical vapor deposition for N/S-doped single Fe site catalysts for the oxygen reduction in direct methanol fuel cells. ACS Catal. 11, 7450–7459 (2021).
Zhao, W. et al. Pt–Ru dimer electrocatalyst with electron redistribution for hydrogen evolution reaction. ACS Catal. 12, 5540–5548 (2022).
Li, Y. et al. Atomically dispersed dual‐metal site catalysts for enhanced CO2 reduction: mechanistic insight into active site structures. Angew. Chem. Int. Ed. 61, e202205632 (2022).
Pei, Z. et al. Highly efficient electrocatalytic oxygen evolution over atomically dispersed synergistic Ni/Co dual sites. Angew. Chem. Int. Ed. 61, e202207537 (2022).
Wang, Y. et al. Precisely constructing orbital coupling-modulated dual-atom Fe pair sites for synergistic CO2 electroreduction. ACS Energy Lett. 7, 640–649 (2022).
Kunwar, D. et al. Stabilizing high metal loadings of thermally stable platinum single atoms on an industrial catalyst support. ACS Catal. 9, 3978–3990 (2019).
Wang, Q. et al. Ultrahigh-loading of Ir single atoms on NiO matrix to dramatically enhance oxygen evolution reaction. J. Am. Chem. Soc. 142, 7425–7433 (2020).
Hejazi, S. et al. On the controlled loading of single platinum atoms as a co‐catalyst on TiO2 anatase for optimized photocatalytic H2 generation. Adv. Mater. 32, 1908505 (2020).
Ling, Y. et al. General strategy toward hydrophilic single atom catalysts for efficient selective hydrogenation. Adv. Sci. 9, 2202144 (2022).
Xu, H. et al. Cation exchange strategy to single-atom noble-metal doped CuO nanowire arrays with ultralow overpotential for H2O splitting. Nano Lett. 20, 5482–5489 (2020).
Zhang, Z. et al. Electrochemical deposition as a universal route for fabricating single-atom catalysts. Nat. Commun. 11, 1215 (2020).
Li, H. et al. Vapor-phase self-assembly for generating thermally stable single-atom catalysts. Chem 8, 731–748 (2022).
Wang, Z. et al. Titania‐supported Cu‐single‐atom catalyst for electrochemical reduction of acetylene to ethylene at low‐concentrations with suppressed hydrogen evolution. Adv. Mater. 35, e2303818 (2023).
Chen, Z. et al. Stabilizing Pt single atoms through Pt−Se electron bridges on vacancy‐enriched nickel selenide for efficient electrocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 62, e202308686 (2023).
Shi, Y. et al. Site-specific electrodeposition enables self-terminating growth of atomically dispersed metal catalysts. Nat. Commun. 11, 4558 (2020).
Chao, T. et al. Atomically dispersed copper–platinum dual sites alloyed with palladium nanorings catalyze the hydrogen evolution reaction. Angew. Chem. Int. Ed. 56, 16047–16051 (2017).
Kong, F. et al. Selectively coupling Ru single atoms to PtNi concavities for high‐performance methanol oxidation via d‐band center regulation. Angew. Chem. Int. Ed. 61, e202207524 (2022).
Liu, S. et al. Awakening (220) as one more active facet of PtMo alloy via single‐atom doping to boost ammonia electrooxidation in direct ammonia fuel cell. Adv. Funct. Mater. https://doi.org/10.1002/adfm.202306204 (2023).
Wang, Y.-R. et al. Reduction-controlled atomic migration for single atom alloy library. Nano Lett. 22, 4232–4239 (2022).
Du, C. et al. Selectively reducing nitrate into NH3 in neutral media by PdCu single-atom alloy electrocatalysis. ACS Catal. 13, 10560–10569 (2023).
Xia, W. et al. Adjacent copper single atoms promote C–C coupling in electrochemical CO2 reduction for the efficient conversion of ethanol. J. Am. Chem. Soc. 145, 17253–17264 (2023).
Cao, S. et al. Single-atom gold oxo-clusters prepared in alkaline solutions catalyse the heterogeneous methanol self-coupling reactions. Nat. Chem. 11, 1098–1105 (2019).
Liu, Y. et al. Unraveling the function of metal–amorphous support interactions in single‐atom electrocatalytic hydrogen evolution. Angew. Chem. Int. Ed. 61, e202114160 (2022).
Shan, J. et al. Integrating interactive noble metal single-atom catalysts into transition metal oxide lattices. J. Am. Chem. Soc. 144, 23214–23222 (2022).
Zeng, R. et al. Atomically site synergistic effects of dual-atom nanozyme enhances peroxidase-like properties. Nano Lett. 23, 6073–6080 (2023).
Yang, S. et al. Metal single-atom and nanoparticle double-active-site relay catalysts: design, preparation, and application to the oxidation of 5-hydroxymethylfurfural. ACS Catal. 12, 971–981 (2022).
Ye, X. et al. Highly selective hydrogenation of CO2 to ethanol via designed bifunctional Ir1–In2O3 single-atom catalyst. J. Am. Chem. Soc. 142, 19001–19005 (2020).
Zhang, J. et al. Competitive adsorption: reducing the poisoning effect of adsorbed hydroxyl on Ru single‐atom site with SnO2 for efficient hydrogen evolution. Angew. Chem. Int. Ed. 61, e202209486 (2022).
Yang, S. & Lee, H. Atomically dispersed platinum on gold nano-octahedra with high catalytic activity on formic acid oxidation. ACS Catal. 3, 437–443 (2013).
Zhang, X. et al. Platinum–copper single atom alloy catalysts with high performance towards glycerol hydrogenolysis. Nat. Commun. 10, 5812 (2019).
Wang, Y. et al. p–d orbital hybridization induced by a monodispersed Ga site on a Pt3Mn nanocatalyst boosts ethanol electrooxidation. Angew. Chem. Int. Ed. 61, e202115735 (2022).
Zhu, Y. et al. Supported ruthenium single‐atom and clustered catalysts outperform benchmark Pt for alkaline hydrogen evolution. Adv. Mater. 35, 2301133 (2023).
Acknowledgements
The authors acknowledge funding under the Australian Research Council’s Discovery Project (R.D.T., DP200100143 and DP230100596; J.J.G., DP210102698), Centre of Excellence (R.D.T., CE230100032) and Training Centre IC210100056 and National Health and Medical Research Council Investigator grant (J.J.G., GNT1196648). S.V.S. acknowledges support from the Australian Government Research Training Program (RTP) Scholarship. They also acknowledge support from Microscopy Australia and the Mark Wainwright Analytical Centre and Electron Microscope Unit at the University of New South Wales.
Author information
Authors and Affiliations
Contributions
L.G., S.V.S. and R.D.T. researched data for the article. L.G. and R.D.T. contributed substantially to the discussion of the content. L.G., J.J.G. and R.D.T. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Materials thanks Dingsheng Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gloag, L., Somerville, S.V., Gooding, J.J. et al. Co-catalytic metal–support interactions in single-atom electrocatalysts. Nat Rev Mater 9, 173–189 (2024). https://doi.org/10.1038/s41578-023-00633-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-023-00633-2
- Springer Nature Limited