Abstract
This study aims to investigate the characteristics of the phenotype and management of chronic obstructive pulmonary disease (COPD) patients in the general population in China. We analyzed spirometry-confirmed COPD patients who were identified from a population-based, nationally representative sample in China. All participants were measured with airflow limitation severity based on post-bronchodilator FEV1 percent predicted, bronchodilator responsiveness, exacerbation history, and respiratory symptoms. Among a total of 9134 COPD patients, 90.3% were non-exacerbators, 2.9% were frequent exacerbators without chronic bronchitis, 2.0% were frequent exacerbators with chronic bronchitis, and 4.8% were asthma-COPD overlap. Less than 5% of non-exacerbators ever had pulmonary function testing performed. The utilization rate of inhaled medication in non-exacerbators, exacerbators without chronic bronchitis, exacerbators with chronic bronchitis, and asthma-COPD overlap was 1.4, 23.5, 29.5, and 19.4%, respectively. A comprehensive strategy for the management of COPD patients based on phenotype in primary care is urgently needed.
Similar content being viewed by others
Introduction
Chronic obstructive pulmonary disease (COPD) is a globally public-health problem, with an estimation of 299.4 million prevalent cases and 3.2 million deaths per year worldwide1,2. In China, the COPD prevalence is 8.6% among adults with a projected 99.9 million patients3,4. COPD management greatly challenge the health-care systems, especially in low- and middle-income countries.
Early diagnosis and appropriate management of COPD patients in primary care play an important role in reducing the risk of adverse outcomes and improving quality of life, but this issue is paid not enough attention previously5. Furthermore, individualized management protocol based on specific COPD phenotype has been advocated instead of a “one size fits all” pattern6,7. Phenotypic strategies based on symptoms, response to therapy, and risk of disease progression are essential to direct the identification and management of COPD patients in routine practice8. However, great variations in terms of epidemiological characteristics and management protocols within ethnicity or nation complicates this goal. Although COPD phenotype distribution associated with clinically meaningful outcomes have been identified in clinical-based studies9,10,11, the issue is less known for COPD patients in general population in China.
Using data from a nationally representative, cross-sectional study in China4, we aimed to investigate the epidemiological and clinical characteristics, and management of COPD patients by phenotype in general population, according to a predefined criterion as well as Global Initiative for Chronic Obstructive Lung Disease (GOLD) classification.
Results
Participants
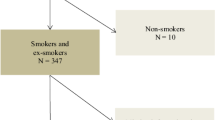
Of the 75,107 participants in the survey, 8355 participants were excluded due to severe diseases or ineligible spirometry, and 57,618 with a FEV1/FVC ≥0.7 were excluded (Fig. 1). Finally, the study detected 9134 spirometry-confirmed COPD patients aged ≥40 years, of whom all were available for classification of predefined phenotype and 7639 for GOLD classification. The mean age was 61.3 years (SD 9.6), 3326 were never-smokers, 246 were ever diagnosed as asthma, and 1132 patients were obesity (Table 1).
Prevalence of COPD by phenotype
The estimated COPD phenotype prevalence among COPD patients according to predefined definition were, NON-AE, 90.3% (95% CI, 89.3–91.3%), AE NON-CB, 2.9% (2.4–3.4%), AE-CB, 2.0% (1.7–2.3%), and ACO, 4.8% (4.2–5.4%), while the pattern according to GOLD was GOLD A, 54.1% (95% CI, 49.3–58.8%), GOLD B 39.5% (34.8–44.1%), GOLD C, 1.7% (1.2–2.1%), and GOLD D, 4.8% (4.1–5.6%) (Fig. 2). All NON-AE patients were in GOLDA through B group, and AE with or without CB were in GOLD C through D group. Of ACO patients, approximately 78% were in GOLD A through B, whereas 22% in GOLD C through D. (Supplementary Table 1). We also did a sensitivity analysis using the lower limit of normal to define COPD and this gave a similar pattern of phenotype prevalence (Supplementary Table 2).
a COPD phenotype prevalence with 95% CI by sex, predefined definition. b COPD phenotype prevalence with 95% CI by geographic locations, predefined definition. c GOLD classification prevalence with 95% CI by sex, GOLD classification. d GOLD classification prevalence with 95% CI by geographic locations, GOLD classification. Error bars represented the standard error of the prevalence and were estimated by Taylor series variances estimation approach. Abbreviations: COPD, chronic obstructive pulmonary disease; GOLD, Global Initiative for Chronic Obstructive Lung Disease; NON-AE, non-exacerbator; AE-CB, exacerbator with chronic bronchitis; AE NON-CB, exacerbator without chronic bronchitis; ACO, asthma-COPD overlap.
Based on ~77.2 million COPD patients aged 40 years or old in China, we project 69.7 million NON-AE patients, 3.8 million with frequent exacerbations, 3.7 million ACO patients, whereas 35.5 million patients with high risk of exacerbation or more syndromes (B through D grades).
NON-AE was more prevalent in men than women (91.0 vs. 88.4%, P < 0.001), whereas ACO (4.2 vs. 6.3%, P < 0.001) was more common in women than men (Fig. 2). Both AE-CB (2.6 vs. 1.4%, P = 0.001) and AE NON-CB (4.1 vs. 1.6%, P < 0.001) were more prevalent in the south than that in the north, whereas the pattern was inverse for ACO (4.0 vs. 5.6%, P = 0.01). In contrast, we did not observe systematically patterns of the GOLD classifications in term of sex and geographic locations. We examined in greater detail the finding of geographic variation with respect to predefined phenotypes; and found that the geographic pattern was associated with regional-level temperature (Supplementary Tables 3 and 4).
Symptom burden and comorbidity of COPD patients by phenotype
Prevalence of risk factors, symptom burden and airflow limitation severity significantly differed by predefined phenotypes and GOLD classifications (Table 1 and Supplementary Table 5). For example, current smoking was more prevalent in NON-AE (48.8%) and AE-CB (44.5%) than that in AE NON-CB (29.2%). Current smoking was more prevalent in GOLD A and B (49.0 and 50.7%) than that in GOLD C and D (31.5 and 34.1%). Among 439 ACO patients, 191 were never-smokers, most of whom were exposure to biomass or occupational pollutants (Supplementary Table 6). A history of severe pulmonary diseases in childhood was most frequent in ACO (12.3%) than that in other phenotypes.
AE-CB had the highest CAT or mMRC scores and the worst lung function as compared with NON-AE. Lung function and symptom burden were worse in patients with ACO as compared with NON-AE, but better than AE regardless of co-existing CB. The total exacerbation events were substantially higher in AE-CB (5.7 events·year−1) and AE NON-CB (4.2 events·year−1), and moderately higher in ACO (1.3 events·year−1) as compared with NON-AE (0.01 events·year−1). Similar systematic patterns in terms of symptom and airway limitation were also observed in GOLD classification.
Overall, 32.1% of COPD patients had comorbidities (Table 2 and Supplementary Table 7), and the common comorbidities among COPD patients were hypertension (21.3%), cerebrovascular disease (5.9%), coronary artery disease (5.7%), diabetes mellitus (5.3%), and osteoporosis (4.4%). AE-CB, AE NON-CB and ACO patients more often had comorbidities or multiple comorbidities as compared with NON-AE (P < 0.001). Similarly, the prevalence of comorbidities was higher in GOLD C and D as compared with GOLD A and B.
Management of COPD patients
Overall, 5.9% of COPD patients ever had pulmonary function performed, 3.4% ever utilized inhaled medications and 2.5% ever had oxygen therapy (Table 3), whereas less than 1% of patients received long-acting or short-acting bronchodilators with more utilization of short-acting bronchodilators (0.9%) than long-acting bronchodilator (0.3%) or multiple therapies (0.4%). By comparison, 10.4% ever received non-inhaled medications, with most commonly antibiotics (5.5%), antitussives (5.3%), traditional Chinese medicines (3.5%), and theophylline (3.1%). Inhaled medication utilization was higher in AE-CB (29.5%), AE NON-CB (23.5%), and ACO (19.4%) as compared with NON-AE (1.4%; P < 0.001). Utilization of non-inhaled medications was also greater in AE-CB and AE NON-CB than in NON-AE or ACO (P < 0.001). The treatment using mono- or multiple corticosteroids-containing inhalers was notably low in ACO patients (2.5%).
Similarly, the utilization rates of inhaled medication were relatively low in GOLD C (26.8%) and D (31.5%), however the proportion in GOLD A and B was greatly decreased (1.1 and 3.1%) (Supplementary Table 8). Additionally, the utilization of corticosteroids in patients who were classified as GOLD C or D and ACO was 10.3%, where the proportion in patients in GOLD A and B was only 1.6% (data no showed).
Discussion
The population-based study provided comprehensive evidence on the phenotypic prevalence, epidemiological and clinical characteristics of COPD patients by phenotype in general population in China. The study estimated that 90% of COPD patients were non-exacerbators, and approximate 5% were frequent exacerbator with high-risk of poor outcomes. Furthermore, the study revealed that the proportions of COPD management with respect to diagnosis, inhaled medications, and risk factors control were substantially low in primary care.
The study displayed the phenotypic pattern of COPD patients by two different classifications: a predefined criterion and the GOLD classification. The GOLD classification is regarded as the best-available management and prediction tool in clinical practice12,13. Nonetheless, some studies noticed that the individual management should be tailed according to the risk and phenotype related to the clinical presentation of COPD14,15. We defined a phenotype algorithm according to the GesEPOC, which was first established in 2012 and adopted by some national guidelines14,16. In the GesEPOC, COPD patients were classified as non-exacerbators, asthma-COPD overlapped, and exacerbators with emphysema or chronic bronchitis. However, the emphysema was difficult to be confirmed in population-based study due to the cost of the chest computed tomography, and therefore AE NON-CB was used as alternative9. The diagnosis of ACO in a given patient included three elements: significant smoking exposure (more than 10 pack-years), irreversible airflow limitation, and asthma. But in our study, the smoking exposure was not an essential element in the ACO patients because the exposure to biomass fuel and occupational pollutants were also reported as main risk factors of COPD in China15,17.
Our results showed that most of the COPD patients in general population of China were in low-risk status with less or no symptoms and mild airway obstruction, consistent with other studies in primary care18,19. Compared with studies using similar definition of exacerbator, the AE prevalence in our study was similar with the PLATINO study in the Latin America (6% in 2009)20, but substantially lower than the POPE cohort in the central and eastern European (30% in 2014–2015)9. The ACO prevalence was similar with that in the USA and European countries (6% in 2012–2013)21, but lower than estimates from Latin America (13% in 2010)20 and Japan (9% in 2017)22. Correspondingly, the proportions of high-risk GOLD categories in our study were comparable to the COPDGene cohort (0.8% in GOLD C; 5% in GOLD D)10, but lower than the ECLIPSE (7 and 15%)23 and POPE (2 and 36%)12 cohorts. Under the similar definition, the discordant pattern may reflect the heterogeneity of population, different epidemiological characteristics of risk factors, diagnosis, and management of patients.
We observed a systematic pattern of the AE and ACO phenotypes by geographic locations, which might be associated with the difference in temperature and humidity between the south and north. It was well-known that the south was hotter and more humid than the north in China. Previous studies linked the excess risk of respiratory viral or bacterial infection to higher temperature and associated humidity with variations in exacerbation frequency14,24. In contrast, cold air exposure is related to bronchial hyperresponsiveness25. Further investigations of potential mechanisms (genetic susceptibility, respiratory infections, and lifestyle patterns) are needed, but the results imply the role of geographic location in the management of COPD patients.
Another important finding was that COPD patients within any phenotype were underdiagnosed or undertreated in China, inconsistent with other studies in western countries26,27. Long-acting bronchodilators alone or in combination were considered as the basis of pharmacological treatment of COPD16,28, however, in our study, less than 5% of patients prescribed inhaled medication and less than 30% of AE phenotype ever used inhaled medications. The underdiagnosis of COPD patients might contribute to the undertreatment. Furthermore, the utilization of short-acting bronchodilators was more common than long-acting bronchodilators, implying the goal of current treatment was preferred for the control of symptoms rather than improvement of quality of life and lung function. In contrast, the prevalence of conventional risk factors associated with exacerbation risk, e.g., current smoking29,30, underweight or obesity27, and indoor or occupational exposure15,17, were substantially high in each phenotype. Additionally, that less than 5% received influenza or pneumococcal vaccines, and less than 3% received rehabilitation or oxygen therapy, underscoring the opportunity for primary care intervention in China.
The ACO phenotype was worth of being independently identified in the management, and guidelines advocated the long-acting bronchodilators combined with ICS-containing medications in ACO treatment16,31. Our results showed the utilization of inhaled bronchodilators and ICS-containing medications among ACO patients in our survey (19 and 5%, respectively) compare unfavorably with other studies, such as Latin American data (49 and 23%, respectively)18. This may be associated with low proportion of ACO patients fell into the GOLD D group (20% of ACO) for which the ICS-containing medications were recommended under the GOLD classifications, but the exact reasons are complicated and still uncertain. Nonetheless, our results showed that the identification of ACO phenotype in management of COPD patients in primary care is of essential not regardless of using GOLD categories or other definitions.
Due to the nature of the study, most of the COPD patients in our study were non-exacerbators, who were underdiagnosed, less symptomatic or asymptomatic, and had mild airway obstruction, indicating the relatively low risk of poor outcomes. Furthermore, the proportion of never-smokers was relatively high (36%). Therefore, the data could not show the real-life treatment of hospital-based COPD patients30,32, but underlined the necessity of management for COPD in primary care, for example, early diagnosis of at-risk patients, smoking cessation, regular pharmacological treatment.
Findings suggested that a fixed ratio (<0.7) might lead to more frequent diagnosis in elderly population and less in younger28, which further affect the estimation of COPD phenotype in the study. Based on the spirometric parameters established for Chinese population, we did a sensitivity analysis using the lower limit of normal, and the proportion of NON-AE slightly increased without statistical significance. Nonetheless, careful analyses are necessary for this issue.
A strength of our study is the large amount of COPD patients from a representative sample enable us to analyze the phenotype prevalence and epidemiological characteristics of COPD patients from general population. Nonetheless, several limitations should be discussed. First, among 8355 excluded participants due to ineligible spirometry, 1084 reported physician-diagnosed COPD or COPD-related symptoms (data not showed), and therefore the cross-sectional study might underestimate the risk of exacerbation in the patients. Our estimates should be considered the minimum values for the high-risk classifications. Second, the inclusion of never-smokers in the ACO phenotype might introduce the misclassification bias in the estimation. Third, in our study, the obese patients accounted for 12% of COPD patients and the obesity state had impact on the measurement and estimation of spirometric parameters (i.e., FEV1, FVC, FEV1/FVC)33. This would affect the diagnosis of COPD and phenotypic characteristics. Final but not the least, the comorbidities and treatment were self-reported but not validated using systematic review of healthcare records.
In conclusions, most of the COPD patients in general population of China were non-exacerbators and approximately 5% were exacerbators. However, early diagnosis and standard treatment were obviously inadequate for each phenotype. A comprehensive strategy for the management of COPD patients based on phenotype in primary care is urgently needed.
Methods
Study design and population
This analysis derived from a previously reported nationally, population-based, cross-sectional study from December 2014 to December 20154. Briefly, the study used a complex, multistage, probability sampling strategy, and included 75,107 Chinese citizens aged 40 years or older who had been living in their current residence for at least 6 months within the year. A total of 66,752 participants had acceptable post-bronchodilator spirometry examinations. The diagnosis of COPD was based on post-bronchodilator spirometry showing a ratio of forced expiratory volume in the first second (FEV1)/forced vital capacity (FVC) <0.7 per the Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018 guidelines28. The participants with an abnormal FEV1/FVC ratio were further examined by chest X-ray to exclude suspicious patients with tuberculosis, bronchiectasis, or tumor. In this study, we included spirometry-confirmed COPD patients with FEV1/FVC <0.7 for this study to reflect the real-world characteristics and management of COPD patients in general population in China.
Measurement and definitions
For each participant, we collected demographic characteristics, respiratory symptoms, risk factors, medical history (i.e., frequency of hospital visit and admission, comorbidities, inhaled or non-inhaled medications, rehabilitation, oxygen therapies, and vaccines), the COPD assessment test (CAT), and the modified Medical Research Council (mMRC) dyspnea scale, through questionnaires. Spirometry was operated by trained technicians using the same brand of spirometer (MasterScreen Pneumo, Jaeger, Germany) according to the criteria of American Thoracic Society. Exacerbations were defined as hospital visits related to acute worsening of respiratory symptoms (increased dyspnea, cough, phlegm, and wheeze) resulting in treatment with antibiotics or oral corticosteroids (moderate exacerbation) or hospital admission (severe exacerbation)29. We divided patients into symptomatic and less symptomatic group based on CAT score (<10 and ≥10), as well as mMRC grade (0–1 and 2–4), respectively.
We defined patients as four phenotypes according to the Spanish COPD guidelines (GesEPOC) and related studies:9,16,31 patients with asthma-COPD overlap (ACO), non-exacerbators (NON-AE), frequent exacerbators with chronic bronchitis (AE-CB) and frequent exacerbators without chronic bronchitis (AE NON-CB). The ACO phenotype was defined as COPD patients who were diagnosed with asthma before 40 years or those who had a positive bronchial dilation test (comprising increases in FEV1 of both ≥ 12% and 200 mL) with asthma or allergy symptoms including wheezing or coughing due to exercise or exposure to cold air or pollutants. Patients who had at least one hospital admissions, two or more moderate exacerbations, or both during the past 12 months were classified as frequent exacerbators (AE). Patients with a maximum of one moderate exacerbation and no hospital admission during the past 12 months were classified as NON-AE. Chronic bronchitis (CB) was identified as sputum production with or without chronic cough more than three months per year during two continuous years. AE Patients with CB were recorded as AE-CB, whereas others were recorded as AE NON-CB. Additionally, we classified patients using the ABCD assessment tool of GOLD 2018 guidelines to describe the characteristics of COPD patients.
The severity of airflow limitation was measured by the absolute FEV1, the predicted FEV1 estimated using reference equations for Chinese population aged 4–80 years old34, and by the ratio of absolute FEV1/the predicted FEV1 (FEV1 % pred.). Also, we estimated the lower limited normal (LLN) of FEV1/FVC to define COPD patient as sensitivity analysis. Occupational exposure was defined as exposure to airborne dust or hazardous chemical gases in the workplace for at least 12 months. Indoor exposure to biomass was defined as household use of biomass fuels (e.g., wood, grass, crop residues, and animal dung) for cooking or heating. Smoking cessation defined as patients who had quitted smoking for at least 12 months. Comorbidities were defined as self-reported solid tumor, coronary heart disease, cerebrovascular disease, diabetes mellitus, hypertension, depression, and osteoporosis.
Statistical analysis
We estimated the phenotypic prevalence with confidential intervals (95% CI) in the overall patients, in subgroups of sex, and geographic locations (north versus south). The phenotypic prevalence was defined as the proportion of patients classified as distinct phenotypes among all COPD patients. We characterized and compared the demographic, conventional risk factors, lung function, symptom burden, comorbidities, and management between/among phenotypes. One-way or multiple comparisons between/among phenotypes were conducted considering the complex sampling. We used analysis of variance (ANOVA) or Kruskal–Wallis test for continuous variables, and Rao-Scott χ² test for categorical variables for comparison. All hypothesis testing was judged at the 0.05 significance level (two-sided). Bonferroni correction for pairwise comparisons was used. All statistical analysis was carried out using SAS version 9.4 and using R software (version 3.5.4).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data that support the findings of this study are included in this published article and its supplementary information files. The datasets are available on request from the corresponding author.
Code availability
Available upon request.
References
GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1736–1788 (2018).
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1789–1858 (2018).
Wang, C. et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study. Lancet 391, 1706–1717 (2018).
Fang, L. W. et al. Chronic obstructive pulmonary disease in China: a nationwide prevalence study. Lancet Respir. Med 6, 421–430 (2018).
Guan, W. J., Ran, P. X. & Zhong, N. S. Prevention and management of COPD in China: successes and major challenges. Lancet Respir. Med 4, 428–430 (2016).
Han, M. K. et al. Chronic obstructive pulmonary disease phenotypes: the future of COPD. Am. J. Respir. Crit. Care Med 182, 598–604 (2010).
Agusti, A. The path to personalised medicine in COPD. Thorax 69, 857–864 (2014).
Agusti, A. & Celli, B. Avoiding confusion in COPD: from risk factors to phenotypes to measures of disease characterization. Eur. Respir. J. 38, 749–751 (2011).
Koblizek, V. et al. Phenotypes of COPD patients with a smoking history in Central and Eastern Europe: the POPE Study. Eur. Respir. J. 49, pii: 1601446 (2017).
Han, M. K. et al. GOLD 2011 disease severity classification in COPDGene: a prospective cohort study. Lancet Respir. Med 1, 43–50 (2013).
Suzuki, M. et al. Clinical features and determinants of COPD exacerbation in the Hokkaido COPD cohort study. Eur. Respir. J. 43, 1289–1297 (2014).
Tudoric, N. et al. GOLD 2017 on the way to a phenotypic approach? Analysis from the Phenotypes of COPD in Central and Eastern Europe (POPE) Cohort. Eur. Respir. J. 49, 1602518 (2017).
Faner, R. et al. Distribution, temporal stability and association with all-cause mortality of the 2017 GOLD groups in the ECLIPSE cohort. Respir. Med 141, 14–19 (2018).
Plaza, V. et al. Consensus on the Asthma-COPD Overlap Syndrome (ACOS) Between the Spanish COPD Guidelines (GesEPOC) and the Spanish Guidelines on the Management of Asthma (GEMA). Arch. Bronconeumol. 53, 443–449 (2017).
Chan, K. H. et al. Solid fuel use and risks of respiratory diseases. A cohort study of 280,000. Am. J. Respir. Crit. Care Med 199, 352–361 (2019).
Miravitlles, M. et al. Spanish Guidelines for Management of Chronic Obstructive Pulmonary Disease (GesEPOC) 2017. Pharmacological treatment of stable phase. Arch. Bronconeumol. 53, 324–335 (2017).
Matteis, S. D. et al. The occupations at increased risk of COPD: analysis of lifetime job-histories in the population-based UK Biobank Cohort. Eur. Respir. J. 54, 1900186 (2019).
Barrecheguren, M. et al. Treatment patterns in COPD patients newly diagnosed in primary care. A population-based study. Respir. Med 111, 47–53 (2016).
McGarvey, L. et al. Characterization of the frequent exacerbator phenotype in COPD patients in a large UK primary care population. Respir. Med 109, 228–237 (2015).
Montes de Oca, M. et al. Frequency of self-reported COPD exacerbation and airflow obstruction in five Latin American cities: the Proyecto Latinoamericano de Investigacion en Obstruccion Pulmonar (PLATINO) study. Chest 136, 71–78 (2009).
Ding, B. & Small, M. Treatment trends in patients with asthma-COPD overlap syndrome in a COPD cohort: findings from a real-world survey. Int J. Chron. Obstruct Pulmon Dis. 12, 1753–1763 (2017).
Inoue, H. et al. Prevalence and characteristics of asthma-COPD overlap syndrome identified by a stepwise approach. Int J. Chron. Obstruct Pulmon Dis. 12, 1803–1810 (2017).
Anderson, G. B., Dominici, F., Wang, Y., McCormack, M. C., Bell, M. L. & Peng, R. D. Heat-related emergency hospitalizations for respiratory diseases in the Medicare population. Am. J. Respir. Crit. Care Med 187, 1098–1103 (2013).
Zanobetti, A., O'Neill, M. S., Gronlund, C. J. & Schwartz, J. D. Summer temperature variability and long-term survival among elderly people with chronic disease. Proc. Natl Acad. Sci. USA 109, 6608–6613 (2012).
Hansel, N. N., McCormack, M. C. & Kim, V. The effects of air pollution and temperature on. COPD COPD 13, 372–379 (2016).
Price, D. et al. Management of COPD in the UK primary-care setting: an analysis of real-life prescribing patterns. Int J. Chron. Obstruct Pulmon Dis. 9, 889–904 (2014).
Make, B., Dutro, M. P., Paulose-Ram, R., Marton, J. P. & Mapel, D. W. Undertreatment of COPD: a retrospective analysis of US managed care and medicare patients. Int J. Chron. Obstruct Pulmon Dis. 7, 1–9 (2012).
Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for Prevention, Diagnosis and Management of COPD (2018 Report). (Accessed 20 May 2018). https://goldcopd.org/archived-reports/2018.
Wedzicha, J. A. & Seemungal, T. A. COPD exacerbations: defining their cause and prevention. Lancet 370, 786–796 (2007).
Jia, G. H. et al. Gender difference on the knowledge, attitude, and practice of COPD diagnosis and treatment: a national, multicenter, cross-sectional survey in China. Int J. Chron. Obstruct Pulmon Dis. 13, 3269–2180 (2018).
Leung, J. M. & Sin, D. D. Asthma-COPD overlap syndrome: pathogenesis, clinical features, and therapeutic targets. BMJ 358, j3772 (2017).
Zeng, Y. et al. Current status of the treatment of COPD in China: a multicenter prospective observational study. Int J. Chron. Obstruct Pulmon Dis. 15, 3227–3237 (2020).
Yamaguchi, K., Omori, H., Tsuji, T. & Aoshiba, K. Classical regression equations of spirometric parameters are not applicable for diagnosing spirometric abnormalities in adipotic adults. World J. Respirol. 8, 1–12 (2018).
Jian, W. et al. Reference values for spirometry in Chinese aged 4-80 years. J. Thorac. Dis. 9, 4538–4549 (2017).
Acknowledgements
This study was supported by the Ministry of Science and Technology of People’s Republic of China (National Key R&D Program of China: 2016YFC1303905, 2016YFC1303900), the Chinese Central Government (key project of public health program, grant no. 2014814).
Author information
Authors and Affiliations
Contributions
L.W.F. and Y.H.C. developed the research protocol, research aim, and supervised the study. H.L.B., G.H.J., S.C., and J.F., contributed to sample collection, preparation, and data analysis. H.L.B. and G.H.J. contributed equally to the writing of the paper. L.W.F., Y.H.C., and J.L.C. revised the manuscript. All authors had intellectual input into the concepts explored, critically reviewed each draft of the full manuscript, and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study protocol was approved by the Ethical Review Committee of the National Center for Chronic and Non-communicable Disease Control and Prevention, China CDC. Written informed consent was obtained from all study participants (NO. 201410).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors are first authors: Heling Bao, Guohua Jia.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bao, H., Jia, G., Cong, S. et al. Phenotype and management of chronic obstructive pulmonary disease patients in general population in China: a nationally cross-sectional study. npj Prim. Care Respir. Med. 31, 32 (2021). https://doi.org/10.1038/s41533-021-00243-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41533-021-00243-x
- Springer Nature Limited