Abstract
This study aimed to develop and validate a deep learning radiomics nomogram (DLRN) for the preoperative evaluation of axillary lymph node (ALN) metastasis status in patients with a newly diagnosed unifocal breast cancer. A total of 883 eligible patients with breast cancer who underwent preoperative breast and axillary ultrasound were retrospectively enrolled between April 1, 2016, and June 30, 2022. The training cohort comprised 621 patients from Hospital I; the external validation cohorts comprised 112, 87, and 63 patients from Hospitals II, III, and IV, respectively. A DLR signature was created based on the deep learning and handcrafted features, and the DLRN was then developed based on the signature and four independent clinical parameters. The DLRN exhibited good performance, yielding areas under the receiver operating characteristic curve (AUC) of 0.914, 0.929, and 0.952 in the three external validation cohorts, respectively. Decision curve and calibration curve analyses demonstrated the favorable clinical value and calibration of the nomogram. In addition, the DLRN outperformed five experienced radiologists in all cohorts. This has the potential to guide appropriate management of the axilla in patients with breast cancer, including avoiding overtreatment.
Similar content being viewed by others
Introduction
Breast cancer ranks as the second leading cause of cancer-related mortality in women and has a high metastasis rate of 20–30%1. As the most frequent site of metastasis, the status of axillary lymph nodes (ALNs) is pivotal for pathological staging, prognosis, and treatment guidance including determining neoadjuvant or adjuvant therapy and surgical planning for patients2. Currently, ALN dissection and sentinel lymph node (SLN) biopsy are the standard methods for determining the metastatic status of ALNs3,4,5. Nevertheless, both methods are invasive and may lead to postoperative morbidity, such as arm numbness and upper limb edema6,7. In addition, 70–80% of patients who undergo SLN biopsy exhibit negative SLNs, indicating the high probability of overtreatment with unnecessary SLN biopsy8,9. Therefore, an accurate, non-invasive approach to predict ALN metastasis would be part of optimal treatment planning in patients with newly diagnosed breast cancer.
Ultrasound represents a widely used imaging tool for preoperative assessment of ALNs in patients with breast cancer. On axillary ultrasound, ALNs with features such as longest/shortest axis ratio < 2, cortical thickening, and loss of fatty hilum in the node are suspected to be malignant10. Previous studies suggested that integration of suspicious features on axillary ultrasound and other clinical factors had the potential to predict ALN metastasis11. However, the performance is unsatisfactory, with a limited area under the receiver operating characteristic (ROC) curve (AUC) of 0.585–0.71912. Additionally, ALNs with suspicious morphology often undergo ultrasound-guided needle biopsy to assist in preoperative planning. However, nearly 35% of metastatic ALNs do not show any suspicious features13, introducing a limitation in the assessment of ALN status using axillary ultrasound.
Several studies have developed noninvasive methods to determine the metastatic status of ALNs, including several clinical nomograms14,15,16. However, few of these studies have taken into account information derived from preoperative imaging of the lesion, and some have incorporated factors that can only be obtained postoperatively. Recently, radiomics has been an effective technology in clinic by converting medical image data into high-throughput imaging features17. The latest studies have revealed improvements in assessing ALN status using conventional radiomics (CR) analysis of primary tumors with mammography, ultrasound, and magnetic resonance imaging18,19,20. However, the limitations of handcrafted features in CR lie in manual labeling and their inability to conform to a specific task21. In contrast, deep learning radiomics (DLR)22 is an innovative method that can learn end-to-end and automatically discover multiple levels of representations for the specific prediction tasks. The DLR process includes feeding raw machine data, such as medical images, and allowing them to learn feature representations, quantify information from images, and discover vectors for prediction tasks using multiple layers of features23. With technological advancements, the application of DLR in breast cancer imaging has rapidly increased, including prediction of ALN status2,24,25,26. However, the clinical utility of these studies remains uncertain as they contained limited sample sizes, lacked external validation, and neglected important clinical information27, which are crucial for classification accuracy28. Therefore, we planned to integrate DLR features extracted from breast ultrasound and preoperative clinical parameters to improve the model performance with a large sample size.
In this study, we developed a deep learning radiomics nomogram (DLRN) based on breast ultrasound, which is a convenient, radiation-free, and favorably repeatable examination for breast cancer29,30, to access ALN status preoperatively. The predictive performance of the DLRN was validated using three external validation cohorts (EVCs). Ultimately, the results indicated that DLRN could detect the metastatic risk of ALNs better than the clinical model and radiologists, enable individualized surgical approaches for the axilla, and minimize overtreatment.
Results
Baseline characteristics
Table 1 summarizes the clinical parameters of the 883 patients with breast cancer in the four hospitals. According to the results of SLN biopsy or ALN dissection, the ALN metastatic rates were 34.6%, 40.2%, 40.2%, and 50.8% in TC, EVC1, EVC2, and EVC3, respectively. The median BMI were 26.3, 25.7, 25.2, and 25.7 in the four cohorts, respectively. The Kappa values of the BI-RADS category were 0.957 and 1 for inter- and intra-observer agreements, respectively (both P < 0.001). For US-ALN, the Kappa values were 0.936 and 1, respectively (both P < 0.001). The metastatic status of ALN showed a significant difference between the TC and EVC3 (34.6% vs. 50.8%, P < 0.001). Other detailed differences in specific clinical parameters are described in Supplementary Notes.
DLR signature construction and validation
In total, 544 handcrafted and 2048 deep learning features were extracted for each patient. Of these, 519 and 1847 features with high reproducibility and stability (ICC > 0.80), respectively, were subsequently combined and analyzed using LASSO logistic regression. Finally, 4 handcrafted and 45 deep learning features with nonzero coefficients in LASSO were selected to derive the DLR signature (Supplementary Fig. 1). A detailed description of the selected features is provided in Supplementary Table 1. The signature achieved the AUCs of 0.886 (95% CI, 0.815–0.958), 0.854 (95% CI, 0.778–0.931), and 0.917 (95% CI, 0.854–0.980) in the three EVCs, respectively. The signature was significantly higher in the metastasis group than in the non-metastasis group in all cohorts (P < 0.001) (Supplementary Fig. 2). The accuracy, sensitivity, specificity, PPV, and NPV of the signature are presented in Table 2.
DLR nomogram construction and performance evaluation
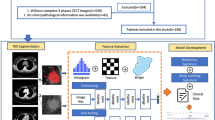
The result of univariate logistic regression analysis of the clinical parameters is presented in Supplementary Table 2. The clinical model was developed based on age, BI-RADS category, nuclear grade, and US-ALN by multivariate logistic regression (Table 3). These four independent clinical parameters were significantly correlated with ALN metastasis. The DLRN combined with the DLR signature and independent clinical parameters is shown in Fig. 1. The optimal cutoff of the ALN metastatic rate for DLRN was 0.407, based on the TC.
The DLRN demonstrated significantly better predictive performance than the corresponding clinical model, with AUCs of 0.914 (95% CI, 0.858–0.971), 0.929 (95% CI, 0.877–0.980), and 0.952 (95% CI, 0.906–0.997) in the EVCs (DeLong P < 0.001). The AUCs of the DLRN in different surrogate molecular subtypes are presented in Supplementary Table 3. The accuracy, sensitivity, specificity, PPV, and NPV of the clinical model, DLR signature, and DLRN are presented in Table 2. The confusion matrices of the DLRN across all cohorts are shown in Supplementary Fig. 3, and the ROC curves demonstrating the comparative results of the AUCs are displayed in Fig. 2a–c.
a–c Receiver operating characteristic area under the curves for the proposed clinical model and DLRN in EVC1, EVC2, and EVC3, respectively. d–f Decision curves of the clinical model and DLRN in EVC1, EVC2, and EVC3, respectively. g–i Calibration curves of DLRN in EVC1, EVC2, and EVC3, respectively. EVC, external validation cohort; CLI, clinical model; DLRN, deep learning radiomics nomogram. Note: In Fig. 2d–f, the purple and blue lines represent the DLRN and clinical model, respectively. The orange line represents the assumption that all cases underwent ALN dissection or SLN biopsy. The green line represents the assumption that no cases underwent ALN dissection or SLN biopsy. The decision curve reveals that the DLRN exhibits better performance than the clinical model over a wide range of threshold probabilities.
In all the EVCs, 25.3% (38/150) of the non-metastatic ALNs had at least one suspicious feature on ultrasound imaging, and 22.7% (34/150) were misdiagnosed as malignant ALNs by experienced radiologists. Nonetheless, 79.4% (27/34) of them were correctly classified as negative ALNs by the DLRN. A detailed comparison of performance of the DLRN and radiologists is presented in Table 2. The result of ALN status assessment by each radiologist is summarized in Supplementary Table 4, and the typical cases evaluated by human experts and the DLRN are shown in Fig. 3.
a Breast ultrasound and b axillary ultrasound of a patient with positive ALN metastasis. The patient was wrongly considered as non-metastatic by the radiologists because there were no suspicious ALNs on axillary ultrasound. In addition, benign characteristics of the primary lesion, including a relatively oval shape, smooth margin, homogeneous hypoechoic matrix, and parallel orientation, added the diagnostic difficulty. However, this case was accurately identified as positive ALN metastasis by the DLRN, with a metastatic possibility of 87%. c Breast ultrasound and d axillary ultrasound of a patient with negative ALN metastasis. The patient was wrongly considered as metastatic type by the radiologists based on the malignant features of the ALN (cortical thickening > 3 mm and microcalcification) and the primary lesion (irregular shape, indistinct margin, heterogeneous echo pattern, and vertical orientation). However, the case was correctly evaluated by the DLRN, with a metastatic possibility of 27%. ALN axillary lymph node, DLRN deep learning radiomics nomogram.
The IDI, NRI, and C-index indicated superior classification accuracy of the DLRN compared with the clinical model and DLR signature (Supplementary Table 5). Decision curve analyses (Fig. 2d–f) demonstrated that the DLRN provided a higher net benefit than the clinical model over a wide range of threshold probability. The calibration curves verified that the predicted ALN status by the DLRN was in good agreement with the actual status (Fig. 2g–i). Additionally, a total of 37 patients were included for reproducibility evaluation of the DLRN. The clinical parameters of the 37 patients are presented in Supplementary Table 6. The inter-observer ICC among the three doctors was 0.82, indicating good reproducibility of the DLRN according to Cicchetti’s guideline.
Discussion
Currently, ALN dissection and SLN biopsy are the standard methods for ALN staging. However, both operations carry varying degrees of postoperative complications and morbidities. Recent researches have concentrated on minimizing unnecessary axillary procedures and avoiding overtreatment for breast cancer. The American Society of Clinical Oncology (ASCO)31 has recommended that SLN biopsy can be omitted for clinically node-negative women aged ≥70 with early-stage invasive breast cancer, that is HER2-negative and hormone receptor-positive. The Sentinel Node vs Observation After Axillary Ultra-Sound (SOUND) trial32 concluded that SLN biopsy can be safely spared in patients with small breast cancer (diameter≤2 cm) and a negative result on axillary ultrasonography. Additionally, several ongoing clinical trials are exploring the possibility of SLN biopsy omission for early breast cancer patients receiving neoadjuvant systemic therapy33 or breast-conserving surgery34. In our study, we successfully developed a DLRN to assess ALN status preoperatively for breast cancer. The DLRN exhibited satisfactory performance in all EVCs, with AUCs of 0.914, 0.929, and 0.952 and accuracies of 88%, 87%, and 89% in EVC1, EVC2, and EVC3, respectively. This represents a promising approach to predict ALN status and avoid unnecessary axillary treatment.
Among the three EVCs, 22.7% (34/150) of the non-metastatic ALNs were misdiagnosed by the five experienced radiologists. However, 79.4% (27/34) of them were correctly classified by the DLRN. On the other hand, 42.0% (47/112) of the metastatic ALNs showed no suspicious features on axillary ultrasound, which is in accordance with the results of another study13. Nonetheless, 72.3% (34/47) of the cases were successfully detected by the DLRN. In addition, the AUC of the DLRN was significantly higher than that of the experienced radiologists (0.665–0.703), consistent with the AUCs of radiologists in other studies (0.585–0.735)10,12,26. The false-negative rate of DLRN was also comparable to that of SLN biopsy, ranging from 7.8 to 27.3%35. Therefore, for patients with a low risk of ALN metastasis by the nomogram, an observational strategy could be recommended instead of invasive axillary treatment. For patients with a high risk of ALN metastasis, axillary operation might be required during management.
Previous studies reported various clinical models for predicting ALN metastasis. Isaac et al. 36 developed a promising multiparametric score to assess the status of nonsentinel lymph nodes in clinically node-negative breast cancer with positive SLNs after systemic therapy. In addition, a representative Memorial Sloan Kettering Cancer Center nomogram14 was developed and commonly used in different populations. Nevertheless, certain variables in this nomogram, such as histological tumor size and lymphovascular invasion, could only be obtained postoperatively, potentially impeding its clinical applicability. In this study, all the factors included in the DLRN can be achieved preoperatively, and the predictive performance was satisfactory when validated in three external hospitals.
Various scholars have explored the additional value of DLR in predicting ALN metastasis. Ding et al. 37 developed a deep learning model based on core needle biopsy specimen to identify ALN status. However, the model performance was expected to be further enhanced (AUC = 0.725) when validated externally. Moreover, biopsy specimens were incapable of capturing the heterogeneity of the entire tumor, and the acquisition of specimens was largely operator-dependent. Consequently, an evaluation of model reproducibility is warranted. Zheng et al. 26 developed a DLR model based on ultrasound and shear wave elastography for breast cancer. The DLR model could predict ALN status (N0 vs. N+[≥1]) and discriminate metastatic burden (N+[1, 2] vs. N+[≥3]) of ALNs with favorable performance. However, the model faced limitation in clinical application due to the nonroutine use of elastography. Compared with the studies, our DLRN was constructed based on a large sample size and with good reproducibility verified by a prospective trial. In addition, to relieve overfitting38 – a common pitfall that may constrain the clinical utility of deep learning models, we initially incorporated handcrafted features to complement DLR features and then utilized LASSO to reduce feature dimensionality. This approach could effectively minimize the risk of overfitting caused by excessive features. The stable and good performance of the DLRN in three external hospitals demonstrated that model overfitting had been mitigated in this study.
In this study, the DLR signature comprised 4 CR and 45 DLR features, which were significantly correlated with ALN status. The four key CR features were local binary pattern (LBP) features, characterized as straightforward, resilient, and efficient texture descriptors39. Some studies have reported that LBP features could promote faster diagnosis of breast malignancies imaged by shear wave elastography40 and the classification of various types of breast lesions based on optical coherence microscopy41. According to these findings, LBP features may reflect different patterns of heterogeneity of breast masses, which may be involved in the occurrence of ALN metastasis.
In the clinical model, young age, high BI-RADS category, high nuclear grade, and positive US-ALN were associated with increased risk of ALN metastasis. The relationship between these variables and ALN status has been confirmed in earlier studies10,14,42. However, these correlations were not consistent across all the studies, and the results were not stable for the EVCs in our study. For example, multivariate analysis in the TC identified age as an independent predictor of ALN status (P = 0.002). However, the correlation was not robust in the EVCs (P = 0.860 for EVC1, 0.737 for EVC2, and 0.287 for EVC3). On the other hand, some studies have identified tumor size, tumor classification, Ki-67, ER, and PR status as independent predictive factors of metastatic ALNs in breast cancer14,43. However, these findings were not observed in the present study. The reason for these inconsistent results may be that clinical parameters only reflect limited aspects of the lesions. Therefore, we integrated the DLR signature with clinical parameters to construct the DLRN and achieved far better predictive performance than the clinical model (P < 0.001) in all cohorts.
Our study still has some limitations. First, inherent variations and shortages were inevitable. For instance, the quality of ultrasonography varied because the operations were performed by different radiologists. Second, the two-dimensional image cannot represent the entire tumor and the information in three-dimensional lesions might be missed. Third, our study exclusively included patients with unifocal breast lesions because it was difficult to identify the lesion responsible for ALN metastasis among multifocal and multicentric lesions. Non-mass-type lesions were also excluded in our study due to the difficulty of ROI segmentation. Fourth, although various researchers have identified the association between different molecular subtypes and ALN status, our study encountered limitations in constructing models for each subtype individually, given the restricted sample size for each subtype. Therefore, further improvements must be made with a more comprehensive analysis in the future.
In summary, we established a deep learning radiomics nomogram to preoperatively evaluate axillary lymph node status in patients with unifocal breast cancer. The nomogram outperformed both the clinical model and radiologists. Therefore, with favorable specificity and sensitivity, this model can offer a potential non-invasive approach to identify lymph node metastasis and guide clinical decision making.
Methods
Patients
This study was approved by the institutional review board of Nanjing Drum Tower Hospital (approval no. 202214201) and compliant with the ethics standards of the regulations of the Declaration of Helsinki. The requirement for informed consent was waived owing to the retrospective nature in this study.
The inclusion criteria were listed as follows: (a) women with histologically diagnosed unifocal breast cancer, (b) patients with confirmed ALN status by ALN dissection/SLN biopsy, and (c) patients received ultrasound examination within 1 week before surgery. The exclusion criteria were as follows: (a) patients received neoadjuvant radiotherapy, chemotherapy, or other therapies preoperatively, (b) patients with ultrasound-invisible or non-mass-type lesions, (c) patients with multifocal lesions or insufficient image quality, (d) patients with metastatic breast cancer, and (e) patients with incomplete clinical or histopathological information. Noteworthily, multifocal lesions were excluded due to the difficulty of distinguishing the responsible lesion which caused metastasis from various masses.
In total, 883 patients with histologically confirmed primary breast cancer from four hospitals were included in this study. A flowchart of the patient recruitment process is shown in Fig. 4. Finally, 621 patients with breast cancer from Hospital I (Nanjing Drum Tower Hospital) between April 1, 2016, and June 30, 2022, were reviewed and identified as the training cohort (TC). From December 30, 2017, to November 1, 2021, 112 patients from Hospital II (Jinling Hospital) were recruited as EVC1. From December 1, 2019, to June 30, 2022, 87 patients from Hospital III (Jiangbei Hospital) and 63 patients from Hospital IV (Taizhou Hospital) were enrolled as two EVCs (EVC2 and EVC3).
Clinical parameters
The preoperative clinical parameters collected for analysis included clinicopathological characteristics and ultrasound findings of the breast and axilla. Clinicopathological characteristics included age, body mass index (BMI), estrogen receptor (ER) status, progesterone receptor (PR) status, human epidermal growth factor receptor 2 (HER2) expression, Ki-67 expression, nuclear grade, tumor classification, and surrogate subtype44. The pathological characteristics were obtained from needle biopsy, which is the standard preoperative procedure for breast cancer. According to the 2017 St Gallen International Expert Consensus45, ER-positive status was identified when ER-positive rate ≥1%, and PR-positive status was identified when PR-positive rate ≥1%. HER2 positivity was identified by an immunohistochemical score of 3+ or a score of 2+ with gene amplification. Cases failing to meet the criteria were classified as HER2-negative. In terms of Ki-67, cases with more than 14% positive nuclei were categorized as high Ki-67 expression, whereas others were classified as low Ki-67 expression.
The ultrasound findings of the breast and axilla included tumor location, ultrasound size of the breast lesion, Breast Imaging Reporting and Data System (BI-RADS) category, and ALN status reported by axillary ultrasound (US-ALN). On axillary ultrasound, suspicious metastatic ALNs were identified if any of the following features were present: (1) longest/shortest axis ratio < 2, (2) cortical thickening > 3 mm, or (3) loss of the fatty hilum in the node. Non-suspicious ALNs were identified when no suspicious features were found. The BI-RADS category and US-ALN were evaluated by two experienced radiologists (B.J. and X.J., with 8 and 9 years of breast US experience, respectively) blinded to pathological results. The inter-/intra-observer agreement of BI-RADS category and US-ALN was evaluated using the Kappa test (detailed in Supplementary Method 1). Landis and Koch’s evaluation46 was utilized to interpret the Kappa value.
Ultrasonography examination and image preprocessing
According to the Guidelines of the American Institute of Ultrasound in Medicine practice47, ten radiologists with at least 5 years of breast ultrasound experience from the four hospitals performed preoperative breast and axillary ultrasound. Patients were kept in the supine position, and the field of view was set to contain the pectoralis muscle at the deepest aspect of breast ultrasound. The detailed equipment for the ultrasound examination is listed in Supplementary Table 7, and the procedures of ultrasound examination are detailed in Supplementary Method 2. For each patient, one single image of the target breast mass was selected at the maximum diameter plane for further analysis.
To compare the performance of ALN status assessment between radiologists and the model, five radiologists with more than 10 years of experience evaluated the ALN status according to breast and axillary ultrasound examinations. Every radiologist assessed the ALN status of all patients independently and was blinded to histopathological status. The consensus or prevailing viewpoint of the five radiologists served as the result of human experts.
The region of interest (ROI) of the primary breast lesion for each ultrasound image was segmented by reader 1 (W.T., with over 15 years of breast US interpretation experience) using the ImageJ software (http://imagej.net). One month later, 60 random patients were selected and delineated again by readers 1 and 2 (H.Y., with 8 years of breast US interpretation experience). The inter- and intra-observer reproducibility of tumor segmentation and DLR/CR feature extraction were analyzed using breast ultrasound in 60 randomly selected patients for ROI-based feature extraction in a blinded manner by the two readers. An inter-/intra-class correlation coefficient (ICC) of the features > 0.80 indicates good agreement with the tumor segmentation and feature extraction, according to Cicchetti’s guidelines48. Based on the ROI of each lesion, the top, bottom, left, and right boundary points were automatically generated to create the bounding box. The rectangular bounding box was then cropped from the original image, resized to 224 × 224 pixels, normalized, and fed into the convolutional neural network as an input layer.
Deep learning radiomics (DLR) feature extraction and signature construction
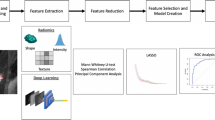
A flowchart of the study is shown in Fig. 5. Handcrafted features including textural and BI-RADS features were extracted in MATLAB 2021b using the Breast Ultrasound Analysis Toolbox49. Deep learning features were extracted using ResNet5050 (Supplementary Fig. 4) and the detailed procedure was presented in Supplementary Method 3. In brief, the fully connected layer and softmax layer of ResNet50 were removed, and the output values of the nodes in last layer were identified as the deep learning features. Subsequently, the handcrafted and deep learning features were combined. The least absolute shrinkage and selection operator (LASSO) logistic regression algorithm51 was used to select the key features related to ALN status and compile the DLR signature.
DLR nomogram construction
The clinical parameters were integrated into the DLR model to improve predictive performance. In the TC, univariate logistic regression analysis was used to identify candidate factors among the clinical parameters. Furthermore, multivariate logistic regression was employed to select independent clinical parameters and construct a clinical model. We then integrated the DLR signature and independent clinical parameters using multivariate logistic regression to construct a combination model. The combination model was finally converted into an individualized DLRN.
Model performance
Integrated discrimination improvement (IDI), net reclassification improvement (NRI), and C-index were used to demonstrate the prediction ability. ROC curve analysis and the AUC with a 95% confidence interval (CI) were used for interpretation. AUCs were compared using the DeLong test52. The AUCs of the DLRN in predicting ALN metastasis for different surrogate molecular subtypes were also calculated. The optimal cutoff value of the DLRN was determined using the Youden index of the TC. Boxplots and confusion matrices were used to visualize the performance of the DLR signature and DLRN, respectively. The accuracy, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) with 95% CIs were also evaluated. Decision curve and calibration curve analyses were performed to assess the clinical value and calibration of each model, respectively.
Reproducibility of the DLRN was assessed in 37 patients who were prospectively enrolled from Hospital I between January 8, 2024, and January 19, 2024. The workflows of the patient recruitment and reproducibility evaluation are shown in Supplementary Fig. 5. Three doctors with varying experience in breast ultrasound (3, 7, and 12 years, respectively) independently performed predictive procedures, including imaging acquisition, ROI segmentation, feature extraction, DLR signature acquisition, clinical data input, and probability calculation for the same lesion in each patient. The inter-observer ICC was calculated to assess model reproducibility.
Statistical analysis
Two-tailed P < 0.05 denoted a significant difference. All statistical analyses were conducted in R 4.1.2, Python 3.6, and MATLAB R2021b. Differences in continuous data were compared using the independent sample t-test or Mann–Whitney exact U test. Categorical variables were compared using the chi-squared or Fisher’s exact test. The code of predicting procedure can be available in https://github.com/ZouLiwen-1999/ALN_metastasis_Pred.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Access to the original images and clinical data in this study are available from the corresponding author on reasonable request.
Code availability
The code for prediction of lymph node metastasis is publicly available in https://github.com/ZouLiwen-1999/ALN_metastasis_Pred.
References
Liang, Y. et al. Metastatic heterogeneity of breast cancer: Molecular mechanism and potential therapeutic targets. Semin. Cancer Biol. 60, 14–27 (2020).
Zhou, L. Q. et al. Lymph node metastasis prediction from primary breast cancer US images using deep learning. Radiology 294, 19–28 (2020).
Veronesi, U. et al. Sentinel-lymph-node biopsy as a staging procedure in breast cancer: update of a randomised controlled study. Lancet Oncol. 7, 983–990 (2006).
Krag, D. N. et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: Overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 11, 927–933 (2010).
Lyman, G. H. et al. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early-stage breast cancer. J. Clin. Oncol. 23, 7703–7720 (2005).
Boughey, J. C. et al. Cost modeling of preoperative axillary ultrasound and fine-needle aspiration to guide surgery for invasive breast cancer. Ann. Surg. Oncol. 17, 953–958 (2010).
Langer, I. et al. Morbidity of sentinel lymph node biopsy (SLN) alone versus SLN and completion axillary lymph node dissection after breast cancer surgery: A prospective swiss multicenter study on 659 patients. Ann. Surg. 245, 452–461 (2007).
Asadi, M. & Krag, D. Internal mammary sentinel lymph node biopsy in clinical practice. Int. J. Surg. 36, 332–334 (2016).
Gentilini, O. & Veronesi, U. Abandoning sentinel lymph node biopsy in early breast cancer? A new trial in progress at the European Institute of Oncology of Milan (SOUND: Sentinel node vs Observation after axillary UltraSouND). Breast 21, 678–681 (2012).
Ecanow, J. S., Abe, H., Newstead, G. M., Ecanow, D. B. & Jeske, J. M. Axillary staging of breast cancer: What the radiologist should know. Radiographics 33, 1589–1612 (2013).
Kim, G. R. et al. Preoperative axillary US in early-stage breast cancer: Potential to prevent unnecessary axillary lymph node dissection. Radiology 288, 55–63 (2018).
Youk, J. H., Son, E. J., Kim, J. A. & Gweon, H. M. Pre-operative evaluation of axillary lymph node status in patients with suspected breast cancer using Shear Wave Elastography. Ultrasound Med Biol. 43, 1581–1586 (2017).
Jiang, M. et al. Radiomics model based on shear-wave elastography in the assessment of axillary lymph node status in early-stage breast cancer. Eur. Radiol. 32, 2313–2325 (2022).
Bevilacqua, J. L. B. et al. Doctor, what are my chances of having a positive sentinel node? A validated nomogram for risk estimation. J. Clin. Oncol. 25, 3670–3679 (2007).
Yeniay, L. et al. A new and simple predictive formula for non-sentinel lymph node metastasis in breast cancer patients with positive sentinel lymph nodes, and validation of 3 different nomograms in Turkish breast cancer patients. Breast Care 7, 397–402 (2012).
Coombs, N., Chen, W., Taylor, R. & Boyages, J. A decision tool for predicting sentinel node accuracy from breast tumor size and grade. Breast J. 13, 593–598 (2007).
Yang, J. et al. Preoperative prediction of axillary lymph node metastasis in breast cancer using mammography-based radiomics method. Sci. Rep. 9, 4429 (2019).
Han, L. et al. Radiomic nomogram for prediction of axillary lymph node metastasis in breast cancer. Eur. Radiol. 29, 3820–3829 (2019).
Yu, F. H. et al. Ultrasound-based radiomics nomogram: A potential biomarker to predict axillary lymph node metastasis in early-stage invasive breast cancer. Eur. J. Radiol. 119, 108658 (2019).
Chai, R. et al. Differentiating axillary lymph node metastasis in invasive breast cancer patients: A comparison of radiomic signatures from multiparametric breast MR sequences. J. Magn. Reson. Imaging 50, 1125–1132 (2019).
Lou, B. et al. An image-based deep learning framework for individualising radiotherapy dose: a retrospective analysis of outcome prediction. Lancet Digit Health 1, e136–e147 (2019).
Wang, K. et al. Deep learning radiomics of shear wave elastography significantly improved diagnostic performance for assessing liver fibrosis in chronic hepatitis B: A prospective multicentre study. Gut 68, 729–741 (2019).
LeCun, Y., Bengio, Y. & Hinton, G. Deep learning. Nature 521, 436–444 (2015).
Lee, Y. W., Huang, C. S., Shih, C. C. & Chang, R. F. Axillary lymph node metastasis status prediction of early-stage breast cancer using convolutional neural networks. Comput Biol. Med. 130, 104206 (2021).
Guo, X. et al. Deep learning radiomics of ultrasonography: Identifying the risk of axillary non-sentinel lymph node involvement in primary breast cancer. EBioMedicine 60, 103018 (2020).
Zheng, X. et al. Deep learning radiomics can predict axillary lymph node status in early-stage breast cancer. Nat. Commun. 11, 1236 (2020).
Xie, Y., Zhang, J., Xia, Y., Fulham, M. & Zhang, Y. Fusing texture, shape and deep model-learned information at decision level for automated classification of lung nodules on chest CT. Inf. Fusion 42, 102–110 (2018).
Lou, B. et al. An image-based deep learning framework for individualizing radiotherapy dose. Lancet Digit Health 1, 136 (2019).
Tucker, N. S. et al. Axillary ultrasound accurately excludes clinically significant lymph node disease in patients with early stage breast cancer. Ann. Surg. 264, 1098–1102 (2016).
Cools‐Lartigue, J. & Meterissian, S. Accuracy of axillary ultrasound in the diagnosis of nodal metastasis in invasive breast cancer: A review. World J. Surg. 36, 46–54 (2012).
Brackstone, M. et al. Management of the Axilla in early-stage breast cancer: Ontario Health (Cancer Care Ontario) and ASCO Guideline. J. Clin. Oncol. 39, 3056–3082 (2021).
Gentilini, O. D. et al. Sentinel lymph node biopsy vs no axillary surgery in patients with small breast cancer and negative results on ultrasonography of axillary lymph nodes: The SOUND randomized clinical trial. JAMA Oncol. 9, 1557–1564 (2023).
Reimer, T., Glass, A., Botteri, E., Loibl, S. & D. Gentilini, O. Avoiding axillary sentinel lymph node biopsy after neoadjuvant systemic therapy in breast cancer: Rationale for the prospective, multicentric EUBREAST-01 trial. Cancers 12, 3698 (2020).
Jung, J. G. et al. No axillary surgical treatment for lymph node-negative patients after ultra-sonography [NAUTILUS]: protocol of a prospective randomized clinical trial. BMC Cancer 22, 189 (2022).
Pesek, S., Ashikaga, T., Krag, L. E. & Krag, D. The false-negative rate of sentinel node biopsy in patients with breast cancer: A meta-analysis. World J. Surg. 36, 2239–2251 (2012).
Cebrecos, I. et al. Nonsentinel axillary lymph node status in clinically node-negative early breast cancer after primary systemic therapy and positive sentinel lymph node: a predictive model proposal. Ann. Surg. Oncol. 30, 5707–5708 (2023).
Ding, Y. et al. Multi-center study on predicting breast cancer lymph node status from core needle biopsy specimens using multi-modal and multi-instance deep learning. NPJ Breast Cancer 9, 58 (2023).
Liu, F. et al. Deep learning radiomics based on contrast-enhanced ultrasound might optimize curative treatments for very-early or early-stage hepatocellular carcinoma patients. Liver Cancer 9, 397–413 (2020).
Gertych, A. et al. Machine learning approaches to analyze histological images of tissues from radical prostatectomies. Comput. Med. Imaging Graph. 46, 197–208 (2015).
Acharya, U. R. et al. Shear wave elastography for characterization of breast lesions: Shearlet transform and local binary pattern histogram techniques. Comput Biol. Med. 91, 13–20 (2017).
Wan, S. et al. Integrated local binary pattern texture features for classification of breast tissue imaged by optical coherence microscopy. Med Image Anal. 38, 104–116 (2017).
Berg, W. A. et al. Shear-wave elastography improves the specificity of breast US: The BE1 multinational study of 939 masses. Radiology 262, 435–449 (2012).
Hu, X. et al. Preoperative nomogram for predicting sentinel lymph node metastasis risk in breast cancer: a potential application on omitting sentinel lymph node biopsy. Front Oncol. 11, 665240 (2021).
Goldhirsch, A. et al. Strategies for subtypes-dealing with the diversity of breast cancer: Highlights of the St Gallen international expert consensus on the primary therapy of early breast cancer 2011. Ann. Oncol. 22, 1736–1747 (2011).
Curigliano, G. et al. De-escalating and escalating treatments for early-stage breast cancer: The St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann. Oncol. 28, 1700–1712 (2017).
Landis, J. R. & Koch, G. G. The measurement of observer agreement for categorical data. Biometrics 33, 159 (1977).
Whitman, G. et al. American Institute of Ultrasound in Medicine & American Society of Breast Surgeons. AIUM practice guideline for the performance of a breast ultrasound examination. J. Ultrasound Med. 28, 105–109 (2009).
Cicchetti, D. V. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol. Assess. 6, 284–290 (1994).
Rodríguez-Cristerna, A., Gómez-Flores, W. & de Albuquerque-Pereira, W. C. BUSAT: A MATLAB toolbox for breast ultrasound image analysis. in Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) 10267 LNCS (2017).
He, K., Zhang, X., Ren, S. & Sun, J. Deep residual learning for image recognition. in Proceedings of the IEEE Computer Society Conference on Computer Vision and Pattern Recognition 770–778 (2016).
Sauerbrei, W., Royston, P. & Binder, H. Selection of important variables and determination of functional form for continuous predictors in multivariable model building. Stat. Med. 26, 5512–5528 (2007).
DeLong, E. R., DeLong, D. M. & Clarke-Pearson, D. L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 44, 837 (1988).
Acknowledgements
This study was funded by the Nanjing Medical Science and Technique Development Foundation (QRX17011), the Ministry of Science and Technology of China (2020YFA0713800) and Clinical Trials from the Affiliated Drum Tower Hospital, Medical School of Nanjing University (2022-LCYJ-MS-24, 2022-YXZX-YX-08).
Author information
Authors and Affiliations
Contributions
Conceptualization, Wentao Kong and Luying Gui; Methodology, Peng Wan and Nan Xu; Software, Yu Zhang and Liwen Zou; Validation, Xiaojing Zhang and Yuhong He; Writing, Han Liu and Shenhai Yun; Han Liu, Liwen Zou, Nan Xu, and Haiyun Shen contributed equally to this work.
Corresponding authors
Ethics declarations
Competing interests
All authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, H., Zou, L., Xu, N. et al. Deep learning radiomics based prediction of axillary lymph node metastasis in breast cancer. npj Breast Cancer 10, 22 (2024). https://doi.org/10.1038/s41523-024-00628-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-024-00628-4
- Springer Nature Limited