Abstract
Mycorrhizae, a form of plant–fungal symbioses, mediate vegetation impacts on ecosystem functioning. Climatic effects on decomposition and soil quality are suggested to drive mycorrhizal distributions, with arbuscular mycorrhizal plants prevailing in low-latitude/high-soil-quality areas and ectomycorrhizal (EcM) plants in high-latitude/low-soil-quality areas. However, these generalizations, based on coarse-resolution data, obscure finer-scale variations and result in high uncertainties in the predicted distributions of mycorrhizal types and their drivers. Using data from 31 lowland tropical forests, both at a coarse scale (mean-plot-level data) and fine scale (20 × 20 metres from a subset of 16 sites), we demonstrate that the distribution and abundance of EcM-associated trees are independent of soil quality. Resource exchange differences among mycorrhizal partners, stemming from diverse evolutionary origins of mycorrhizal fungi, may decouple soil fertility from the advantage provided by mycorrhizal associations. Additionally, distinct historical biogeographies and diversification patterns have led to differences in forest composition and nutrient-acquisition strategies across three major tropical regions. Notably, Africa and Asia’s lowland tropical forests have abundant EcM trees, whereas they are relatively scarce in lowland neotropical forests. A greater understanding of the functional biology of mycorrhizal symbiosis is required, especially in the lowland tropics, to overcome biases from assuming similarity to temperate and boreal regions.



Similar content being viewed by others
Data availability
ForestGEO plot data can be obtained upon request via the ForestGEO portal at http://ctfs.si.edu/datarequest/. All data sources are listed in Extended Data Table 1. PCA axes and the contribution (proportion) of EcM trees to basal area can be found at https://doi.org/10.5281/zenodo.10044772 ref. 93.
Code availability
The code to run the analyses at both coarse and fine scales can be found at https://doi.org/10.5281/zenodo.10044772 ref. 93.
References
Lambers, H., Mougel, C., Jaillard, B. & Hinsinger, P. Plant–microbe–soil interactions in the rhizosphere: an evolutionary perspective. Plant Soil 321, 83–115 (2009).
Smith, S. E. & Read, D. J. Mycorrhizal Symbiosis (Academic Press, 2008).
Tedersoo, L. & Bahram, M. Mycorrhizal types differ in ecophysiology and alter plant nutrition and soil processes. Biol. Rev. 94, 1857–1880 (2019).
Branco, S. et al. Mechanisms of stress tolerance and their effects on the ecology and evolution of mycorrhizal fungi. New Phytol. 235, 2158–2175 (2022).
Jiang, Y. et al. Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science 356, 1172–1175 (2017).
Howard, N. et al. The potential role of Mucoromycotina ‘fine root endophytes’ in plant nitrogen nutrition. Physiol. Plant. 174, e13715 (2022).
Hoysted, G. A. et al. Direct nitrogen, phosphorus and carbon exchanges between Mucoromycotina ‘fine root endophyte’ fungi and a flowering plant in novel monoxenic cultures. New Phytol. 238, 70–79 (2023).
Brundrett, M. C. & Tedersoo, L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 220, 1108–1115 (2018).
Phillips, R. P., Brzostek, E. & Midgley, M. G. The mycorrhizal-associated nutrient economy: a new framework for predicting carbon–nutrient couplings in temperate forests. New Phytol. 199, 41–51 (2013).
Wurzburger, N. et al. Mycorrhizal fungi as drivers and modulators of terrestrial ecosystem processes. New Phytol. 213, 996–999 (2017).
Soudzilovskaia, N. A. et al. Global mycorrhizal plant distribution linked to terrestrial carbon stocks. Nat. Commun. 10, 5077 (2019).
Gadgil, R. L. & Gadgil, P. D. Mycorrhiza and litter decomposition. Nature 233, 133–133 (1971).
Rozmoš, M. et al. Organic nitrogen utilisation by an arbuscular mycorrhizal fungus is mediated by specific soil bacteria and a protist. ISME J. 16, 676–685 (2022).
Wang, L. et al. A core microbiome in the hyphosphere of arbuscular mycorrhizal fungi has functional significance in organic phosphorus mineralization. New Phytol. 238, 859–873 (2023).
Tedersoo, L., Bahram, M. & Zobel, M. How mycorrhizal associations drive plant population and community biology. Science 367, eaba1223 (2020).
Becquer, A. et al. in Advances in Botanical Research (ed. Cánovas, F. M.) 77–126 (Academic Press, 2019).
Averill, C. et al. Global imprint of mycorrhizal fungi on whole-plant nutrient economics. Proc. Natl Acad. Sci. USA 116, 23163–23168 (2019).
Seyfried, G. S., Dalling, J. W. & Yang, W. H. Mycorrhizal type effects on leaf litter decomposition depend on litter quality and environmental context. Biogeochemistry 155, 21–38 (2021).
Steidinger, B. S. et al. Climatic controls of decomposition drive the global biogeography of forest–tree symbioses. Nature 569, 404–408 (2019).
Read, D. J. Mycorrhizas in ecosystems. Experientia 47, 376–391 (1991).
Braghiere, R. K. et al. Mycorrhizal distributions impact global patterns of carbon and nutrient cycling. Geophys. Res. Lett. 48, e2021GL094514 (2021).
Barceló, M. et al. Climate drives the spatial distribution of mycorrhizal host plants in terrestrial ecosystems. J. Ecol. 107, 2564–2573 (2019).
Lokonda, M. et al. Are soils under monodominant Gilbertiodendron dewevrei and under adjacent mixed forests similar? A case study in the Democratic Republic of Congo. J. Trop. Ecol. 34, 176–185 (2018).
Lee, H. S. et al. Floristic and structural diversity of mixed dipterocarp forest in Lambir Hills National Park, Sarawak, Malaysia. J. Trop. Sci. 14, 379–400 (2002).
Newbery, D. M. et al. Transient dominance in a central African rain forest. Ecol. Monogr. 83, 339–382 (2013).
Beard, J. S. The Mora forests of Trinidad, British West Indies. J. Ecol. 33, 173–192 (1946).
Steidinger, B. S. et al. Variability in potential to exploit different soil organic phosphorus compounds among tropical montane tree species. Funct. Ecol. 29, 121–130 (2015).
Koele, N. et al. No globally consistent effect of ectomycorrhizal status on foliar traits. New Phytol. 196, 845–852 (2012).
Godoy, R. & Marín, C. in Mycorrhizal Fungi in South America (eds Pagano, M. C. & Lugo, M. A.) 315–341 (Springer International Publishing, 2019).
Marín, C. et al. Geological history and forest mycorrhizal dominance effects on soil fungal diversity in Chilean temperate rainforests. J. Soil Sci. Plant Nutr. 23, 734–745 (2023).
Weemstra, M. et al. Lithological constraints on resource economies shape the mycorrhizal composition of a Bornean rain forest. New Phytol. 228, 253–268 (2020).
Davies, S. J. et al. ForestGEO: understanding forest diversity and dynamics through a global observatory network. Biol. Conserv. 253, 108907 (2021).
Liu, F. & Eugenio, E. C. A review and comparison of Bayesian and likelihood-based inferences in beta regression and zero-or-one-inflated beta regression. Stat. Methods Med. Res. 27, 1024–1044 (2018).
Zuur, A. F. & Ieno, E. N. Beginner’s Guide to Spatial, Temporal and Spatial-temporal Ecological Data Analysis with R-INLA: GAM and Zero-inflated Models (Highland Statistics Limited, 2018).
Barceló, M. et al. Mycorrhizal tree impacts on topsoil biogeochemical properties in tropical forests. J. Ecol. 110, 1271–1282 (2022).
Newbery, D. M., Alexander, I. J. & Rother, J. A. Phosphorus dynamics in a lowland African rainforest: the influence of ectomycorrhizal trees. Ecol. Monogr. 67, 367–409 (1997).
Henkel, T. W. Monodominance in the ectomycorrhizal Dicymbe corymbosa (Caesalpiniaceae) from Guyana. J. Trop. Ecol. 19, 417–437 (2003).
Hasselquist, N. J. et al. Greater carbon allocation to mycorrhizal fungi reduces tree nitrogen uptake in a boreal forest. Ecology 97, 1012–1022 (2016).
Franklin, O. et al. Forests trapped in nitrogen limitation – an ecological market perspective on ectomycorrhizal symbiosis. New Phytol. 203, 657–666 (2014).
Näsholm, T. et al. Are ectomycorrhizal fungi alleviating or aggravating nitrogen limitation of tree growth in boreal forests? New Phytol. 198, 214–221 (2013).
Treseder, K. K. & Allen, M. F. Direct nitrogen and phosphorus limitation of arbuscular mycorrhizal fungi: a model and field test. New Phytol. 155, 507–515 (2002).
Allen, M. F. Mycorrhizal Dynamics in Ecological Systems (Cambridge Univ. Press, 2022).
Martin, F., Kohler, A., Murat, C., Veneault-Fourrey, C. & Hibbett, D. S. Unearthing the roots of ectomycorrhizal symbioses. Nat. Rev. Microbiol. 14, 760–773 (2016).
Tedersoo, L. & Smith, M. E. in Biogeography of Mycorrhizal Symbiosis (ed. Tedersoo, L.) 125–142 (Springer International Publishing, 2017).
Spatafora, J. W. et al. A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. Mycologia 108, 1028–1046 (2016).
Dickie, I. A. & Moyersoen, B. Towards a global view of ectomycorrhizal ecology. New Phytol. 180, 263–265 (2008).
Pellitier, P. T. & Zak, D. R. Ectomycorrhizal fungi and the enzymatic liberation of nitrogen from soil organic matter: why evolutionary history matters. New Phytol. 217, 68–73 (2018).
Peay, K. G. et al. Lack of host specificity leads to independent assortment of dipterocarps and ectomycorrhizal fungi across a soil fertility gradient. Ecol. Lett. 18, 807–816 (2015).
Anthony, M. A. et al. Forest tree growth is linked to mycorrhizal fungal composition and function across Europe. ISME J. 16, 1327–1336 (2022).
Plett, K. L. et al. Intra-species genetic variability drives carbon metabolism and symbiotic host interactions in the ectomycorrhizal fungus Pisolithus microcarpus. Environ. Microbiol. 23, 2004–2020 (2021).
Corrales, A. et al. Variation in ectomycorrhizal fungal communities associated with Oreomunnea mexicana (Juglandaceae) in a Neotropical montane forest. Mycorrhiza 26, 1–17 (2015).
Tedersoo, L. in Biogeography of Mycorrhizal Symbiosis (ed. Tedersoo, L.) 469–531 (Springer International Publishing, 2017).
Corrales, A. et al. Diversity and distribution of tropical ectomycorrhizal fungi. Mycologia 114, 919–933 (2022).
Pena, R. & Polle, A. Attributing functions to ectomycorrhizal fungal identities in assemblages for nitrogen acquisition under stress. ISME J. 8, 321–330 (2014).
Hazard, C. et al. Contrasting effects of intra- and interspecific identity and richness of ectomycorrhizal fungi on host plants, nutrient retention and multifunctionality. New Phytol. 213, 852–863 (2017).
Hortal, S. et al. Role of plant–fungal nutrient trading and host control in determining the competitive success of ectomycorrhizal fungi. ISME J. 11, 2666–2676 (2017).
Corrales, A., Henkel, T. W. & Smith, M. E. Ectomycorrhizal associations in the tropics—biogeography, diversity patterns and ecosystem roles. New Phytol. 220, 1076–1091 (2018).
Nouhra, E. R. et al. in Mycorrhizal Fungi in South America (eds Pagano, M. C. & Lugo, M. A.) 73–95 (Springer International Publishing, 2019).
Cazzolla Gatti, R. et al. The number of tree species on Earth. Proc. Natl Acad. Sci. USA 119, e2115329119 (2022).
Karst, J., Jones, M. D. & Hoeksema, J. D. Positive citation bias and overinterpreted results lead to misinformation on common mycorrhizal networks in forests. Nat. Ecol. Evol. 7, 501–511 (2023).
Soudzilovskaia, N. A. et al. FungalRoot: global online database of plant mycorrhizal associations. New Phytol. 227, 955–966 (2020).
Brundrett, M. & Tedersoo, L. Misdiagnosis of mycorrhizas and inappropriate recycling of data can lead to false conclusions. New Phytol. 221, 18–24 (2019).
Bueno, G. et al. Conceptual differences lead to divergent trait estimates in empirical and taxonomic approaches to plant mycorrhizal trait assignment. Mycorrhiza 29, 1–11 (2019).
Bueno, C. G. et al. Misdiagnosis and uncritical use of plant mycorrhizal data are not the only elephants in the room: a response to Brundrett & Tedersoo (2019) ‘Misdiagnosis of mycorrhizas and inappropriate recycling of data can lead to false conclusions’. New Phytol. 224, 1415–1418 (2019).
Tedersoo, L. et al. Global database of plants with root-symbiotic nitrogen fixation: NodDB. J. Veg. Sci. 29, 560–568 (2018).
John, R. et al. Soil nutrients influence spatial distributions of tropical tree species. Proc. Natl Acad. Sci. USA 104, 864–869 (2007).
Hendershot, W. H., Lalande, H. & Duquette, M. in Soil Sampling and Methods of Analysis (eds Carter, M. R. & Gregorich, E. G.) 197–206 (CRC Press, 2008).
Mehlich, A. Mehlich 3 soil test extractant: a modification of Mehlich 2 extractant. Commun. Soil Sci. Plant Anal. 15, 1409–1416 (1984).
Bray, R. H. & Kurtz, L. T. Determination of total, organic, and available forms of phosphorus in soils. Soil Sci. 59, 39–46 (1945).
Wolf, J. A. et al. Geospatial observations on tropical forest surface soil chemistry. Ecology 96, 2313–2313 (2015).
Fukuda, M. et al. Evaluation of the Mehlich 3 reagent as an extractant for cations and available phosphorus for soils in Mozambique. Commun. Soil Sci. Plant Anal. 48, 1462–1472 (2017).
Bibiso, M. et al. Evaluation of universal extractants for determination of selected micronutrients from soil. Bull. Chem. Soc. Ethiop. 29, 199–213 (2015).
Tran, T. S. et al. Evaluation of Mehlich‐III extractant to estimate the available P in Quebec soils. Commun. Soil Sci. Plant Anal. 21, 1–28 (1990).
Josse, J. & Husson, F. Handling missing values in exploratory multivariate data analysis methods. J. Soci.été Fr. Stat. 153, 79–99 (2012).
Lê, S., Josse, J. & Husson, F. FactoMineR: an R package for multivariate analysis. J. Stat. Softw. 25, 1–18 (2008).
Ribeiro, P. J. Jr et al. geoR: analysis of geostatistical data. R. package version 1, 8–1 (2020).
Ferrari, S. & Cribari-Neto, F. Beta regression for modelling rates and proportions. J. Appl. Stat. 31, 799–815 (2004).
Blangiardo, M. et al. Spatial and spatio-temporal models with R-INLA. Spat. Spatiotemporal Epidemiol. 4, 33–49 (2013).
Valencia, R. et al. Tree species distributions and local habitat variation in the Amazon: large forest plot in eastern Ecuador. J. Ecol. 92, 214–229 (2004).
Lepore, M. et al. Fgeo: analyze forest diversity and dynamics. R package version 1.1.4 (2019).
Peña-Claros, M. Changes in forest structure and species composition during secondary forest succession in the Bolivian Amazon. Biotropica 35, 450–461 (2003).
Dormann, C. F. et al. Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36, 27–46 (2013).
Riebler, A. et al. An intuitive Bayesian spatial model for disease mapping that accounts for scaling. Stat. Methods Med. Res. 25, 1145–1165 (2016).
Gómez-Rubio, V. Bayesian Inference with INLA (CRC Press, 2020).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2023).
Carpenter, B. et al. Stan: a probabilistic programming language. J. Stat. Softw. 76, jss.v076.i01 (2017).
Gabry, J. & Češnovar, R. Cmdstanr: R interface to ‘CmdStan’. R package version 0.4.0 (2021).
Bürkner, P.-C. Brms: an R package for Bayesian multilevel models using Stan. J. Stat. Softw. 80, jss.v080.i01 (2017).
Bürkner, P.-C. Advanced Bayesian multilevel modeling with the R package brms. R. J. 10, 395–411 (2018).
Gelman, A. et al. Bayesian Data Analysis (CRC press, 2013).
Rue, H., Martino, S. & Chopin, N. Approximate Bayesian inference for latent gaussian models using integrated nested laplace approximations (with discussion). J. R. Stat. Soc. B 71, 319–392 (2009).
Gabry, J. et al. Visualization in Bayesian workflow. J. R. Stat. Soc. A 182, 389–402 (2019).
Medina-Vega, J. A. et al. Dataset and code accompanying the study by Medina-Vega et al. in Nature Ecology & Evolution: tropical tree ectomycorrhiza are distributed independently of soil nutrients. Zenodo https://doi.org/10.5281/zenodo.10044772 (2023).
Quesada, C. A. et al. Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences 7, 1515–1541 (2010).
Chave, J. et al. Above-ground biomass and productivity in a rain forest of eastern South. Am. J. Trop. Ecol. 24, 355–366 (2008).
Fanin, N. et al. Does variability in litter quality determine soil microbial respiration in an Amazonian rainforest? Soil Biol. Biochem. 43, 1014–1022 (2011).
Libalah, M. B. et al. Shift in functional traits along soil fertility gradient reflects non-random community assembly in a tropical African rainforest. Plant Ecol. Evol. 150, 265–278 (2017).
Nkongolo, N. V., Mbuyi, J. J. K. & Lokonda, M. W. Quantification of soil carbon in Ituri forest, Democratic Republic of Congo. In Proc. Global Symposium on Soil Organic Carbon 151–153 (Food and Agriculture Organization of the United Nations, 2017).
Baillie, I. C. et al. Stoichiometry of cationic nutrients in Phaeozems derived from skarn and acrisols from other parent materials in lowland forests of Thailand. Geoderma Reg. 12, 1–9 (2018).
Sukri, R. et al. Habitat associations and community structure of dipterocarps in response to environment and soil conditions in Brunei Darussalam, Northwest Borneo. Biotropica 44, 595–605 (2012).
Ross, S. M. & Dykes, A. in Tropical Rainforest Research—Current Issues: Conf. Proc (eds Edwards, D. S. et al.) 259–270 (Springer Netherlands, 1996).
Dent, D. H. et al. Nutrient fluxes via litterfall and leaf litter decomposition vary across a gradient of soil nutrient supply in a lowland tropical rain forest. Plant Soil 288, 197–215 (2006).
Anderson-Teixeira, K. J. et al. CTFS-ForestGEO: a worldwide network monitoring forests in an era of global change. Glob. Change Biol. 21, 528–549 (2015).
Moraga, P. Geospatial Health Data: Modeling and Visualization with R-INLA and Shiny (CRC Press, 2019).
Acknowledgements
We express our gratitude to the dedicated field and data technicians whose meticulous data-gathering efforts were indispensable to this research. Their pivotal contributions formed the backbone of our study. Our gratitude also extends to the teams of scientists behind the papers and datasets that enriched our primary dataset. This research and J.A.M.-V. were supported as part of the Next Generation Ecosystem Experiments-Tropics, funded by the US Department of Energy, Office of Science, Office of Biological and Environmental Research. For site-specific acknowledgements, please refer to Supplementary Table 3.
Author information
Authors and Affiliations
Contributions
J.A.M.-V. and S.J.D. conceptualized the study, coordinated the data compilations, designed the analysis and interpreted the data. J.A.M.-V. performed data analyses. J.A.M.-V. led the writing of the paper with inputs from S.J.D., D.F.R.P.B., J.W.D., S.E.R. and D.Z. S.A., A.A., P.B., W.Y.B., S.B., N.C., J.C., A.A.d.O., Á.D., S.E., C.E.N.E., J.F., S.P.H., A.I., S.K., S.K.Y.L., J.-R.M., H.M., D.M., M.B.M., A.N., R.N., N.V.N., V.N., M.J.O., R.P., N.P., G.R., S.T., J.T., M.U., R.V., A.V., T.L.Y., J.K.Z. and D.Z. provided coordination and leadership, data management and quality control, travel, consumables and commented on the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Leho Tedersoo, César Marín and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
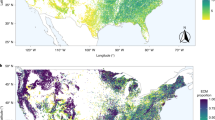
Extended Data Fig. 1 Location of the 31 study sites.
Letters indicate the tags used to identify plots in the principal component analysis (PCA) of soil data, constructed using coarse scale soil data (Fig. 1b and Extended Data Table 1). The names of each study site are enclosed in parentheses. Thirty sites were used in the analysis at a coarse scale, whereas 16 sites (shown in bold and italics) were used for the fine scale analysis (see Methods). Map made with Natural Earth.
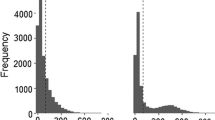
Extended Data Fig. 2 Variation in quadrat-level basal area and its association with the probability of observing EcM trees and their relative abundance in 16 lowland tropical forests.
a, Shows the distribution of the quadrat-level total basal area (BA) after applying a natural logarithm transformation. The x-axis represents the transformed total BA for each 20 × 20 m quadrat, whereas the y-axis indicates the study site. Vertical lines at the base of each density curve indicate individual observations. b,c, Present mean site-level coefficients, with panel b representing the probability (Prob.) of observing EcM trees and panel c for their relative abundance (conditional on the presence of EcM trees; Cd. Rel. Abun.) in relation to the quadrat-level total BA. The x-axes show the value of the coefficient on the logit scale, with the y-axes again showing the study site. Error bars show the 95% credible interval of the coefficient. These coefficients and their credible intervals derive from 200 draws from the Zero-Altered Beta (ZABE) regression’s posterior predictive distribution. This regression estimated the probability of observing EcM trees and their conditional relative abundance in BA, with the total quadrat-level basal area being logarithmically transformed before the analyses. The study includes 16 sites from lowland tropical regions in Africa (Af., two sites), Asia (As., eight sites), the neotropics (Neo., five sites), and Oceania (O., one site). Dashed lines indicate that the coefficients are not different from zero.
Supplementary information
Supplementary Information
Supplementary discussion, Note 1, Tables 1–3 and Figs. 1 and 2.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Medina-Vega, J.A., Zuleta, D., Aguilar, S. et al. Tropical tree ectomycorrhiza are distributed independently of soil nutrients. Nat Ecol Evol 8, 400–410 (2024). https://doi.org/10.1038/s41559-023-02298-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-023-02298-0
- Springer Nature Limited