Abstract
DNA binding transcription factors possess the ability to interact with lipid membranes to construct ion-permeable pathways. Herein, we present a thiazole-based DNA binding peptide mimic TBP2, which forms transmembrane ion channels, impacting cellular ion concentration and consequently stabilizing G-quadruplex DNA structures. TBP2 self-assembles into nanostructures, e.g., vesicles and nanofibers and facilitates the transportation of Na+ and K+ across lipid membranes with high conductance (~0.6 nS). Moreover, TBP2 exhibits increased fluorescence when incorporated into the membrane or in cellular nuclei. Monomeric TBP2 can enter the lipid membrane and localize to the nuclei of cancer cells. The coordinated process of time-dependent membrane or nuclear localization of TBP2, combined with elevated intracellular cation levels and direct G-quadruplex (G4) interaction, synergistically promotes formation and stability of G4 structures, triggering cancer cell death. This study introduces a platform to mimic and control intricate biological functions, leading to the discovery of innovative therapeutic approaches.
Similar content being viewed by others
Introduction
Bio-inspired peptides exhibit a remarkable ability for self-assembly into nanostructures in specific microenvironments, enabling the formation of membrane channels upon insertion into biological or model lipid bilayers1,2. Some of these peptides induce cytotoxicity or potent antitumor activity2. Transmembrane channel proteins play a vital role in cellular homeostasis by transporting ions across lipid bilayer membranes1,2,3. However, their practical utility is limited by structural complexity and critical molecular mechanisms4,5,6. Efforts have been directed towards the development of synthetic ion channels with nucleic acids, natural transcription factors and peptides, and synthetic molecules by mimicking characteristics of biological ion channels7,8,9,10,11,12,13,14,15,16. Only a few small molecule ion transporters have been reported to exhibit therapeutic properties17,18,19,20,21,22,23. For instance, Zhang et al. reported a small molecule based cation transporter with the ability to kill cancer cells19. Crafting synthetic molecular ion transporters with therapeutic potential and understanding their biophysical and biological properties could provide critical insights into their functional mechanisms24,25,26,27,28,29,30,31,32,33,34,35,36. Peptide mimetics37,38,39,40,41, due to their unique structural diversity and potential role in biological systems, hold promise as essential chemotherapeutic agents by targeting DNA or its secondary structures.
Non-B-DNA four stranded secondary structures e.g., G-quadruplexes (G4s) play a crucial role in cellular growth by regulating the replication and transcription machinery42,43,44. G4s are commonly found in the promoter G-rich region of various proto-oncogenes (e.g., c-MYC, c-KIT), as well as in telomeres (h-TELO) of cancer cells. The c-MYC proto-oncogene predominantly controls cell proliferation, apoptosis, and drug resistance in various cancer types such as cervical, breast, and lung cancers44,45,46,47,48. The NHE (nuclease hypersensitive element) III1 region of c-MYC promoter is responsible for approximately 90% of its transcriptional activity, contains G4-forming sequences that act as transcriptional repressors45,46,47,48,49,50,51,52. Stabilizing these G4 structures with synthetic molecules has emerged as a potential strategy for cancer therapeutics, offering promising avenues for targeted cancer treatment53,54,55,56,57,58,59.
In this work, we show the development of a thiazole-based dimeric peptidomimetic (TBP2), which exhibits the unique ability to form self-assembled nanostructures in lipid microenvironments and effectively transport ions across model lipid bilayer membranes (Fig. 1). Moreover, TBP2 inhibits cancer cell growth by modulating intracellular ion concentrations and G4-mediated transcriptional regulation of oncogenes. This study represents the illustration of a DNA binding artificial ion channel, introducing an approach for engineering self-assembling peptidomimetics that can mimic and regulate essential biological functions (Fig. 1).
Results and discussion
Design and synthesis of self-assembling peptidomimetics
Thiazole peptides (TBP) were synthesized by the cycloaddition of azide and alkyne functionalized bis-thiazoles 1 and 2 in the presence of copper sulfate and sodium ascorbate in a solvent mixture of tert-butanol/water (Fig. 2(i), Fig. S1 and details in Supplementary Information). Bis-thiazole azide 1 and alkyne 2 were prepared using stepwise amide coupling, resulting in high overall yields. The dimeric peptide TBP2 was obtained by Boc-deprotection of TBP1. Due to the presence of dimeric bis-thiazole amide units connected via a triazole linkage, these peptidomimetics were expected to self-assemble into supramolecular nanoarchitectures, capable of transitioning between open and folded conformations in response to an external stimulus (Fig. 2i). Moreover, the thiazole units and aliphatic amino group were anticipated to facilitate the permeation of TBPs into cellular nuclei and target DNA secondary structures via stacking or groove binding mode39,40.
(i) Design and synthesis of dimeric thiazole peptidomimetics. (ii) Self-assembling properties of TBP2: (A) TEM micrograph of TBP2 showing distribution of vesicles (indicated with purple arrow). The TEM imaging were performed independently three times (n = 3). B The average size distribution (±SD) and abundance of TBP2 formed vesicles as measured by DLS method. C Vertically stacked supramolecular arrangement of six TBP2 molecules forming the ion channel, as obtained from the periodic propagation of an optimized TBP2 dimer followed by molecular dynamics minimization. D Top and (E) side views of the optimized structure of a single TBP2 molecule at the M06-2x/6-31 G(d,p) level of theory. The hydrogen bond distances are indicated in the Figure (F, G) Optimized structure of Na+ and K+ encapsulated TBP2, respectively. Each of the cations is coordinated with three donor atoms and the corresponding distances are indicated in the Figure. (iii) High Resolution TEM micrographs of TBP2 in NaCl (A–D) and KCl (E–H) buffers (pH 7.4); showing nanofibre and vesicular structures. The experiments were performed independently at least three times (n = 3).
Supramolecular self-assembly of TBPs into vesicles and nanofibres
Transmission Electron Microscopy (TEM) imaging revealed that TBP2 could self-organize into nanofibrous and vesicle like structures in both NaCl and KCl buffers. The dynamic light scattering (DLS) study further revealed the relative abundance of vesicles formed through the supra-molecular assembly of the peptidomimetic TBP2, showing an average size distribution of approximately 10 nm (Fig. 2(ii)B). The formation of multiple vesicular structures could presumably be due to both the hydrophobic and hydrophilic parts of the compounds and strong aggregation of aromatic surfaces.
However, in the case of peptidomimetic TBP1 with the Boc protecting group, no ordered structures similar to those of TBP2 were observed in the presence of either Na+ or K+ ions (see the Supplementary Information, Fig. S2). In the TEM image of TBP1, only a limited number of vesicular structures were observed, and they were not as distinct as those seen in TBP2. Consequently, the presence of the -NH2 group in TBP2 played a crucial role in promoting the formation of well-organized non-covalent structures. The supramolecular structure of TBP2 was determined by optimizing the molecule using quantum chemistry calculations at different levels (M06-2x/6-31 G(d,p) and B3LYP/6-31 G(d,p)). The optimized structure revealed that TBP2 became folded and stabilized by three hydrogen bonding interactions, one of which involves the terminal -NH2 group (Fig. 2(ii)D, (ii)E). The hydrogen bonding distances were calculated to be 1.85 Å, 1.88 Å, and 2.31 Å, respectively.
The structure exhibited an almost elliptical cavity, with dimensions of approximately 5.8 Å for the long axis and 4.6 Å for the short axis. Subsequently, Na+/K+ ions were placed within this cavity, and their coordination with the molecular structure was investigated through DFT optimization (Fig. 2(ii)F, (ii)G). The cations were observed to be well-contained within the cavity without causing significant changes to the surrounding molecular arrangement. The cations were coordinated to three donor atoms, a triazole nitrogen, a nitrogen atom from a thiazole ring and a carbonyl oxygen from a peptide linkage, and the corresponding distances are shown in Fig. 2(ii) F, (ii) G.
To quantify ion encapsulation, the binding energies of Na+ and K+ were calculated using the formula Ebinding = E(M-ion)-EM-Eion, where E(M-ion), EM, and Eion represent the electronic energies of the optimized structures of the molecule-ion composite system, the free molecule, and the cation, respectively. The binding energies for Na+ and K+ were calculated to be −75.6 kcal/mol and −55.9 kcal/mol, respectively at the M06-2x/6-31 G(d,p), and −74.6 kcal/mol, and −67.7 kcal/mol, respectively at B3LYP/6-31 G(d,p) level. The high binding energies indicate the strong stability of the folded molecular structure to accommodate Na+ and K+ ions within its cavity, and are similar to those found for these metal ions bound to the active sites of various proteins60. We further optimized the structure of two such folded molecules on top of each other, following periodic propagation to create a configuration which contains six vertically stacked molecules. Molecular dynamics energy minimization was performed for geometry relaxation, resulting in a structure where the vertical stacking of TBP2 maintained the pore arrangement observed in a single TBP2 molecule (Fig. 2(ii)C). The top view of this stacked arrangement confirmed the preservation of the pore structure (see Supplementary Information, Fig. S9d).
TBP2 transports cations across model membrane bilayers
Fluorescence based HPTS (8-hydroxypyrene-1, 3, 6-trisulfonic acid trisodium salt) assay was employed27 to investigate the potential formation of supra-molecular channel-like nanostructures and ion transport activity across lipid membranes (Fig. 3). HPTS was encapsulated in large unilamellar vesicles (LUVs) of average diameter ~70 nm (dynamic light scattering method), prepared from egg yolk L-α-phosphatidylcholine (EYPC) in HEPES NaCl or HEPES KCl buffer (10 mM HEPES, 100 mM NaCl or KCl, pH 6.4).
A–F HPTS assay for measuring the ion transport activity of TBP2 in the presence of Na+ and K+ ions. Buffer composition: Internal – (A, B) 10 mM HEPES 100 mM KCl (pH 6.4), (D, E) 10 mM HEPES 100 mM NaCl (pH 6.4), External – (A, E) 10 mM HEPES 100 mM KCl (pH 7.4), (B, D) 10 mM HEPES 100 mM NaCl (pH 7.4). C EC50 value determination of TBP2 in the presence of external buffer – 10 mM HEPES 100 mM NaCl or KCl (pH 7.4), internal buffer – 10 mM HEPES 100 mM KCl (pH 6.4). Data are presented as means ± SD (n = 3). Source data are available. F EC50 value determination of TBP2 in the presence of external buffer – 10 mM HEPES 100 mM NaCl or KCl (pH 7.4), internal buffer – 10 mM HEPES 100 mM NaCl (pH 6.4). Data are represented as means ± SD (n = 3). Source data are available. G Lucigenin assay of TBP2 for Cl- transport. Change in fluorescence intensity as a function of time in 225 mM NaNO3 buffer. H Safranin O assay for membrane polarization in the presence of HEPES-NaCl (external) and HEPES-KCl (internal) buffers. I CF release assay; determination of CF release percentage in the presence of TBP1 and TBP2 (External: 10 mM HEPES 100 mM NaCl, pH 7.4; Internal: 10 mM HEPES 100 mM NaCl, pH 7.4) after 8 minutes. Error bars represent ± SEM (n = 3) (J) Membrane colocalization of TBP1 and TBP2 in GUVs. Scale bars represent 5 µM (top row) and 2 µM (bottom row). The imaging experiments were performed independently at least three times (n = 3). HPTS, 8-hydroxypyrene-1, 3, 6-trisulfonic acid trisodium salt; CF Carboxyfluorescein, LUVs large unilamellar vesicles, GUVs giant unilamellar vesicles.
The liposomes were then suspended in 10 mM HEPES buffer (pH 6.4) containing 100 mM of MCl2 (Mn+ = Na+, K+, Cs+, Rb+, Li+). After incorporating TBP1 and TBP2 (dissolved in DMSO) into the bilayer membrane, an external pH gradient was generated by adding NaOH. The change in HPTS fluorescence intensity was recorded over time. TBP1 exhibited minimal changes in HPTS fluorescence up to 20 µM, while TBP2 displayed high transport activity for K+ (~70%) and Na+ (~65%) in the presence of HEPES NaCl or HEPES KCl internal buffer (pH 7.4) (Fig. 3).
Using the Hill equation, EC50 (concentration of molecules to achieve 50% ion transport activity) values were determined to be 3.1 µM and 14 µM for TBP2, whereas TBP1 showed EC50 values of 43 µM and 56 µM for K+ and Na+, respectively (Internal buffer: HEPES NaCl, pH 6.4) (Fig. 2 and see the Supplementary Information, Fig. S3). Substituting the internal buffer with KCl (10 mM HEPES, 100 mM KCl, pH 6.4) resulted in TBP2 displaying EC50 values of 5.8 µM and 7 µM for K+ and Na+, respectively. TBP2 exhibited relatively greater transport efficiency for K+ and Na+ in the presence of alternate internal buffers, possibly due to the exchange of K+ and Na+. The Hill coefficient of TBP2 for K+ (nK+ = 1.64) indicates that more than one molecule (i.e., positive co-operativity) might form supramolecular ion channels to transport K+. Thus, HPTS studies revealed that TBP2 could form channel like structures, facilitating efficient transportation of K+ and Na+, whereas TBP1 did not show channel-forming activities. The HPTS data further revealed EC50 values of 54 ± 2.7 µM, 26 ± 1.3 µM, and 14 ± 0.7 µM for Li+, Cs+, and Rb+, respectively, indicating greater transport efficiency of TBP2 for K+ (EC50 = 5.8 ± 0.3 µM) and Na+ (EC50 = 7 ± 0.4 µM) (see the Supplementary Information, Fig. S3.2). Based on HPTS data, the transport efficiency of TBP2 for different alkali cations can be inferred as K+ > Na+ > Rb+ > Cs+ » Li+.
Furthermore, we conducted a lucigenin assay to observe Cl− transport activity of TBP2, where EYPC-LUVs were filled with the lucigenin dye in 225 mM NaNO3 solution and the fluorescence intensity of the lucigenin (λem = 535 nm; λex = 455 nm) was monitored until 500 sec (Fig. 3G). No significant changes in lucigenin fluorescence were observed with incremental addition of TBP2; indicating no Cl- flux across the lipid membrane. Altogether, HPTS and lucigenin assay demonstrate that TBP2 preferentially transports cations over anions across unilamellar vesicles.
Membrane integrity and polarization evaluation
To assess the impacts of TBP1 and TBP2 on membrane integrity, a CF leakage assay was conducted using a self-quenched carboxyfluorescein dye (CF) (<10 Å in size) (Fig. 3I, see Supplementary Information, Fig. S4). The membrane-impermeable CF dye (40 mM) can efflux from the vesicles upon pore formation (>10 Å) or disruption of the LUVs, leading to an increase in fluorescence intensity. After addition of TBP1 and TBP2, the CF discharge percentage was calculated as 52% and 2.7% after 8 minutes, respectively. The lower CF release in the presence of TBP2 indicated conserved membrane integrity of LUVs, while the higher CF discharge suggested that TBP1 could disrupt the membrane structure or create larger openings in the vesicles.
Furthermore, a safranin O assay was performed to investigate TBP2-dependent membrane polarization using unilamellar vesicles. Upon addition of safranin O to the vesicular solution, the fluorescence (with or without TBP2) (1 µM) was monitored for 600 seconds at an excitation of 522 nm and emission of 581 nm (Fig. 3H). The data revealed a significant decrease in the fluorescence intensity of safranin O with increasing ligand concentration, indicating TBP2-induced membrane polarization in the presence of alternate NaCl or KCl buffers. These results demonstrate that TBP2 embeds within the membrane and forms channels to facilitate ion transport via the vesicular system.
Membrane localization and conductance measurement
Confocal microscopy was employed to examine the membrane embedding feature of TBP1 and TBP2 using Giant Unilamellar Vesicles (GUVs) and HeLa (cervical carcinoma) cells stained with nile red. The microscopic imaging results revealed that TBP2 could effectively embed within the GUV membrane and co-localize with nile red, the membrane staining dye (Fig. 3J). In contrast, TBP1 appeared to disrupt the membrane structure, corroborating the findings from the CF release assay, where TBP2 preserved the membrane integrity of the vesicles. Further, the imaging data confirmed the membrane insertion properties of the peptidomimetics TBP1 and TBP2 in HeLa cells, showing their rapid colocalization with nile red within the cell membrane (see the Supplementary Information, Fig. S5).
To gain real-time insights into the channel-forming behavior of the thiazolyl peptidomimetic TBP2, patch clamp experiments were performed using planar lipid bilayers. Cis and trans compartments, containing 1 M NaCl or 1 M KCl solutions, were separated by a planar lipid bilayer membrane composed of EYPC lipid. Currents were measured over time against different applied potentials (+ve and −ve). Upon adding TBP2 to the planar bilayer, distinct channel openings and closings were observed at +80 mV and −80 mV, providing the formation of channels across planar lipid bilayer membrane in the presence of both Na+ and K+ (Fig. 4A–D). When 1 M NaCl was present on both the cis and trans sides, TBP2 demonstrated multiple channel openings for Na+ transport at −80 mV (Fig. 4A). At the holding potential of +80 mV, it displayed multiple square-top behavior in the presence of either NaCl or KCl (Fig. 4C, D). TBP2 formed stable channel openings while transporting Na+, whereas its channel openings fluctuated more rapidly between closed and open conformations during K+ transport (Fig. 4). Furthermore, TBP2 demonstrated continuous channel openings and closings for a longer duration (measured up to ~15 seconds) in the presence of 1 M KCl at the applied potential of +100 mV, suggesting its potential to form channel-like structures within the lipid membrane (see the Supplementary Information, Fig. S6). TBP2 exhibited efficient transport of Na+ and K+ across planar lipid bilayer membrane with high conductance. The I-V plot of TBP2 exhibited an ohmic-linear relationship between current vs. voltage (Fig. 4G). The average conductance values for transporting Na+ and K+ were measured to be ~0.56 nS and ~0.68 nS in the presence of 1 M NaCl and KCl (Fig. 4E–J), respectively, indicating robust ion transport capabilities of TBP2 across the planar lipid bilayer membrane.
Current measurement in the presence of 1 M NaCl at (A) −80 mV, (C) + 80 mV or 1 M KCl at (B) −80 mV and (D) + 80 mV, respectively. Bar diagram for frequency vs current of TBP2 in the presence of 1 M NaCl at (E) −80 mV, (H) + 80 mV or 1 M KCl at (F) −80 mV, (I) + 80 mV. G I-V plot of TBP2 in the presence of 1 M MCl [M = Na+, K+, Cs+, Li+, Rb+] buffers (pH 7). Data are represented as means ± SD (n = 3) (J) Drop line plot of conductance of TBP2 in the presence of different metal ions. Data are represented as means ± SD (n = 3).
The current behavior of TBP2 was also examined in the presence of CsCl, LiCl, RbCl buffers to explore its voltage-dependent gating characteristics for other monovalent cations (e.g., Cs+, Li+, Rb+) besides Na+ or K+ (see the Supplementary Information, Fig. S7). The I-V analysis revealed that TBP2 exhibited comparatively lower conductance values for these metal ions (e.g., Cs+: 0.29 nS, Li+: 0.08, Rb+: 0.15 nS), in relation to applied positive or negative voltages. However, no significant currents were observed through TBP2 incorporated lipid membrane in the absence of applied potentials. These results indicate that TBP2 possesses distinct preferences for Na+ and K+ ions over other monovalent cations, and its conductance behavior is affected by the specific ions present in the buffer solution.
Molecular dynamics of ion transport
Molecular dynamics simulation studies were conducted using a vertically stacked arrangement of six TBP2 molecules embedded within a lipid bilayer of dimensions 10 × 8 nm in the XY plane (Fig. 5A). Subsequently, 500 ns production simulations were initially performed for the ion channel-embedded lipid membrane in 0.15 M NaCl and KCl aqueous solutions, respectively. Throughout the simulations, we monitored the number of Na+ and K+ ions present within the ion channel. Initially, the channel was devoid of any cations. However, within a few picoseconds of simulations (Fig. 5C), cations started entering the channel, and their number fluctuated continuously throughout the entire duration of the simulation.
A Structure of the ion-channel embedded lipid bilayer showing the vertical arrangement of the TBP2 molecules and Na+ within the channel. Lipid molecules have been blurred for clarity. B Schematic illustration of ion channel formation. C Number of ions within the ion channel for 500 ns production simulations for the channel-embedded lipid membrane in 0.15 molar NaCl and KCl medium. D Free energy profiles (in kcal/mol) in terms of potential of mean forces (PMF’s) for the passage of Na+, K+, and Cl- ions through the ion-channel at 310 K, calculated employing the adaptive biasing force (ABF) module implemented in NAMD 2.12.
Most of the time, we observed one or two Na+ and K+ ions inside the channel, with three cations present at fewer time instances. This dynamic variation in the number of cations within the ion channel affirmed the spontaneous passage of ions through the channel, where old ions exited, and new ions entered. The average interaction energy of a single cation with the entire ion channel was calculated to be −23.8 kcal/mol and −14.7 kcal/mol for Na+ and K+, respectively, which aligns with the trends obtained from our quantum chemistry calculations. A sodium ion demonstrated greater stability compared to a potassium ion within the channel. Thus, molecular dynamics simulations shed light on the intricate ion transport dynamics and the preferential stability of Na+ and K+ ions within the TBP2-induced ion channel. Additionally, we performed a 2500 ns long simulation with the same system as described above to observe the stability of the assembly of six TBP2 molecules inside the lipid membrane (Fig. S10, Supplementary Information). The results suggested that TBP2 was stable inside the lipid membrane throughout the entire duration of MD simulation, Supplementary Movie 1.
To unravel the thermodynamics of ion transport, we calculated the free energy profile for the passage of Na+, K+ and Cl- through the ion-channel (Fig. 5D). To this regard, we first placed an ion at one end of the ion channel, and then applied a biasing force, to facilitate its entry and passage through the channel. The resulting free energy profiles exhibit periodic patterns, representing the symmetry in the arrangement of the TBP2 molecules. The maximum free energy penalty observed for translocation was ~1.8 kcal/mol, ~3 kcal/mol, and ~32 kcal/mol for Na+, K+, and Cl−, respectively. While the magnitudes for the cations were well within the diffusion-controlled limit, favouring their spontaneous passage through the channel; the free energies were significantly high for Cl-, hindering the movement of chloride ions through the channel. Notably, the smaller free energy barriers observed for Na+ can be attributed to its smaller size and greater degree of stabilization within the channel, facilitating its spontaneous passage. On contrary, K+ faced more repulsion to move from the center of one TBP2 molecule to the next, resulting in relatively sharp free energy barriers for its transport. Given its larger ionic radius and negative charge, chloride experienced pronounced repulsion within the channel, leading to elevated free energy magnitudes. These results suggest that the ion channel had a higher affinity for incorporating smaller Na+ ions, allowing their easier transport compared to K+ ions, while the possibility of Cl- transport remained weak. The MD simulation study corroborates the findings from HPTS, lucigenin, and patch clamp data, collectively indicating that TBP2 facilitates the efficient transport of cations like Na+ and K+ with high ionic conductance, while it does not favor the transport of Cl- ions. Ngo et al. determined the free energies associated with the water-mediated ion transport through a transmembrane ion channel gramicidin A (gA), and found the magnitudes to be 3–5 kcal/mol for both Na+ and K+60,61. Our results corroborate with their findings, even though on the lower side as compared to those observed for gA.
TBP2 modulates intracellular Na+ and K+ concentrations in cancer cells
The in-cellulo characteristics of TBP1 and TBP2 were first evaluated by monitoring their cellular localization by confocal microscopy. The colocalization study with membrane staining dye nile red revealed that TBP2 efficiently embedded into the cell membrane immediately after treatment (see the Supplementary Information, Fig. S5). As TBP2 localized in the cell membrane and exhibited cation transportation (e.g., Na+ and K+) through vesicles, we continued to measure the intracellular concentrations of Na+ and K+ in cancer cells.
To accomplish this, we utilized well-known fluorescent dyes that bind to sodium and potassium, namely sodium-binding benzofuran isophthalate acetoxymethyl ester (SBFI-AM, Na+ probe) and potassium-binding benzofuran isophthalate acetoxymethyl ester (PBFI-AM, K+ probe), respectively20. The intracellular fluorescence measurements revealed that TBP2 (8 µM) significantly20 increased the concentration of Na+ up to ~ 37% and slightly reduced cytosolic K+ concentration by ~ 15% in HeLa cells (Fig. 6A). Similarly, TBP2-embedded A549 cells exhibited a ~19% increase in Na+ concentration and a ~10% decrease in intracellular K+ concentration compared to the control. These results can be rationalized by the different distributions of Na+ and K+ across cell membranes, where intracellular Na+ concentrations (~12 mM) are substantially lower than extracellular Na+ concentrations (~145 mM), and extracellular potassium concentrations (~4 mM) are significantly lower than its intracellular counterpart (~150 mM) under physiological conditions.
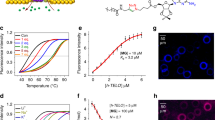
A TBP2 dependent intracellular fluorescence intensity measurements of Sodium-binding benzofuran isophthalate acetoxymethyl ester (SBFI-AM, Na+ probe) and Potassium-binding benzofuran isophthalate acetoxymethyl ester (PBFI-AM, K+ probe) in (i) HeLa and (ii) A549 cells. Error bars represent mean ± SD (n = 3). B Confocal images of HeLa cell (fixed) stained with TBP2 (blue) and BG4 (red); scale bars represent 10 µM. C Determining the average number of BG4 foci in untreated and ligand treated HeLa cells after 24 h. At least five independent fields were considered to quantify the BG4 foci. D qRT-PCR analysis for transcriptional regulation of c-MYC, BCL-2, c-KIT oncogene after treatment with TBP2 in HeLa cells for 24 h. Quantification was done in terms of fold change by double delta CT method using 18 s rRNA as a housekeeping gene. Fold change of ligand treated relative gene expression is normalized with control. Error bars represent mean ± SD (n = 3). **P = 0.0038, 0.0007 and *P = 0.0074, 0.0042, 0.0233, 0.0019; as obtained using paired sample statistical analysis (Student’s t test), versus untreated or control HeLa cells. E Relative luciferase expression of c-MYC promoter normalized with the Renilla plasmid pRL-TK after treatment with TBP2 at two different doses for 48 h. Percentage change of ligand treated relative luciferase expression is normalized with control. Error bars stand for mean ± SD (n = 3). **P = 0.0007, ***P = 0.00002; as obtained using paired sample statistical analysis (Student’s t test), versus untreated or control HeLa cells.; F Schematic illustration of TBP2-G4 interaction to mediate cancer cell death. G4; G-quadruplex.
However, after 24 h of incubation in HeLa cells, a significant accumulation of TBP2 within cellular nuclei was observed via confocal microscopy, possibly due to the molecule’s amphipathic nature. Given previous studies demonstrating that thiazole derivatives can bind to non-canonical DNA structures, we investigated the potential of both TBP1 and TBP2 to stabilize G4 DNAs in the cellular system. Interestingly, in TBP2-treated cells, the number of G4 binding antibody (BG4) foci significantly increased compared to both control cells and cells treated with TBP1 (Fig. 6B, C). This result suggests that TBP2 possessed the ability to stabilize G4 structures in cancer cells. Consequently, the data indicated that the significant intracellular increase of monovalent cation Na+ might disrupt the ionic balance, thereby synergistically contributing to the formation and stabilization of G-quadruplex structures. This effect could be a result of substantial accumulation of TBP2 within the nuclei of cancer cells after an extended incubation period.
The cell growth inhibition assay (see the Supplementary Information, Fig. S11) showed that TBP2 exhibited IC50 values of ~8.2, ~10 and ~16 µM in cervical carcinoma (HeLa), leukemia (K562) and lung carcinoma (A549) cells after 24 h, respectively. Intriguingly, it did not inhibit the growth of normal kidney epithelial (NKE) and human embryonic kidney epithelial (HEK293T) cells up to 100 µM. In contrast, TBP1 was found to be non-toxic to cancer cells as well as NKE cells up to 100 µM. The non-toxicity towards normal cell lines may be attributed to the low abundance of G4 structures in non-cancerous cell lines. As the proto-oncogenes like MYC, BCL-2 and c-KIT are overexpressed in human cancers, the regulatory role of TBP2 on the proto-oncogenic transcription was analysed in HeLa cells (Fig. 6D, see the Supplementary Information, Fig. S12). The qRT-PCR analysis revealed that TBP2 reduced c-MYC mRNA levels by ~44% and ~88% at 4 µM and 8 µM, respectively after 24 h while TBP1 reduced the relative c-MYC mRNA level only up to 42% at 40 µM (see the Supplementary Information, Fig. S12). TBP2 decreased c-KIT expression up to ~6% at 4 µM and ~59% at 8 µM; and BCL-2 up to ~20% at the highest dose after 24 h as compared to the control HeLa cells (Fig. 6D). Thus, the qRT-PCR data correlated with other biological studies and demonstrated the potential of the peptide mimic TBP2 to preferentially downregulate the c-MYC oncogene expression in cancer cells.
A dual luciferase assay, using wild type and mutant pGL3.0 variants of Del4 c-MYC promoter constructs and a renilla plasmid construct (pRL-TK) as the control vector, further confirmed the ability of TBP2 to alter c-MYC oncogene expression in HeLa cells by interaction with G4 harboring promoter region. TBP2 reduced the luciferase activity of c-MYC by ~67% and ~76% at 4 µM and 8 µM, respectively for the wild type promoter after 48 h (Fig. 6E). Intriguingly, TBP2 did not inhibit the luciferase expression of c-MYC mutants. These results demonstrate that TBP1 was not involved in the regulation of transcription or translation machinery while the synthetic ion transporter, TBP2 reduced the c-MYC oncogene expression through its interaction with the promoter region of the wild type c-MYC luciferase reporter plasmid (Fig. 6F).
TBP2 preferentially binds to c-MYC22 G4
As TBP2 interacts to the promoter region and alters oncogene expression, biophysical assays were conducted to study its binding potential with promoter G-quadruplexes. FRET based melting technique was used to evaluate TBP1 and TBP2 triggered stabilization of 5’-FAM and 3’-TAMRA conjugated G4 forming sequences (c-MYC22, c-KIT1, c-KIT2, BCL-2, h-TELO) and a duplex DNA (ds26 DNA) by comparing the melting temperature (TM) of control DNAs (without ligand) and ligand treated DNAs. TBP1 (2 µM or 10 eq. concentration of DNA) did not show significant changes in melting temperatures (TM) for the examined G4s and ds26 (ΔTM values; c-MYC22 = 3.5 ± 0.2 °C, BCL-2 = 1 ± 0 °C, c-KIT1 = 0 °C, c-KIT2 = 0.5 ± 0 °C, h-TELO = 6 ± 0.3 °C, ds26 DNA = 0 °C; Fig. 7A). Intriguingly, TBP2 displayed ΔTM values of 17 ± 0.9 °C for the c-MYC22 G4 DNA at 2 µM.
A 3D bar diagram of FRET based melting experiments to determine stabilization potenetial (ΔTM) of TBP1 and TBP2 for G4s and ds26 DNA. B FRET titration of TBP2 with G4s and ds26 DNA in 60 mM potassium cacodylate buffer (pH 7.4); Error bars represent mean ± SD (n = 3). Fluorescence titration of (C) TBP2 and (D) TBP1 in 60 mM potassium cacodylate buffer (pH 7.4); Error bars stand for mean ± SD (n = 3). 1D 1H NMR spectra of the (E) imino region of c-MYC22 in 25 mM Tris∙HCl buffer containing 100 mM KCl at different [TBP2:DNA] molar ratios. F NMR-structure of c-MYC22 (pdb-code: 1XAV).
However, mild or no alterations in ΔTM values were detected for other G4s and ds26 DNA (ΔTM values; BCL-2 = 3.8 ± 0.2 °C, c-KIT1 = 1.3 ± 0.1 °C, c-KIT2 = 4.1 ± 0.2 °C, h-TELO = 4.2 ± 0.2 °C, ds26 DNA = 0 °C; Fig. 7A, B). These results indicated that TBP2 selectively stabilized the c-MYC22 G4 with high stabilization potential (ΔTM) values over other G4s and ds26 DNA. Both TBP1 and TBP2 did not show any stabilizing effect for ds26 (duplex DNA). Fluorometric measurements revealed that TBP1 and TBP2 exhibit maxima at 417 nm upon excitation at 325 nm in potassium cacodylate buffer (pH 7.4). A significant enhancement (up to ~ 8 fold) of fluorescence emission intensity of TBP2 was observed upon gradual addition of pre-annealed c-MYC22 G4 DNA (Fig. 7C, see the Supplementary Information, Fig. S17B), while no noticeable changes in its intensity were observed with other DNA quadruplexes like c-KIT1, c-KIT2, BCL-2, h-TELO and ds26 DNA (Fig. 7C, see the Supplementary Information, Fig. S17). TBP2 exhibited a dissociation constant (Kd) of 1.6 µM for the c-MYC22 G4 (Fig. 7C) and higher Kd values for c-KIT1 (9.5 µM), h-TELO (8.2 µM) and ds26 DNA (8.2 µM), illustrating preferential binding of TBP2 to c-MYC G4 over other experimented G4s and ds26 DNA. Peptidomimetic TBP1 did not show affinity towards G4s or ds26 DNA as revealed from fluorescence titrations (Fig. 7D, see the Supplementary Information, Fig. S17).
The interaction of TBP2 with c-MYC22 was investigated by recording 1D 1H NMR spectra at different [TBP2]:[DNA] ratios (Fig. 7E, F, see the Supplementary Information, Fig. S18). The titrations were performed in 25 mM Tris∙HCl buffer containing 100 mM KCl. The c-MYC22 DNA formed a definite G-quadruplex structure that was described by Yang et al. (PDB: 1XAV). The DMSO effect on the c-MYC22 G-quadruplex was previously discussed62 and the assignment for the target DNA was followed from Yang et al.63. Clear changes in the 1D 1H spectrum upon addition of the ligand to the G-quadruplex formed by the sequence c-MYC22 were detected. The addition of 0.25 equivalents TBP2 led to general line broadening of the imino signals (Fig. 7E). However, further addition revealed stronger effects for the imino signals of G4, G9, G10, G13 and G17. For the imino signal of G6, a second signal for the bound state seemed to appear that was present in a 1:1 ratio at a [ligand:DNA] molar ratio of 1. In the aromatic region, the signals of A3, G2 or G4, G5, G13, T20, A21H2, A22, A22H2 and some signals of the overlapping region were mostly affected by ligand-binding (see the Supplementary Information, Fig. S18). The imino and aromatic signals showing the largest changes upon addition of TBP2 are marked in red and with spheres on the NMR-structure of c-MYC22 (pdb-code 1XAV). The data demonstrates that the strongest effect was observed for nucleotides on the lower and upper tetrad as well as both capping structures and G5 and G9 from the middle tetrad (Fig. 7E, F, see the Supplementary Information, Fig. S18). Thus, the interaction of TBP2 took place on the bottom and top of the G-quadruplex and there was probably a direct interaction with G6 as well.
We further employed classical MD simulations to gain insights and compare how TBP2 interacts with two topologically distinct G4s c-MYC22 (PDB: 1XAV) and h-TELO (PDB: 1KF1). For each of these G4s, two sets of MD simulations were conducted, placing the TBP2 molecule ~12 Å away from both the upper and lower tetrads of the quadruplex. Figure 8A displays the top and side views of the final composite structures for c-MYC and h-TELO G4s in both sets of simulations. The simulations results revealed that the TBP2 molecule binds to the upper and bottom tetrad motif of both the G4s; aligning with NMR findings. Further analysis showed that in the case of c-MYC; A2, G4, G5, G13, G17, T20, A21, and A22 residues interacted with TBP2, while for h-TELO; G2, G3, G4, T5, T11, G15, T17, G21, and G22 were involved in the interaction with TBP2.
A Top and side views of the final structures of TBP2, interacting with the top and bottom tetrad of c-MYC (1XAV) and h-TELO (1KF1) G-quadruplexes, as observed from two MD simulation trajectories. B Number of H-bonds between TBP2 and G-quadruplexes. C Free energies of binding between TBP2 and the two G-quadruplexes. Since the TBP2 molecule undergoes adsorption at different distances from the two G-quadruplexes, therefore, for a direct comparison, the reaction coordinate is represented as δ = d-d0, where, d and d0 are the distances at any time instant and the initial distance of adsorption, respectively.
The assessment of hydrogen bonds revealed that c-MYC formed 2-3 hydrogen bonds with TBP2, whereas h-TELO only formed a single hydrogen bond (Fig. 8B). However, the connections between TBP2 and the G4s mainly originated from van der Waals and long-range electrostatic interactions. The free binding energies of TBP2 were −11.5 kcal/mol for c-MYC and −3.9 kcal/mol for h-TELO G4s, suggesting a more favorable binding affinity with c-MYC, consistent with our biophysical findings. The simulation images indicate that TBP2 gets fitted onto the top or bottom of c-MYC, resulting in a higher binding energy, while the fitting cavity of h-TELO could not adequately accommodate the TBP2 molecule, leading to a comparatively weaker interaction (Fig. 8A).
In summary, we have designed a peptide mimic that forms vesicular and nanofibrillar non-covalent framework in response to specific microenvironments. The supramolecular nanostructures of TBP2 can span the lipid membrane, displaying distinct channel behavior in the presence of Na+ or K+, thereby regulating intracellular ion concentration. TBP2, possibly in its monomeric form, enters cell membranes and binds to non-canonical DNA structures in cell nuclei, and reduces cellular transcription.
Moreover, TBP2 rapidly localizes in model lipid bilayers and the plasma membrane to facilitate ion transport, while it accumulates in the nuclei of HeLa cells after prolonged duration (e.g., 24 h) in a time-dependent manner, showing enhanced fluorescence in cellular system. This versatility makes TBP2, a promising molecular scaffold for studying membrane related and intracellular processes. The formation of channels by TBP2, increase intracellular concentration of monovalent cations like Na+, the essential component of DNA G4 structures. The time-dependent membrane or nuclear localization, subsequent increased intracellular cation concentration and direct interaction with G4 structures promote the formation and stabilization of G4s, leading to cancer cell death. This class of synthetic peptide nanostructures provides structural and functional insights into ion channels and represents a paradigm for developing artificial transporters with therapeutic potential.
Methods
TEM imaging
TEM experiments were carried out in bright-field mode on a JEOL 1200 EX electron microscope, operated at an acceleration voltage of 120 keV. TBP1 or TBP2 were diluted in HEPES NaCl or HEPES KCl buffer (pH 7.4) at the con. of 20 µM. The sample was prepared by placing a drop (5 μL) of aqueous dispersions of TBP1 and TBP2 on a carbon-coated copper grid and air-dried at room temperature overnight.
Ion transport study by fluorescence spectroscopy
Large unilamellar vesicles (LUVs) were formed using a 200 μL 9:1 mixture of 10 mM EYPC (L-α-Phosphatidylcholine egg yolk) and cholesterol in chloroform. After solvent removal and vacuum drying, the resulting thin film was hydrated with 500 μL of buffer (10 mM HEPES, 100 mM NaCl or KCl, pH 6.4) containing 100 μM HPTS (8-hydroxypyrene-1,3,6- trisulfonic acid trisodium salt). Next, the suspension was subjected to six freeze–thaw cycles (liquid nitrogen/water at room temperature) during hydration. The resulting white suspension was then extruded 19 times through a 100 nm polycarbonate membrane to obtain large unilamellar vesicles (LUVs) with an average diameter of ~ 60 nm (as measured by DLS method). The LUVs suspension was separated from extravesicular HPTS dye by using size-exclusion chromatography (Econo-Pac 10DG column, Bio-rad; mobile phase: 10 mM HEPES, 100 mM NaCl or KCl, pH 6.4) and diluted with mobile phase for the desired working concentration.
TBP1 or TBP2 (0–20 µM) was added to a EYPC· LUVMs⊃ HPTS suspension ([EYPC] = 10 mM, [HPTS] = 100 μM) in 10 mM HEPES buffer containing 100 mM MCl [M = Na+, K+, Cs+, Rb+, Li+] (490 µL, pH 6.4) followed by subsequent addition of an aqueous solution of NaOH (0.5 M, 5 μL, ΔpH = 1) in a clean and dry fluorescence cuvette. Fluorescence intensity of HPTS at 510 nm upon excitation with 460 nm-light was monitored as a function of time until the addition of 1.0 wt% Triton X-100 (40 μL) at 450 s. Relative fluorescence intensity of HPTS was evaluated by the equation of
Where I0, and It represent the initial, final and ILysed represent the fluorescence intensities before addition of NaOH, after addition of NaOH and 1 wt% Triton X as lysis buffer, respectively.
The Hill equation was used to determine the EC50 values (concentration of molecules to achieve 50% ion transport activity) of the peptidomimetics:
I = relative fluorescence intensity, [Channel] = concentration of TBP1 or TBP2, n = Hill co-efficient
Formation of giant unilamellar vesicles (GUVs)
GUVs were prepared by electroformation technique (Vesi Prep Pro, Nanion, Germany). 10 µL of a 10 mM solution of EYPC and cholesterol (9:1) in chloroform was spread evenly on the indium tin oxide (ITO) coated glass slides within the “O” ring area. The solvent was evaporated at room temperature and the slides were dried under vacuum. Then, ITO slides were assembled in the Vesi Prep Pro and filled with 275 µL of sorbitol solution (1 M). A sinusoidal AC field of 3 V and 5 Hz was applied for 2 h at 25 °C temperature. The prepared GUV solution was collected and subjected to patch-clamp experiments.
Confocal imaging in GUVs and cells
GUVs were suspended in 10 mM HEPES, 100 mM KCl or 100 mM NaCl (pH 7.4) buffer and incubated with TBP1 or TBP2 (20 μM) and nile red (Sigma, USA) for 5 minutes. After incubation, the mixture was placed on the fluodish and observed under Confocal Microscope (Zeiss, Germany). For control slides, GUVs were incubated with either TBP2 or Nile red. At least 4 fields per slide and three independent sets were examined. HeLa cells were treated with TBP1 or TBP2 (8 µM) following nile red (10 µM) incubation 10 minutes prior to imaging. The images obtained were processed using ImageJ software.
Conductance measurements by automated patch-clamp technique
Conductance measurements were carried out using the Port-a-Patch setup (Nanion, Munich, Germany). First, a borosilicate glass chip (NPC chip, Nanion, Germany) with 3–5 mΩ was loaded with symmetrical working buffer containing 10 mM HEPES 1 M MCl [M = Na+, K+, Cs+, Li+, Rb+] buffers (pH 7.0) in both cis and trans compartments and Ag/AgCl electrodes were placed on both sides of the NPC chip. Next, bilayer membrane with >1 Giga Ohm resistance was constructed across the micrometer-sized aperture in the NPC chip by adding GUV suspension and applying a mild negative pressure (−10 mbar). TBP2 (20 μM) was added to the cis-side of the chamber. Current traces were recorded using an HEKA EPC 10 patch clamp amplifier with a built-in 1 kHz 4 pole Bessel low-pass filter and a Digidata 1322 A digitizer. I-V curve was generated using a voltage ramp from -80 mV to +80 mV. Data analysis was performed using Clampfit 10.2 software.
Measurement of intracellular Na+ and K+ concentration
HeLa or A549 cells were cultured in 96 well plates and incubated with 10 µM fluorescent Na+ probe [sodium-binding benzofuran isophthalate acetoxymethyl ester (SBFI-AM)] or K+ probe [potassium-binding benzofuran isophthalate acetoxymethyl ester (PBFI-AM)] in culture media for 1.5 h at 37 °C. Cells were washed with 1x PBS to remove the free extracellular SBFI-AM or PBFI-AM following treatment with TBP2 at different concentrations (0, 1, 2, 5, 8, 20 µM) with 2 h incubation period at 37 °C. The fluorescence of SBFI-AM (λex = 350 nm, λem = 527 nm) or PBFI-AM (λex = 350 nm, λem = 527 nm) was measured in a microplate reader (Molecular Devices, USA).
NMR titration
The c-MYC22 DNA was purchased from Eurofins MWG Operon in HPSF grade followed by purification with HPLC. The titration was carried out with 100 µM DNA in 25 mM Tris•HCl buffer (pH 7.4), which contains 100 mM KCl in 5% d6-DMSO/95% H2O. Low volume of TBP2 stock solution in 100% d6-DMSO was added directly into the NMR tube (7.5% d6-DMSO at the end of the titration). 2,2-dimethyl-2-silapentane-5-sulfonate (DSS) was kept as reference. For water suppression, gradient-assisted excitation sculpting or jump-return-Echo was used.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data that support the findings of this study are available in the manuscript and Supplementary Information file. The reported PDB codes and crystal structures of c-MYC (1XAV) and h-TELO (1KF1) are given in the manuscript. The atomic coordinates of the optimized computational models are deposited in the Zenodo OpenAIRE database under doi: 10.5281/zenodo.11118221. All other information is available from the corresponding authors upon request. Source data available with this manuscript. Source data are provided with this paper.
References
Montenegro, J., Ghadiri, M. R. & Granja, J. R. Ion channel models based on self-assembling cyclic peptide nanotubes. Acc. Chem. Res. 46, 2955–2965 (2013).
Otis, F., Auger, M. & Voyer, N. Exploiting peptide nanostructures to construct functional artificial ion channels. Acc. Chem. Res. 46, 2934–2943 (2013).
Fuertes, A., Juanes, M., Granja, J. R. & Montenegro, J. Supramolecular functional assemblies: dynamic membrane transporters and peptide nanotubular composites. Chem. Commun. 53, 7861–7871 (2017).
Kourie, J. & Shorthouse, A. Properties of cytotoxic peptide-formed ion channels. Am. J. Physiol. Cell Physiol 278, C1063–C1087 (2000).
Hille, B. Ionic Channels of Excitable Membranes 3rd edn (Sinauer Associates, Inc., 2001).
Thompson, M. J. & Baenziger, J. E. Ion channels as lipid sensors: from structures to mechanisms. Nat. Chem. Biol. 16, 1331–1342 (2020).
Sakai, N. & Matile, S. Synthetic ion channels. Langmuir 29, 9031–9040 (2013).
Sato, K., Muraoka, T. & Kinbara, K. Supramolecular transmembrane ion channels formed by multiblock amphiphiles. Acc. Chem. Res. 54, 3700–3709 (2021).
Howorka, S. Building membrane nanopores. Nat. Nanotechnol. 12, 619–630 (2017).
Burns, J. R., Al-Juffali, N., Janes, S. M. & Howorka, S. Membrane-spanning DNA nanopores with cytotoxic effect. Angew. Chem. Int. Ed. 53, 12466–12470 (2014).
Tosteson, M. T., Kim, J. B., Goldstein, D. J. & Tosteson, D. C. Ion channels formed by transcription factors recognize consensus DNA sequences. Biochim Biophys Acta 1510, 209–218 (2001).
Zheng, S. P., Huang, L. B., Sun, Z. & Barboiu, M. Self-assembled artificial ion-channels toward natural selection of functions. Angew. Chem. Int. Ed. 60, 566–597 (2021).
Yang, J. et al. Artificial transmembrane ion transporters as potential therapeutics. Chem. 7, 3256–3291 (2021).
Picci, G., Marchesan, S. & Caltagirone, C. Ion channels and transporters as therapeutic agents: from biomolecules to supramolecular medicinal chemistry. Biomedicines 10, 885 (2022).
Xin, W., Jiang, L. & Wen, L. Engineering bio‐inspired self‐assembled nanochannels for smart ion transport. Angew. Chem. Int. Ed. 134, e202207369 (2022).
Haynes, C. J. et al. Blockable Zn10L15 ion channels through subcomponent self-assembly. Angew. Chem. Int. Ed. 56, 15388–15392 (2017).
Trabocchi, A. Principles and applications of small molecule peptidomimetics. Small Molecule Drug Discovery 163–195 (Elsevier, 2020).
Spruijt, E., Tusk, S. E. & Bayley, H. DNA scaffolds support stable and uniform peptide nanopores. Nat. Nanotechnol. 13, 739–745 (2018).
Zhang, H., Ye, R., Mu, Y., Li, T. & Zeng, H. Small molecule-based highly active and selective K+ transporters with potent anticancer activities. Nano Lett. 21, 1384–1391 (2021).
Park, S.-H. et al. Synthetic Na+/K+ exchangers promote apoptosis by disturbing cellular cation homeostasis. Chem. 7, 3325–3339 (2021).
Roy, A. & Talukdar, P. Recent advances in bioactive artificial ionophores. ChemBioChem 22, 2925–2940 (2021).
Rodríguez-Vázquez, N., Amorín, M. & Granja, J. Recent advances in controlling the internal and external properties of self-assembling cyclic peptide nanotubes and dimers. Org. Biomol. Chem. 15, 4490–4505 (2017).
Alfonso, I. & Quesada, R. Biological activity of synthetic ionophores: ion transporters as prospective drugs? Chem. Sci. 4, 3009–3019 (2013).
Shen, F.-F. et al. Mediating K+/H+ transport on organelle membranes to selectively eradicate cancer stem cells with a small molecule. J. Am. Chem. Soc. 142, 10769–10779 (2020).
Debnath, M. et al. Ionophore constructed from non-covalent assembly of a G-quadruplex and liponucleoside transports K+-ion across biological membranes. Nat. Commun. 11, 1–12 (2020).
Si, W., Xin, P., Li, Z.-T. & Hou, J.-L. Tubular unimolecular transmembrane channels: construction strategy and transport activities. Acc. Chem. Res. 48, 1612–1619 (2015).
Gilles, A. & Barboiu, M. Highly selective artificial K+ channels: an example of selectivity-induced transmembrane potential. J. Am. Chem. Soc. 138, 426–432 (2016).
Krishnan R, S. et al. Autonomously assembled synthetic transmembrane peptide pore. J. Am. Chem. Soc. 141, 2949–2959 (2019).
Thomson, A. R. & Brady, R. L. Constructing ion channels from water-soluble α-helical barrels. Nat. Chem. 13, 643–650 (2021).
August, D. P. et al. Transmembrane ion channels formed by a star of David [2] catenane and a molecular pentafoil knot. J. Am. Chem. Soc. 142, 18859–18865 (2020).
Qi, S. et al. Foldamer-based potassium channels with high ion selectivity and transport activity. J. Am. Chem. Soc. 143, 3284–3288 (2021).
Su, G., Zhang, M., Si, W., Li, Z. T. & Hou, J. L. Directional potassium transport through a unimolecular peptide channel. Angew. Chem. Int. Ed. 55, 14678–14682 (2016).
Ye, R. et al. Molecular ion fishers as highly active and exceptionally selective K+ transporters. J. Am. Chem. Soc. 141, 9788–9792 (2019).
Matile, S., Sakai, N. & Hennig, A. Transport experiments in membranes. Supramol. Chem. From Mol. NanoMater. 2, 473-500 (2012).
Chen, S. et al. An artificial molecular shuttle operates in lipid bilayers for ion transport. J. Am. Chem. Soc. 140, 17992–17998 (2018).
El Ghoul, Y. et al. Biomimetic artificial ion channels based on beta-cyclodextrin. Chem. Commun. 49, 11647–11649 (2013).
Paul, R., Dutta, D., Paul, R. & Dash, J. Target‐directed azide‐alkyne cycloaddition for assembling HIV‐1 TAR RNA binding ligands. Angew. Chem. 132, 12507–12511 (2020).
Madhu, C., Voshavar, C., Rajasekhar, K. & Govindaraju, T. Cyclic dipeptide based cell-penetrating peptidomimetics for effective DNA delivery. Org. Biomol. Chem. 15, 3170–3174 (2017).
Dutta, D. et al. Cell penetrating thiazole peptides inhibit c-MYC expression via site-specific targeting of c-MYC G-quadruplex. Nucleic Acids Res. 46, 5355–5365 (2018).
Paul, R., Dutta, D., Das, T., Debnath, M. & Dash, J. G4 sensing pyridyl-thiazole polyamide represses c‐KIT expression in leukemia cells. Chem. Eur. J. 27, 8590–8599 (2021).
Asamitsu, S., Obata, S., Yu, Z., Bando, T. & Sugiyama, H. Recent progress of targeted G-quadruplex-preferred ligands toward cancer therapy. Molecules 24, 429 (2019).
Bochman, M. L., Paeschke, K. & Zakian, V. A. DNA secondary structures: stability and function of G-quadruplex structures. Nat. Rev. Genet. 13, 770–780 (2012).
Siddiqui-Jain, A., Grand, C. L., Bearss, D. J. & Hurley, L. H. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-MYC transcription. Proc. Natl. Acad. Sci. USA 99, 11593–11598 (2002).
Balasubramanian, S., Hurley, L. H. & Neidle, S. Targeting G-quadruplexes in gene promoters: a novel anticancer strategy? Nat. Rev. Drug Discov 10, 261–275 (2011).
Yang, D. & Hurley, L. H. Structure of the biologically relevant G-quadruplex in the c-MYC promoter. Nucleosides Nucleotides Nucleic Acids 25, 951–968 (2006).
González, V. & Hurley, L. H. The c-MYC NHE III1: function and regulation. Annu. Rev. Pharmacol. Toxicol. 50, 111–129 (2010).
Hänsel-Hertsch, R. et al. Landscape of G-quadruplex DNA structural regions in breast cancer. Nat. Genet. 52, 878–883 (2020).
Phan, A. T., Kuryavyi, V., Gaw, H. Y. & Patel, D. J. Small-molecule interaction with a five-guanine-tract G-quadruplex structure from the human MYC promoter. Nat. Chem. Biol. 1, 167–173 (2005).
Wang, W. et al. Human MYC G-quadruplex: from discovery to a cancer therapeutic target. Biochim. Biophys. Acta Rev. Cancer 1874, 188410 (2020).
Carvalho, J., Mergny, J.-L., Salgado, G. F., Queiroz, J. A. & Cruz, C. G-quadruplex, friend or foe: the role of the G-quartet in anticancer strategies. Trends Mol. Med. 26, 848–861 (2020).
Wang, K.-B. et al. Indenoisoquinoline topoisomerase inhibitors strongly bind and stabilize the MYC promoter G-quadruplex and downregulate MYC. J. Am. Chem. Soc. 141, 11059–11070 (2019).
Long, W. et al. Rational design of small-molecules to recognize G-quadruplexes of c-MYC promoter and telomere and the evaluation of their in vivo antitumor activity against breast cancer. Nucleic Acids Res. 50, 1829–1848 (2022).
Zheng, B.-X. et al. A small-sized benzothiazole–indolium fluorescent probe: the study of interaction specificity targeting c-MYC promoter G-quadruplex structures and live cell imaging. Chem. Commun. 56, 15016–15019 (2020).
Minard, A. et al. A short peptide that preferentially binds c-MYC G-quadruplex DNA. Chem. Commun. 56, 8940–8943 (2020).
Müller, D., Saha, P., Panda, D., Dash, J. & Schwalbe, H. Insights from binding on quadruplex selective carbazole ligands. Chem. Eur. J. 27, 12726–12736 (2021).
Zuffo, M. et al. More is not always better: finding the right trade-off between affinity and selectivity of a G-quadruplex ligand. Nucleic Acids Res. 46, e115–e115 (2018).
Krafcikova, M. et al. Monitoring DNA–ligand interactions in living human cells using NMR spectroscopy. J. Am. Chem. Soc. 141, 13281–13285 (2019).
Ma, Y. et al. Vinylnaphthalene-bearing hexaoxazole as a fluorescence turn-on type G-quadruplex ligand. Org. Biomol. Chem. 19, 8035–8040 (2021).
Yang, M. et al. Targeting a noncanonical, hairpin-containing G-quadruplex structure from the MYCN gene. Nucleic Acids Res. 49, 7856–7869 (2021).
Li, H. et al. Representation of ion–protein interactions using the drude polarizable force-field. J. Phys. Chem. B 119, 9401–9416 (2015).
Ngo, V. et al. Polarization effects in water-mediated selective cation transport across a narrow transmembrane channel. J. Chem. Theory Comput. 17, 1726–1741 (2021).
Kumar, Y. P. et al. A fluorescent guanosine dinucleoside as a selective switch-on sensor for c-myc G-Quadruplex DNA with potent anticancer activities. Chem. Eur. J. 19, 11502–11506 (2013).
Ambrus, A., Chen, D., Dai, J., Jones, R. A. & Yang, D. Solution structure of the biologically relevant G-quadruplex element in the human c-MYC promoter. Implications for G-quadruplex stabilization. Biochemistry 44, 2048–2058 (2005).
Acknowledgements
R.P. thanks DST-India for INSPIRE fellowship [IF160996]. D.D. and B.L. thank CSIR-India for a research fellowship. The authors thank Mrs. Debapriya Ghatak, IACS for Confocal Microscopy. We thank Sri Gopal Manna, IACS for the 3D artwork. This work was supported by Wellcome Trust-DBT India Alliance [Grant Number, IA/S/18/2/503986] fellowship grant and DST-SERB CRG research grant [CRG/2021/004525]. We also thank Technical Research Committee (TRC) grant, IACS for the additional funding.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. J.D. conceived the study; J.D. and R.P. designed the experiments; J.D. and R.P. wrote the paper; D.D. and B.L. synthesized the molecules; R.P. conducted the biophysical and biological experiments; T.M. and A.D. performed the quantum chemistry calculations and molecular dynamics simulations; D.M. and H.S. contributed to the NMR experiments and data analysis. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Van Ngo and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Paul, R., Dutta, D., Mukhopadhyay, T.K. et al. A non-B DNA binding peptidomimetic channel alters cellular functions. Nat Commun 15, 5275 (2024). https://doi.org/10.1038/s41467-024-49534-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-49534-0
- Springer Nature Limited